Abstract
Mitochondrial dysfunction and oxidative stress play essential roles in the pathogenesis of diabetic nephropathy (DN). The respiratory oxygen consumption and oxidative stress status of kidney mitochondria are closely associated with the development of DN. In this study, raspberry ketone (RK), the predominant bioactive component extracted from raspberry, was applied to treat the established DN mice model. This study investigated whether RK protects the kidneys of high-fat and high-sugar/streptozotocin (STZ)-induced diabetic rats by inhibiting oxidative stress and ameliorating mitochondrial dysfunction. Besides, the DN mice models were established by injecting high-fat and high-sugar/STZ (130 mg/kg, intraperitoneal injection). The animals were randomly divided into the control group (normal saline, ig), DN group (normal saline, ig), DN + RK group (200 mg/kg RK + normal saline, ig), DN + RK group (400 mg/kg RK + normal saline, ig), and DN + Metformin (Met) (200 mg/kg Met + normal saline, ig). Regular monitoring of fasting blood glucose (FBG) levels was observed in mice. After 10 weeks of drug treatment, the kidneys of mice in each group were analyzed using ultrasound, and the mice were euthanized humanely. Kidney weight (KW)/body weight (BW) and kidney injury, mitochondrial function, and oxidative stress indicators were determined. The histopathological changes in renal tissue were observed after hematoxylin and eosin (H&E) staining. The results recommended that RK has a renoprotective function on DN mice by improving mitochondrial dysfunction and inhibiting oxidative stress.
Introduction
Diabetes is a chronic disease with abnormal blood glucose and fat metabolism as the principal clinical features.1,2 Diabetic nephropathy (DN) is among the most prevalent microvascular complications in diabetic patients and more than 80% of end-stage renal disease (ESRD) is caused by abnormal elevation of blood glucose.3,4 DN causes substantial morbidity and mortality in diabetic patients.5,6 Controlling glucose and lipid levels in the blood is the primary method for preventing the development of DN. Nonetheless, the therapeutic effect of controlling blood glucose or lipid levels alone is not enough. Due to the intricate pathogenesis of diabetes, researchers have also introduced other therapeutic approaches, such as glycolipid control and anti-oxidative stress.3,4,7,8 In accordance with these studies, a combination of distinct treatment methods is more effective and can alleviate some complications of diabetes to some extent, especially in the prevention and treatment of DN. Therefore, the search for new treatment options has become a crucial research direction in the prevention and treatment of DN.
The kidney is rich in mitochondria which provide energy during metabolism and play an significant role in the physiology and pathophysiology of the kidney.9,10 Meanwhile, mitochondrial dysfunction plays a pathogenic role in kidney injury.11,12 And acute kidney injury can be significantly improved by regulating mitochondrial homeostasis in renal tubules and renal mitochondrial fission.13,14 In addition, studies have demonstrated that mitochondrial dysfunction leads to the overproduction of reactive oxygen species (ROS), a major cause of oxidative stress, which plays an essential role in the initiation and progression of DN.9,15 Currently, studies have revealed that by regulating mitochondrial fission, the oxidative stress of the kidney is inhibited, and the homeostasis of the kidney mitochondria is maintained, which ultimately attenuates renal failure induced by ischemic acute kidney injury. 16 Meanwhile, excessive production of ROS during oxidative stress reduces the efficiency of the electron transport chain (ETC) and causes irregular mitochondrial protein synthesis. 17 In addition, the overproduction of ROS in kidney mitochondria has a negative impact on insulin sensitivity, which will contribute to the decline of DN.
Raspberry is a plant of the genus raspberry in the family Rosaceae, which has many Chinese pharmacological effects such as anti-cancer, anti-diabetes, and kidney protection.15,18,19 As a functional compound, raspberry ketone (RK) is one of the core components of raspberry fruit, which has abundant pharmacological effects, namely antioxidant, anti-inflammatory, anti-diabetic, etc15,18,19 RK markedly enhanced insulin sensitivity, decreased lipid profile parameters, increased nuclear factor erythroid-derived 2-related factor 2 (Nrf-2) expression, and GSH and p-Irs1/p-Akt/GLUT4 signaling while it decreased malondialdehyde (MDA), Leptin/Stat-3, and Srebp-1c in animal models of diet-induced insulin resistance. 18 Additionally, RK has been reported to activate peroxisome proliferation-activated receptor α (PPARα) and alleviate oxidative damage and hyperlipidemia through its antioxidant properties.15,19 Nonetheless, there are few reports that RK exerts a protective effect on the kidney by enhancing mitochondrial dysfunction and inhibiting oxidative stress. Moreover, our research will offer a new experimental foundation for the clinical treatment of DN.
Materials and Methods
Drugs and Reagents
The RK extracted from the plant raspberry produced in Changbai Mountain, in the Jilin area of China, was acquired from Shaanxi Hebu Biological Co., Ltd. Glucose assay kit, creatinine (CRE) assay kit, mice urine microalbumin-linked immunoassay kit, superoxide dismutase (SOD) assay kit, glutathione peroxidase (GSH-Px) assay kit, Catalase (CAT) assay kit, and MDA assay kit, triglyceride (TG) assay kit, total cholesterol (TC) assay kit, and low-density lipoprotein (LDL) cholesterol assay kit were purchased from Nanjing Jiancheng Bioengineering Institute, Jiangsu, China. Streptozotocin (STZ) was acquired by Sigma. The high-fat and high-sugar feed were purchased from Changchun Yisi Experimental Animal Technology Co., Ltd. D-glucose was purchased from Biotech Bioengineering (Shanghai) Co., Ltd.
Animal Experiments
Male, 8-week-old C57 BL/6J mice, weighing 20 to 22 g (purchased from Liaoning Changsheng Biotechnology Co., Ltd). The animals were housed in a standard animal room (temperature: 21-23 °C, humidity: 50%, light: 12: 12 h light/dark cycles). All animals received a standard diet and were allowed free access to water ad libitum. Mice were acclimatized for 1 week prior to initiating the experiment. The entire experimental process was approved by the Animal Ethics Committee of Jilin Medical University (Approval number: 2019-lw001).
Experimental Design
The mice were fed with ordinary maintenance fodder for a week to assist them to adapt to the environment. Mice were fed with the high-glucose-high-fat diet for 4 weeks and injected with a once coeliac injection of freshly prepared STZ solution (130 mg/kg). Fasting blood glucose (FBG) level was measured a week after STZ administration with the aid of a glucose assay kit (fasting and free drinking water for 12 h). Mice with fasting plasma glucose levels≥ 240 mg•dL−1 were used in the experimental research.
The mice were randomly divided into 5 groups according to blood glucose level, and followed these intervention regimens (n = 10): Group I (n = 10) Normal control group (Con), Group II (n = 10) DN model group (DN), Group III (n = 10) DN + 200 mg/kg/day RK group (RK 200), Group IV (n = 10) DN + 400 mg/kg/day RK group (RK 400), and Group V (n = 10) DN + 200 mg/kg/day Metformin (Met).
Groups I and II were administered with an equal amount of normal saline daily, Group III and Group IV were administered with RK (200, 400 mg/kg/day), and Group V was administered with Met (200 mg/kg/day) for a total of 10 weeks. At the end of the experiment, the blood samples were collected from the venous plexus after the eyeball and were executed by cervical dislocation. The blood samples were centrifuged at 3000 rpm for 10 min to obtain the serum and detect biochemical indicators. The kidneys were dissected and rinsed with cold saline, and a portion of the kidneys was kept in 10% formalin and utilized for hematoxylin and eosin (H&E)-stained and Masson-stained. Urine was harvested from each group for examination of urine and ultrasonic inspection before the mice were executed. The other portion of the kidneys was minced and homogenized in an ice-cold phosphate buffer saline solution (0.02 M, pH 7.4). The homogenate obtained from the kidneys was centrifuged and the supernatant obtained was stored at 4 °C for further analysis.
Oral Glucose Tolerance Test (OGTT)
FBG levels were determined at 0 and 10 weeks by a commercial kit (Jiancheng Bioengineering Institute, Nanjing, Jiangsu, China) upon mice had fasted for 12 h. The mice fasted and freely drank water for 12 h, and D-glucose (2 g/kg body weight) was intragastrically administered. The blood glucose levels were observed at 0, 30, 60, 90, and 120 min after gavage administration in accordance to the manufacturer's kit instructions.
Kidney Organ Coefficient Calculations
At the beginning of the 10th week, the size of the kidneys was produced by an animal diagnostic ultrasound system. Mice that had fasted for 1 night were weighed. The kidneys were immediately removed and weighed after humane euthanasia. The organ coefficient of kidneys was calculated by the following formula: KW/BW = kidney weight (g)/body weight (g).
Mitochondrial Functional Assessment
Mitochondrial function was measured using OROBOROS O2K high-resolution mitochondrial function/cell energy metabolism measurement system. After oxygen calibration, 2mL miR05-kit was added into the glass chamber, and then various substrates, uncoupling agents, inhibitors, and other substances were added, respectively. Furthermore, the data were analyzed with O2K software, and the maximum respiratory oxygen consumption, ADP-related oxygen consumption, ADP-irrelevant oxygen consumption, and the ROS generation rate could be comprehensively detected in real time.
Assessment of Oxidative Stress and Antioxidation
The left renal tissues were homogenized with ice-cold normal saline (W/V = 1 g/9 mL). Homogenates were centrifuged (4000 rpm, 20 min, 4 °C) and the aliquots of supernatant were collected. The expressive activity with SOD, GSH-Px, CAT, and the content of MDA was determined with the help of a commercially obtainable assay kit (Jiancheng Bioengineering Institute, Nanjing, Jiangsu, China) using multifunctional high-end enzyme labeling instrument (TECAN, Austria) according to the manufacturer's kit instructions.
Assessment of Serum Creatinine (Scr)
Blood samples were drawn from the retrobulbar venous plexus and serum samples were separated from blood samples collected in the 10th week by centrifugation (3500 rpm, 15 min, 4 °C). CRE levels needed to be determined in serum using a multifunctional high-end enzyme labeling instrument (TECAN, Austria) with the assistance of a commercially obtainable assay kit (Jiancheng Bioengineering Institute, Nanjing, Jiangsu, China) based on the kit manufacturer' instructions.
Assessment of Urine Parameters
At the end of the experiment, the urine of mice within 24 h was collected to detect urinary microalbumin (mALB) and CRE. The creatinine clearance (Ccr) was calculated by combing urine creatinine (Ucr) with Scr. Ccr = urinary creatinine × 24 h urine output/Scr /2440 min.
Histopathological Assessments
The left kidney samples were fixed in 10% formalin for 24 h, then dehydrated and integrated into paraffin. Sections of 4 μm thicknesses were sliced from each embedded sample and stained with H&E and Masson. The examination of renal tissue was performed under optical microscopy with the magnification of 400x by a pathologist in a blind manner for assessment.
Statistical Analysis
The experimental results are indicated as mean ± SD. Statistical analysis of data was executed making use of GraphPad Prism 8 software (San Diego, California, USA). Statistical dissimilarity significance analysis was determined by Student's t-test or one-way ANOVA. A P value < .05 was examined as statistically significant.
Results
Effect of RK on Blood Glucose and Blood Lipids in DN Mice
For the purpose of assessing the effect of RK on FBG and glucose tolerance, we tested the effect of RK on FBG levels in DN mice. In comparison with the Con mice, the FBG of the DN mice was substantially increased, and the FBG was significantly decreased after treatment with RK. Consequently, we performed an OGTT at 10 weeks of dosing to confirm the hypoglycemic effect of RK (Figure 1A-C). We also tested the effect of RK on serum TG, TC, and LDL in DN mice. Compared with the Con mice, the TG, TC, and LDL in the serum of the DN mice were increased and enhanced after treatment with RK (Figure 1D-F).

Effect of RK on blood glucose and blood lipids. (A) FBG, (B) OGTT, (C) OGTT(AUC), (D) serum TG, (E) Serum TC, and (F) Serum LDL-c.
Effect of RK on Kidney Mass-to-Body Weight Ratio and Cross-Sectional Area in DN Mice
The kidney cross-sectional area of each group of mice was measured using ultrasound imaging technology, and the results indicated that the kidney cross-sectional area of the DN mice was substantially increased, and the kidney cross-sectional area was significantly reduced upon treatment with RK (Figure 2A-F). The KW and BW of each group of mice were recorded and the KW/BW was compared, and the results demonstrated that the KW/BW of the DN mice increased in comparison with the Con mice, and the KW/BW was significantly decreased after treatment with RK (Table 1).

Ultrasound images of the kidney of mice. (A) Control, (B) DN, (C)RK 200, (D) RK 400, (E) Met, and (F) renal transverse plane area.
Comparison of the BW, KW, and KW/BW (%) in Each Group.

*P < 0.05, **P < .01 and ***P < .001 versus Con #P < .05 and ##P < .01 versus DN.
Abbreviations: BW: body weight; DN: diabetic nephropathy; KW: kidney weight; Met: Metformin; RK: raspberry ketone.
Effect of RK on mALB, Scr, Ucr, and Ccr Expression in the Kidney
To further verify the effect of RK on kidney damage in DN mice, we studied the indicators related to kidney damage. In comparison to the Con mice, the content of mALB and Scr in DN mice was significantly increased, and the content of Ucr and Ccr was substantially decreased, indicating that kidney damage occurred in high-fat diet/STZ-induced mice. After RK treatment, the levels of mALB and Scr in mice were significantly decreased, and the content of Ccr was markedly decreased. Taken together, RK can ameliorate renal injury in mice (Figure 3A-D).

Effect of RK on serum and urine biochemical parameters. (A) mALB, (B) Scr, (C) Ucr, and (D) Ccr.
Effect of RK on Kidney Mitochondrial Dysfunction
Intending to evaluate the effect of RK on the dysfunction of the kidney mitochondria in DN mice, we measured the oxygen consumption of the kidney mitochondria at various stages. The maximum respiratory oxygen consumption and the ADP-related oxygen consumption of the kidney mitochondria of the DN mice were considerably lower than those of the Con mice. After RK treatment, the maximum respiratory oxygen consumption and the ADP-related oxygen consumption of the DN mice were noticeably improved.
Compared with Con mice, the ADP-independent oxygen consumption in the DN mice was significantly increased. After RK treatment, correspondingly, the ADP-independent oxygen consumption in the DN mice was significantly reduced. It has been founded that RK could improve mitochondrial dysfunction in DN mice (Figure 4A-C).

Effect of raspberry ketone (RK) on kidney mitochondrial dysfunction. (A) Maximum respiratory oxygen consumption, (B) ADP-related oxygen consumption, and (C) ADP-independent oxygen consumption.
Effect of RK on Kidney Oxidative Stress
For the purpose of further revealing the effect of RK on oxidative stress in the kidneys of DN mice, we examined the rate of ROS production and the expression levels of SOD, GSH-Px, CAT, and MDA in the kidneys of DN mice. After treatment of DN mice with RK, the production rate of ROS and the content of MDA in the kidneys of DN mice were substantially decreased, and the expressions of SOD, GSH-Px, and CAT were markedly increased. In conclusion, it is proved that RK can improve the antioxidant capacity of the kidney and has an antioxidant effect (Figure 5A-E).
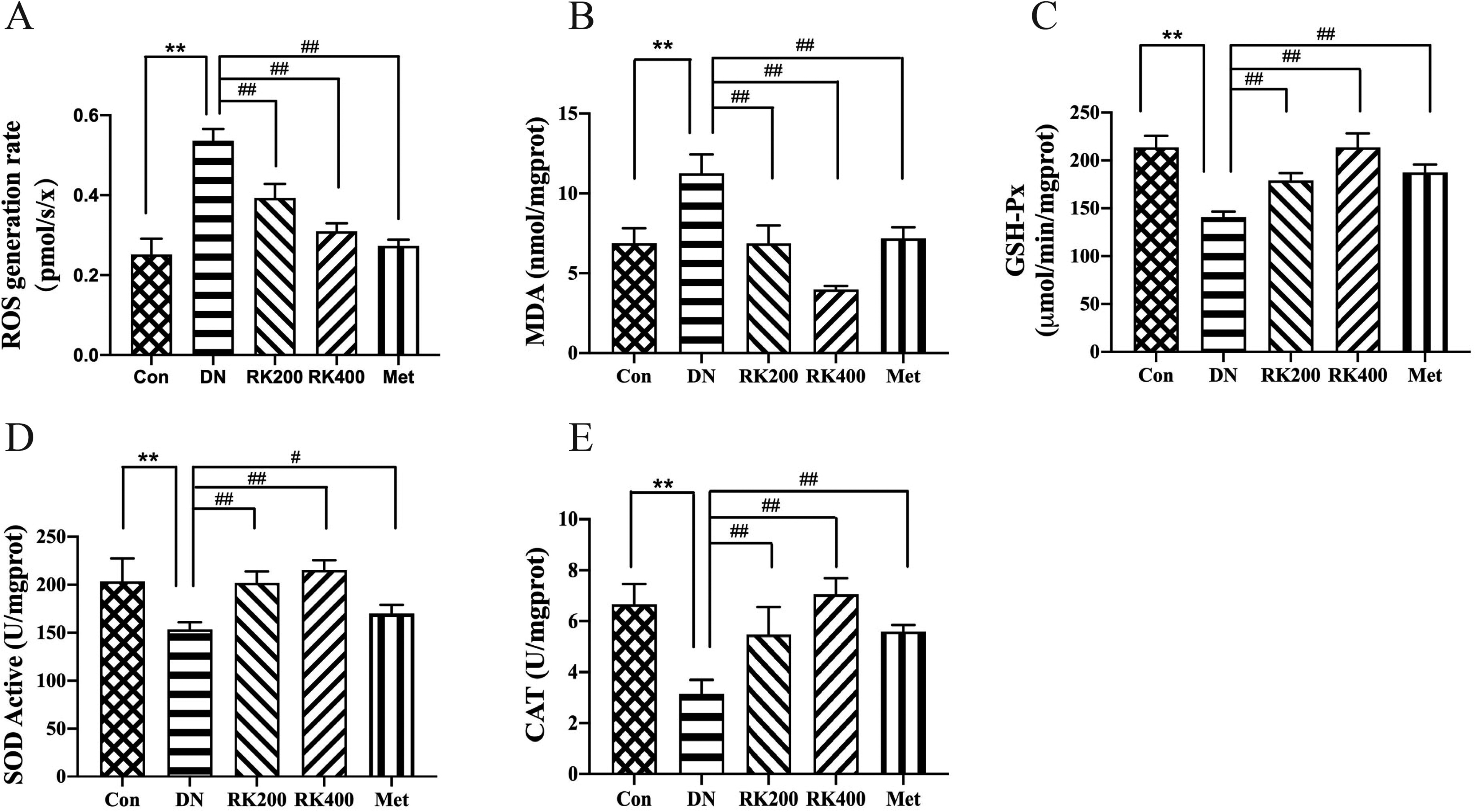
Effect of RK on oxidative stress indicators. (A) ROS generation rate, (B) MDA, (C) GSH-Px, (D) SOD, and (E) CAT.
Effect of RK on Kidney Pathological Changes
H&E staining and Masson staining were used to observe the pathological changes in the kidneys of DN mice. Comparing the kidneys of DN mice to those of Con mice, H&E staining revealed that the kidneys of DN mice had obvious epithelial cell vacuoles and nuclear degeneration of renal tubular epithelial cells, which were significantly ameliorated by RK treatment (Figure 6A-E). The results of Masson staining showed that compared with the Con mice, the glomerular basement membrane and glomerular capsule of DN mice were thickened and renal interstitial fibrosis was observed. After RK treatment, the renal interstitial fibrosis disappeared, and the glomeruli returned to normal (Figure 7A-F). The results indicated that RK can improve the kidney damage caused by DN.

H&E staining of the kidney of mice in each group (400x). (A) Con, (B) DN, (C) RK200, (D) RK 400, and (E) Met.

Masson staining of the kidney of mice in each group (400x). (A) Con, (B) DN, (C) RK200, (D) RK 400, (E) Met, and (F) quantitative analysis.
Discussion
DN is a severe public health problem, which is thought to be among the grievous life-threatening complications of diabetes mellitus. 20 Recently, several researchers were focused on the use of natural products and functional food as viable alternatives for the development of novel anti-diabetic and reno-protective agents.3,21–23 Numerous reports have emphasized the positive effect of food compounds from natural products, particularly medicinal plants, in mitigating the harmful effects and holding back the development/progression of chronic diseases such as DN.24–26 Most of these food compounds have antioxidant and anti-inflammatory effects and exert preventive abilities by inhibiting or ameliorating hyperglycemia-induced oxidative stress and inflammatory response.25,27–30 Consequently, our study explored the kidney protective effect of RK in HFD/STZ-induced diabetic mice (DM). We found typical clinical manifestations of DN in HFD/STZ-induced DM, including kidney hypertrophy, kidney damage, mitochondrial dysfunction, and oxidative damage, which are very similar to DN in humans. The results of this study showed that RK can reduce FBG, KW/BW, and kidney volume in DN mice, and improve the abnormal expression of mALB, Scr, and Ccr in DN mice.
Oxidative stress plays a major role in the occurrence and development of DN. 31 Mitochondrial dysfunction in the kidney causes the kidney to metabolize excess ROS and ultimately leads to oxidative stress. Studies have demonstrated that increasing the activity of antioxidant enzymes can reduce oxidative stress. In the STZ-induced DN mice, some marker antioxidant enzymes decreased, while lipid peroxidation products increased. 32 In DN mice, kidney tissue can generate superoxide, which will be neutralized by SOD to form H2O2 immediately, to reduce its destructive effect on renal tissue.33,34 Under the action of GSH-Px, H2O2 can be further converted into H2O and O2. 35 It has the same biological function as SOD. The study found that MDA with lipid peroxidation increased dramatically, while the biological activities of SOD, GSH-Px, and CAT indicated a downward trend in DN mice.21,22 Our experiment found that RK therapy reduced the rate of ROS generation and ADP-irrelevant oxygen consumption in the mitochondria of DN mice, and increased the maximum respiratory oxygen consumption and ADP-related oxygen consumption, indicating that RK therapy can improve kidney mitochondrial dysfunction in DN. This study also revealed that after treatment with RK, DM significantly increased the biological activity of internal SOD, GSH-PX, and CAT, while simultaneously causing lipid peroxidation to generate MDA progeny. It confirms that RK has beneficial antioxidant effects, which can reduce lipid peroxidation and decrease the production of oxidative stress.
Nrf2 plays an essential role in the anti-oxidative stress of DN. Through activating the Nrf2-mediated antioxidant pathway, the activity of antioxidant enzymes in DN mice is significantly increased. 36 RK can inhibit oxidative stress in ethanol-induced gastric ulcer mice by activating the Nrf2 pathway. 37 Based on the above results, it is hypothesized that RK may inhibit oxidative stress in DN mice by activating the Nrf2 antioxidant pathway. The antioxidative effect inhibited the mitosis of renal mitochondria in DN mice, maintained mitochondrial homeostasis, and effectively improved renal mitochondrial dysfunction in DN mice. Therefore, RK protects DN mice from kidney damage induced by mitochondrial dysfunction by inhibiting oxidative stress.
In our experimental animal model with DN, diabetes-associated kidney damage was further confirmed by the histological changes, and animal ultrasound analysis of DM indicated noteworthy that the renal lesion was serious in DN mice, including vacuoles in renal tubular epithelial cells, nuclear degeneration of renal tubular epithelium, thickening of glomerular basement membrane and glomerular balloon and renal interstitial fibrosis. Treatment of DM with RK remarkably ameliorated the kidney function markers, and kidney size and prevented hyperglycemia-induced structural alterations in the kidney.
Conclusion
In conclusion, our experimental findings indicate that RK may have a protective effect on the kidney by enhancing mitochondrial function and the biological activity of antioxidant enzymes. RK has numerous functions in the prevention and treatment of DN, which is consistent with the clinical treatment concept of DN and is an ideal DN therapeutic drug. Nonetheless, the target of RK in DN has not yet been discovered, and further research is required.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author’s work was supported by Project Agreement for Science & Technology Development, Jilin Province(grant numbers 20190304027YY and 20180201033YY).
Ethical Approval
The Administration Committee of Experimental Animals, Jilin Province, China, approved this study.
Statement of Human and Animal Rights
All experimental procedures involving the animals were conducted in accordance with the Committee of Jilin Medical College (Approval number: 2019-LW001), China, and approved by the Administration Committee of Experimental Animals, Jilin Province, China.
Data Availability
All data used to support the findings of this study are included in the article.
