Abstract
Keywords
Introduction
Obstructive sleep apnea-hypopnea syndrome (OSAHS) is characterized by recurrent partial collapse of the upper airway during sleep, resulting in decreased airflow or pauses in breathing lasting at least 10 s, accompanied by cortical arousal and a subsequent decrease in oxygen saturation. 1 According to a recent epidemiological survey, there are 425 million OSAHS patients worldwide, with China having the largest number of cases globally. The prevalence of OSAHS among Chinese people aged 30–69 years is 23.6%, and 8.8% of these cases are classified as moderate to severe. 2 Notably, the incidence of OSAHS continues to rise, likely due in part to lifestyle changes and the increasing prevalence of obesity. 3
Common risk factors for OSAHS include obesity, aging, male sex, anatomical abnormalities of the upper airway, family history, smoking and alcohol use.4–6 As the disease progresses, it can lead to various complications, such as hypertension, coronary artery disease, heart failure, stroke, and other cardiovascular diseases, metabolic syndrome, cognitive impairment, and damage to multiple organ systems, 7 all of which significantly impact health and quality of life. Currently, there are no specific pharmacological treatments for OSAHS. Although continuous positive airway pressure (CPAP) is usually the first-line treatment, some patients may be intolerant or have poor compliance due to patient-ventilator asynchrony. Other treatment options also have limitations. For example, prolonged use of oral appliances may cause temporomandibular joints pain, and some surgical treatments can result in complications, such as infections and may not be suitable for all patients. 8 Therefore, there is a pressing need for more practical treatment alternatives.
Traditional Chinese Medicine (TCM) has shown promising results in treating OSAHS using a holistic and evidence-based approach. Based on the clinical manifestations of OSAHS, it is believed that its onset is closely associated with the phlegm-dampness constitution, 9 which was also confirmed by a study on the TCM constitution in patients with OSAHS. 10 XHLQ, a formula developed by Professor Wang Qi, is specifically designed for treating patients with damp-phlegm OSAHS. It can not only remove dampness and dissolve phlegm to address the underlying phlegm-dampness constitution but also improve the pharynx and eliminate snoring, thus addressing both the symptoms and the underlying cause. Previous studies have demonstrated that XHLQ can reduce daytime sleepiness and decrease nocturnal apnea. 11 After XHLQ treatment based on basic interventions, the phlegm-dampness constitution score of patients decreased from 25.64 to 19.21, the TCM syndrome score dropped from 17.86 to 10.11, the Epworth Sleepiness Scale score was reduced from 11.18 to 9.21, the apnea-hypopnea index was decreased from 16.56 times/hour to 10.65 times/hour, and the lowest nocturnal pulse oxygen saturation was increased from 83% to 87.93%, all of which were statistically significant (P < 0.05). 12 Additionally, the serum levels of C-reactive protein, TNF-α, and IL-6 were lower in patients with OSAHS after treatment. 13 Thus, XHLQ is effective in alleviating the clinical symptoms of OSAHS, including reducing daytime sleepiness and nocturnal apnoea, and it decreases serum levels of inflammatory factors. However, its specific mechanism of action remains unclear.
This study aims to combine network pharmacology analysis and experimental validation using an intermittent hypoxia (IH)-induced rat model to investigate potential key targets and related signaling pathways for the treatment of OSAHS with XHLQ, which not only provides a solid experimental foundation for elucidating the pharmacological mechanism of XHLQ, but also opens new avenues for further exploring the potential intervention pathways of TCM in OSAHS.
Material and Methods
Drugs
The composition of XHLQ was derived from various herbal ingredients, specifically: bamboo shavings (Caulis Bambusae in Taenia, known as “Zhuru” in Chinese, ZR) 15 g, Ban Xia (Rhizoma Pinelliae, “Banxia” in Chinese, BX) 9 g, Tangerine Peel (Pericarpium Citri Reticulatae, “Chenpi” in Chinese, CP) 15 g, Cortex Magnolia Officinalis (“Houpo” in Chinese, HP) 10 g, Bulbus Fritillariae Thunbergii (“Zhebeimu” in Chinese, ZBM) 10 g, Bunge Pricklyash Seed (“Chuanjiaomu” in Chinese, CJM) 10 g, Chinese Clematis Root (Radix Clematidis, “Weilingxian” in Chinese, WLX) 10 g, Fructus Aurantii (“Zhi Qiao” in Chinese, ZQ) 10 g, Peach Kernel (Semen Persicae, “Taoren” in Chinese, TR) 10 g, Semen Raphani (“Laifuzi” in Chinese, LFZ) 10 g, Rhizoma Acori Graminei (“Shichangpu” in Chinese, SCP) 10 g, and Rhizoma Belamcandae (“Shegan” in Chinese, SG) 10 g. The daily dose of XHLQ Granules was formulated into 24 g of granules. Subsequently, these granules were dissolved in 400 ml of boiling water (100 °C), allowed to cool, and then stored at 4 °C.
Animals
Eighteen healthy male Sprague Dawley rats, aged 4 weeks and weighing 120 ± 3 g, were supplied by SPF Biotechnology (Beijing, China; lot number DM190623060102). They were accommodated in the animal facility of the Clinical Research Institute of China-Japan Friendship Hospital, maintained under standard environmental conditions with a temperature of 22 ± 2°C, relative humidity of 55 ± 5%, and a 12-h light/dark cycle. The animals had unrestricted access to food and water, and the experiment commenced one week following an adaptation period. The animal experiment comply with the National Research Council's Guide for the Care and Use of Laboratory Animals and the ethical approval for the experiment was granted by the Animal Ethical and Welfare Committee of the Clinical Research Institute of China-Japan Friendship Hospital (No. 190107).
Experimental Materials, Reagents and Instruments
The experimental materials and reagents are listed in Table S1 in the Supplemental Materials. Furthermore, the experiment primarily required modeling equipment and laboratory instruments. Specifically, the modeling equipment encompassed a self-made IH animal chamber, a programmable control system, and a gas supply system. Meanwhile, the laboratory instruments were mainly used for specimen preparation and experimental detection, as detailed in Table S2 in the Supplemental Materials.
Network Pharmacology
Active Ingredients Search and Target Prediction for XHLQ
The Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP) was used to identify the active ingredients present in the XHLQ formula. The screening process involved evaluating the oral bioavailability (OB) and drug-likeness index (DL) of these ingredients, applying threshold of OB ≥ 30% and DL ≥ 0.18. Following this, we queried the relevant targets for these active ingredients and standardized the target names using the UniProt database. To gather information on active ingredients not listed in the TCMSP database, we supplemented our data with details from the Traditional Chinese Medicine Integrated Database (TCMID).
The active ingredients were imported into the PubChem database to obtain the corresponding chemical structures and information, which were then saved in SDF format. The active ingredients were screened by Swiss ADME with screening criteria set to “High” gastrointestinal (GI) absorption and at least three “Yes” to the DL rule of five (Lipinski, Ghose, Veber, Egan, Muegge). Finally, the ingredients meeting the criteria were uploaded into Swiss Target Prediction in SDF file format for target prediction. Relevant parameters were set to include the organism “Homo sapiens” and to ensure a correlation possibility between ingredients and targets greater than 0. The targets identified for these ingredients were pooled and deduplicated to obtain the drug targets of XHLQ.
OSAHS Target Prediction
The targets for OSAHS were identified through a search of bioinformatics and chemometrics databases, specifically GeneCards, DisGeNET, DrugBank, and OMIM, using the search term “obstructive sleep apnea-hypopnea syndrome”. The resulting data were then aggregated and deduplicated.
Construction of the Protein-Protein Interaction (PPI) Network for the Common Targets of XHLQ and OSAHS and Screening of Core Targets and Ingredients
The Venny2.1 website was used to analyze the common targets of XHLQ and OSAHS to create a database and Venn diagram. Then the common targets were imported into the STRING database, with the highest confidence level set to ≥0.9 and the species specified as “Homo sapiens”. From this, the PPI network of common targets was constructed. The active ingredients in XHLQ for OSAHS were obtained by removing the ingredients that did not intersect with the disease target. The PPI results were downloaded in TSV file format and imported into Cytoscape 3.7.2. Core targets were identified through analysis and calculation of the median values utilizing CytoNCA plugins, including betweenness centrality (BC), closeness centrality (CC), degree centrality (DC), eigenvector centrality (EC), the local average connectivity-based method (LAC), and network centrality (NC). These core targets were visualized using Cytoscape, with larger and darker nodes indicating higher tightness. Ingredients lacking intersecting targets were further eliminated. The remaining active ingredients and common targets were then imported into Cytoscape 3.7.2 to construct an ingredient-target interaction network. A topology operation was performed to select the top 5 core ingredients based on DC value, where a higher DC value signifies a greater number of intersections with other nodes and the closer connection.
Gene Ontology (GO) Enrichment Analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Enrichment Analysis
GO enrichment analysis was performed using R software (Version 4.1.1) and data packages including “DOSE”, “cluster Profiler”, and “enrich plot”. Analysis included biological process (BP), cellular component (CC), and molecular function (MF) analyses, and the top 10 significant items in each category were visualized as bar charts and bubble charts. In the bubble chart, the horizontal coordinate represents the gene enrichment ratio, while the vertical coordinate corresponds to the GO terms. The size of the bubble is proportional to the number of genes, and the color indicates the significance of enrichment, with redder shades denoting higher significance. In the bar chart, the length of the bars is proportional to the number of enriched genes, while color again indicates the significance of enrichment. Additionally, KEGG enrichment analysis was conducted utilizing the same software and data packages. The top 20 significantly enriched pathways were subsequently visualized through bar charts and bubble charts.
Animal Experiment
Preparation of IH Animal Chambers
The IH animal chamber consisted of three main components: the chamber itself, the control system, and the gas delivery channel. The chamber featured a gas inlet on one side and an outlet on the other. The outlet served to balance the air pressure within the chamber and was connected to an oxygen meter. The inlet was linked to two gas flowmeters, which allowed precise control over the nitrogen and compressed air flow into the chamber. This arrangement ensured that the gas flow could be adjusted based on the oxygen meter readings, thereby regulating the oxygen concentration within the chamber. Carbon dioxide and water vapor produced inside the chamber were absorbed by sodium lime and anhydrous calcium chloride placed at the chamber's base. The control system's primary function was to regulate the flow of nitrogen and compressed air through the gas transporting channels, simulating the cyclical conditions of IH and reoxygenation. Using the gas transporting channel, nitrogen (99.99% pure, contained in medical cylinders) and compressed air (supplied by an air compressor, entering the gas transport channel after decompression by a valve) were delivered into the chamber, with their flow rates adjusted by the flowmeters.
Animal Grouping and Modeling and Pharmacological Interventions
In this study, an OSAHS animal model was established using the IH method. In OSAHS research, several approaches for constructing OSAHS models have been proposed, such as upper airway obstruction, fragmented sleep, and IH. The IH method is commonly preferred in experimental studies because it is non-invasive and can accurately replicate the IH and reoxygenation cycles, which are key pathophysiological mechanisms of OSAHS.14–16 Our study followed this widely accepted approach.
The rats were randomly assigned into the normoxia group (N), moderate IH group (IH-1), mild IH group (IH-2), XHLQ-1group and XHLQ-2 group, with six rats in each group. The IH-induced rat model of OSAHS was established based on previous studies.16,17 Briefly, the IH animal chambers were used to expose rats in the IH and XHLQ groups to intermittent hypoxia conditions. The oxygen concentration within the chamber was continuously monitored using an oxygen meter. Ventilation cycles were set to 120 s, with each cycle comprising two distinct phases: hypoxia and normoxia. During the hypoxia phase, 99.99% pure nitrogen gas was infused into the the IH-1 and XHLQ-1 chambers at a flow rate of 5 L/min, while 99.99% pure nitrogen gas was infused into the chambers of the IH-2 and XHLQ-2 groups at a flow rate of 3 L/min. As a result, the oxygen concentration in chambers of the IH-1 and XHLQ-1 groups gradually decreased to a minimum of 10% over a 30-s period, while the oxygen concentration in chambers of the IH-2 and XHLQ-2 gradually decreased to a minimum of 12.5% over a 30-s period. During the reoxygenation phase, which followed the hypoxia phase, compressed air was introduced into all the animal chambers and the air flow rate was 5 L/min, leading to a gradual increase in oxygen concentration to a maximum of 21% for 90 s. The animals were alternately exposed to hypoxic and normoxic gases to induce periodic reductions and recoveries in blood oxygen saturation (SpO2), simulating the process of repeated hypoxia and reoxygenation experienced by OSAHS patients during nighttime. At the same time each day, the rats in the N group were placed in identically sized animal chambers connected to an air compressor. Detailed information regarding this procedure is provided in Table 1, while the IH chamber schematic diagram and the oxygen concentration curves within the IH chambers are depicted in Figure 1. The rats were exposed to IH in the animal chambers for 8 h daily. Beginning on the 36th day, the XHLQ groups received XHLQ granules at a dose of 3.78 g/kg via gastric administration daily, prior to the IH experiment procedures. Meanwhile, the N and IH groups received an equal volume of saline. On day 51, rats were anesthetized with an intraperitoneal injection of 2% pentobarbital sodium (50 mg/kg). Immediately thereafter, samples were collected, followed by euthanized of the rats.

A: IH chamber schematic diagram. B: Oxygen concentration curves within IH animal chambers.
Gas Release Settings and Oxygen Concentration in Animal Chambers (n = 6).

Recording General Characteristics of Rats
This study aims to mimic the intermittent hypoxic state of OSAHS and evaluate the therapeutic effectiveness of XHLQ on OSAHS by observing the general characteristics such as mental state, fur luster, activity level, and respiratory movement, along with the pathological alterations in rats.
The general characteristics of the rats, including mental state, fur luster, activity level, and respiratory movement, were recorded regularly at the same time every day.
Performing Hematoxylin and Eosin (H&E) Staining on Lung Tissue
Lung tissues were fixed overnight in a 4% paraformaldehyde solution, dehydrated, and embedded in paraffin. Tissue sections were cut to a thickness of 4 μm and subsequently stained with H&E. The tissue sections were examined under a light microscope, and images were captured at a consistent bronchial level to compare the pathological changes of lung tissue across groups.
Enzyme-Linked ImmunoSorbent Assay (ELISA) Detection of Serum IL-6 and TNF-α
The Rat Interleukin-6 ELISA Test Kit and Rat Tumor Necrosis Factor-α ELISA Test Kit were used to conduct the ELISA detection of IL-6 and TNF-α in the serum of each group of experimental rats following the instructions.
Performing Immunohistochemistry (IHC) on Lung Tissue
IHC was conducted to identify the positive expression of PI3K and Akt in the lung tissue. Detailed steps were as follows: Tissue sections were baked in a 60°C oven for 4 h, then they were washed with phosphate-buffered saline (PBS) solution before being boiled in a bath of sodium citrate antigen repair solution for 10 min. After the sections had cooled down, they were washed again with PBS solution and then blocked endogenous peroxidase with 3% hydrogen peroxide for 10 min. A 5% bovine serum blocking solution was used to block non-specific binding of the antibodies for 1 h, after which sections were incubated overnight at 4°C with PI3K primary antibodies (1:75) or Akt primary antibodies (1:150). The next day, the sections were incubated with secondary antibodies for 1 h after being washed with PBS solution. The sections were stained and developed with DAB after being incubated with streptavidin peroxidase for 30 min. Subsequently, after nuclear staining with hematoxylin, the sections were observed under the microscope at 200× and 400× magnification. Finally, images were collected, and the average OD was determined using ImageJ (version 1.53).
Quantitative Real-Time PCR (RT-qPCR) to Determine the Relative Expression Level of PI3K and Akt mRNA
In total, 100 mg of lung tissue was taken. Total RNA was extracted using Trizol reagent, and cDNA was obtained by reverse transcription. Real-time PCR was conducted using a fluorescence quantitative PCR instrument (MA-6000, Suzhou Molarray Co. Ltd) as follows: the reaction mixture consisted of 7 µl dd H2O, 2 µl cDNA, 0.5 µl of forward primer (10 pmol/µl) and 0.5 µl of reverse primer (10 pmol/µl), and 10 µl SYBR mix. Then, RT-qPCR was performed after centrifugation. The reaction conditions were set as follows: 1 cycle of pre-denaturation at 95 °C for 2 min, followed by 40 PCR cycles at 95 °C for 15 s and 60 °C for 30 s. The specific primer sequences are presented in Table S3 in the Supplemental Materials.
Statistical Analysis
SPSS 26.0 statistical software was used for statistical analysis. Data are expressed as the mean ± standard deviation (x̄ ± s). One-way analysis of variance (ANOVA) was used to analyze multiple sets of measurement data. For pairwise comparisons, post-hoc tests such as Tukey HSD or Bonferroni were applied. The Kruskal-Wallis test, a nonparametric test, was used for data that did not follow a normal distribution. A P-value < 0.05 was considered statistically significant.
Results
Network Pharmacology
Active Ingredients of XHLQ, XHLQ-OSAHS Common Targets and Venn Diagram
After the removal of duplicates and elimination of non-targeted ingredients, 96 active ingredients of XHLQ with potential therapeutic effects on OSAHS were identified (Table S4 in the Supplemental Materials). In total, there were 671 targets associated with the active ingredients of the drug and 847 targets related to OSAHS. Sixty-three common targets were obtained, including ACE, ALDH5A1, BCL2, CLK1, CXCL8, CYP1A2, CYP2C19, ESR1, FDFT1, FGFR1, GABBR1, HIF1A, MIF, MMP9, NFE2L2, NR1H2, NR1H3, PGR, PPARG, PTGS2, RELA, SLC6A2, SLC6A4, SLC9A1, TLR4, VDR, ADRA2A, CYP2C9, EDNRA, FABP4, FFAR4, ICAM1, IL6, LTB4R, NR3C1, NR3C2, PDE4D, TGFBR1, SLC6A3, SCN5A, TNNT2, MAPT, REN, HSPA1A, PIK3CD, DNMT3A, MTOR, ALB, GC, APOB, CYP3A4, PON1, SELE, VCAM1, FECH, FGFR2, IGF1, PGF, TGFB2, CDC42, CHIT1, NOS3, and SELP. The Venn diagram is presented in Figure 2A.

A: Venn diagram of common targets. B: PPI map of XHLQ-OSAHS common targets. C: Core target network diagram and screening process. The final core targets were obtained after two rounds of screening utilizing Cytoscape topology analysis, where the larger node size and redder color indicate a closer connection to other nodes. D: Ingredient-target network map. The green nodes located in the center represent the common targets of XHLQ and OSAHS. The outer circles depict the 67 active ingredients, selected after excluding those without overlapping targets. Nodes with the same color pertain to the same drug, and nodes marked with red inverted triangles are common to multiple drugs.
Construction of XHLQ-OSAHS Common Targets Network and the Screening of Core Targets and Ingredients
A PPI network, comprising 58 nodes and 395 edges, was constructed (Figure 2B). Nine core targets were identified, namely ALB, IL6, CXCL8, HIF1A, MMP9, IGF1, PPARG, NOS3, and TLR4 (Figure 2C). Sixty-seven active ingredients of XHLQ relevant to OSAHS were identified (Figure 2D), with the top five core ingredients being beta-sitosterol, palmitoleic acid, α-linolenic acid (ALA), kaempferol, and linoleic acid (Table S5 in the Supplemental Materials).
GO Enrichment Analysis
A total of 1789 GO functional analysis results were obtained, including 1600 BPs, such as regulation of epithelial cell proliferation, response to lipopolysaccharide, response to oxygen levels, and response to hypoxia. Additionally, 31 CCs were identified, including membrane raft, membrane microdomain, and caveola. Furthermore, 98 MFs were characterized, including nuclear receptor activity, ligand-activated transcription factor activity, and steroid binding. The top ten significant terms from each category were selected and are presented in Table S6 in the Supplemental Materials. These data were visualized as bubble and bar charts by the R software, as depicted in Figures 3.

Go enrichment analysis of common targets (top 10).
KEGG Pathway Enrichment Analysis
A total of 111 pathways were identified, and the top 20 most significant pathways encompassed HIF-1, TNF-α, NF-κB, PI3K/Akt, and MAPK signaling pathways (Table S7 in the Supplemental Materials). As illustrated in Figures 3 and 4, these were visualized as bubble and bar charts using the R software.

KEGG signaling pathway enrichment analysis of common targets (top 20).
Animal Experiments
General Characteristics of Rats
As the exposure to IH prolongs, in comparison with the N group, the rats in the IH groups and the XHLQ groups exhibit deteriorated mental states, decreased fur luster, and gradually become lethargic and less active. Additionally, their respiratory movements are weakened, and some even present paradoxical respiration of the chest and abdomen. After intervention with XHLQ granules through gavage, the rats in the XHLQ groups exhibited an improved mental state and enhanced fur luster, along with increased activity and strengthened respiratory movement.
Pathological Changes of Lung Tissue in Rats
The results of the H&E staining revealed significant inflammatory cell infiltration and pulmonary interstitial thickening in the IH groups of rats, whereas these findings were absent in the N group. In comparison to the IH groups, the XHLQ groups exhibited a significant reduction in inflammatory cell infiltration and pulmonary interstitial thickening (Figure 5).

H&E-stained sections of lung tissue from rats in each group (200× magnification). A, B, C, D and E represent the N group, IH-1 group, IH-2 group, XHLQ-1 group and XHLQ-2 group, respectively.
XHLQ Reduced the Expression Levels of IL-6 and TNF-α in rat serum
Compared with the N group, the IL-6 and TNF-α levels in the IH groups increased (P < 0.05). Comparisons between the IH-1 and IH-2 groups showed that serum IL-6 and TNF-α levels increased with decreasing oxygen concentration (P < 0.05). In the pairwise comparisons between the IH-1 and XHLQ-1 groups as well as between the IH-2 and XHLQ-2 groups, the levels of serum IL-6 and TNF-α were diminished in the lung tissues of rats in the XHLQ groups under equivalent oxygen concentrations (P < 0.05) (Supplemental Material Table S8 and Figure 6).

Serum levels of IL-6 and TNF-α. * Indicates a significant difference compared to the N group (P < 0.05). # Indicates a significant difference compared to the IH-1 group (P < 0.05). Δ Indicates a significant difference compared to the IH-2 group (P < 0.05).
XHLQ Inhibited the Expression Levels of PI3K and Akt in the Lung of IH-Induced Model
We performed IHC staining and RT-qPCR of PI3K and Akt in the PI3K/Akt signaling pathway. The corresponding AOD values in each IH group were significantly higher than those in the N group (P < 0.05). When comparing IH-1 group with IH-2 group, it was observed that the AOD in IH-1 group increased significantly (P < 0.05); in other words, as the oxygen concentration decreased, the AOD value increased further. Following treatment with traditional Chinese medicine, the AOD values in XHLQ-1 were significantly lower compared to those in IH-1 group, and those in XHLQ-2 group were significantly lower than those in IH-2 group (P < 0.05) (Table S9 in the Supplemental Materials and Figure 7). When compared with the N group, the relative expression levels of Akt and PI3K mRNA were significantly upregulated in the IH groups (P < 0.05). A comparison between the IH-1 and IH-2 groups revealed that the relative expression levels of PI3K and Akt mRNA increased as the oxygen concentration decreased (P < 0.05). Pairwise comparisons conducted between the IH-1 and XHLQ-1 groups, as well as between the IH-2 and XHLQ-2 groups, demonstrated that the XHLQ groups exhibited a significant downregulation of the relative expression levels of PI3K and Akt mRNA in the lung tissues of rats under equivalent oxygen concentrations (P < 0.05) (Table S10 in the Supplemental Materials and Figure 8).
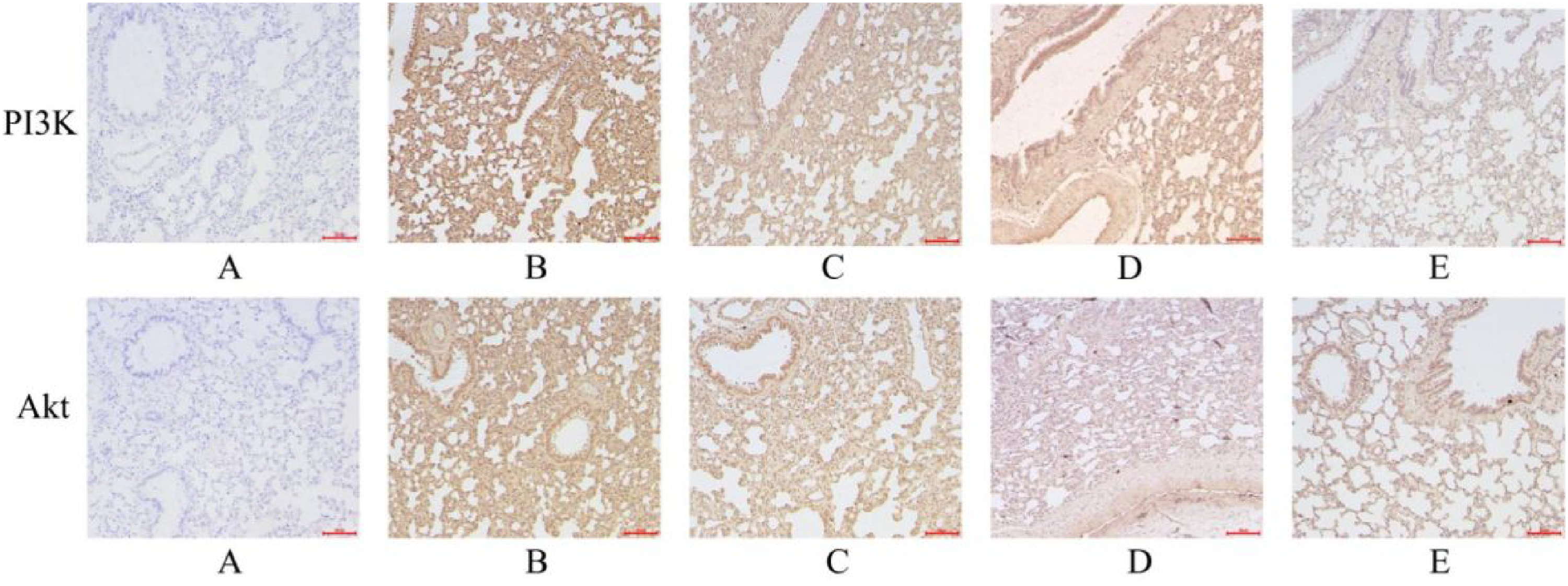
IHC staining results of PI3K and Akt (200× magnification) A, B, C, D and E represent the N group, IH-1 group, IH-2 group, XHLQ-1 group and XHLQ-2 group, respectively.

The relative mRNA expression levels of PI3K and Akt. * Indicates a significant difference compared to the N group (P < 0.05). # Indicates a significant difference compared to the IH-1 group (P < 0.05). Δ Indicates a significant difference compared to the IH-2 group (P < 0.05).
Discussion
Prediction of the Mechanism of XHLQ for the Treatment of OSAHS
The results of network pharmacology showed that XHLQ contains 67 active ingredients, including β-sitosterol, palmitoleic acid, ALA and kaempferol. β-sitosterol has anti-inflammatory and antioxidant effects and can reduce the expression levels of IL-6, TNF-α and other inflammatory factors, and increased intake of β-sitosterol may also prevent obesity-related chronic inflammatory diseases.18,19 Palmitoleic acid is a functional fatty acid that exerts a significant influence on inflammation, immune response and metabolism-related diseases. 20 Additionally, it downregulates TNF-α-stimulated inflammatory factors, including NF-κB, cyclooxygenase-2, MCP-1, and IL-6. 21 Clinical studies have shown that serum levels of C-reactive protein in patients decreased after taking 6 g of ALA orally per day for 2 months. 22 Moreover, prior research has demonstrated that dietary intake rich in ALA can reduce the expression of IL-1β, IL-6, and TNF-α, resulting in anti-inflammatory effects. 23 Kaempferol, a bioactive compound renowned for its anti-inflammatory, antioxidant and antitumor properties, has exhibited remarkable efficacy in the management of diverse acute and chronic inflammatory diseases. 24 Pretreatment with kaempferol has been shown to effectively attenuate oxidative stress in vivo, while concurrently inhibiting NF-κB/p65 activation and reducing the release of pro-inflammatory cytokines, such as TNF-α and IL-6. 25 OSAHS is characterized by oxidative stress and inflammatory responses.26–28 In light of the anti-inflammatory, antioxidant and metabolic regulatory effects of the active ingredients in XHLQ, we rationally postulate that XHLQ holds the potential to ameliorate OSAHS by mediating or regulating the aforementioned biological processes.
The analysis of GO and KEGG indicated that the significant pathways of XHLQ for OSAHS include HIF-1, TNF-α, NF-κB, PI3K/Akt, MAPK signaling pathways, which are intricately associated with biological processes such as immune inflammation, hypoxic stress, and epithelial cell proliferation. HIF-1α is an important hypoxia-regulating protein. 29 The HIF-1 signaling pathway has been observed to be activated in the pathogenesis of OSAHS, and studies have shown that HIF-1α was increased in patients with OSAHS compared to healthy subjects. 30 Additionally, IH plays an important role in OSAHS-related vascular inflammation and angiogenesis induced by IH through the upregulation of NF-κB-dependent HIF-1 production. 31 The TNF signaling pathway activates downstream pathways such as NF-κB, thereby participating in the inflammatory response and apoptosis. 32 The expression level of serum TNF-α was significantly increased in patients with OSAHS compared to healthy subjects and positively correlated with the severity of OSAHS. 33 NF-κB regulates the transcription and activation of cytokines such as TNF-α and IL-6, playing a critical role in inflammation. 34 Studies have shown that the expression levels of NF-κB, TNF-α and IL-6 were significantly higher in OSAHS patients compared to healthy individuals, and a similar conclusion has been reached in animal IH models and in vitro experiments.35,36 The MAPK pathway is an important link in cell growth, differentiation, inflammation, and stress. 37 Animal experiments have shown that the MAPK signaling pathway is activated in IH-induced obese mice. 38
Previous studies reported that the PI3K/Akt signaling pathway is activated in OSAHS combined with hypertension, which can be effectively alleviated by inhibiting the PI3K/Akt pathway. 39 In addition, activation of the PI3K-AKT pathway was also found to play a role in suppressing OSAHS-associated insulin resistance, suggesting that the PI3K pathway may play an important role in the multiple metabolic complications associated with OSAHS. 40 Moreover, in the context of OSAHS pathophysiology, IH-induced changes are closely related to the PI3K/AKT pathway. Chronic intermittent hypoxia is a crucial pathophysiological characteristic of OSAHS. Studies have shown that exposure to IH leads to ROS production, and that ROS induces HIF-1α expression through the PI3K/AKT-related pathways, which further regulates the expression of downstream proteins involved in glucose metabolism and angiogenesis. 41 In a mouse model of IH, the PI3K/AKT pathway exhibited activation, as evidenced by a significant increase in PI3K mRNA expression and concurrent overactivation of p-PI3K and p-AKT. 42 Complementarily, previous studies demonstrated that the expression and activity of PI3K pathway-related molecules were altered in genioglossus muscle myofibroblasts under hypoxia, which further confirmed the involvement of PI3K pathway in the cellular stress response to hypoxia. 43 Furthermore, additional studies revealed that the PI3K pathway was also activated in an IH cell model and was shown to regulate the expression of inflammation-related genes, especially interleukin-8 (IL-8). Considering the importance of inflammation in the development of OSAHS, this implies that the PI3K pathway participates in the regulation of inflammation associated with OSAHS at the cellular level and may be a key factor in the pathophysiological process of OSAHS. 44
Based on the aforementioned findings, we suggest that XHLQ may exert antioxidative activity and suppress inflammatory responses by modulating diverse signaling pathways, including HIF-1, TNF, NF-κB, PI3K-Akt and MAPK. Consequently, it may play a significant role in the treatment of OSAHS.
Validation of the Mechanism of XHLQ for the Treatment of OSAHS
Results of Network pharmacology and previous studies have shown that the PI3K/Akt signaling pathway plays an important role in OSAHS. It is involved in the regulation of inflammation, oxidative stress, cell survival, apoptosis, as well as vascular endothelial function.45,46 All these biological processes have a vital influence on the development and progression of OSAHS and its associated complications. Hence, in this study, experimental approaches were employed to further explore and confirm the role of the PI3K/Akt signaling pathway in the modulation within the context of OSAHS.
An artificial, non-invasive simulated IH environment was used to induce OSAHS in rats in this experimental study. The lung tissues of rats in the IH group showed massive inflammatory cell infiltration, and compared with those in the N group, the expression of serum IL-6 and TNF-α in the IH group was elevated. These results suggest that the IH condition induced an inflammatory response, which is consistent with previous animal experiments and pathological changes in patients with OSAHS in the presence of chronic inflammation.27,45 Furthermore, immunohistochemistry and PCR experiments revealed upregulated expression of PI3K and Akt, as well as their corresponding mRNA expression, in the IH-induced rat model. This upregulation became more pronounced when decreasing oxygen concentration during the hypoxic period. An integrated in vivo and in vitro study demonstrated that the PI3K/Akt pathway was activated in both IH-induced animal and cell models, aligning with the findings of this study. 46 Notably, in this study, the XHLQ groups showed a significant downregulation of PI3K and Akt expression, coupled with a reduction in relative mRNA expression levels, in the lung tissue of rats maintained at the same oxygen concentration as the IH groups. Based on these findings, it is suggested that XHLQ may exert a potential therapeutic effect on OSAHS by inhibiting the PI3K/Akt signaling pathway.
However, it should be noted that certain limitations exist in the experimental conditions. Specifically, the sleep of experimental rats was not monitored in the present study, and the animal experiments may not comprehensively replicate the pathophysiological process of OSAHS. Hence, it is essential to further optimize the modelling methods and assessment tools to obtain more precise scientific conclusions. Moreover, this study merely provided a preliminary exploration of the PI3K/Akt pathway. The detailed upstream and downstream regulatory mechanisms and interactions with other pathways require further investigation. In subsequent studies, further monitoring of key molecules in the PI3K pathway is needed to fully elucidate its complex role in OSAHS. Meanwhile, the scope of research should be broadened to include other related pathways and molecular targets, and multi-level data should be integrated to provide a theoretical basis for formulating effective treatment strategies.
Conclusion
In conclusion, the results of network pharmacology suggest that XHLQ may intervene in pathological processes such as inflammation and oxidative stress through multiple targets and multiple pathways to achieve therapeutic effects in OSAHS. Moreover, by establishing a non-invasive IH-induced rat model of OSAHS, it was demonstrated that XHLQ could attenuate airway inflammation and downregulate the expression of PI3K and Akt in the IH-induced rat model. Based on these findings, it can be inferred that XHLQ holds potential as an adjunctive therapy for OSAHS.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X251317618 - Supplemental material for Xiaohan Liqi Granules Alleviate Inflammation and Modulate PI3K/Akt Pathway in an Intermittent Hypoxia-Induced Rat Model of OSAHS: A Network Pharmacology and Experimental Study
Supplemental material, sj-doc-1-npx-10.1177_1934578X251317618 for Xiaohan Liqi Granules Alleviate Inflammation and Modulate PI3K/Akt Pathway in an Intermittent Hypoxia-Induced Rat Model of OSAHS: A Network Pharmacology and Experimental Study by Shiyu Zhang, Qi Guo, Ruifeng Jin, Hongsheng Cui, Qiuyi Chen, Guirui Huang, Mingsheng Lv, Shuaiyang Huang, Xuefeng Gong, Zhijie Zhang and Shuan Liu in Natural Product Communications
Footnotes
Acknowledgments
Not applicable.
Authors’ Contributions
Shiyu Zhang and Qi Guo: Conceive ideas, complete experiments, and animal experiment implementation. Xuefeng Gong, Zhijie Zhang and Shuan Liu: Data collection and arrangement. Qiuyi Chen, Guirui Huang, Mingsheng Lv and Shuaiyang Huang: Article revision and supportive work. Ruifeng Jin and Hongsheng Cui: Design and fund experiments.
Availability of Data and Material Statement
All relevant data are contained within the article: The original contributions presented in the study are included in the article/Supplementalal material, further inquiries can be directed to the corresponding author.
Ethics Approval
This study was reviewed by the Ethics Committee of the China-Japan Friendship Hospital (No. 190107), Beijing China. The experiments were conducted by the Chinese Government's “Guidelines on the Care and Use of Laboratory Animals” to minimize their suffering.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article. This work was supported by the National Natural Science Foundation of China (No. 82074390), the National Natural Science Foundation of China (No. 82274497), and the Young Teachers Program of Beijing University of Chinese Medicine (BUCM) (No. 2019-JYB-JS-170).
ORCID iDs
Statement of Human and Animal Rights
All the experimental procedures involving animals were conducted in accordance with the Chinese Government's “Guidelines on the Care and Use of Laboratory Animals”, and approved by the Ethics Committee of the China-Japan Friendship Hospital, Beijing China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
