Abstract
Yang He Decoction (YHD), a classical Chinese medicine prescription, is used to treat bone and joint diseases. However, there are few mechanism studies for YHD on the use of YHD to treat bone tuberculosis (BT) and the corresponding mechanism of action of YHD. In the present study, the chemical ingredients of YHD and targets of the ingredients were revealed by a network pharmacology method, and an ingredient–target–disease network was visualized and analyzed. Then, gene ontology enrichment analysis and Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis were carried out. In addition, molecular docking was performed to clarify the binding of the key active ingredients of YHD to the key targets. Further, an in vitro model of Mycobacterium tuberculosis-induced BT was established, and a mechanism for the inhibitory effect of YHD on the differentiation of RAW 264.7 cells into osteoclasts was investigated. A total of 138 active ingredients in YHD and 50 targets between YHD ingredients and BT were identified. The phosphatidylinositol 3-kinase–protein kinase B (PI3K–Akt) and mitogen-activated protein kinase (MAPK) signaling pathways were the key pathways involved in the anti-BT effect of YHD. Moreover, the in vitro results showed that YHD inhibited the differentiation of RAW 264.7 cells into osteoclasts. YHD decreased the levels of tumor necrosis factor-α and interleukin-1β, increased the levels of superoxide dismutase and glutathione peroxide, and decreased the level of malondialdehyde. Further, YHD inhibited the protein and messenger RNA expression of PI3K/Akt, p38 MAPK, and c-Jun N-terminal kinase. These findings show that YHD is a promising anti-BT agent that suppresses the PI3K/Akt and MAPK signaling pathways to inhibit the differentiation of RAW 264.7 cells into osteoclasts and ameliorate inflammation and oxidative stress.
Introduction
Mycobacterium tuberculosis (MTB), one of the greatest threats to human health, can spread from the blood circulatory system to the skeletal system, which causes bone or joint lesions, and eventually bone tuberculosis (BT). 1 BT, also known as osteotuberculosis or osteoarticular tuberculosis, is disseminated by the MTB that infects the bones and joints, which causes the necrosis of the joints with the rich blood supply in negative or active joints. The incidence of BT accounts for the largest proportion (35%) of the total incidence of extrapulmonary tuberculosis.2,3 Also, BT is a disease that may occur in people of any age. In developed countries, BT occurs more commonly in the elderly. On the other hand, in developing countries, where epidemics are not well controlled, BT occurs in teenagers, young children, and infants. It is difficult to differentiate and diagnose. Thus, it is often misdiagnosed. And the long-term use of common anti-tuberculosis drugs leads to damage to the liver, kidneys, gastrointestinal tract and has other toxic side effects. 4
Traditional Chinese medicine (TCM) categorizes BT into the “multiple abscesses with damp phlegm” category. The use of TCM to treat BT can not only avoid damage to the liver, but also enhance human immunity. 5 Yang He Decoction (YHD), a classical TCM prescription, is recorded in the Life-saving Manual of Diagnosis and Treatment of External Diseases written during the period of the Qing dynasty of China. 6 YHD contains 7 herbs 6 : Shudihuang (SDH, Scrophulariaceae, Rehmanniae Radix Praeparata); Rougui (RG, Lauraceae, Cinnamomi Cortex); Mahuang (MH, Ephedreae, Ephedrae Herba); Baijiezi (BJZ, Cruciferae, Sinapis Semen); Ganjiang (GJ, Zingiberaceae, Zingiberis Rhizome); Gancao (GC, Leguminosae, Glycyrrhizae Radix et Rhizoma); Lujiaojiao (LJJ, Cervidae, Cervi Cornus Colla). It has been reported that modified YHD can be used to treat ankylosing spondylitis treatment, 7 and YHD is regarded as a safe and effective alternative therapy for preventing chondrocyte apoptosis, and as an antiinflammatory in treating knee osteoarthritis. 8 TCM considers BT an autoimmune disease.7,8 YHD may regulate the immune system to treat BT. However, the therapeutic effect of YHD on BT and the corresponding mechanism of action of YHD need to be investigated.
The network pharmacology method is increasingly being applied to study the interaction between a multicomponent drug and its targets.9,10 Due to the development of bioinformatics, the ingredient–target interaction can be studied from the molecular to organismic level. Furthermore, the network pharmacology method can be used to understand the mechanisms of action of a multicomponent TCM prescription. 11 In addition, the effectiveness of TCM prescriptions can be increased by using databases for targeted diseases to elucidate the intervention by the prescriptions in the key targets that promote the occurrence and development of the diseases.
In the present study, we investigated the main active ingredients of YHD and used the network pharmacology method to investigate the targets of YHD for the treatment of BT and the mechanism of action of YHD. Then, a cell model of MTB-induced BT was established to verify the mechanism of action of YHD in vitro.
Materials and Methods
Establishment of the Herb–Ingredient–Target Interaction
The chemical ingredients of YHD were extracted from the TCM databases, including the Traditional Chinese Medicine Systems Pharmacology Database (https://tcmsp-e.com/) and Bioinformatics Analysis Tool for Molecular Mechanism of Traditional Chinese Medicine (http://bionet.ncpsb.org/batman-tcm/). On the basis of the absorption, distribution, metabolism, and excretion (ADME), oral bioavailability (OB), drug-likeness (DL), and Caco-2 permeability values of the chemical ingredients of YHD, the ingredients with OB ≥30%, and DL ≥0.18 were screened out. The UniProt database (https://www.uniprot.org/) was used to find the gene names for the targets by setting the species as “Homo sapiens.” Moreover, in the GeneCards database (https://www.genecards.org/), ‟bone tuberculosis” was used as a keyword to find the common targets for the treatment of BT.
The STRING database (https://string-db.org/) was used to identify the potential molecular targets of each chemical ingredient of YHD. Cytoscape software (version 3.7.2) was used to construct the ingredient–target–disease networks of the herbs of YHD. The target genes were subjected to the Database for Annotation, Visualization, and Integrated Discovery (DAVID, https://david.ncifcrf.gov/) to obtain information via the gene ontology (GO) enrichment analysis and the Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis (KEGG; https://www.kegg.jp/kegg/) of the key target genes.
Molecular Docking
The 5 chemical ingredients of YHD that have the highest ADME, OB, DL, and Caco-2 permeability values were selected as the ligands for molecular docking analysis using a zinc database (http://zinc.docking.org/). The chemical composition files were used to select the top 5 core targets for the treatment of BT, and the UniProt numbers to download the protein files from the PDB database (http://www.rcsb.org/). Then, AutoDock software (version 4.2) was used to analyze the docking of the active ingredients at the key targets. The results were visualized using PyMOL software (version 2.2.0).
Preparation of YHD
The 7 herbs of YHD (SDH [30 g, Lot: 20190813], RG [3 g, Lot: 20180517], MH [2 g, Lot: 20200110], BJZ [6 g, Lot: 20190421], GJ [2 g, Lot: 20200207], GC [3 g, Lot: 20181221] and LJJ [9 g, Lot: 20170615]) were purchased from Shandong Qihuang Traditional Chinese Medicine Pieces Co., Ltd. The herbs were mixed and soaked in 10 times the amount of water (wt/wt) for 30 min, boiled twice for 30 min, extracted 3 times, and concentrated. The YHD extract was evaporated at 60 °C under vacuum conditions to obtain a dry YHD powder. The dried powder was dissolved in dimethylsulfoxide (DMSO) at a concentration of 1 g/mL for further experiments.
Culture of RAW 264.7 Cells
The RAW 264.7 cell line was purchased from the Cell Center of the Institute of Foundation, Chinese Academy of Sciences. The RAW 264.7 cells were cultured in high-glucose Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum and incubated in a 37 °C thermostatic incubator under 5% CO2 conditions. According to the morphology and wall growth of the cells, the culture medium was changed every 2 to 3 days. The cells (1.0 × 106 cells/well) were inoculated into a 6-well plate and observed under an inverted microscope.
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyl Tetrazolium bromide Assay
RAW 264.7 cells (5 × 103 cells/well) were cultured in a 96-well plate. Various concentrations of YHD (0, 75, 150, 300, or 600 μg/mL) were added to the cells, and the cells were incubated for various periods (24, 48, or 72 h). Six replicates were prepared for each combination of YHD concentration and cell incubation period. Twenty microliters of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (5 mg/mL) were added to each well, and the plate was incubated at 37 °C for 4 h. Then, 150 μL of DMSO was added to each well. Optical density was measured at 490 nm using a multimode microplate reader (Thermo Scientific).
Cell Culture and BT-Related Osteoclast Induction
Preparation of calf bones. Fresh cortical calf bones were selected. A chainsaw was applied perpendicularly to the bones at moderate force. Then, about 1.5 cm2 areas were cut at a thickness of about 4 to 5 mm, polished with abrasive stone, and fixed with 2.5% glutaraldehyde for 2 h. The bones were cut into pieces with a 30 µm thickness using a Leica hard-tissue cutter, and the bone pieces were placed in an ethanol gradient (75%-95% ethanol, 0.25 mol/L) and sonicated on an ultrasonic water bath for 10 min. Then, the bone pieces were subjected to natural air drying, γ-ray disinfection, and soaking in the 4 °C phosphate-buffered saline containing 1000 units/mL of penicillin and 1000 units/mL of streptomycin.
Preparation of MTB ultrasonic spallation (MTUS) using MTB H37Rv. Standard MTB H37Rv was obtained from the National Institute for the Control of Pharmaceutical and Biological Products. On a sterile operating table, the standard MTB H37Rv was revived and inoculated into 10 cm × 1.5 cm sized tubes containing the Lowenstein–Jenden medium. The tubes were incubated at 37 °C for 4 weeks. To prepare the MTUS, a certain number of living bacteria in the logarithm growth stage were placed in 50 mL centrifuge tubes, added with sterile ultra-pure water, and precooled for 20 min. Then, an ultrasonic disintegration device was used to lyse MTB on an 0 °C ice bath. The ultrasonic power was 300 W, the length of each disintegration cycle was 9 s each time, and the intermittent disintegration time was 1 h. After being centrifuged under frozen conditions at 3000 rpm for 1 h, the product was passed through a 0.22 μm filter. The MTUS was stored at −20 °C.
Induction of differentiation of RAW 264.7 cells into osteoclasts. RAW 264.7 cells (2.0 × 106 cells/well) were placed in a 24-well plate. The induction of the differentiation of RAW 264.7 cells into osteoclasts was performed with MTUS (20 μg/mL) and previously prepared calf bone pieces. The MTUS-induced RAW 264.7 cells were cultured in a 37 °C incubator under 5% CO2 conditions. The medium was replaced every day, and on the third day, the cells were challenged using MTUS for induction of differentiation into osteoclasts.
Cell Treatment and Observation of Tartrate-Resistant Acid Phosphatase Staining
Cells were pretreated with YHD (150 or 300 mg/mL) for 24 h, and the induction of the cells into osteoclasts was continued for 3 days. In the meantime, control and model groups were also established. To observe the effect of YHD on the differentiation of RAW 264.7 cells into osteoclasts, a tartrate-resistant acid phosphatase (TRAP) staining kit (Thermo Fisher Scientific) was used according to the manufacturer’s instructions. The TRAP staining solution and hematoxylin (150 μL) were added to each well and maintained in the well for 2 min. Then, the plate was incubated at 37 °C under dark conditions for 60 min before being rinsed with deionized water and dried. The distrene plasticizer xylene (DPX) mounting medium was added to the cells, and TRAP-positive cells (positive nuclei ≥3) were observed and counted under a microscope (Leica, Olympus).
Statistical Analysis
The data were processed using SPSS19.0 software, and the results were expressed as the mean ± standard deviation (
Results
Main Bioactive Ingredients and Predicted Targets of YHD
There were 138 potential active ingredients in YHD that were screened out, and 543 action targets of the ingredients that were collected. Then, 198 relevant disease targets for BT were retrieved and collected using the GeneCards database (Selection score > 18). A Venn chart was drawn to find the 50 intersecting targets of the ingredient targets and disease targets (Figure 1A). In succession, using Cytoscape software (3.7.2 version), a “drug–ingredient–target–disease” visualization network of the intersection targets was constructed (Figure 1B).

Prediction of YHD main bioactive ingredients and targets on BT by network pharmacology. (A) Venn diagram of targets for YHD treatment of BT. (B) Drug–ingredient–target–disease network of YHD on BT (green nodes represent the herb medicines, blue nodes the effective ingredients of the herb medicines, orange nodes identify the interaction targets of herb medicines and disease, and the red node indicates the disease). (C) PPI network of the core targets.
The STRING online analysis platform was used to download the tab separated value (TSV) files of the intersecting targets, which were based on the network constructed using Cytoscape. The results exhibited that there were 23 key core targets: insulin (INS), interleukin-6 (IL6), vascular endothelial growth factor A (VEGFA), tumor necrosis factor (TNF), protein kinase B (AKT1), epidermal growth factor (EGF), matrix metalloproteinase 9 (MMP9), CXC motif chemokine ligand 8 (CXCL8), interleukin 10 (IL10), TP53, interleukin 2; IL2, interleukin 1B (IL1B), interferon-gamma (IFNG), epidermal growth factor receptor (EGFR), mitogen-activated protein kinase 1 (MAPK1), matrix metalloproteinase 2 (MMP2), Fabry outcome survey (FOS), interleukin 4 (IL4), secreted phosphoprotein 1 (SPP1), C-reactive protein (CRP), transforming growth factor-beta 1 (TGFB1), myeloperoxidase (MPO), and catalase (CAT) (the target code obtained from the UniProt database). The protein–protein interaction (PPI) network diagram is shown in Figure 1C.
Targets and Pathways That are Affected by YHD to Treat BT
The 50 intersecting targets were analyzed by GO enrichment analysis using DAVID database and screened on the basis of their by P values, Figure 2A showed that YHD relieved of BT mainly through the modulation of the inflammatory response, positive regulation of transcription from the RNA polymerase II promoter and immune responses. The results of KEGG pathway enrichment analysis (Figure 2B) indicated that the core pathway involved in the anti-BT effect of YHD was the PI3K–Akt signaling pathway, the mitogen-activated protein kinase (MAPK) signaling pathway, and cytokine–cytokine receptor interaction.
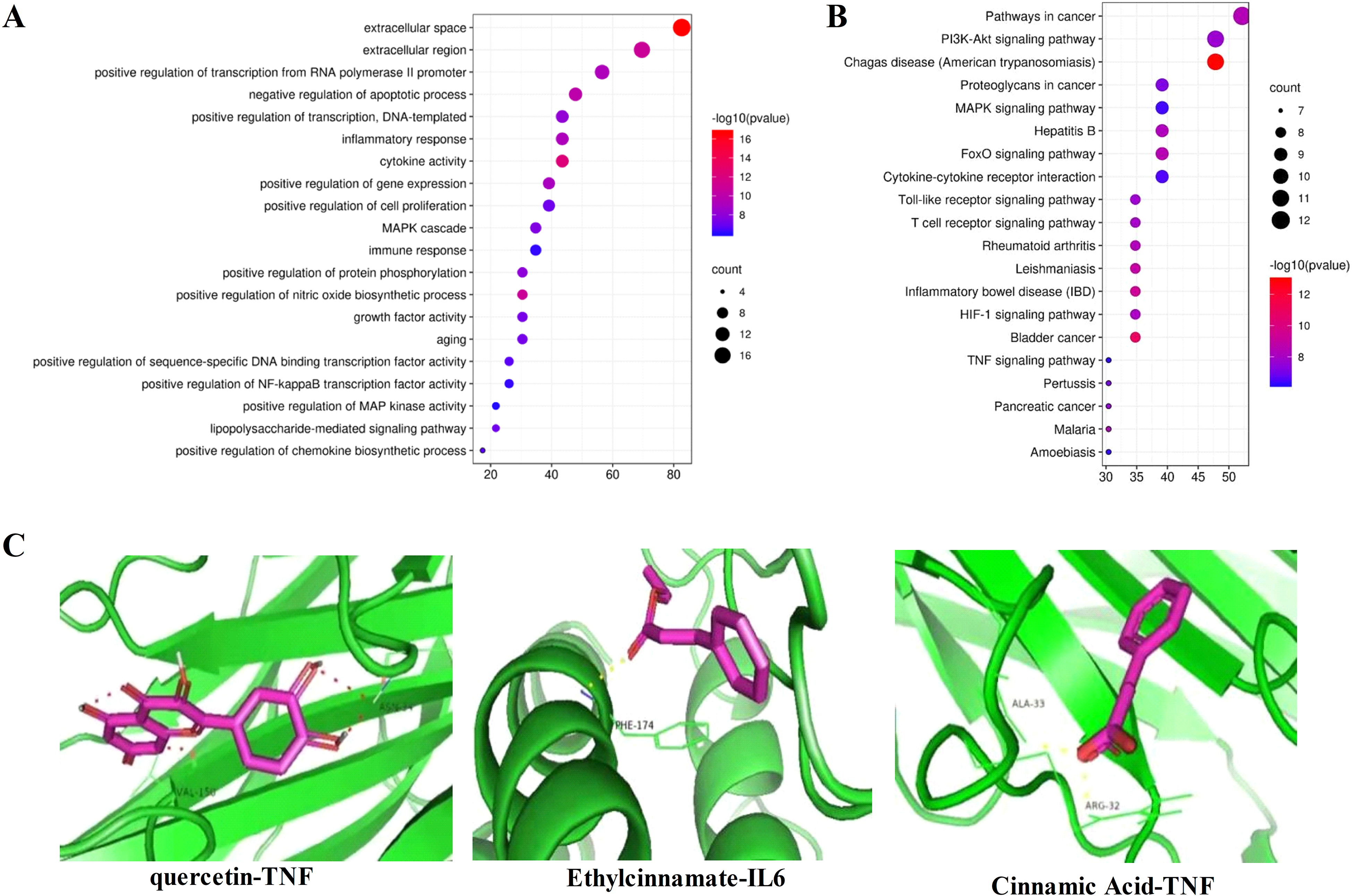
Enrichment analysis and molecular docking results. (A) GO functional enrichment analysis about molecular function results. (B) KEGG pathway enrichment analysis. (C) Dominant conformation of the bioactive components with target proteins.
After screening, the genes TNF, VEGFA, IL6, AKT1, and INS were selected as the docking targets, and kaempferol, quercetin, trans-cinnamic acid, ethyl cinnamate, and cinnamic acid were identified as the active ingredients of the docking targets. The above genes were linked to the associated active ingredients using Autodock software, and the minimum binding energy was calculated. The docking results were visualized with PyMOL software. The results are shown in Table 1 and Figure 2C.
Main key Targets and Active Ingredient Docking Energy of YHD on BT.

Abbreviations: YHD, Yang He Decoction; BT, bone tuberculosis; INS, insulin; IL6, interleukin-6; VEGFA, vascular endothelial growth factor A; TNF, tumor necrosis factor; AKT1, protein kinase B.
Effect of YHD on Proliferation of RAW 264.7 Cells and MTUS-Induced Differentiation of RAW 264.7 Cells into Osteoclasts
At a concentration of ≥150 mg/mL, YHD significantly inhibited the proliferation of RAW 264.7 cells (P<.01) in a concentration-dependent and duration-dependent manner (Figure 3A), which suggested that YHD could affect the proliferation and differentiation of RAW 264.7 cells.
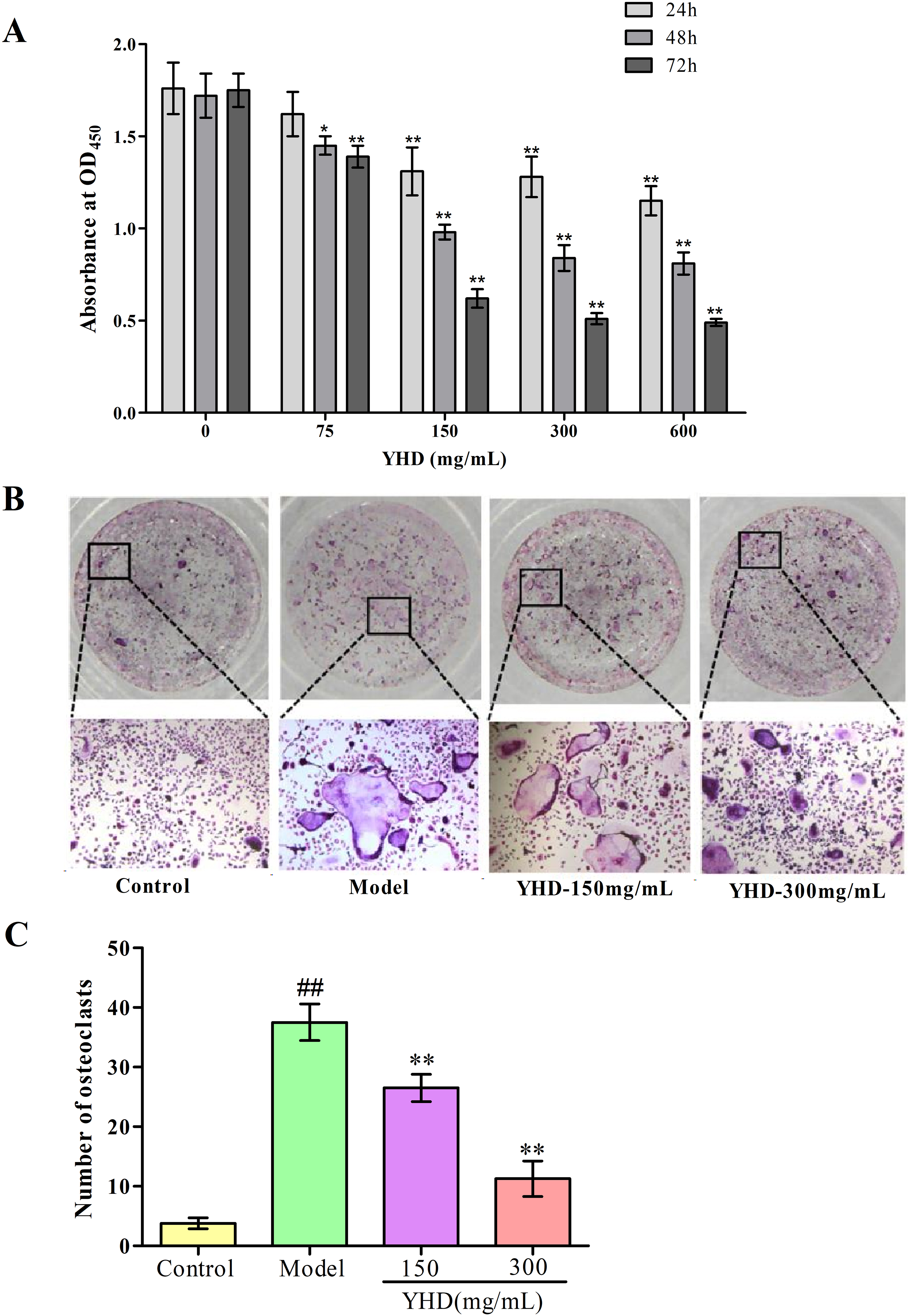
Effect of YHD on osteoclast differentiation of RAW264.7 cells. (A) RAW264.7 cell proliferation at different concentrations of YHD after treatment for 24, 48, and 72 h. (B) Observation of cellular morphology with MTUS post YHD intervention on day 3 via TRAP staining (magnification ×100). (C) Comparison of TRAP stained positive cells between different groups. Note: Compared with model group, *P < 0.05, **P < 0.01; Compared with control group, #P < 0.05, ##P < 0.01.
As illustrated in Figure 3B, RAW264.7 cells were small cells with uniform size and distribution. After 3 days, the number of cells on culture plates increased. The cells had irregular edges, irregular protrusions, and pseudopods. The TRAP staining solution stained the cytoplasm and nuclei of osteoclasts purplish-red and blue, respectively (Figure 3B). The number of TRAP-positive cells in the culture of RAW 264.7 cells that was added with 150 or 300 mg/mL of YHD significantly decreased compared to the number of TRAP-positive cells in the control culture of RAW 264.7 cells (P < .01, Figure 3C). This suggested that YHD inhibited the differentiation of RAW 264.7 cells into osteoclasts.
YHD Lowered Inflammatory Cytokines and Regulated Antioxidative Factors
The differentiation of RAW 264.7 cells into osteoclasts is associated with inflammation and oxidative stress. YHD significantly reduced the levels of inflammatory cytokines (TNF-α and IL-1β) and regulated the levels of antioxidative factors (superoxide dismutase [SOD], glutathione peroxidase (GSH-Px), and malondialdehyde [MDA]) in MTUS-induced RAW 264.7 cells (P<.01), which suggested that YHD inhibited inflammation and oxidative stress to inhibit the differentiation of RAW 264.7 cells into osteoclasts. The results are shown in Figure 4.

Effect of YHD on inflammatory cytokines (TNF-α, IL-1β) and antioxidation factors (SOD, GSH-Px, and MDA) in MUTS-induced RAW264.7 cells. (A) Inflammatory cytokines. (B) Antioxidation factors. Note: Compared with model group, *P < .05, **P < .01; Compared with the control group, #P < .05, ##P < .01.
YHD Suppressed the PI3K/Akt and MAPK Signaling Pathways
Recent studies have indicated that YHD inhibited the differentiation of RAW 264.7 cells into osteoclasts by inhibiting PI3K/Akt and MAPK signaling pathways.12,13 Similar to the reported results, the results of our network pharmacology analysis (Figure 5) revealed that YHD affected BT by inhibiting the above-mentioned pathways. The levels of PI3K, Akt, p38 MAPK, and c-Jun N-terminal kinase (JNK) in the cells of the YHD group were significantly lower than those in the cells of the control group (P<.01). The results suggested that YHD inhibited the differentiation of RAW 264.7 cells into osteoclasts by inhibiting the activation of the PI3K/Akt and MAPK signaling pathways.

Effect of YHD on PI3K/Akt–MAPK signaling pathway in the osteoclast differentiation process. (A) Comparison of mRNA expression. (B) Western blot photograph. (C) Comparison of protein expression. Note: Compared with model group, *P < .05, **P < .01; compared with the control group, #P < .05, ##P < .01.
Discussion
Throughout the pathogenesis of BT, bone destruction and formation occur significantly. Osteoclasts play an important role in the pathogenesis of BT. 14 Scientists have explored the origin of osteoclasts for nearly 100 years. Nowadays, most scholars recognize that osteoclasts originate from mononuclear macrophages. The main role of osteoclasts is to perform both the physiological and pathological bone absorption in the body. Osteoclasts, which are dispersed throughout bone tissue, consist of multinucleated giant cells and produce positive results when stained with TRAP staining solution.15,16 The dynamic balance between osteoclasts and osteoblasts maintains the morphology of healthy bones. However, under certain conditions, the number and function of osteoclasts may change, which causes bone absorption and destruction. 17 RAW 264.7 cells are tumor cells induced by the Abelson murine leukemia virus. The cells have a circular shape and grow fast.18,19 Cell strains are easy to culture and proliferation. RAW 264.7 cells express macrophage colony-stimulating factor (M-CSF), oncogene receptors (C-fms receptor), and RANK in bone metabolism. The in vitro cell model can basically simulate the characteristics of tuberculosis lesions and can cause pathological changes similar to bone and joint tuberculosis, which avoid the spread of live bacteria and has a good application value in research for tuberculosis-related diseases. 20 MTB H37rv was chosen in this study because it is a highly aggressive and infectious strain, which antigenicity is similar to that of clinical MTB strains. Previous studies have found that the pyrolysis products of MTB can induce the differentiation of RAW 264.7 cells into osteoclasts, and IL-1β, TNF-α, and transforming growth factor 1 (TGF 1) are involved in the differentiation process.21–23 The roles of antioxidative factors in the differentiation of RAW 264.7 cells into osteoclasts have been examined.24,25 YHD decreased the number of TRAP-positive cells, which indicated that YHD reduced the levels of inflammatory cytokines (TNF-α and IL-1β) and regulated the levels of antioxidative factors (SOD, GSH-Px, and MDA) in MTUS-induced RAW 264.7 cells. These results indicated that YHD inhibited inflammation and oxidative stress to block the differentiation of RAW 264.7 cells into osteoclasts.
Recently, the network pharmacology approach, a systematic analytical technology that is based on the development of bioinformatics, network biology, and pharmacology, has been broadly applied to TCM and emerges as an indispensable method for TCM development.6,26 Considering the commendable ability of the approach to explore the multicomponent, multitarget, and multipathway characteristics of TCM, 27 we conducted a network pharmacology analysis to clarify the anti-BT ingredients of YHD. As they are closely related to the inflammatory response, the PI3K/Akt and MAPK signaling pathways may have some roles in the differentiation of RAW 264.7 cells into osteoclasts. In the current study, through network pharmacology analysis and further experiments, we found that YHD can act on both signaling pathways. Therefore, as demonstrated, the anti-BT effect of YHD may be associated with the inhibition of PI3K/Akt and MAPK signaling pathways.
The effect of YHD on the signal transduction system is a too complex network to fully clarify the mechanism of YHD. Through network pharmacology analysis, the inhibitory effect of YHD on the PI3K/Akt and MAPK signaling pathways and thus differentiation of RAW 264.7 cells into osteoclasts was further investigated. The differentiation of RAW 264.7 cells into osteoclasts can be activated or suppressed by staggered interactions among signaling factors, and the activation or inhibition of these factors can regulate the balance between bone formation and absorption. As the report goes, the MAPK and PI3K/Akt pathways are associated with osteoclast differentiation. 28 The present study revealed that YHD could block both the PI3K/Akt and MAPK pathways.
The PI3K/Akt and MAPK signaling pathways have been shown to play a crucial role in the differentiation and maturation of osteoclasts and osteoblasts.12,13 However, research on the downstream target protein is relatively lacking. As is known, the signaling transduction pathway is a complex but valuable network system, which can be used to understand the downstream target proteins affected by the differentiation and maturation of osteoclasts. And it may diagnose the disease and even activate and inhibit target proteins. Our future work is going to focus on studying the downstream target proteins that regulate the differentiation and maturation of osteoclasts and osteoblasts, and further establishing a theoretical basis for the dynamic regulation of the balance between osteoclasts and osteoblasts. In addition, similar to a previous study, 29 the present study confirms that the PI3K/Akt signaling pathway is associated with inflammation and oxidative stress.
Conclusion
YHD suppressed the MTUS-induced differentiation of RAW 264.7 cells into osteoclasts by inhibiting the PI3K/Akt and MAPK signaling pathways with modulation of inflammation and oxidative stress. Collectively, YHD is a promising complementary drug for BT, which inhibits the PI3K/Akt and MAPK signaling pathways. Further studies are needed to explore the relationship between each active ingredient of YHD and each disease target.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X221075080 - Supplemental material for Effect of Yang He Decoction on Treatment of Bone Tuberculosis via Phosphoinositide 3-Kinases/Protein Kinase B and Mitogen-Activated Protein Kinase Signaling Pathways
Supplemental material, sj-doc-1-npx-10.1177_1934578X221075080 for Effect of Yang He Decoction on Treatment of Bone Tuberculosis via Phosphoinositide 3-Kinases/Protein Kinase B and Mitogen-Activated Protein Kinase Signaling Pathways by Qiang Zhang, Wanli Ma, Qing Wang, Hua Gao and Jianmin Sun in Natural Product Communications
Supplemental Material
sj-xlsx-2-npx-10.1177_1934578X221075080 - Supplemental material for Effect of Yang He Decoction on Treatment of Bone Tuberculosis via Phosphoinositide 3-Kinases/Protein Kinase B and Mitogen-Activated Protein Kinase Signaling Pathways
Supplemental material, sj-xlsx-2-npx-10.1177_1934578X221075080 for Effect of Yang He Decoction on Treatment of Bone Tuberculosis via Phosphoinositide 3-Kinases/Protein Kinase B and Mitogen-Activated Protein Kinase Signaling Pathways by Qiang Zhang, Wanli Ma, Qing Wang, Hua Gao and Jianmin Sun in Natural Product Communications
Footnotes
Author Contributions
QZ and WLM wrote the manuscript and carried out the experiments. QW and HG performed data analysis and revised the manuscript. JMS contributed to the design of this study and was responsible for the overall planning of the manuscript. All the authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Traditional Chinese Medicine Science and Technology Development Plan of Shandong Province (grant number No.2019-0524).
Ethical Approval
This study was approved by the Shandong Provincial Hospital, Cheeloo College of Medicine, Shandong University, China.
Statement of Human and Animal Rights
There are no human and animal subjects in this article and informed consent is not applicable.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
