Abstract
Keywords
Introduction
Metabolic associated fatty liver disease (MAFLD), once known as non alcoholic fatty liver disease (NAFLD). 1 With the increasing research on NAFLD, it has been found that metabolic disorders are more important than etiology in the evolution of NAFLD. And it has been clarified that its definitive diagnosis is a pathology characterized by hepatic lipid accumulation with metabolic disorders. 2 According to the latest criteria, patients with hepatic steatosis can be diagnosed with MAFLD as long as they fulfill at least one of the criteria of overweight or obesity, type 2 diabetes, or the presence of evidence of metabolic dysregulation.3,4 The global pooled prevalence of MAFLD was estimated to be 32.4%, with a higher prevalence in males (39.7%) than in females (25.6%), and the prevalence has been increasing year by year, reaching as high as 37.8% after 2016 5 ; the highest pooled prevalence was found in Latin America (44.4%), followed by the Middle East, North Africa, South Asia, Southeast Asia, North America, and East Asia, with the lowest prevalence in Western Europe (25.1%). 6 The pooled prevalence of adult MAFLD in China over the past 20 years was 29.6%, and was higher in men (34.8%) than in women (23.5%). 7 MAFLD is not only a high risk factor for cirrhosis and liver cancer, but also for coronary heart disease, diabetes mellitus and hyper lipidaemia.8-11 Currently, in addition to lifestyle interventions, the main treatment for MAFLD is symptomatic therapy, including statins and GIPR/GLP1R,12,13 so it is crucial to explore new therapeutic agents.
The high incidence of MAFLD is closely related to contemporary people's lifestyles such as poor diet and staying up late with little movement. “Diet since double, stomach and intestines are injured”, overeating fat, sweet, spicy, cold products can damage the spleen and stomach, resulting in spleen qi deficiency, malfunction of the spleen. On the one hand, “fluid, water and grain odor is also born”. If the spleen is not able to distribute the essence, the dietary water and grain can not be transformed into essence, but stagnation for the disease, turbid evil within; on the other hand, spleen deficiency generates dampness, aggravating the stagnation of water and liquid, moisture into phlegm, resulting in dampness, phlegm, stasis and blood and other pathology products. 14 According to Traditional Chinese medicine (TCM), the key factor in the development of MAFLD is a deficiency of the spleen. Damage to the spleen and stomach will result in nutrients not being able to be transported properly and stagnating in the body, leading to an imbalance of liver qi and the accumulation of dampness, turbidity, stasis, and heat in the liver, resulting in the formation of MAFLD. Shenling Baizhu powder (SLBZP) is derived from the Song Dynasty's “Taiping Huimin Hejiju Prescription”, which consists of herbs for strengthening the spleen, resolving dampness, and draining the liver and qi. It has been used for thousands of years as a representative formula for the treatment of diseases related to weakness of the spleen and stomach. SLBZP is recommended for the treatment of metabolism-related diseases, 15 and clinical practice also generally recognizes SLBZP as a good treatment for MAFLD patients with spleen deficiency and dampness.16,17
Network pharmacology seen as a new way to explore TCM. The complex interactions between drugs, diseases and biological systems can be systematically expressed using network pharmacology, 18 thereby systematically revealing the biological basis of the overall diagnosis and treatment of TCM. 19 Molecular docking simulates the ability of a drug component to bind to a protein target and is one of the most common means of developing drugs and is the primary modality used in this study to validate network pharmacology results. MAFLD is a metabolic syndrome caused by various factors such as dietary disorders, lack of exercise, etc. At present, there is no specific drug for the treatment of MAFLD, and network pharmacology can combine traditional Chinese medicine prescriptions with modern biotechnology, which not only preserves the culture and therapeutic concepts of traditional medicines, but at the same time verifies and analyzes their mechanisms of action through scientific methods, which is a new way of modernization and internationalization of traditional medicines through this interdisciplinary approach. This interdisciplinary research method provides a new way for the modernization and internationalization of traditional medicines.
This study used a combination of network pharmacology, molecular docking and experiment to analyze the active ingredients, potential targets and molecular mechanisms of SLBZP in the treatment of MAFLD, and we conducted preliminary verification by experiments. The workflow is shown in graphical abstract Figure 1.

The experimental flow chat.
Methods
Screening of Active Components and Their Targets in SLBZP
The components of all herbal medicines in SLBZP were obtained by searching the traditional Chinese medicine systematic pharmacology database and analysis platform 20 (TCMSP, http://tcmspw.com/). Oral bioavailability (OB) and drug similarity (DL) 21 are key parameters for the evaluation of drug effectiveness. In this study, the active components and their corresponding targets were screened for both OB ≥ 30% and DL ≥ 0.18,22,23 and the results were imported into the UniProt database 24 (http://www.Unitprot.org/) and transformed into uniform gene names.
Predicting the Target of MAFLD
The search terms “non-alcoholic fatty liver disease” or “metabolism-related fatty liver disease” were used to search the Gene Card 25 (https://www.genecards.org/), TTD 26 (http://db.idrblab.net/ttd/) and the DisGeNET 27 (https://www.disgenet.org/) database for disease-related targets. In which, disease genes with a score >1 in the Genecards database were selected. The obtained targets were pooled to remove duplicate values to obtain targets for MAFLD. From these, the targets were matched to the active components of SLBZP to obtain crossover targets, which are the potential targets of SLBZP for MAFLD. Venn diagram of the crossover targets of SLBZP and MAFLD using the Venny platform 28 (https://bioinfogp.cnb.csic.es/tools/venny/index.html).
Constructing the “Herb - Active Component - Crossover Targets” Network
The network and type files were prepared from the data collected and imported into Cytoscape 3.7.2 software 29 to construct the ‘herb-active component-crossover targets’ network, which was topologically analyzed by the plug-in Network Analyzer. The degree value reflects the importance of the nodes in the network, and the core active components in the network are identified according to the degree value of the active component.
Constructing the “Protein–Protein Interaction” (PPI) Network
Upload crossover targets to the STRING database 30 (version 11.5, https://string-db.org), set the species to “Homo sapiens” with a confidence score of ≥0.4 between targets, and download the PPI network and TSV files. The TSV files were imported into Cytoscape 3.7.2, and topological analysis of PPI parameters was performed using its plugin CytoNCA, and the core targets for SLBZP treatment of MAFLD were screened based on the median of the three main parameters of “Degree Centrality (DC)”, “Betweenness Centrality (BC)” and “Closeness Centrality (CC)”.
GO and KEGG Enrichment Analyze
To elucidate the biological functions and associated signaling pathways involved in the crossover targets, they were imported into the David database
31
(version 6.8, https://david.ncifcrf.gov/) for GO function and KEGG pathway enrichment analysis. The statistical significance threshold was set at
Molecular Docking
Based on degree values, the top four active compounds were selected as ligands for molecular docking with the core crossover targets. Download the mol2 file of the 2D structure of the active compound in the TCMSP database. The RCSB-PDB database 32 (http://www.rcsb.org/pdb/) provides 3D structural data on large biomolecules. After determining the PDB ID of the core target gene by reviewing the literature, the PDB file of the 3D protein receptor structure of the core targets was downloaded. The ligand and water molecules were removed from the crystal structure of the PDB files using Pymol 2.4 software. 33 The PDB files and mol2 files add hydrogen and add charge in Auto Dock Vina 1.1.2 software 34 and are saved in PDBQT format as receptors and ligands for molecular docking and binding energy statistics. It is generally accepted that the binding energy is negatively related to the stability and binding activity of the structure. When the binding energy is <0 (kJ/mol), meaning that there is binding activity between the active compound and the core crossover target, and when the binding energy is < −5 (kJ/mol), suggesting that there is good binding activity between the two. 35 The results of molecular docking were visualised in Pymol 2.4 software.
Experimental Validation
Reagents
SLBZP was purchased from Guangdong Yifang Pharmaceutical Co. Ltd; HepG2 cells (CL-0103), fetal bovine serum (164210), PBS buffer (PB180327), MEM medium (PM150410), double antisera (PB180120) were purchased from Wuhan Punuosai Life Science and Technology; trypsin (Gibco 25300-054), DMSO (Solabio Technology, D8371), palmitic acid (Sigma, P0500), CCK-8 (Servicebio, G4103), and Triglyceride (TG) assay kit (A110-1-1),total cholesterol (TCH/T-CHO) assay kit (A111-1-1) were purchased from Nanjing Jiancheng Bioengineering Institute, AMPKα (Signalway Antibody, #40585), p-AMPKα (Signalway Antibody, #11183), SREBP-1C (Signalway Antibody, #41458).
Animal Pharmacodynamic Study
Experimental Animals and Drugs
48 SPF-grade male SD rats, all weighing (180 ± 20) g, were purchased from Changsha Tianqin Biotechnology Co. Ltd, License No.: SCXK (Xiang) 2019-0013; the high-fat diet was purchased from Suzhou Shuangshi Experimental Animal Sheds Food Science and Technology Co. Ltd, License No.: Su 20160102 (the high-fat diet consisted of 20% lard, 4% sugar, 2% powdered milk, 1% cholesterol and 73% normal diet).
Preparation of Pharmaceutical Solution
The human clinical dose (Drug1) was converted to rat equivalent dose (Drug2) on the basis of body surface area using the formula:
Animal Grouping, Modeling and Treatment
The 48 SD male rats were acclimatized and fed for 1 week. Based on the randomized numeric subtabulation method, divided into blank control group (NC group, n = 8) and model group (MAFLD group, n = 40) according to the method of random number table. The rats in the NC group were fed with normal diet and the rats in the MAFLD group were fed with high-fat diet according to the pre-modeling method of the research group. All rats were given free access to water, and the MAFLD rat model was successfully established after 4 weeks. In order to test whether the MAFLD model was successfully established. The pathological changes of rat livers after 4 weeks of modeling were examined by H&E and oil red O staining, and the results of H&E staining showed that the hepatocytes in group M were abnormally arranged, with small vacuoles in some of the cells and a large number of vacuole-like structures around the hepatocytes compared with those in group NC. In addition, the results of oil red O staining showed that there were a lot of oil red O precipitates and a lot of lipid droplets in the livers of group M compared with that of group NC. The above results indicated that the MAFLD animal model had been successfully established (see Appendix Figure A1 for details). The rats in the MAFLD group were again randomized numeric subtabulation method, divided into the HFD group (n = 8), the SLBZP low-dose group (SLBZP-L, n = 8), the SLBZP medium-dose group (SLBZP-M, n = 8), the SLBZP high-dose group (SLBZP-H, n = 8), and the silybin group (Silybin group, n = 8). Feeding was kept unchanged in all groups, and the SLBZP-L, SLBZP-M, SLBZP-H and Silybin groups were given gavage treatment with the corresponding drug solution (10 m L/kg/d) for 3 weeks, and the HFD and NC groups were treated with saline gavage in equal volume. They were fasted for 12 h before the last dose and anesthetized with 1% sodium pentobarbital, the rats were sacrificed and dissected to collect the liver.
Staining of Liver Pathology Sections
Oil Red O Staining
Slices of the same parts of the livers were cut off and frozen for staining with Oil-red O with standard method.
H&E Staining
The liver tissues were fixed in 10% paraformaldehyde for 48 h and embedded in paraffin, then the tissues were cut into 3∼5 μm sections and stained with hematoxylin and eosin. All the specimens were examined under a light microscop.
Cellular Experimental Validation
The HepG2 hepatocyte cell line was cultured in MEM base medium,supplemented with 0.5% double antibody mixture at 37 °C in a 5% CO2 atmosphere. For induction of hepatic steatosis, HepG2 cells were incubated with 0.3 mM of PA in MEM base medium to stimulate lipid accumulation for 24 h. 38 Serum samples containing SLBZP were obtained from the SLBZP 5.4 g/mL normal diet-fed SD rats, and serum samples containing Silybin were obtained from the Silybin 9.4 mg/mL normal diet-fed SD rats, respectively. Then, drug-containing serotoxicity was tested by cck-8, for details, see Table 1. Lipid droplet changes of HepG2 cells were detected through oil red O staining; the levels of TG and TC in HepG2 cells were detected; the expressions of AMPK, p-AMPK, and SREBP-1c were detected through Western blot. The grouping and specific operations are shown in Tables 2 and 3.
Drug-Containing Serotoxicity.

Common Culture medium Preparation System.

Cell Grouping and Intervention.

Statistical Analysis
Data were analyzed using GraphPad Prism 8.0, and results were expressed as mean ± standard deviation. One-way ANOVA was used to differentiate between the different groups.
Results
Network Pharmacology Results
Active Compounds and Targets of SLBZP
Up to 950 components of SLBZP were retrieved, of which 204 active components met the screening criteria. Of these, the active components without targets were removed, resulting in 161 active components and 265 targets. Table 4 shows the number of active components with OB ≥ 30%, DL ≥ 0.18 and their corresponding targets for each herb in the SLBZP.
Number of Active Ingredients and Targets of Each Herb in SLBZP.

Identification of Targets for SLBZP Treating MAFLD
In all 263 disease genes were retrieved, of which 179 genes for MAFLD with a score of >1 was retrieved from the Gene Card database. After removing duplicate targets, 243 targets of MAFLD were identified (Figure 2a). A total of 41 targets overlapped between active component targets and MAFLD targets (Figure 2b), which were considered as potential targets for SLBZP treating MAFLD.

Venn diagrams. (a) Venn diagram of MAFLD-related targets. (b) Venn diagram of SLBZP targets and MAFLD targets.
Regulatory Network Analysis of “Herb - Active Compound - Crossover Targets” Network
These 41 crossover targets were matched to the 122 active compounds of SLBZP. The “herb - active compound - crossover targets” network contains 175 nodes (including 12 herbs, 122 compounds and 41 overlapping genes) and 461 edges (Figure 3). Node-to-node connectivity called degree, the more other nodes connected to a node, the higher its degree value, indicating that it is more central in that network. Table 5 shows the main information on the top 10 active compounds, the highest degrees of active ingredients in the network are B1(quercetin), A2(naringenin), A6(luteolin) and A5(kaempferol), which are all compounds belonging to a wide range of herbs, suggesting that they are the main active components of SLBZP in treating MAFLD.

The herb - active compound - crossover targets network of SLBZP (the hexagon node indicates the herbal name of SLBZP, the diamond node indicates the active compound and triangular node indicates the active compound from multiple herbs (A: from two herbs; B: from three herbs; C: from four herbs). Each colour represents an herb and its active compound. The green rectangular node represents the crossover targets of SLBZP and MAFLD.).
Basic Information on the top 10 Degrees of Active Compounds of SLBZP.

Construction of PPI Networks and Identification of Core Targets
The 41 crossover targets were subjected to PPI network analysis in the STRING database, with 41 nodes and 402 edges (Figure 4a), and plot a bar chart of the top 20 targets of the PPI network degree (Figure 4b). Topology analysis of PPI parameters was performed in Cytoscape 3.7.2 by the plugin CytoNCP, selecting medians greater than DC, BC, and CC to create sub-networks for screening core targets (Figure 5a). The conditions of the first sub-network were set to DC > 18, BC > 8.88 and CC > 0.64, and a total of 19 nodes and 147 edges were obtained (Figure 5b), which could be the core targets of SLBZP in treating MAFLD. To narrow the scope, we created a second sub-network: DC > 27, BC > 29.73, CC > 0.75. We finally obtained 9 nodes and 35 edges, namely ALB, TNF, IL1B, PPARα, PPARγ, HMOX1, PTGS2, IL6 and ADIPOQ (Figure 5c), the specific DC, BC, and CC of these can be seen in Table 6.

(a) PPI network of the core targets of SLBZP against MAFLD. (b) The top 20 significant genes in PPI network.

The network topology analysis of the PPI network. (a) The PPI network of the crossover targets was generated by Cytoscape, which is comprised of 41 nodes and 402 edges. (b) The subnetwork after DC, BC, and CC filtration, which is comprised of 19 nodes and 147 edges. (c) The core network after DC, BC, and CC filtration, which is comprised of 9 nodes and 35 edges.
PPI Core Target BC, CC, DC Values.

GO and KEGG Enrichment Analyze
A total of 263 GO entries were enriched (
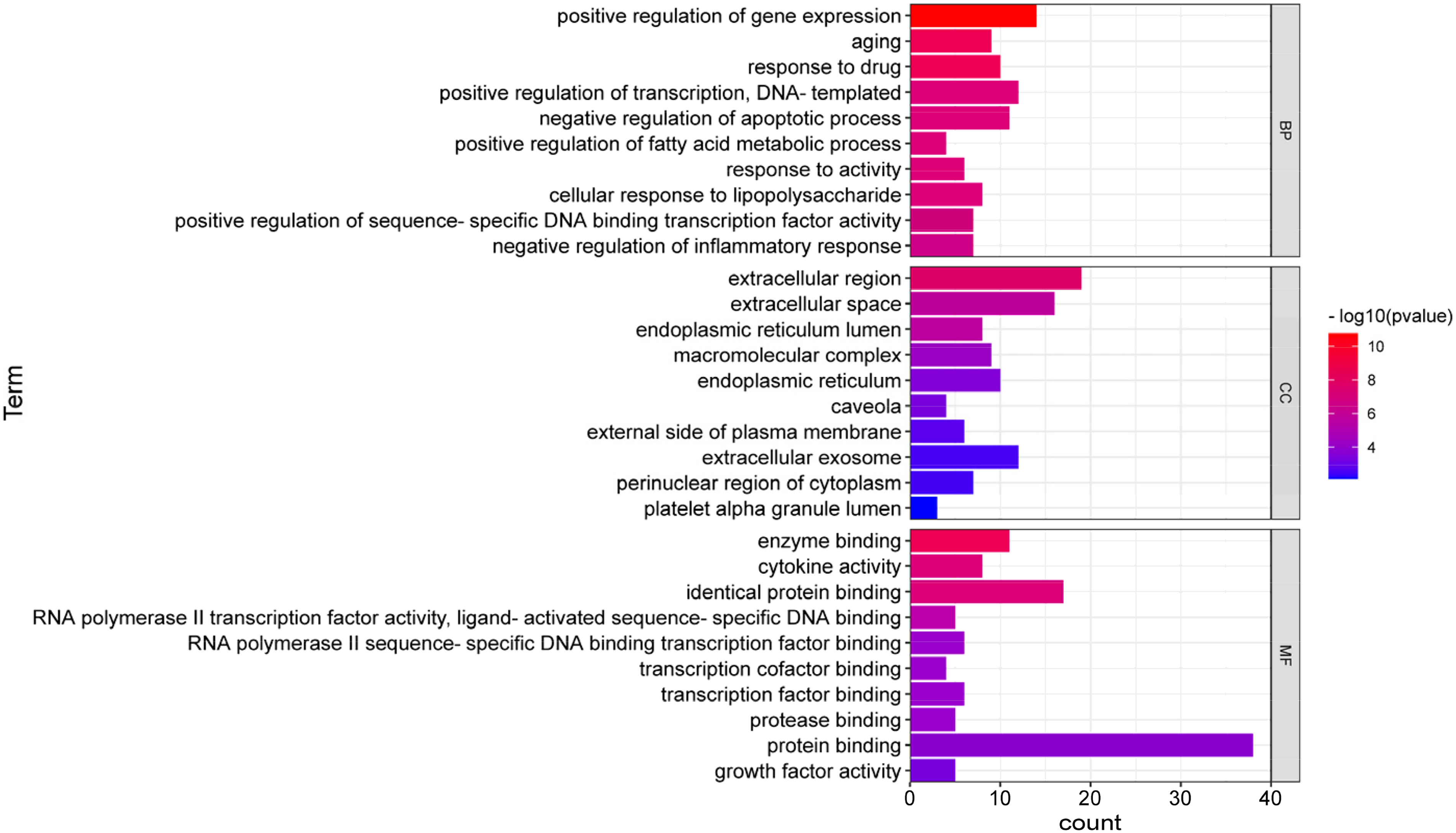
Gene ontology terms of 41 crossover targets. The top 10 GO functional terms were selected (
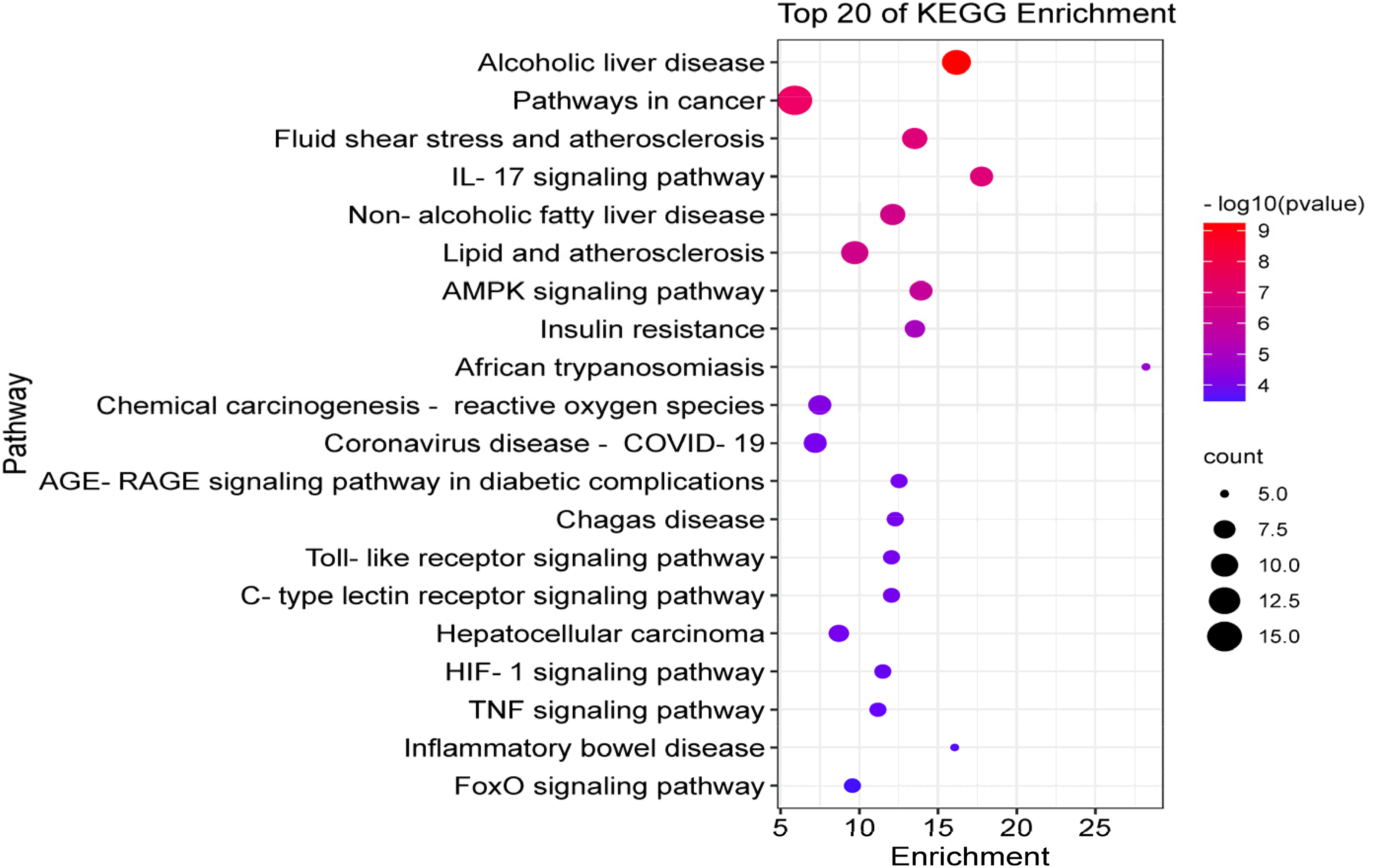
KEGG pathway enrichment of 41 crossover targets. The top 20 pathways were identified. Color represented P value, and size of the spot represented count of genes.

AMPK signaling pathway diagram. The red labeled genes represent the pathway on which SLBZP therapeutic target of this pathway.
Molecular Docking Results
4 core targets and pathway targets were randomly selected for molecular docking with key active compounds, and the lower their binding energies indicated higher affinity. Table 7 show the docking results of the AMPK pathway targets AMPK, SREBP-1c with quercetin and naringenin, as well as plots of the binding energies and simulated docking results of quercetin, naringenin, lignocerol, and kaempferol with ALB, TNF, IL1B, and ADIPOQ, respectively. The results showed that all docking binding energies were < −5 kcal/mol, It indicates that the coordination and the receptor can form a relatively stable spatial conformation, and it also indicates that the SLBZP active ingredient has a strong binding ability to the MAFLD core gene. In Figure 9, the yellow dashed line represents hydrogen bonding, where the ligand molecule binds to the receptor molecule through hydrogen bonding and other interactions, forming a relatively stable spatial conformation. This indicates a strong binding ability and stable spatial conformation among the active component of SLBZP and the core target gene of MAFLD. This indicates that the molecular docking results initially validate the results of network pharmacology and enhance the reliability of it.

Molecular dockings of the top 4 active compounds and core targets. (a–d) Molecular docking plots of ALB with quercetin, naringenin, lignans, and kaempferol. (e–h) Molecular docking plots of TNF with quercetin, naringenin, lignans, and kaempferol. (i–l) Molecular docking plot of IL1B with quercetin, naringenin, lignans, and kaempferol. (m–p) Molecular docking plot of ADIPOQ with quercetin, naringenin, lignans, and kaempferol. (q–r) Molecular docking plots of AMPK with quercetin and naringenin; (s–t) Molecular docking plots of SREBP-1c with quercetin and naringenin.
the optimum Model for Molecular Docking.

In Vivo and in Vitro Experimental Results
Effect of SLBZP on the Morphology of the Liver Tissue of Rats Suffering from HFD-Induced MAFLD
Oil red O (Fig. 10a) and H&E (Fig. 10b) staining as shown in the figure showed no obvious lesions and lipid droplets in the liver sections of the NC group. Compared with the NC group, the MAFLD group showed a significant increase in steatosis and lipid droplets, pronounced hepatic steatosis, and inflammatory cell infiltration in the center of lobules and portal vein area, which also confirmed the success of the metabolism-associated steatohepatopathy model. The SLBZP-L, SLBZP-M, and SLBZP-H groups showed varying degrees of improvement in lipid droplets and lesions compared with the model group. The results indicated that SLBZP could improve lipid accumulation and reduce hepatic steatosis in rat liver induced by high-fat diet.

Effect of SLBZP on lipid accumulation and hepatic steatosis in HFD-fed rats. (a) Oil red O staining of liver tissue (×200). Blue arrows: lipid droplets. (b) H&E staining of liver tissue (×200). Red arrow: fat vacuoles, Blue arrow: inflammatory infiltrate.
Cell Viability of HepG2 Cells Treated with SLBZP
In order to determine the optimal administration concentration, the effects of different concentrations of drug-containing serum on cell viability were detected by CCK-8. there was no inhibitory effect on cell viability with 10% drug-containing serum in the SLBZP and Silybin groups compared to 10% drug-containing serum in the K group (Figure 11a).

Effect of SLBZP on HepG2 cells. (a) Effect of different concentrations of drug-containing serum on HepG2 cell viability. (b) Intracellular TG and TC levels in HepG2 cells treated with SLBZP and PA. (c) Oil red O staining images of HepG2 cells treated with SLBZP and PA (×200).
SLBZP Decreased Intracellular Lipid Accumulation in PA-Induced HepG2 Cells
Significantly high intracellular TG and TC level was induced by an exposure to 300 μM PA for 24 h of HepG2 cells (Figure 11b), whereas in SLBZP-treated cells, TG and TC accumulation showed a significant decrease compared to that in PA-only treated cells. Oil red O staining showed that there was significant lipid droplet accumulation in PA-treated HepG2 cells, but SLBZP along with PA treatment,the lipid accumulation was significantly decreased compared to those for the PA-only treated cells (Figure 11c).
SLBZP Ameliorates Lipid Accumulation and Hepatic Steatosis in PA-Induced HepG2 Cells via the Regulation of AMPK/SREBP-1C-Dependent Signaling
The protein expression results are shown in Figure 12. Compared with the NC group, the expression of SREBP-1c protein was significantly higher in the PA group (

Changes in the levels of AMPK/SREBP-1c pathway-related proteins in HepG2 cells in each group.
Discussion
Lipid accumulation in the liver causes restriction of liver function and exacerbates metabolic imbalances in the liver, causing inflammatory cell infiltration and activation of liver stellate cells, leading to the formation of liver fibrosis and structural changes within the liver, contributing to the aggravation of MAFLD into cirrhosis or even liver cancer, this severely limits the lives and health of patients and increases the burden on society. Dyslipidemia is a risk factor for metabolism-associated fatty liver disease (MAFLD). 39 SLBZP significantly improves symptoms, restores liver function, and lowers blood lipids in patients with NAFLD. 17 The “enterohepaticcirculation” is a key part of the liver's metabolic function. According to the TCM, most of the functions of the small intestine, such as digestion and absorption, fall under the category of “the spleen is responsible for transport and transformation”. There is evidence that SLBZP improves fibrosis in the liver by regulating intestinal flora and inhibiting the expression of intestinal inflammatory cells. 40
Based on the herb-compound-target network, we found that the top 6 active compounds, quercetin, naringenin, kaempferol, luteolin, beta-carotene and nobiletin, inhibited adipogenesis and oxidative stress and prevented further development of MAFLD. Quercetin has been widely shown to be an antioxidant that alleviates lipid peroxidation. 41 kaempferol and luteolin inhibit the transcriptional activity of inflammatory factors.42,43 Hua YQ 44 et al demonstrated that naringin improved liver function in NASH mice by promoting the expression of genes related to lipid oxidation, inhibiting ROS production, and up regulating the activity of antioxidant enzymes. Naringenin could promote the expression of lipid oxidation-related genes, inhibit ROS production and up-regulate antioxidant enzyme activity in NASH mice, and alleviate lipid accumulation and oxidative stress in the liver. Supplementation with naringin significantly reduced serum lipid levels in patients with NAFLD. 45 Mitochondrial and metabolic adaptations are associated with hepatic UPR dysfunction, systemic oxidative stress and altered hepatic lipid metabolism. 46 It was shown that altered lipid profiles in patients with NAFLD are associated with reduced levels of beta-carotene. Nobiletin inhibits autophagy through activation of the IL-6/STAT3/FOXO3a signaling pathway 47 and promotes degradation of lipid droplets in hepatocytes. 48
The PPI network identified 9 genes, including ALB, TNF, IL1B, and ADIPOQ, as the core targets of SLBZP for the treatment of MAFLD, and the molecular docking results showed that they had a strong affinity for the key active compounds quercetin, kaempferol, lignocerol, and naringenin.Several clinical studies have shown42,49,50 that albumin levels are negatively correlated with the severity of MAFLD 42 and are the earliest indicator of liver function impairment. 49 Habib SA 51 et al revealed that increasing serum albumin levels significantly improved impaired liver function due to drugs. The presence of an inflammatory response is the main cause of exacerbation of MAFLD, 52 with excessive lipid accumulation leading to lipotoxicity and oxidative stress leading to inflammatory factor infiltration and damage to hepatocytes. TNF, IL10 and IL1B are important inflammatory factors which exert a cytotoxic effect in the body's immune damage. 53 The levels of TNF-α, considered a crucial cytokine in the evolution of NAFLD to NASH, are markedly increased in patients with NASH compared to those with NAFLD. 54 Inflammation induced by a high-fat diet can be suppressed and impaired liver function restored with the use of TNF antagonists. 55 The latest studies have shown that IL1B engaged in the development of obesity 56 and stimulates the expression of other pro-inflammatory factors, 57 playing a major role in initiation of inflammation and immune reaction. IL-1β also reduces the ability of insulin to respond to glucose, leading to IR. 58 As an anti-inflammatory cytokine, IL10 is negatively related to MAFLD. Knockdown of IL10 was shown to result in impaired inflammation and mitochondrial functional architecture in mice. 59 In contrast, overexpression of IL10 induced regulatory T cell expression, inhibited TNFα and IL1β production, ameliorated hepatic steatosis and fibrosis.60,61 ADIPOQ is shown to be an adipocytic factor capable of limiting the risk of obesity. 62 ADIPOQ expression is suppressed in both serum and liver of NAFLD patients. 63 In addition, inflammatory adipokines such as TNFα, IL10 and IL1B further exacerbate the reduction of ADIPOQ leading to liver fibrosis. 64 Therefore, ADIPOQ has also been used as a clinical indicator to predict the progression of MAFLD.65,66
GO enrichment results suggest that SLBZP regulates crossover targets involved in the response to lipopolysaccharide and inflammation, enzyme binding and cytokine activity for therapeutic action against MAFLD. KEGG enrichment results showed that cancer pathways, alcoholic liver disease (ALD) signaling pathways, lipid and atherosclerosis, and NAFLD signaling pathways were significantly enriched. The higher the number of crossover targets enriched for a pathway, the more important the pathway was. The pathways most relevant to MAFLD and enriched for a high number of crossover targets were the ALD signaling pathways and NAFLD signaling pathways, hitting 11 and 9 crossover targets, respectively. Mainly involved in AMPK signaling pathways, TNF signaling pathways and Adipocytokine signaling pathways, and their functions are focused on fatty acid anabolism and cellular inflammatory processes. AMPK is involved in maintaining cellular energy homeostasis and is also considered a prospective target for inhibition and therapy of MAFLD. 67 Activation of the AMPK signaling pathway can inhibit the activity of fatty acid synthesis factors, upregulate the expression of fatty acid oxidation factors such as PPARα 68 and improve lipid levels in patients with NAFLD. 69 The Adipocytokine signaling pathway inextricably linked to lipid metabolism and inflammatory reactions. Marinovic MP 70 and Choi H 71 et al demonstrated that knockdown of ADIPOQ exacerbated steatosis in the mouse liver and that overexpression of ADIPOQ reversed fat accumulation and inflammatory responses. TNFα promotes NF-κB activation, and NF-κB activation enhances the transcriptional process of inflammatory factors such as TNFα, further exacerbating the inflammatory response. Zhang R 72 et al treated NASH by inhibiting macrophage activation through inhibition of the TNF/NF-κB signaling pathway. Of interest is that the AMPK pathway was also significantly enriched individually, and AMPK has been recognized as a potential target for the prevention and treatment of MAFLD, capable of mediating multiple signaling cascades to treat MAFLD. In this study, by labeling the location of potential therapeutic targets in the AMPK pathway, we found that SLBZP may reduce adipogenic genes FAS and ACC and upregulate the expression of fatty acid oxidizing factors, such as PPARα, through the activation of AMPK and inhibition of its downstream transcription factor SREBP-1c into the nucleus.Meanwhile, molecular docking results also showed that both quercetin and naringenin, the active components of SLBZP, were able to spontaneously bind to AMPK and SREBP-1c, and the binding energies were < −5 kcal/mol. Through experimental results, SLBZP was shown to be effective in ameliorating lipid accumulation and hepatic steatosis in high-fat diet-fed rats and PA-induced HepG2 cells to prevent MAFLD.In cellular experiments, we demonstrated that SLBZP could promote AMPK and reduce the expression of SREBP-1c protein to treat MAFLD. Therefore, the mechanism of action of SLBZP in treating MAFLD may be to improve lipid metabolism disorders by activating the AMPK signaling pathway.
It should be noted that there are limitations in the current study. The use of network pharmacology, although an innovative strategy for generating hypotheses, tends to oversimplify large and complex biological systems and interactions. Consequently, this may lead to an exaggerated perception of SLBZP efficacy, ignoring the dynamics and complexity of human biological networks. Although this study is in line with the advantages of TCM's multi-target and multi-pathway treatment of diseases, it is difficult to fully Zen master its principles due to the variety of drugs in the formula and the complexity of its effectiveness, but this also happens to be the greatest advantage of TCM, which is never confined to the treatment of a single disease, but rather, it has a holistic view of the discernment of the evidence. And It is worth noting that the underlying mechanisms are certainly more complex than those described here. In addition, our results do not exclude that other signaling pathways and mechanisms may be involved in the inhibition of MAFLD by SLBZP. In this study, we have only explored a single mechanism of action of SLBZP at the cellular level, which needs to be explored from other aspects. In the future, MAFLD animal and cellular models will be used to validate the SLBZP components and further clarify its SLBZP on MAFLD mechanisms to be explored.
Conclusion
SLBZP is a formula that has been used by TCM for thousands of years to treat MAFLD. The mechanism of action involves multiple active ingredients, targets and signaling pathways, which is in line with the “multi-component, multi-target and multi-pathway” therapeutic characteristics of TCM. This study aimed to explore the therapeutic potential of SLBZP against MAFLD using a combination of network pharmacology and experimental validation. SLBZP effectively ameliorates lipid accumulation and hepatic steatosis in MAFLD, and the mechanism may be related to the AMPK signaling pathway. However, while our results provide a solid foundation for further exploration, the study's reliance on in vitro assays highlights the need for in vivo validation and clinical trials to fully elucidate the therapeutic potential of SLBZP in the clinical setting. Future studies should focus on validating these findings in vivo and exploring the clinical feasibility of SLBZP for the treatment of patients with MAFLD.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241306938 - Supplemental material for Potential Mechanisms for Shenling Baizhu Powder in the Treatment of MAFLD Based on Network Pharmacology and Experimental Validation
Supplemental material, sj-docx-1-npx-10.1177_1934578X241306938 for Potential Mechanisms for Shenling Baizhu Powder in the Treatment of MAFLD Based on Network Pharmacology and Experimental Validation by Anni Zheng, Sufei Song, Qiuling Xu and Tao Liu in Natural Product Communications
Footnotes
Acknowledgments
We are grateful for the platform provided by Hainan Medical University.
Author's Contributions
Tao Liu and Quling Xu supervised the study. Anni Zheng and Sufei Song designed and performed experiments. Sufei Song analyzed the data. Anni Zheng and Sufei Song wrote the original manuscript. Tao Liu and Quling Xu made manuscript revisions. All the authors review the manuscript. All authors read and approved the final manuscript.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Youth Qi Huang scholar project of state administration of traditional Chinese medicine, the National Natural Science Foundation of China, the Natural Science Foundation of Hainan Province, (grant number No. [2022] 256 of the National TCM Human Resource , no. 81760795, no.821RC579).
Ethical Approval
The study protocol was approved by the Ethics Committee of Hainan Medical College, approval number: HYLL-2021-109.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
Appendix
See Figure A1.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
