Abstract
Background
Scientific and ethnopharmacological studies have shown that Justicia insularis leaves are used in the treatment of oxidative stress, heart problems, labour pains, and dysmenorrhoea. Therefore, this study was geared towards the investigation of the antioxidant claims of this plant leaves as well as the isolation and characterization of the components responsible for its antioxidant activities.
Method
The leaves of the plant were collected, air-dried, pulverized, and subjected to successive extraction with n-hexane, dichloromethane, ethyl acetate, and methanol for 72 h; thereafter, the fractions were subjected to in vitro antioxidant assays. The dichloromethane fraction (10 g) that had the best activity underwent thin layer and column chromatography, and the eluents were characterized with spectroscopic techniques (1H-NMR, 13C-NMR, and EI-MS).
Results
The results obtained showed that the dichloromethane fraction had the best activity in all the assays except for the DPPH-radical scavenging assay. The chromatographic analyses yielded three oily sub-fractions (F1, F7, F8) and one pure compound (JID2) identified as lauryl alcohol (1-dodecanol). The GC-MS analysis of the oils revealed the presence of 1-Nonadecene, 1-Heneicosanol, Octacosanol, phytol, 1,54-dibromo-tetrapentacontane, 1-Eicosanol, hexadecanoic acid methyl ester, 2-methyltetracosane, and 2,4-Bis(1,1-dimethylethyl)-Phenol with already reported pharmacological activities.
Conclusion
The high levels of antioxidant activity seen in this study could be exclusively attributed to 1-dodecanol or to its beneficial interactions with other substances. (Word counts: 210)
Introduction
Justicia insularis T. Anders (Acanthaceae) is commonly called hunter's weed, Mmeme in Ibibio, and Isepe-akere and Esisi in Yoruba. It is a perennial herbaceous plant with opposed climbing branches that grows to a height of 30–75 cm. It has simple, opposite leaves and white, pink, or purple blooms 1 (Figure 1). The ethnomedicinal importance of this plant is well known as the phytochemical reports revealed its richness in phenolics, alkaloids, flavonoids and glycosides. 2 These phytochemicals are responsible for its diverse biological activities which include antioxidant activities; hence, it is beneficial therapeutically for health issues brought on by oxidative stress as it serves as a source of natural antioxidants. 3 This study was aimed at the evaluation and validation of the antioxidant activities of the plant, the isolation, and characterization of its antioxidant compounds.

Justicia insularis in its habitat.
Results
The Antioxidant Activities of the Plant Extract/Fractions/Isolated Compounds
The results (Figures 2–8) showed that the dichloromethane (DCM) fraction had the highest antioxidant activity, as observed from the ferric ion chelating activity (Figure 2), hydrogen peroxide scavenging activity (Figure 4), nitric oxide assay (Figure 5), and ferric ion reducing power (FRAP) (Figure 6) as compared with their various standards. Whereas methanol fraction showed the highest activity on the 1, 1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging activity (Figure 3). Moreover, the isolated compounds (lauryl alcohol) with the oils possessed significant antioxidant activities as compared with the standard drug (ascorbic acid) based on the DPPH-radical scavenging activity and ferric iron reducing power (FRAP) assays as shown in Figures 7 and 8, respectively.

Ferric ion chelating activity of Justicia insularis leaves. Values represent mean ± SEM; Values with different superscript compared to the control (ASA: Ascorbic acid) is significantly different (p < 0.001) (n = 3).
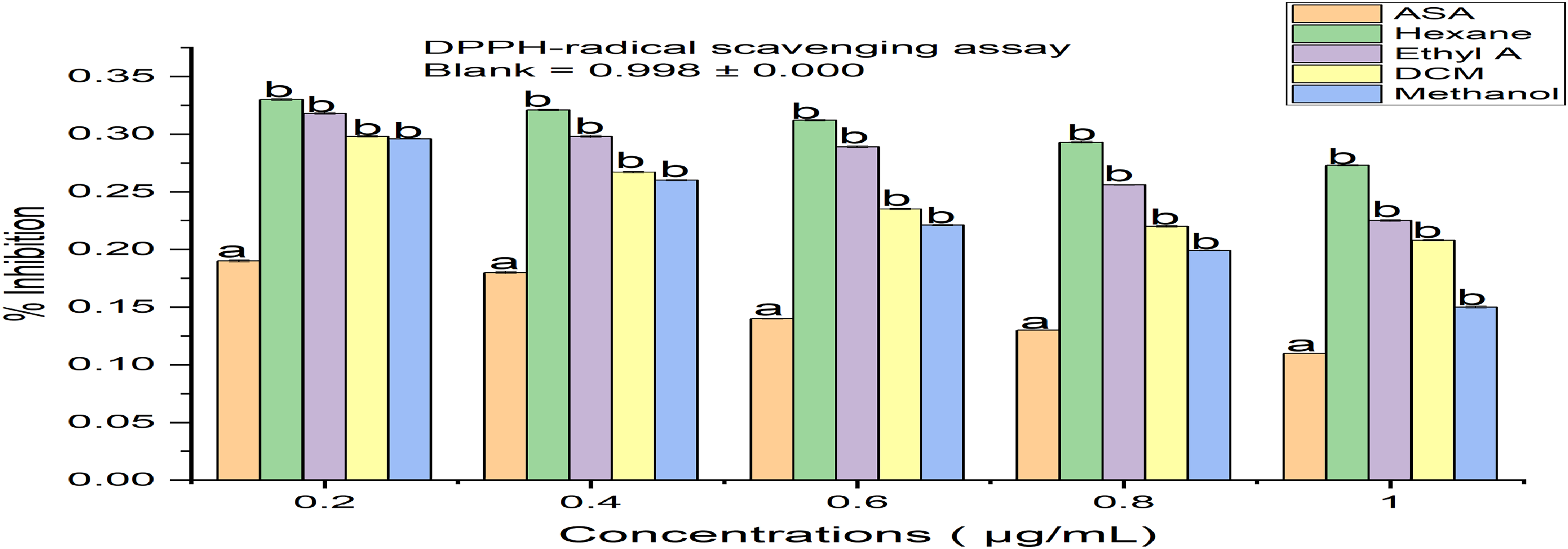
DPPH-radical scavenging activity of Justicia insularis leaves. Values represent mean ± SEM; Values with different superscript compared to the control (ASA: Ascorbic acid) is significantly different (p < 0.001) (n = 3).

Hydrogen peroxide scavenging activity of Justicia insularis leaves. Values represent mean ± SEM; Values with different superscript compared to the control (ASA: Ascorbic acid) is significantly different (p < 0.001) (n = 3).

Nitric oxide scavenging activity of Justicia insularis leaves. Values represent mean ± SEM; Values with different superscript compared to the control (ASA: Ascorbic acid) is significantly different (p < 0.001) (n = 3).

Ferrous ion reducing power (FRAP) of Justicia insularis leaves. Values represent mean ± SEM; Values with different superscript compared to the control (ASA: Ascorbic acid) is significantly different (p < 0.001) (n = 3).
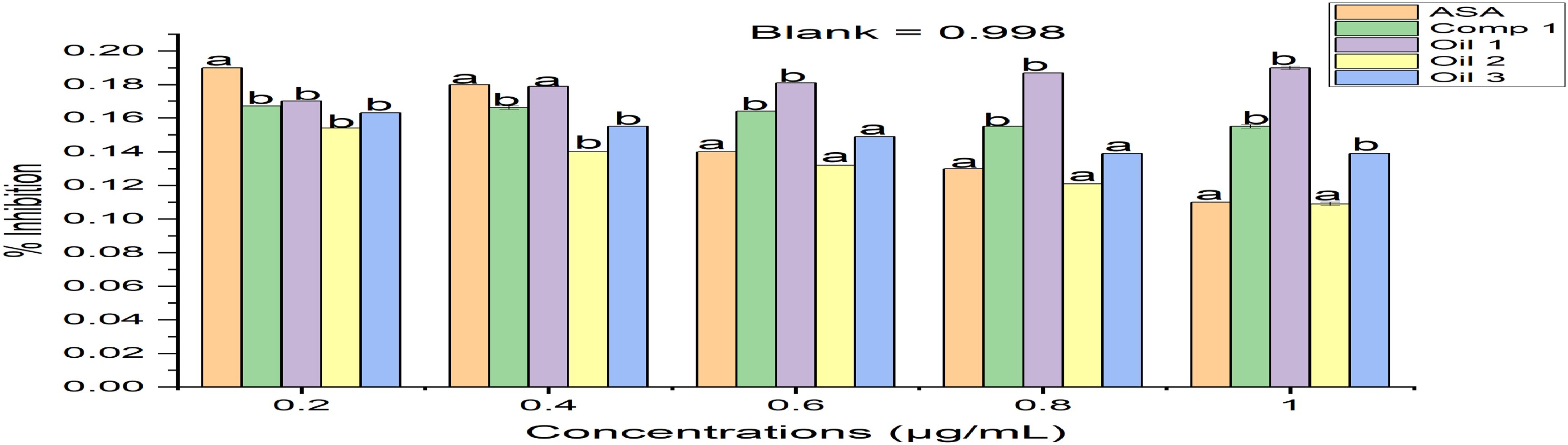
DPPH-radical scavenging activity of isolated compound JID2 (lauryl alcohol) and oils (sub-fractions F1, F7, F8) from DCM fraction of Justicia insularis leaves. Values represent mean ± SEM; Values with different superscript compared to the control (ASA: Ascorbic acid) is significantly different (p < 0.001) (n = 3).

FRAP assay for isolated compound JID2 and oils (sub-fractions F1, F7, F8) from DCM fraction of Justicia insularis leaves. Values represent mean ± SEM; Values with different superscript compared to the control (ASA: Ascorbic acid) is significantly different (p < 0.001) (n = 3).
NMR Spectral Presentation for Compound JID2 and GC-MS of Oil
Figure 9 are the various NMR spectra of compound JID2 whereas its 13C-NMR spectral characteristics is presented in Table 1. However, Figure 10 revealed the chemical structure and formula of the isolated compound JID2 (1-dodecanol). Spectroscopic analysis (Tables 2–4, and Figures 11–13) through GCMS revealed the total ion chromatogram and chemical constituents in the oil 1, 2, and 3 to include but not limited to phytol, 1-Nonadecene, 1-Heneicosanol, Octacosanol, 1,54-dibromo-tetrapentacontane, 1-Eicosanol, hexadecanoic acid methyl ester, 2-methyltetracosane and 2,4-bis (1,1-dimethyl ethyl)-phenol.

NMR Spectra of compound JID2.

Chemical structure of isolated compound JID2 (1-dodecanol/lauryl alcohol).
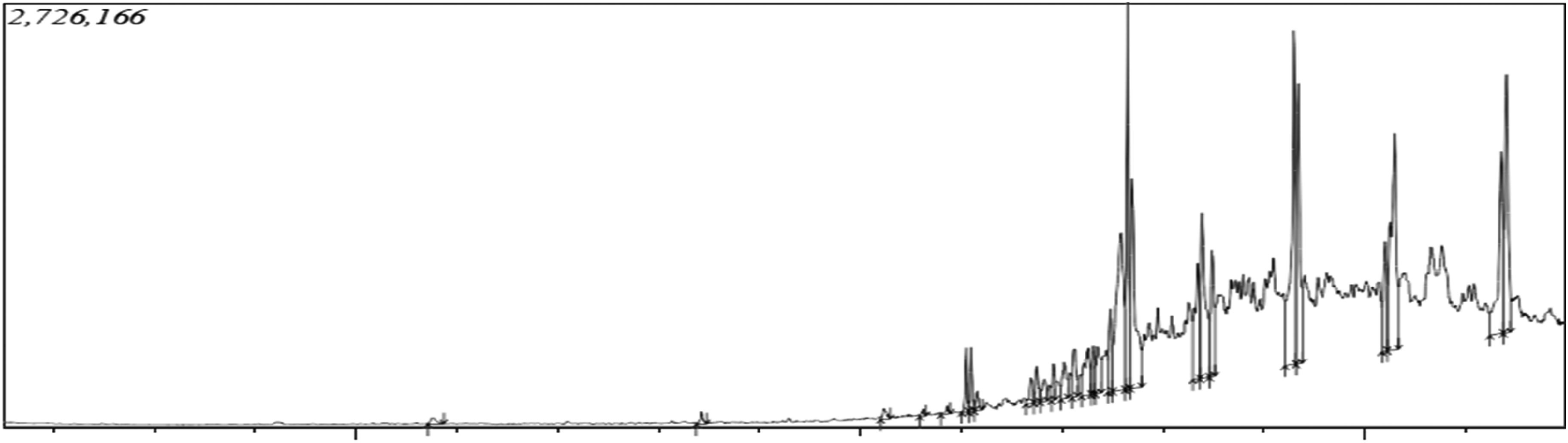
GC-MS analysis revealing total ion chromatogram (TIC) of oil 1 (sub-fraction F1).

GC-MS analysis revealing total ion chromatogram (TIC) of oil 2 (sub-fraction F7).

GC-MS analysis revealing total ion chromatogram (TIC) of oil 3 (sub-fraction F8).
Compound JID2 13C-NMR Spectral Characteristics.

GC-MS Analysis of Oil 1 (sub-Fraction F1).
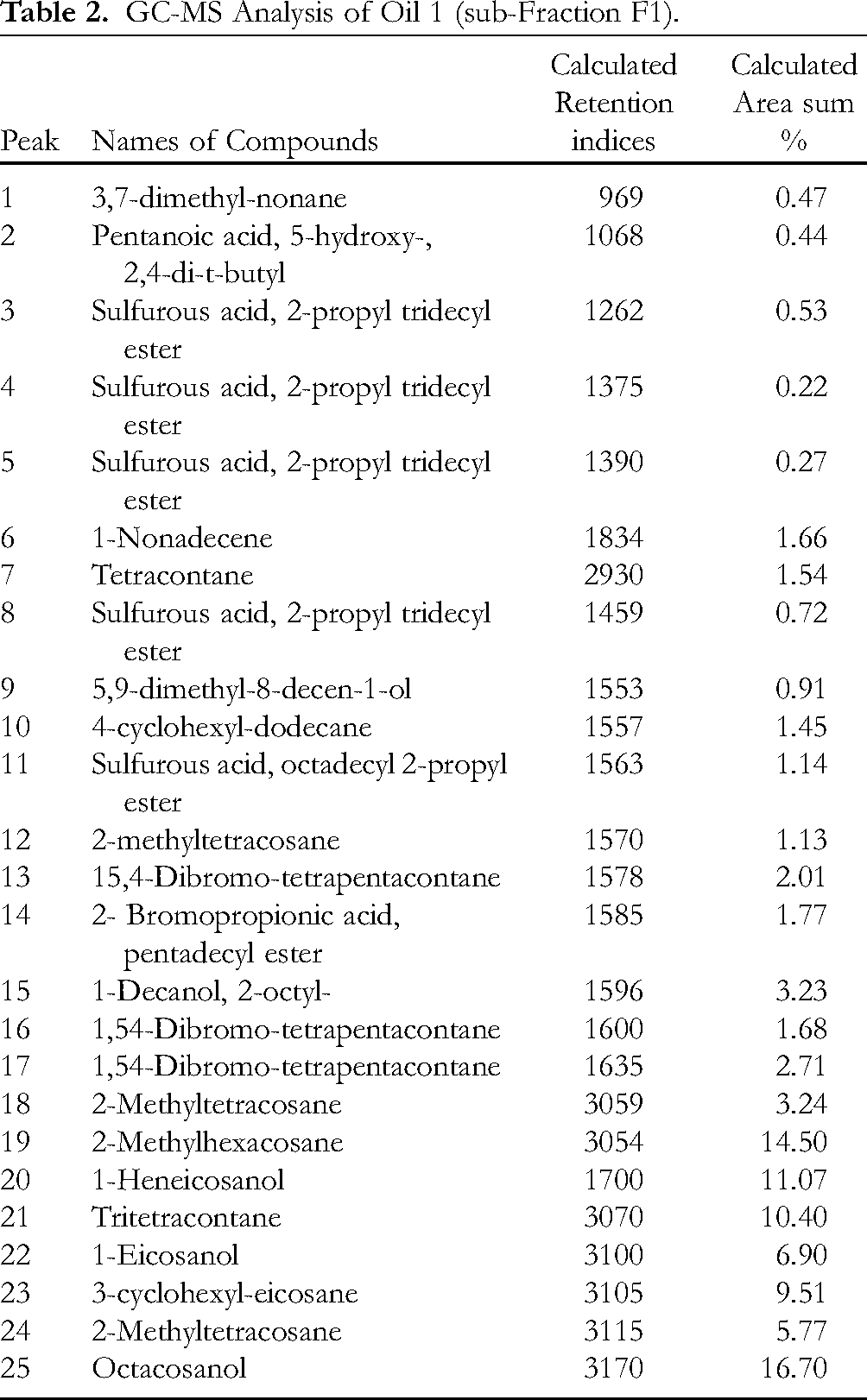
GC-MS Analysis for Oil 2 (sub-Fraction F7).

GC-MS Analysis for Oil 3 (sub-Fraction F8).

Discussion
The generation of an abundance of reactive oxygen species (ROS) can result in oxidative stress, contributing to various health issues such as hypertension, diabetes, arthritis, and cancer 5 ; however, antioxidants (exogenous and endogenous) protect the body from oxidative stress and may help to combat oxidative damages. 6 It's important to highlight that plants serve as abundant reservoirs of natural antioxidants. Consequently, these antioxidants enhance the plasma's antioxidation capability; thereby, lowering the susceptibility to some diseases like cancer, heart disease, and stroke. 7 The result obtained from the antioxidant activity assay showed that all the fractions had substantive activity. Moreover, the dichloromethane fraction in all the assays had the best activity except for the DPPH-radical scavenging assay, where the DPPH-radical scavenging activity decreased as the concentrations were increased as compared to other fractions; their order of activity were as follows: methanol > dichloromethane > ethyl acetate > n-hexane. Similarly, the isolated compound JID2 (1-dodecanol) revealed significant free radical scavenging activity that compared favourably with aspirin. However, oil 2 (sub-fraction F7, yellowish oil) revealed the best DPPH-radical scavenging activity when compared with other oils in the order as follows: oil 2 > oil 3 >oil 1. The FRAP assay showed that the lauryl alcohol and the three oils in a concentration-dependent manner had increased reducing ability, with oil 3 (sub-fraction F8) being the best, and their order of reducing activity was as follows: oil 3 > oil 2 > oil 1.
Moreover, the GC-MS analysis of the various oils indicated the presence of 30, 16, and 28 bioactive compounds. They include but not limited to the following compounds with already reported pharmacological and biological activities; 1-Nonadecene (antifungal and anticancer activity), 1-Heneicosanol, and Octacosanol (anticancer activity, hypocholesterolemic agent, anticoagulant activity, energy boosting agent), 7 phytol (antimicrobial agent), 1,54-dibromo-tetrapentacontane (anticancer activity), 1-eicosanol, hexadecanoic acid methyl ester, 2-methyltetracosane, and 2,4-bis(1,1-dimethylethyl)-phenol are used as antioxidants. 8
Furthermore, the isolated compound JID2 from sub-fraction F6c is a saturated unbranched alcohol with molecular weight (186.34 g/mol), melting point (24 °C), boiling point (256 °C), and chemical formula (C12H25OH). Moreover, the spectral data obtained from 13C-NMR (DEPT-135, DEPT-45, and DEPT-90) indicated the presence of 12 carbon atoms, comprising one methyl, and eleven methylene atoms. The analysis also revealed the existence of single bonds, along with one hydroxyl group in the molecule. The 1H-NMR (COSY, NOESY, HSQC, and HMBC) spectra showed a singlet methyl and a carbinolic proton with an endo-linear methylene group and the EI-MS fragmentation pattern displayed characteristics consistent with lauryl alcohol, featuring a base peak at 55 m/z. Based on these comprehensive spectral findings, the compound JID2 was identified as 1-dodecanol (Lauryl alcohol). In addition, I-dodecanol is reported for the first time to possess antioxidant activities, but some researchers have reported that the compound has a floral odour and is used in detergents, lubricating oil, and pharmaceuticals. 9
Conclusion
Oxidative stress is known to aggravate cardiovascular disorders due to the generation of free radicals; thereby, promoting the development of many non-communicable diseases (NCDs) and necessitating drug research on bio-activity fractionation, isolation, and characterization of the antioxidant constituents in the leaves of J. insularis. The plant leaves extracts/isolated compounds were bio-actively screened through in vitro antioxidant studies, whereas the DCM fraction with the most favourable therapeutic indices underwent column chromatography. The eluents (sub-fractions F1, F7, and F8) characterized with GC-MS were oils with characteristic colours and good antioxidant activities. However, sub-fraction F6c being the isolated pure compound JID2 underwent characterization, which was conducted through the use of spectroscopic techniques (1H, 13C-NMR, and EI-MS) and identified as 1-dodecanol (Lauryl alcohol). The isolation of 1-dodecanol (Lauryl alcohol) from the J. insularis leaf is reported here for the first time and the saturated unbranched alcohol has never been reported to have antioxidant activities. Therefore, this study's high antioxidant activities may be solely due to 1-dodecanol or its synergistic interaction with other compounds.
Limitations
Few samples and methodology in the antioxidant studies because of financial constraints.
Unreliability of some data as the in vitro studies may be unreliable as compared to in vivo studies.
Inaccessibility of the NMR and EI-MS equipment that caused impurities to the samples on transit.
Inadequate prior research studies on isolation of the compounds based on our study design
List of Abbreviations
GC-MS Gas chromatography mass spectrometry
NMR Nuclear magnetic resonance
EI-MS Electron impact mass spectrometry
DCM Dichloromethane
CC Column chromatography
TLC Thin layer chromatography
ROS Reactive oxygen species
NCD Non communicable disease
COSY Correlation spectroscopy
FRAP Ferric reducing antioxidant power
DPPH 1, 1-diphenyl-2-picrylhydrazyl
Experimental (Materials and Methods)
Ethics Approval
The study was approved by the Akwa Ibom State Educational Research Board, Akwa Ibom State, Nigeria.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Collection and Identification of Plant Material
The plant Justicia insularis was collected in November 2022 from a location in Itiam/Ewet Housing Estate, Uyo Local Government Area, Akwa Ibom State, Nigeria. It was identified by Professor (Mrs.) U. A. Essiett, a taxonomist in the Department of Botany and Ecological Studies, Faculty of Science, University of Uyo. The Herbarium Specimen with the Voucher Number UUPH1C was deposited in the Department of Pharmacognosy and Natural Medicine, Faculty of Pharmacy, University of Uyo Herbarium.
Preparation and Extraction of Plant Material
The leaves of the plant were collected, air-dried, and pulverized using an electric blender. The powdered leaves (601.96 g) were subjected to successive extraction with n-hexane (5 L), dichloromethane (4 L), ethyl-acetate (3.5 L), and methanol (4 L) at room temperature for 72 h, respectively; thereafter, the fractions were filtered and concentrated to dryness in a water bath at 30 °C. The resultant extract/fractions were weighed and their percentage yields were calculated as follows: (21.11 g) of n-hexane fraction (3.51%), (13.23 g) of dichloromethane fraction (2.20%), (5.26 g) of ethyl acetate fraction (1.01%), and (62.34 g) of methanol fraction (10.36%). These extracts/fractions were stored in a refrigerator at 4 °C until required for use.
Evaluation of Antioxidant Activities of the Fractions
The methods described by Shekhar and Anju 10 ; Shekhar and Anju 10 ; Oyaizu 11 ; Ruch et al; 12 Marcocci et al 13 ; were used to measure the ferric ion chelating activity, DPPH-radical scavenging activity, ferrous ion reducing antioxidant power, H2O2 scavenging activity, and nitric oxide radical scavenging assay, respectively.
Chromatographic Analysis
After being analysed by column chromatography using silica gel (60-120 mesh) and eluted with the suitable solvent systems (n-hexane, dichloromethane, ethyl acetate, and methanol), the dichloromethane fraction (10 g) with the highest antioxidant activity produced 93 fractions. Based on similarities in Rf values, these fractions were subjected to two distinct rounds of thin layer chromatography (TLC) analysis (the pooling and bulking processes), yielding twenty-six (26) and nine (9) bulked fractions, respectively. Eight (8) sub-fractions (F1-F8) were obtained by subjecting bulk fraction 6 (A31-51) (0.82 g) labelled F to further column chromatography (CC) using silica gel and eluting it with the proper solvent systems. The oils with the following characteristic colours were recognized as sub-fractions: sub-fraction F1 (colorless), sub-fraction F7 (yellow), and sub-fraction F8 (blackish yellow). In order to produce six sub-fractions (F6a-F6f), sub-fraction F6 was additionally analysed by column chromatography employing Sephadex gel and washed with the proper solvent systems. Compound JID2 as designated from the sub-fraction F6c (0.4 g, a pure white powder), which appeared as a single spot on the surface of the TLC plate (using different solvent systems).
General Procedures
An Avance AV-400 MHz spectrometer was used to perform 1H and 13C-NMR (nuclear magnetic resonance) spectra. Trimethyl silane was used as the referential (δH = δC = 0), with coupling constants in Hertz, and dimethyl sulfoxide (DMSO)-d6 improved the breakdown of sub-fraction F6c (JID2). Electron ionization mass spectrometer (EI-MS) (positive mode) improved mass spectrometry quantitation. Using analytical already coated TLC silica gel 60 from Sigma-Aldrich, thin-layer chromatography was performed and seen under UV illumination with both long (366 nm) and short (254 nm) wavelengths. Additionally, Sephadex LH-20 and Sigma-Aldrich's silica gel Kiesel gel 60 (200-400 mesh, Merck) improved column chromatography (CC) evaluation. 14
Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
The oil samples (F1, F7, and F8) were analyzed using Shimadzu GC-MS equipment. The analysis was carried out using a Clarus 500 Perkin Elmer gas chromatograph equipped with an Elite-5 capillary column (30 nm × 0.25 mm ID × 0.25 m df) containing 5% phenyl and 95% dimethyl polysiloxane. 22,4-trimethylpentane was used for the stabilization. The mass detector turbo-mass gold of the company was operated in EI mode. The injector temperature was set at 290 °C, and the oven temperature was controlled as follows: starting at 50 °C and increasing at a rate of 8 °C/min until it reached 200 °C, then increasing at a rate of 7 °C/min until it reached 290 °C and held for 10 min. Helium was used as the medium for the gas at a flow rate of 1 ml/min. The mass spectrum was interpreted using the National Institute of Standards and Technology (NIST) database, which contains over 62,000 patterns. The constituents were identified based on their calculated retention indices and their representation are shown as calculated % of the area sum obtained from the mass spectra of unknown compounds and those of known compounds stored in the library. 15
Statistical Analysis
Using the Statistical Package for Social Sciences (SPSS) 17.0, the data obtained from antioxidant assays were analyzed using a One-way Analysis of Variance (ANOVA) and reported as mean ± SEM. A confidence level of p < 0.05 was deemed significant.
Footnotes
Acknowledgment
We extend our gratitude for the dedication and technical support provided by Mr Nsikan Malachy Department of Pharmacology and Toxicology, University of Uyo, Nigeria.
Consent for Publication
Not applicable.
Data Availability Statement
All datasets generated in this study are available from the corresponding author on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The experimental protocol was strictly followed and approved by the Akwa Ibom State Research Board, Uyo, Nigeria.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
The experimental procedures neither involve human beings nor animals; therefore, no guidelines, rights, nor approval was sort in this regards.
Statement of Informed Consent
Human subjects were not involved in this study; therefore, informed consent is not applicable.
