Abstract
Introduction
Asthma is a prevalent respiratory disease characterized by chronic airway inflammation. The global prevalence of adult asthma is about 4.4%, with significant variability across different countries and regions. 1 Notably, the prevalence of asthma in the grassland areas surrounding northern China reaches as high as 9.8%. 2 Annually, asthma imposes a global economic burden of 3 billion US dollars, 3 and in China, the average asthma patient incurs healthcare costs of approximately $300. 4 The clinical manifestations of asthma typically encompass dyspnea, wheezing, coughing, sputum production, exacerbation tendencies, and lung function decline. 5 Recent immunological studies have categorized asthma into “type 2-high” and “type 2-low”, with allergic asthma belonging to the former. 6 Type 2-high asthma is characterized by eosinophilia, mucus hypersecretion, bronchial hyperresponsiveness, Immunoglobulin (Ig) E production, and a susceptibility to exacerbations. Conversely, type 2-low asthma is more closely associated with obesity, neutrophil presence, and corticosteroid unresponsiveness. Research indicates that 50% to 70% of patients with mild to moderate asthma exhibit abnormalities in type 2 immune responses. 7 Based on the level of disease control, treatment options for asthmatics encompass inhaled corticosteroids, β2-agonists, muscarinic receptor antagonists, leukotriene receptor antagonists, and anti-IgE antibodies. 8 The objectives of asthma management are to achieve optimal symptom control and minimize the future risk of asthma-related mortality. However, current management practices face challenges such as inadequate control rates, diverse adverse drug reactions, substantial economic burdens, and non-standard use of inhalation devices.9,10
Traditional Chinese Medicine (TCM) is an ancient and effective complementary and alternative medical therapy that exhibits multi-level therapeutic effects on asthma. A large retrospective study revealed that, compared to non-TCM asthmatics, TCM patients experienced a significant reduction in emergency visits and hospital admissions. 11 In the clinic, certain TCM formulations have been employed as substitutes in the treatment of asthma. For instance, Pingchuan Yiqi granules are capable of remarkably improving lung function and alleviating symptoms of acute asthma, while partially reducing the dosage of short-acting β2 agonists (SABAs). 12 Wumeiwan serves as an alternative therapy for steroid-dependent asthma, reducing airway inflammation, and its mechanism may be linked to the Interleukin (IL)-17 and mitogen-activated protein kinase (MAPK) signaling pathways. 13 Dingchuan decoction can mitigate inflammatory pathological damage to the lung and airway, modulate lung microbiota, and effectively reduce the levels of multiple inflammatory factors, while inhibiting the expression of Toll-like receptor (TLR) 4 mRNA in the lung tissue of asthmatic mice. 14 Xiaoqinglong decoction plays a crucial role in alleviating lung inflammation and asthma, potentially by regulating the thymic stromal lymphopoietin (TSLP) signaling pathway. 15
SZG is comprised of ten herbal medicines, and its specific composition is detailed in the Traditional Chinese medicine materials section. Previous studies conducted by our research group have indicated that SZG can improve lung function and inhibit chronic airway inflammation in a rat model of cough variant asthma (CVA) by modulating the expression of IκBα/NF-κB in lung tissue.. 16 Clinical randomized controlled trials have demonstrated that SZG can significantly alleviate and reduce clinical cough symptoms in patients with CVA, improve clinical efficacy and quality of life, and have a high safety profile. Additionally, SZG exhibits certain long-term therapeutic effects, making it suitable for widespread clinical application. 17 Although the clinical efficacy of SZG has been demonstrated, the underlying molecular mechanisms remain poorly understood and require further investigation due to the complexity and diversity of its composition. UPLC-MS/MS is a powerful analytical technique widely used for the identification and quantification of bioactive compounds in complex herbal formulations. 18 This method excels in separating and identifying compounds based on their retention times and mass spectrometric signatures, allowing for precise profiling of complex mixtures. Network pharmacology systematically predicts the interconnected relationships between the identified components and their potential biological targets, providing insights into the pharmacological effects of these formulas. 19 Molecular docking, as a computational approach, further aids in visualizing the interactions between active compounds and target proteins at the molecular level, offering a basis for hypothesis-driven validation. 20
In this study, we applied a combination of UPLC-MS/MS, network pharmacology, and molecular docking techniques to investigate the active components of SZG and their potential molecular mechanisms in treating allergic asthma. This integrated approach significantly enhances the exploration of complex herbal formulations, deepening our understanding of their therapeutic effects.
Results
The Active Ingredient Absorbed into the Blood
Through the utilization of UPLC-MS/MS, 70 components absorbed into the serum of SZG were detected. Detailed information on these components is provided in Schedule 1 of the Supplemental Materials, and the corresponding mass spectra are presented in Figure 1, Additional active ingredients are shown in Figure S1 in Supplemental Material. 28 active compounds were identified and screened based on intestinal absorption, compliance with Lipinski's Rule of Five, and target probabilities greater than 0 as predicted by the SwissTargetPrediction platform. The blood-absorbed prototype components of SZG were retrieved from the Marvin JS database (https://marvinjs-demo.chemaxon.com/). Additionally, the metabolic processes of these metabolites were thoroughly analyzed. Figure 2 illustrates the structure and metabolism of some of these components, while Figure S2 in the Supplemental Material provides the structure and metabolism diagrams of all blood-absorbed components that possess target proteins related to allergic asthma.
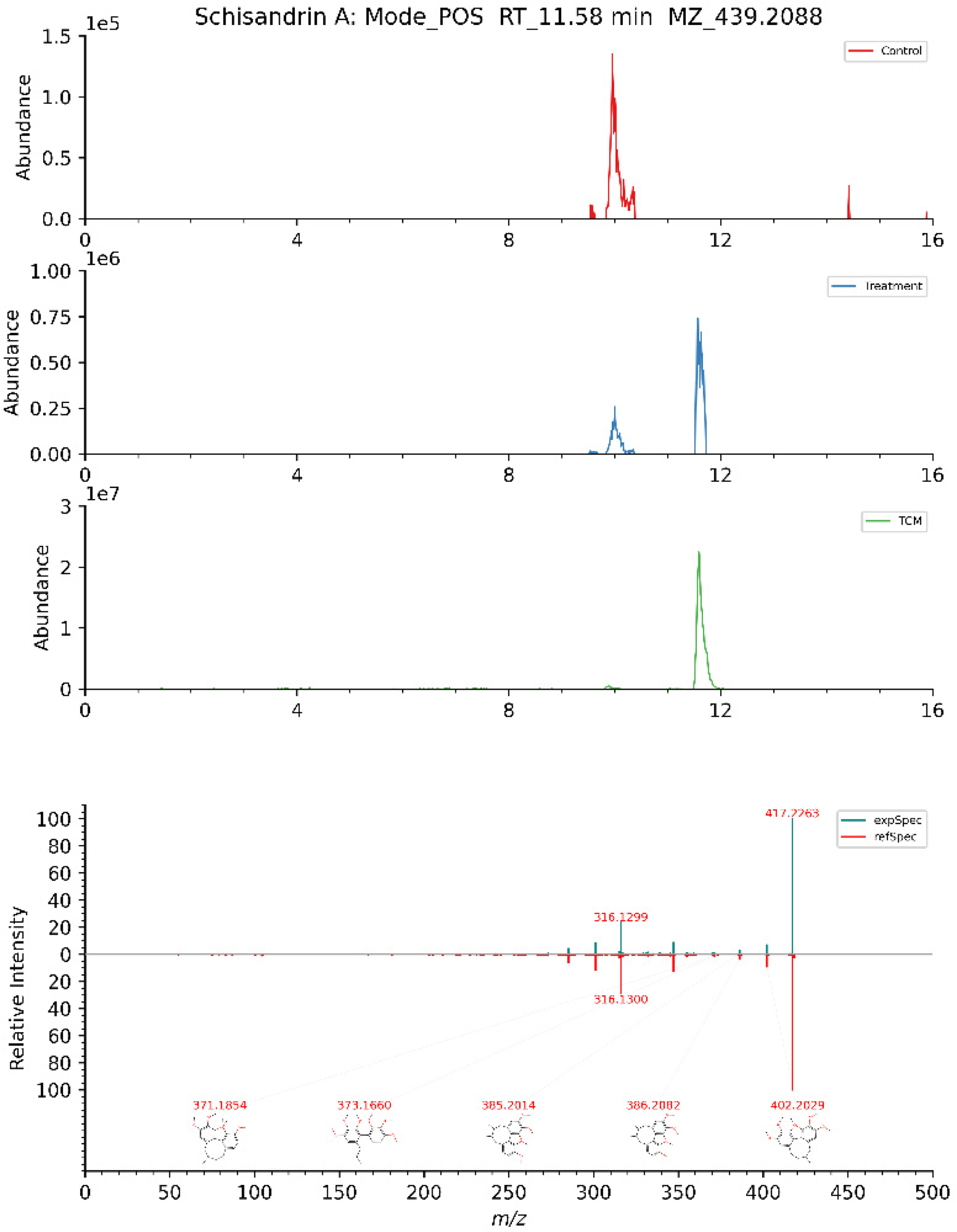
Secondary mass spectra of schisandrin A. Control: Blank serum sample. Treatment: Serum samples after SZG gavage. TCM: Sample of SZG. expSpec: Experiment Specification. refSpec: Reference Sepcification. RT: Retention time (min). MZ: Measured mass-to-charge ratio (m/z).

Examples of molecular structure diagram and metabolic diagram of components. (A) Molecular structure of Schisandrin A. (B) Magnoflorine_metabolite (M) 1 formed after demethylation of magnoflorine. (C) Arglabin_M1was formed after reduction and hydroxylation of arglabin.
SZG Against Allergic Asthma Targets Identification
Upon integration and statistical analysis of open-source databases, 584 potential targets were identified in the blood components of SZG. 826 targets related to allergic asthma were collected from DrugBank (2 genes/proteins), GeneCards (694 genes/proteins), NCBI Gene (139 genes/proteins), OMIM (88 genes/proteins), and PharmGkb (100 genes/proteins). The merged targets revealed 113 intersectional molecules between SZG and allergic asthma. Furthermore, 22 active ingredients were identified as sharing common targets with allergic asthma, and their specific information is presented in Table 1.
Information of 22 Active Ingredients.

PPI Network and the Pivotal Targets
Further analysis and computation were conducted on the 113 shared targets between SZG and allergic asthma using the String database. The results were then imported into CytoNCA software for network construction, centralization, and visualization. Isolated nodes were removed due to their lack of intersection with the target genes. This process resulted in a protein-protein interaction (PPI) network composed of 97 nodes and 456 interactions (edges) among the remaining nodes (see Figure 3A). The PPI network underwent two rounds of filtering using R software, and 13 targets with values greater than the median were selected as the pivotal targets (see Figure 3B) after screening. In the PPI network, the nodes represent proteins, while the edges represent the interactions between these proteins.

(A) The compounds-disease intersecting target PPI network, with yellow nodes representing the core targets identified after two rounds of screening. (B) PPI network of 13 pivotal targets.
GO and KEGG Pathway Enrichment
Through GO function enrichment analysis, 2122 Biological Process (BP) terms with statistical significance (P-value < .05) were obtained, including regulation of inflammatory response in the circulatory system, vascular processes, leukocyte migration, and peptidyl-tyrosine phosphorylation. Additionally, 65 Cellular Component (CC) terms were identified, covering membrane microdomain plasma membrane raft, plasma membrane signaling receptor complex, and vesicle lumen. 127 Molecular Function (MF) terms were also detected, such as non-membrane spanning protein tyrosine kinase activity, protein serine/threonine kinase activity, and MAP kinase activity. The top 10 items for each of BP, CC, and MF were visualized (see Figure 4A).

Enrichment analysis results of SZG against asthma according to adjusted P-value and counts screening. (A) Results of GO Ontology enrichment analysis. (B) The top 30 pathways were visualized by KEGG enrichment analysis.
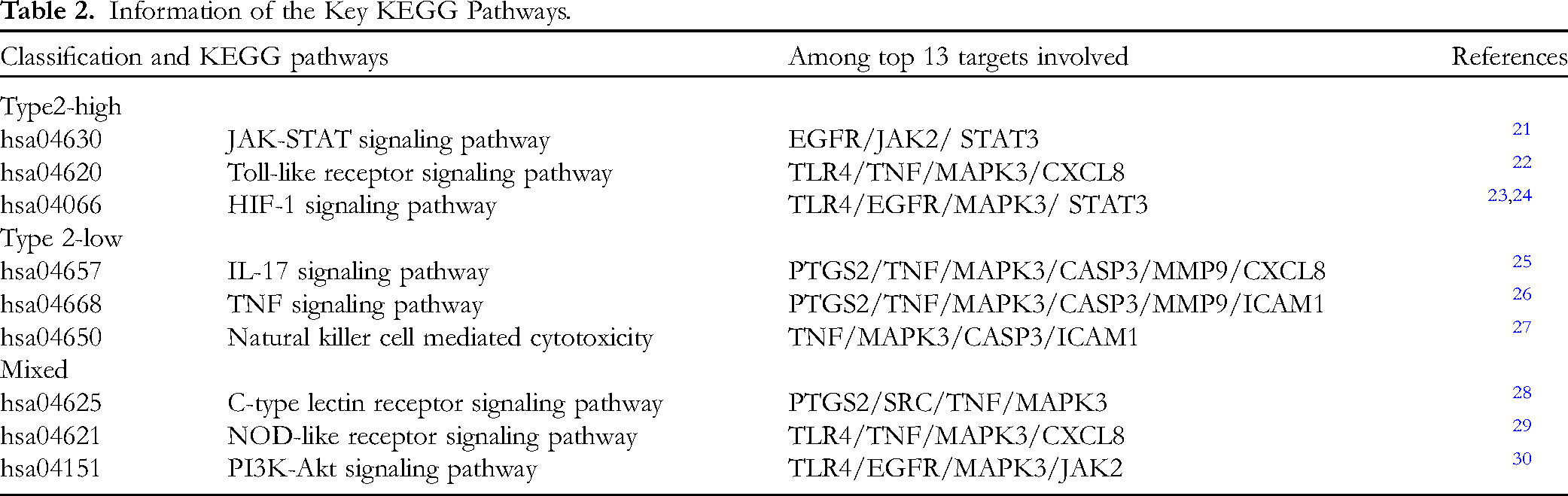
Through KEGG pathway analysis, 163 pathways were identified (P-value < .05). The top 10 pathways were subsequently visualized (see Figure 4B). 36 signal pathways were determined to be associated with asthma following literatures (see Schedule 2 in the Supplemental Material), encompassing signal pathways related to the phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) signaling pathway, IL-17 signaling pathway, and NOD-like receptor (NLR) signaling pathway. Based on the available data, we identified the cell types associated with asthma within each of these pathways. The pathways were subsequently categorized as type 2-high, type 2-low, or mixed asthma based on the diverse cell types they involved (see Table 2).
Information of the Key KEGG Pathways.
“Compound-Target-Pathway-Cell Type” Network Analysis
Based on the above results, Excel was employed to generate the type file and network file for the “Compound-Target-Pathway-Cell Type” network.The “Compound-Target-Pathway-Cell Type” network was constructed using Cytoscape 3.8.0 software (see Figure 5). The top 10 core active components were identified based on the number of matching disease targets, which include (25R)-12alpha-Hydroxyspirost-4-en-3-one_M1, 24-Hydroxyglycyrrhetic acid, arglabin_M1, combretastatin A4_M1, curdione_M1, magnoflorine_M1, neocurdione_M1, phaeocaulisin E_M1, schisandrin A, and toddaculin_M1.
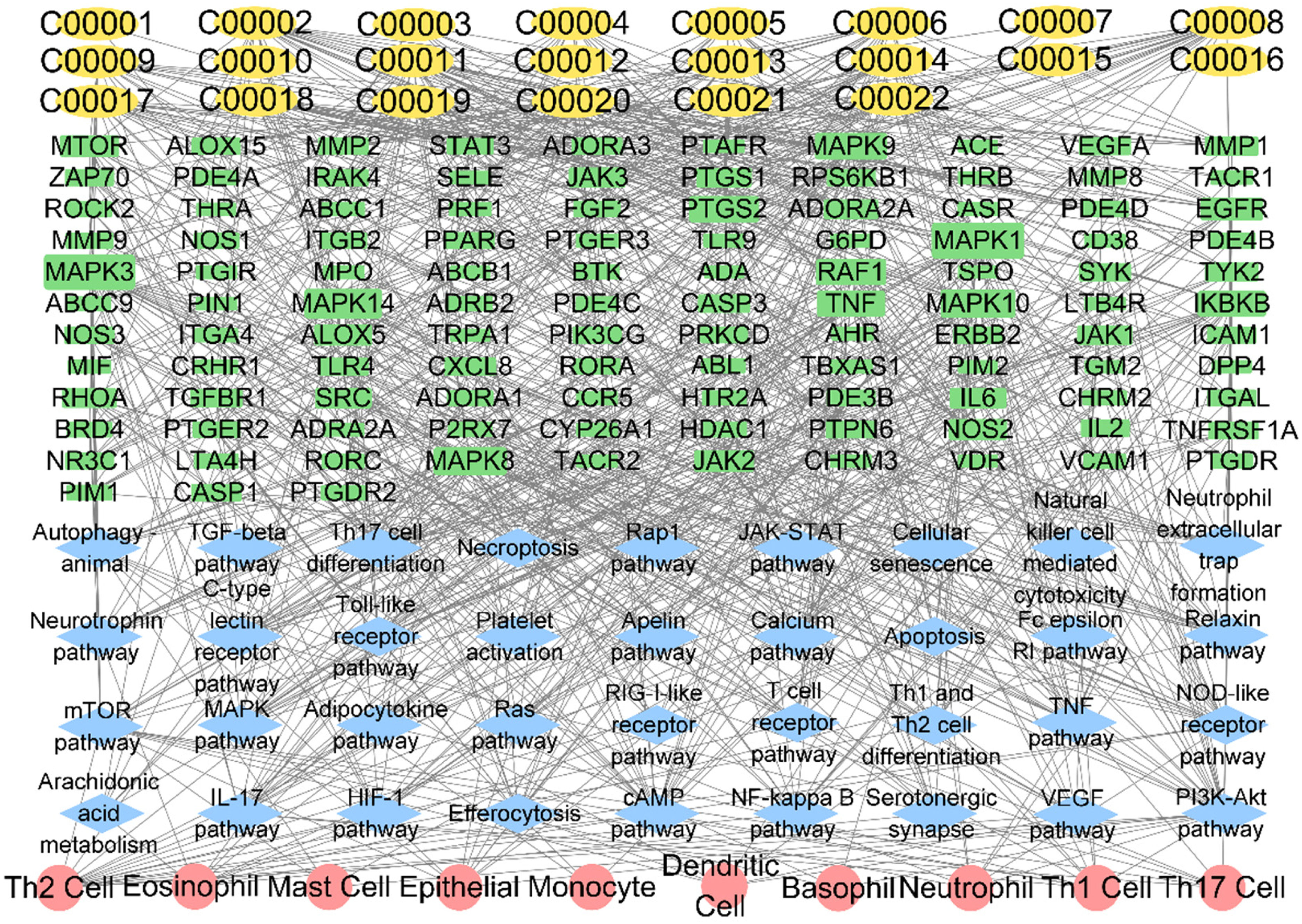
“Compound-target-pathway-cell type” network. C00001 (compound00001): 3-O-Caffeoylquinic acid methyl. The other sorted components are the same as Table 1.
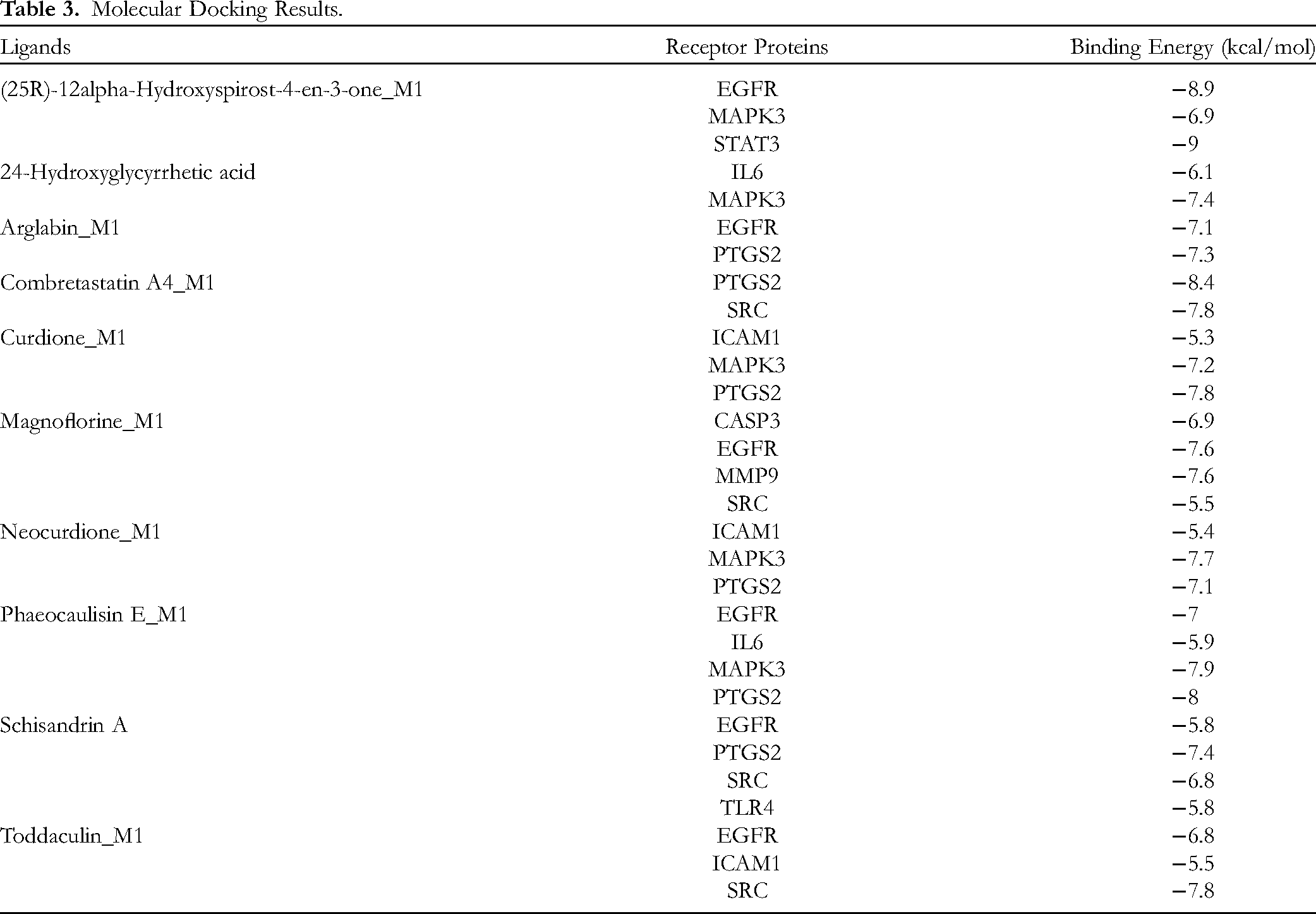
Molecular Docking of top 13 Critical Targets and key Compounds of SZG
Molecular docking was conducted on 13 pivotal targets and 10 core components to validate the findings of the network analysis. Their binding affinity (kJ/mol) was used to assess the extent of ligand binding to the receptor protein (see Table 3). A binding energy of ≤ -5.0 kJ/mol is considered indicative of good binding affinity between the ligand and receptor. Among these targets, Janus kinase (JAK) 2, C-X-C motif chemokine ligand (CXCL) 8, and TNF exhibited a binding energy of >-5.0 kJ/mol with their respective core components, indicating poor binding affinity. Conversely, the remaining core targets and compounds displayed superior binding affinity. Figure 6 depicts the specific images of the simulated docking between (25R)-12alpha-Hydroxyspirost-4-en-3-one_M1 and its receptor proteins. Additional simulated docking conditions are presented in Figure S3 in the Supplemental Material.
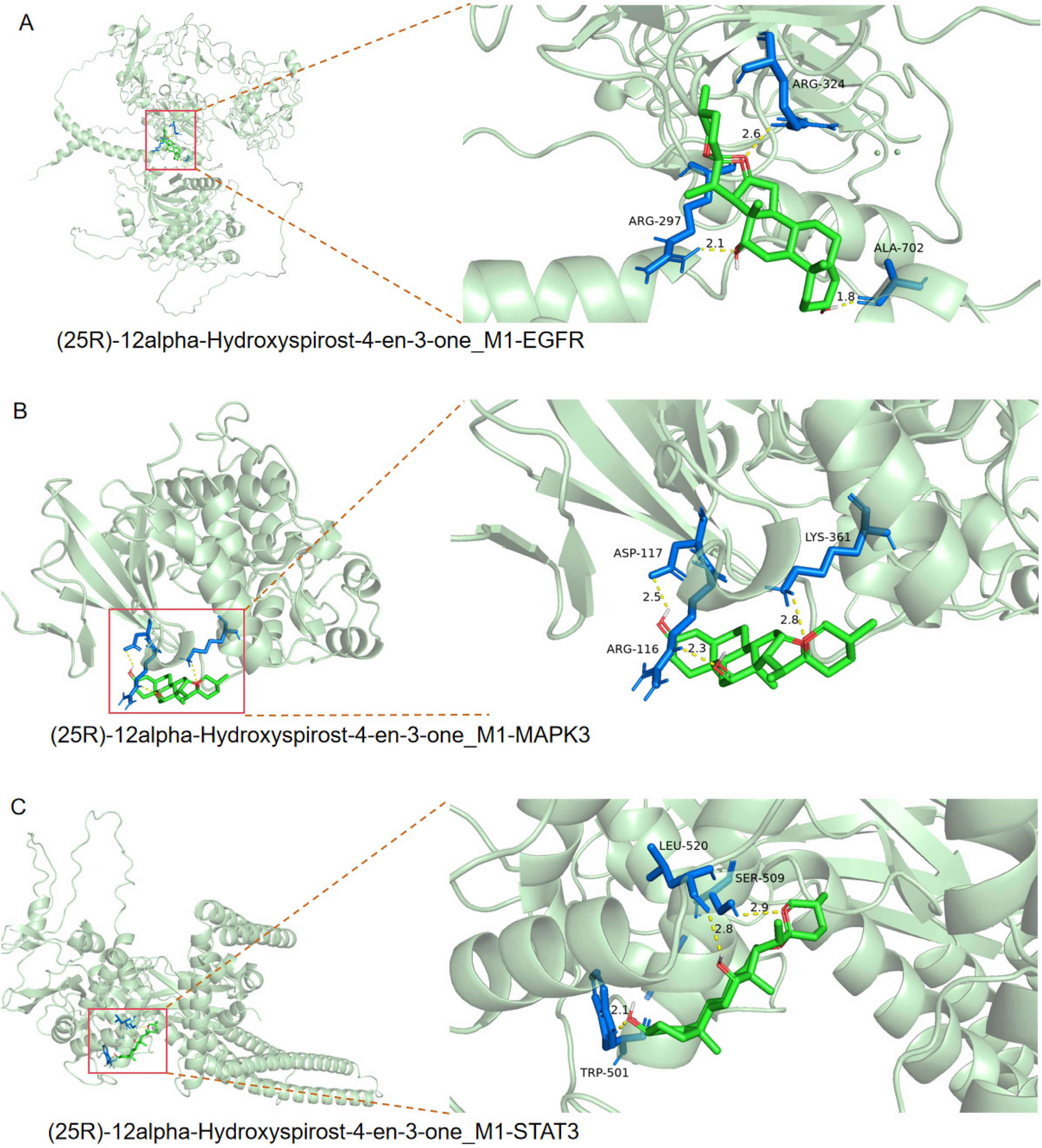
The specific pictures of simulated docking of (25R)-12alpha-hydroxyspirost-4-en-3-one_M1 and its receptor proteins (EGFR, MAPK3 and STAT3). The green part represents curcumin, and the blue part represents the amino acid residues of the target in the red box.
Molecular Docking Results.
Discussion
Allergic asthma is classified as type 2-high asthma, which is an airway inflammatory condition mediated by T helper 2 cells (Th2), group 2 innate lymphoid cell (ILC2), and related cytokines. 6 Upon exposure to allergens, such as pollen and dust mites, dendritic cells (DCs) present antigens to naïve T cells, inducing their differentiation into Th2 cells, which then secrete type 2 inflammatory cytokines in the human respiratory tract, including IL-4, IL-5, and IL-13. Alternatively, the release of alarm cytokines such as IL-25, IL-33, and TSLP by epithelial cells, macrophages, dendritic cells, and other cell types activate ILC2 cells. They subsequently induce the production of type 2 cytokines, which promote B cell maturation and differentiation, stimulate the secretion of specific IgE antibodies, and recruit and activate mast cells, basophils, and eosinophils. Allergic asthma manifests as disruption of the bronchial epithelial barrier, increased vascular permeability, infiltration of inflammatory cells and mediators, goblet cell metaplasia, excessive mucus secretion, smooth muscle dysfunction, and airway remodeling. Furthermore, neutrophils are implicated in the development of type 2-high asthma. Type 2-low asthma is characterized by airway inflammation, with or without neutrophils, and a low responsiveness to corticosteroids. 6 It is associated with female gender, obesity, smoking, and adult-onset symptoms. 31 Airway epithelial cells, dendritic cells (DCs), and alveolar macrophages can be activated by environmental factors such as microorganisms or pollutants. These cells produce pro-inflammatory cytokines, including IL-1β and IL-6, while epithelial cells also produce CXCL8. Under the influence of the cytokine milieu, Th1 and Th17 cells are induced and further involved in the recruitment and activation of neutrophils. Activated neutrophils release cytokines such as neutrophil elastase, myeloperoxidase, or reactive oxygen species (ROS), which induce epithelial cell damage, increase mucus production, and jointly promote the progression of asthmatic pathological characteristics. 32 Matrix metalloproteinase (MMP)-9 is increased in airway samples from smokers with asthma and severe neutrophilic asthma, and MMP family is considered as targets for the treatment of asthma.21,22
SZG is a traditional Chinese herbal compound consisting of multiple medicinal herbs, and it has shown promising application in chronic airway diseases, including asthma. Previous studies have demonstrated that when used as an adjuvant to budesonide-formoterol inhalation powder, SZG significantly enhanced the clinical effectiveness in treating patients with CVA, resulting in more pronounced symptom improvement. 17 Another clinical study reached similar conclusions, indicating that SZG, when combined with budesonide and ipratropium bromide, markedly improved the treatment efficacy during acute exacerbations of chronic obstructive pulmonary disease (COPD), reducing airway resistance and lowering serum levels of IL-17 and HIF-1α. 23 Furthermore, in cases of post-infectious cough, SZG showed superior efficacy compared to pseudoephedrine hydrochloride oral solution, with a more marked improvement in cough relief and throat irritation. 24
In this study, we investigated the molecular underpinnings of SZG in the treatment of asthma, which were verified via molecular docking. Through UPLC-MS/MS, we identified 22 effective blood-borne ingredients of SZG for asthma. Schisandrin A has been found to effectively alleviate asthma symptoms by reducing the activation of the nuclear factor kappa B (NF-κB) signaling pathway, while simultaneously lowering the Th2 cell ratio and mitigating the inflammatory damage to epithelial cells. 25 Magnoflorine exhibits anti-inflammatory and anti-allergic effects by inhibiting the MAPK signaling pathway, thereby reducing the expression of phosphorylated proteins such as p38, extracellular signal-regulated kinase (ERK), and c-Jun N-terminal kinase (JNK). It also significantly inhibits NF-κB expression and inflammatory cytokines, including IL-6 and IL-8. 26 Phaeocaulisin E is considered a potential anti-inflammatory agent to inhibit the formation of nitric oxide (NO), which plays a crucial role in the acidic inflammatory process. 27 Arglabin has been proven to possess anti-inflammatory activity by inhibiting the NLR Family pyrin domain-containing (NLRP) 3 inflammasome andpro-inflammatory cytokines, including IL-1β and IL-18. 28 Curdione exhibits therapeutic effects on inflammation-induced lung injury by inhibiting the recruitment and infiltration of neutrophils, as well as the formation of neutrophil extracellular traps (NETs). 29 Todaclin is a promising anti-inflammatory agent to significantly inhibits the production of nitric oxide, the phosphorylation of ERK1/2, and LPS-induced activation of NF-κB. 30 This research also identified several novel active components not previously reported for use in asthma and other chronic airway inflammatory diseases, such as neocurdione, 24-Hydroxyglycyrrhetic acid, and combretastatin A4, which may provide new avenues for drug development targeting respiratory disorders.
IL-8, IL-5, IL-33, and EOS levels exhibit a positive correlation with Proto-Oncogene Tyrosine-Protein Kinase Src (Src) mRNA expression. 33 Inhibition of Src expression may normalize barrier integrity and cytokine release in airway diseases associated with barrier dysfunction. 34 MAPK3 has been shown to facilitate Th2 differentiation and airway remodeling in experimental asthma. 35 Members of the MAPK family possess numerous pro-eosinophilic functions, 36 and can upregulate the expression of intercellular adhesion molecule-1 (ICAM-1) in pulmonary endothelial cells, leading to the release of TNF-α from neutrophils and dysfunction of airway barriers, 37 thus mediating the development of type 2-low asthma. Caspase-3 participates in pyroptosis and releases IL-1β and IL-18 by cleaving Gasdermin D/E and inducing the formation of cell pores, which subsequently mediates the development of asthma. 38 Epidermal growth factor receptor (EGFR) is a pivotal regulatory factor involved in the migration, proliferation, and differentiation of airway epithelial cells, playing a crucial role in the repair of airway epithelial barriers. 39 In asthma, the overactivation of EGFR leads to epithelial cell injury, goblet cell metaplasia, and airway remodeling. 40 EGFR phosphorylation can induce the expression of signal pathways of thymus and activate the chemokines of Th2 cells, and is positively correlated with the expression of IL-17 and IL-8, indicating that EGFR can synergistically promote airway inflammation through eosinophil and neutrophil pathways. 41 TLR4 is an essential immune pattern recognition receptor that modulates innate and adaptive immune responses, promoting the synthesis and release of cytokines. 42 TLR4 can also regulate the inflammasome and induce pyroptosis by activating NF-κB and upregulating the expression levels of NLRP3, IL-1β, and IL-18,. 43 Prostaglandin-endoperoxide synthase 2 (PTGS2) can be induced by a variety of mediators present in the airway microenvironment, including IL-1β, TLRs, and bradykinin, and produces diverse prostaglandin compounds. These compounds exert the effects of desensitization of β2-adrenergic receptors, mucus secretion, matrix metalloproteinase production, cough reflex, bronchial protection, and anti-inflammatory. 44
TLR is a membrane-bound receptor expressed in dendritic cells and macrophages, recognizing antigens through pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs), facilitating antigen presentation to naive CD4+ T cells and inducing their differentiation into Th2 cells. 45 The TLR signaling pathway can be categorized into two groups: the MyD88-dependent pathway, which activates NF-κB and MAPK and induces the production of pro-inflammatory cytokines, and the MyD88-independent pathway, which induces IFN-α and IFN-β gene expression and exhibits slower activation of NF-κB and MAPK. Recent studies demonstrated that the TLR4/MyD88/NF-κB cascade reaction potentially may potentially lead to allergic reactions by stimulating the production of Th2 cytokines and IL-13 synthesis in lipopolysaccharide (LPS)-induced mast cells. 42 Additional experiment demonstrated that inhibiting the TLR4/MyD88/ROS/NF-κB cascade reduces the number of eosinophils and the levels of IL-4, IL-5, IL-6, IL-13, and TNF-α in bronchoalveolar lavage fluid (BALF), which mitigates airway inflammation in mast cell-dependent mouse models of allergic asthma. 46 The hypoxia-inducible factor (HIF) 1 pathway can be activated by airway hypoxia, NO, and various growth factors resulting from asthmatic inflammation. This activation stabilizes HIF-1α and translocates it to the nucleus, which subsequently promotes the expression of genes such as glucose transporter 1, erythropoietin, vascular endothelial growth factor (VEGF), and other hypoxia response genes. 47 Numerous studies demonstrated that HIF-1α can regulate the activity of eosinophils and enhance airway epithelial inflammation by activating NF-κB.48,49 Recent studies further revealed that inhibiting the PI3K/Akt/mTOR/HIF-1α/VEGF signaling cascade within the HIF-1 pathway significantly ameliorates airway hyperresponsiveness and OVA-induced pathological changes in asthmatic models. 50 In the JAK-STAT signaling pathway, STAT proteins dimerize and translocate to the nucleus upon the activation of JAK family members by cytokines, which regulates the expression of genes involved in Th cell differentiation and promotes the development of asthma. 51 A study suggested that IL-13 can regulate the expression of certain key inflammatory genes through the IL-4Rα/Jak2/Stat3 cascade in activated monocytes. 52 STAT3 has been shown to promote M2 macrophage activation, and facilitate STAT6 binding to target genes, which is crucial for the expression of Th2 cell-related cytokines and transcription factors. 53
The IL-17 signaling pathway is hypothesized to contribute to the development of severe asthma by recruiting and activating neutrophils, mediating chronic airway inflammation, excessive airway mucus secretion, and airway remodeling. 54 A clinical study indicated that the expression of inflammatory factors associated with IL-17 is upregulated in bronchial and nasal biopsies of neutrophilic asthma patients, suggesting that IL-17 plays a significant role in recurring asthma exacerbations. 55 Interestingly, the enhanced expression of IL-17E (also known as IL-25) was observed to support the release of IL-33 and TSLP, and the production of Th2-related cytokines, which facilitate the allergic airway inflammation. 56 However, multiple studies have reported that IL-17 and Th2 signaling pathways are mutually inhibitory.57,58 The TNF signaling pathway regulates cell apoptosis, survival, inflammation, and immune response via the MAPK cascade and the PI3K/AKT/NF-κB pathway. Furthermore, This pathway is also associated with airway inflammation, smooth muscle contraction, and airway remodeling in asthma. 59 Among these, TNF-α, being a Th1 cytokine, is regarded as a key marker for non-type 2 asthma. A randomized, controlled clinical trial observed a significant increase in TNF, TNFR1, and TNFR2 levels in sputum during neutrophilic asthma and severe asthma, suggesting that dysregulation of the TNF signaling pathway could play a key role in the pathogenesis of neutrophilic and severe asthma. 60 Recent studies have found that members of the tumor necrosis factor superfamily (TNFSF) and their receptors promote ILC2 activity in eosinophilic asthma, identifying potential therapeutic targets for type 2 asthma within the TNFSF.61,62 Natural killer (NK) cell-mediated cytotoxicity primarily eliminates target cells via the death receptor pathway and the perforin-granzyme pathway. In this process, Caspase-3 triggers DNA fragmentation, and induces target cell apoptosis. 63 NK cells mitigate the inflammatory response in asthmatic patients by inducing eosinophil apoptosis. A clinical observation revealed that despite an increase in CD56dim NK cells in severe asthma patients, their cytotoxicity declined. This finding suggests that lung NK cells are versatile immune effectors, playing both inhibitory and promotive roles in asthma pathogenesis. 64
C-type lectin receptors (CLRs) constitute a protein superfamily expressed in dendritic cells (DCs), macrophages, NKcells, and other immune cells, which are characterized by the presence of a C-type lectin-like domain. 65 CLRs can generate reactive oxygen species (ROS), trigger intracellular signaling pathways involving spleen tyrosine kinase (Syk), and activate downstream proteins such as caspase recruitment domain-containing protein 9 (CARD9), MAPK, NF-κB, and interferon regulatory factors (IRFs). Through their interplay with other pattern-recognition receptors (PRRs), CLRs play a pivotal role in allergic asthma caused by allergens associated with fungi and house dust mite (HDM) components. 66 Mouse models of airway inflammation induced by HDM and ovalbumin (OVA) have demonstrated that, compared to wild-type mice, Dectin-1-deficient mice exhibit reduced Th2 and Th17 cell responses, as well as decreased eosinophilic and neutrophilic inflammation. 67 Members of the NOD-like receptor (NLR) family in the NLR signaling pathway can recognize diaminopimelic acid, peptidoglycan, and muramyl dipeptide in bacteria. They activate receptor-interacting protein 2 (RIP2) and subsequently induce the activation of MAPK and NF-κB, cytokine production, and apoptosis. Meanwhile, NLRP family members, such as NLRP3, can be activated by bacterial RNA, uric acid crystals, ATP, bacterial toxins, and lead oxide. This activation leads to the maturation of IL-1β and IL-18 and drives pyroptosis via the induction of Caspase-1. 68 A study demonstrated that in HDM-induced airway inflammation in mice, inhibition of Estrogen Receptor 1 (ESR1)/NLRP3/Caspase-1 signaling can significantly reduce the total IgE level and the concentrations of IL-4, IL-6, and IL-17A in serum. 69 Another study suggested that human placenta extract can regulate macrophage polarization by inhibiting the NLRP3 inflammasome, thus exhibiting an anti-allergic effect. 70 Moreover, several medicinal plants and natural products have been shown to inhibit the NLRP3 inflammasome for the treatment of asthma or airway inflammation, such as Citrus peel and Suhuang.71,72 The PI3K/AKT signaling pathway can be activated by a wide range of cellular stimuli and toxic insults. PI3K is activated by receptor tyrosine kinases (RTKs), toll-like receptors 2/4 (TLR2/4), and G-protein-coupled receptors (GPCR), then the activated PI3K phosphorylates PIP2 to generate PIP3, which recruits PDK1 and AKT to the plasma membrane. Subsequently, PDK1 phosphorylates AKT, which in turn activates downstream effectors, regulating cell cycle progression, apoptosis, angiogenesis, migration, and invasion. 73 The PI3K/AKT signaling pathway participates in the pathogenesis of asthma through the regulation of miR-107 and Forkhead box C1 (FOXC1), a hypoxia-induced transcription factor. 74 It also orchestrates downstream protein phosphorylation and activation of NF-κB and glycogen synthase kinase 3β (GSK3β), increasing the expression of IL-4, IL-6, IL-8, TNF-α, and IgE. This ultimately contributes to smooth muscle hypertrophy and fibrosis, airway inflammation, and airway remodeling. 74 Additionally, when stimulated and activated by inflammation, PI3K and AKT activate mTOR, which then regulates protein synthesis and T cell growth. The PI3K/mTOR signaling pathway is crucial for the proliferation of airway smooth muscle, and its inhibition can attenuate airway remodeling in asthma. 75
This study utilized UPLC-MS/MS technology to comprehensively analyze the active blood-permeable components of SZG, thereby providing foundational data for subsequent pharmacological investigations. The outcomes of network pharmacology and molecular docking analyses offer robust predictions and support for the potential targets and mechanisms underlying its therapeutic effect on allergic asthma. However, this study possesses certain limitations. The intricate nature of SZG may encompass synergistic or antagonistic interactions among its components, which network pharmacology and molecular docking may not fully elucidate. Furthermore, these techniques are dependent on existing databases and algorithms, and the accuracy of predictions may be limited by the completeness and precision of these resources. Additionally, the absence of in vivo or in vitro experimental validation introduces uncertainty to the predictions, thereby necessitating further biological experiments to validate their authenticity.
Conclusions
The primary active components in SZG for the treatment of allergic asthma comprise Schisandrin A, Magnolol, Eudesmin, and Isorhamnetin, among the 22 identified bioactive compounds. These constituents target multiple key molecules, including MMP9, Src, MAPK3, Caspase-3, TLR4, and EGFR. They exert their therapeutic effects by modulating various signaling pathways, specifically PI3K/AKT, HIF-1, TNF, and NK cell-mediated cytotoxicity. However, due to the theoretical nature of this study, experimental evidence is still lacking to support these predictions. Therefore, future research will employ in vivo animal models to further validate the specific mechanisms and therapeutic effects of SZG in allergic asthma. Through animal experimentation, we aim to elucidate the pharmacological actions of this formulation and provide more robust experimental evidence for its clinical application.
Materials and Methods
UPLC-MS/MS of SZG Analysis
Reagent
Acetonitrile (HPLC-grade), methanol (HPLC-grade) and formic acid (HPLC-grade) were acquired from Thermo Fisher Scientific (CAS: A998-4, A452-4, A117-50). Additionally, pure water was used in the analysis.
Instrument
Ultrasonic Cleaning Machine from Shenzhen Fuyang Technology Group Co., Ltd (SPEC: F-060SD). Eddy current oscillator from Shanghai Hannuo Instrument Co., Ltd (SPEC: TYXH-I). High speed refrigerated centrifuge from Shanghai Lu Xiangyi Centrifuge Instrument Co., Ltd (SPEC: TGL-16MS). HPLC from Waters (SPEC: ACQUITY UPLC I-Class HF). Chromatographic column from Waters (SPEC: ACQUITY UPLC HSS T3). PDA Detector from Waters (SPEC: ACQUity UPLC). High resolution liquid mass spectrometer from Thermo Fisher Scientific (SPEC: Thermo-Obritrap-QE).
Traditional Chinese Medicine Materials
SZG is comprised of 10 Chinese herbal medicines, including Dark Plum, Mulberry Leaf, Mori Cortex, Paeoniae Radix Alba, Schisandra chinensis (Turcz.) Baill., Glycyrrhizae Radix et Rhizoma Praeparata Cum Melle, Bitter Apricot Seed, Loquat Leaf, Balloonflower Root, and Chinese Wolfberry Root-bark. These herbs were mixed in a weight ratio of 10:10:10:10:6:6:10:15:10:10. SZG is produced and prepared by Beijing Kangrentang Pharmaceutical Co., Ltd and purchased from the traditional Chinese medicine pharmacy of the Third Affiliated Hospital of Beijing University of Chinese Medicine.
Preparation of Medicated serum and Control serum
6 SPF-grade male Sprague-Dawley (SD) rats, weighing 220-250 g, were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd (Animal License No: SYXK (Beijing) 2021-000). The rats were housed in the SPF animal laboratory at Beijing University of Chinese Medicine [SYXK (Beijing) 2020-0033], maintained at a temperature of (23 ± 2) °C, and a humidity of (55 ± 10)%, with free access to food and water, and fed standard nutritional pellet rat feed. This study was approved by the Ethics Committee for Medicine and Experimental Animals at Beijing University of Chinese Medicine, with the animal experiment ethics approval number: BUCM-2023020802-1188. 3 rats were randomly selected as the treatment group and administered SZG via gavage at a concentration of 1.5 g·mL⁻¹, 4 mL per dose, twice daily for seven consecutive days. 76 The remaining 3 rats were assigned to the control group and received an equal volume of normal saline via gavage. Food was withheld for 12 h before the final administration, while water was freely accessible. Blood samples (0.5 mL) were collected at 30 min, 60 min, and 120 min post-gavage. The plasma samples were mixed uniformly and placed in heparin sodium anticoagulant tubes, then centrifuged at 4000 rpm for 10 min to obtain serum, which was stored at −80 °C. 77
Preparation of serum Sample
The serum samples were thawed at 4 °C. Then, 150 μL of each sample was transferred to a 1.5 mL EP tube and vortexed for 1 min to mix thoroughly. Next, 500 μL of the supernatant was placed into LC-MS vials and concentrated to dryness. The residues were reconstituted in 150 μL of water-methanol-acetonitrile (V:V:V = 1:2:1). Then, 100 μL of the supernatant was transferred into vials for analysis. 78 Additionally, an equal volume of each sample's supernatant was pooled to create a quality control sample for UPLC-MS/MS analysis.
Preparation of Extracts from SZG
The samples were ground uniformly, and 100 mg was transferred into a 1.5 mL centrifuge tube. Then, 1 mL of purified water was added, followed by vortexing for 1 min to mix thoroughly. The mixture was centrifuged for 10 min at 12 000 rpm and 4 °C. The supernatant was filtered through a 0.22 μm aqueous phase filter membrane and transferred into vials for analysis.
Liquid Chromatography- Mass Spectrometry Conditions
The UPLC-MS/MS system consisted of an ACQUITY UPLC I-Class HF ultra-performance liquid chromatography coupled with a Q Exactive high-resolution mass spectrometer equipped with a heated electrospray ionization source (Thermo Fisher Scientific, Waltham, MA, USA). The chromatographic separation was performed on an ACQUITY UPLC HSS T3 column (100 mm × 2.1 mm, 1.8 μm) in both positive and negative ion modes, with the column temperature set at 45 °C. The mobile phase consisted of (A) water with 0.1% formic acid and (B) acetonitrile with 0.1% formic acid. The elution gradient was as follows: 0.01 min, 5% B; 2 min, 5% B; 4 min, 30% B; 8 min, 50% B; 10 min, 80% B; 14 min, 100% B; held at 100% B for 1 min; decreased to 5% B in 0.1 min and held for 1 min. The flow rate was 0.35 mL/min with an injection volume of 5 μL. The PDA scanning range was set to 210-400 nm. Mass spectrometry signals were collected in both positive and negative ion scanning modes. The operating parameters of the mass spectrometer were as follows: spray voltage, 3800 V (+) and 3000 V (−); capillary temperature, 320 °C; auxiliary gas heater temperature, 350 °C; sheath gas flow rate, 35 Arb; auxiliary gas flow rate, 8 Arb; S-lens RF level, 50; mass range, 100-1200 m/z; full MS resolution, 70 000; MS/MS resolution, 17 500; NCE/stepped NCE, 10, 20, and 40 eV.
Data Processing and Analysis
The raw LC-MS data underwent baseline filtering, peak identification, integration, retention time correction, peak alignment, and normalization using the Progenesis QI V2.3 software (Nonlinear Dynamics, Newcastle, UK). The primary parameters comprised a 5 ppm precursor tolerance and a 10 ppm product tolerance. Based on precise mass-to-charge ratios (M/z), secondary fragments, and isotopic distributions, compounds were identified using LuMet TCM database, which is an exclusive database of traditional Chinese medicine ingredients established for plant samples and contains 5000 + information of traditional Chinese medicine ingredient standard. In addition, in order to ensure the accuracy of the results, Animal-DB and Herb databases are utilized to supplement the compound identification.
In the QI database, substances qualitatively identified were deemed as original components of SZG if their total score exceeded 50 points or if their total score was greater than 40 points and the secondary match score exceeded 50 points. In the treatment group (administered serum) and the control group (blank serum), substances with an FC value greater than 10 were considered incoming medicinal ingredients.
Network Pharmacology Analysis
Screening of SZG Blood Components and Target Genes
Screened the prototype components and metabolites of TCM absorbed into the blood through UPLC-MS/MS. Subsequently, the SwissADME online platform (http://www.swissadme.ch/index.php) was used to remove the components with poor gastrointestinal absorption (GI absorption labeled as Low) and further screen for components that adhere to Lipinski's Rule of Five. The SwissTargetPrediction platform (http://www.swisstargetprediction.ch/) was employed to predict the potential targets for components that meet the criteria.
Screening of Asthma Genes
The databases of Genecards (https://www.genecards.org/), DrugBank (https://go.drugbank.com/), NCBI (https://www.ncbi.nlm.nih.gov/), OMIM (https://omim.org/) and PharmGKB (https://www.pharmgkb.org/) were adopted to search with “allergic asthma” as the key term. Duplicate entries of the allergic asthma-related target genes were removed using R software (Version 4.0.2).
Component-Disease Intersection Targets
The component targets and disease genes were analyzed using the Venny 2.1.0 platform (https://bioinfogp.cnb.csic.es/tools/venny/) to identify the intersection targets, which were subsequently exported.
“Compound-Target” and PPI Network Construction
The “component-target” network graph was generated using Cytoscape 3.8.0 software. The top 10 compounds were selected as the core components of SZG based on the number of matches with targets related to allergic asthma. The intersection targets were imported into the String database (https://cn.string-db.org/) for PPI network interaction analysis. The resulting data file was then imported into Cytoscape 3.8.0 software for topological analysis and filtering out the core components.
GO and KEGG Enrichment Analysis
The mechanism of SZG's anti-asthma effects can be comprehensively elucidated through Gene Ontology (GO, https://geneontology.org/) and Kyoto Encyclopedia of Genes and Genomes (KEGG, https://www.kegg.jp/) pathway enrichment analysis. Utilizing the GO resource, the hub targets of SZG in asthma were identified, classified, and annotated into three biological aspects: MF, CC, and BP. R 4.0.2 software was used to obtain the Entrez gene IDs of the intersection target genes. Subsequently, based on the preceding steps, all relevant pathways and GO terms were screened using R 4.0.2 software. A P-value less than 0.05 was considered statistically significant.
Molecular Docking Verification of SZG and Targets
The 2D structures of the core active ingredients were retrieved from the PubChem platform (https://pubchem.ncbi.nlm.nih.gov/). In ChemBio3D Ultra 14.0 software, the molecules were converted into 3D format with energy minimization. The structure of the target proteins was obtained from the Protein Data Bank database (https://www.rcsb.org/). Protein structures with self-ligands resembling small molecules were preferred and saved in PDB format. Water molecules and isolated small molecules were removed from the protein structures using PyMOL 1.0.0.0 software.The spatial sites for molecular docking were determined using the target protein and its endogenous ligand. Molecular docking was performed on the obtained receptors and corresponding ligands using AutoDockTools 1.5.6 software. Affinity (kJ/mol) is a parameter that quantifies the spontaneous binding energy between a ligand and receptor. Finally, the results were imported into PyMOL 1.0.0.0 software for further processing and visualization.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241301999 - Supplemental material for Molecular Mechanism of Sangmei Zhike Granules in Treating Allergic Asthma: Insights from UPLC-MS/MS and Network Pharmacology
Supplemental material, sj-docx-1-npx-10.1177_1934578X241301999 for Molecular Mechanism of Sangmei Zhike Granules in Treating Allergic Asthma: Insights from UPLC-MS/MS and Network Pharmacology by Kai Li, Shi-yu Zhang, Gui-rui Huang and Hong-sheng Cui in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Medical and Laboratory Animal Ethics Committee of Beijing University of Chinese Medicine (BUCM-2023020802-1188).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All of the experimental prcedures involving animals were conducted in accordance with the Medical and Laboratory Animal Ethics Committee of Beijing University of Chinese Medicine (BUCM-2023020802-1188) approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
