Abstract
Objective
Yin-Qiao-Qing-Re Tablets (YQT) are clinically used for the treatment of wind-heat type common cold. However, YQT is composed of nine herbs, containing complex and multiple chemical components, and the metabolism of YQT in vivo lacks a more in-depth study.
Methods
In this study, based on targeted and non-targeted UPLC-Q-TOF-MS/MS data analyzing strategy, the prototypes and metabolites of YQT in normal rats were systematically studied for the first time, and the metabolic pattern of its chemical components was summarized. Finally, to identify the target and signaling pathway of YQT in the treatment of fever, the prototypes in plasma were analyzed through network pharmacology and molecular docking.
Results
A total of 238 components, including 39 prototypes and 198 metabolites, were identified from drug-containing plasma, urine and feces. The metabolic pathways of flavonoids, phenylethanols, organic acids, lignans and alkaloids in the TCM compound were summarized. The main metabolic reactions included one-phase metabolism (dehydroxylation, demethylation, dehydration, deglycosylation, hydrogenation) and two-phase metabolism (methylation, hydroxylation, glucuronidation, sulfation, alanine binding). Network pharmacology predicted 112 key targets for YQT treatment of fever, and KEGG enrichment analysis yielded 163 pathways. The molecular docking results indicated that there is good affinity between the core targets and the key compound molecules.
Conclusion
This study provides a rapid qualitative analysis of the prototypes and metabolites of YQT in rats, revealing its potential active ingredients and mechanism of action in the treatment of fever. Furthermore, it provides a solid database for the subsequent pharmacokinetic and pharmacodynamic studies of YQT, promoting the further clinical application of this marketed drug.
Keywords
Introduction
Yin-Qiao-Qing-Re Tablets (YQT) was a clinically experienced formula optimized on the basis of Yin-Qiao-Bai-Du Decoction (Shen Hanqing’s Wen-Re-Jing-Jie). The prescription, consisting of Lonicerae Japonicae Flos (LJF), Puerariae Lobatae Radix (PLR), Forsythiae Fructus (FF), Anemarrhenae Rhizoma (AR), Isatidis Radix (IR), Arctii Fructus (AF), Menthae Haplocalycis Herba (MHH), Cimicifugae Rhizoma (CR) and Cicadae Periostracum (CP), manifested effects in resolving exterior with pungent and cool natured drugs. This prescription was used in treating wind-heat type common cold, such as fever, sore throat, chills, nasal congestion and runny nose. 1 LJF, on the one hand, known as “a good medicine for clearing heat-toxin”, had significant effects on improving the symptoms of various feverish diseases. PLR, on the other hand, had the effects of relieving thirst and was beneficial for fever and cold. These two herbs functioned as the sovereign drugs in the prescription. On top of this, FF and AR were incorporated into the prescription as the minister drugs to relieve exterior symptoms and clear heat, and when combined with other assistant drugs, they further strengthened the medicinal effects.
In recent years, serum pharmacochemistry of traditional Chinese medicine (TCM) has garnered widespread attention and application from scholars both domestically and internationally, leading to the achievement of several significant research outcomes. 2 The synergistic effects of multi-components, multi-targets, and multi-pathways in TCM allow for flexible adjustment of prescriptions and medications, 3 thus reducing adverse reactions and drug resistance. However, it is precisely because of the material basis of TCM’s pharmacological effects remains one of the major bottlenecks in the development and modernization of TCM. Furthermore, YQT, as a type of Chinese patent medicine, exhibiting significant differences in chemical components compared to single herbs due to their unique preparation process. Currently, there are relevant research reports on the metabolic study of single herbs in vivo, for example, PLR, 4 FF 5 and CP, 6 as well as studies on the metabolism of single component like Timosaponin AIII, 7 Arctiin, 8 Goitrin. 9 Nevertheless, the pharmacological efficacy of TCM is frequently not attributed solely to a single components but arises from the intricate interplay and synergistic effects among multiple components. The mechanism of this multi-component synergistic effect is complex and not fully understood at present, which limits the process of modernizing TCM. Therefore, single herbs or single components cannot fully explain the overall metabolic characteristics of TCM.
To address these issues, it is necessary to strengthen the research on the material basis of TCM’s efficacy and conduct more scientific studies on its efficacy and safety. UPLC-Q-TOF-MS/MS can meet the analytical needs of different types of samples and compounds, including but not limited to natural drugs and biological samples.10,11 Meanwhile, this technology is a high-resolution, high-mass accuracy, and high-efficiency analytical tool that can accurately and rapidly determine the structure of compounds. 12 In recent years, network pharmacology has been widely applied in the research of TCM, which can elucidate the mechanism of action of diseases and drugs by constructing biological network models such as drug-target network and gene-disease network. 13 In this study, our team has previously reported a non-targeted and targeted strategy that successfully characterized the metabolic pathways of TCM in rats. 12 Therefore, this strategy were continued in this study to investigate the absorption, distribution, metabolism and excretion of exogenous components of YQT in rats. This study lays the foundation for subsequent research on YQT in various aspects such as quality control, pharmacokinetics, pharmacodynamics, etc Additionally, we have also conducted preliminary explorations into the network pharmacological mechanisms of YQT in treating fever.
Materials and Methods
Materials and Reagents
Yin-Qiao-Qing-Re Tablets (NO. 230303) was provided by Jiangsu Kanion Co., Ltd. The reference standards were purchased from the following suppliers: National Institutes for Food and Drug Control (Beijing, China): goitrin (≥100%, Lot No. 111753-202007), caffeic acid (≥99.7%, Lot No. 110885-201703), mangiferin (≥98.3%, Lot No. 111607-202305), puerarin (≥96.8%, Lot No. 110752-202217), daidzin (≥96.3%, Lot No. 111738-202305), ferulic acid (≥99.6%, Lot No. 110773-201313), isoferulic acid (≥99.3%, Lot No. 111698-201904), forsythoside A (≥96.4%, Lot No. 111810-202209). Shanghai Yuanye Bio-Technology Co., Ltd (Shanghai, China): 3’-hydroxypuerarin (≥98%, Lot No. A10HB191367), sweroside (≥98%, Lot No. F21IB207682), 3’-methoxypuerarin (≥98%, Lot No. J12HB173942), mirificin (≥98%, Lot No. J13HB173936), isoforsythiaside (≥98%, Lot No. Z30J9L65411), forsythoside H (≥98%, Lot No. F27GB140183), Arctigenin (≥98%, Lot No. M2OIB210166), timosaponin AI (≥98%, Lot No. M21J12S138178), timosaponin AII (≥98%, Lot No. D08IB234733), and timosaponin AIII (≥98%, Lot No. A11GB157567). The National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China): cimifugin (≥98%, Lot No. 111710-200602). HPLC-grade acetonitrile and methanol were purchased from Merk (Darmstadt, Germany) and HPLC-grade formic acid was purchased from Roe Scientific Inc (Newark, USA). The water was prepared as deionized water using a Milli-Q water purification system (Millipore, USA). Carboxymethylcellulose Sodium (CMC-Na, NO. F20100126) was purchased from Nanjing Chemical Reagent Co., Ltd.. Ethyl carbamate (NO. N11IS232062) was purchased from Shanghai Yuanye Bio-Technology Co., Ltd and heparin sodium was (NO. B805BA0008) acquired from BBI Life Sciences Corporation.
Instrument
Agilent 1290 Infinity Ultra High Performance Liquid Chromatography, Agilent 6538 Q-TOF Mass Spectrometer (Agilent Technologies, USA); MIKRO 200 High-Speed Centrifuge (Andreas Hettich GmbH & Co.KG, Germany); Milli-Q Water Purification System (Millipore, USA); KQ-500DE Digital Control Ultrasonic Cleaner (Kunshan Ultrasonic Instrument Co.,Ltd, China); Mettler AL204 Electronic Analytical Balance (Mettler Toledo, China); Auto EVA-Mini Parallel Concentrator (RayKol Group Corp., Ltd, China).
Preparation of Samples for YQT and Reference Standards
The powder of YQT (1.0 g) was dissolved in 50% methanol (25.0 mL) and refluxed for 30 min. Afterwards, the supernatant was centrifuged at 14,000 rpm for 10 min before use. In addition, reference standards were dissolved in 50% methanol to prepare solutions with a concentration of 20-50 ug/mL. Finally, these solutions were centrifuged as above, collecting the supernatants.
Preparation of Samples for Drug Administration
The CMC-Na (5.0 g) was dissolved in water (1000.0 mL) to prepare a 0.5% CMC-Na solution. The powder of YQT (26.0 g) were dissolved in 0.5% CMC-Na solution (100.0 mL) to prepare a concentration of 0.26 g/mL (1 mL/100 g/day, 6 times equivalent to the oral dose of humans).
Animals and Administration
SD rats (SPF grade, male, weighing 180-200 g) were provided by Hangzhou medical college, Hangzhou City, China. The qualification certificate number was 20231114Aazz0100000452 and the animal production license number was SCXK-(Zhejiang) 2019-0002. The study also received ethical approval, with the approval number 2023111420. The rats were raised in separate cages, 2/cage, under a controlled environment of room temperature 22 ± 2 °C and humidity 50-60%. They had free access to water and were adaptively fed for 7 days. All experiments were conducted in accordance with guidelines for the management and use of laboratory animals. The reporting of this study conforms to the ARRIVE 2.0 guidelines. 14
The rats were randomly divided into control group (n = 3) and YQT group (n = 6). Seven days later, the control group was gavaged with 0.5% CMC-Na solution, while the YQT group was gavaged with YQT solution, and the dosage (based on body weight) was administered for three consecutive days, and the drug was administered once a day at 9:00 am.
Biological Samples Collection and Pretreatment
All rats were fasted 12 h prior to the collection of bio-samples and were placed in metabolic cages for free access to water overnight. After the fourth administration, the rats’ urine and feces were collected through the metabolic cage from 0 to 12 h, 12 h to 24 h. After the sixth administration, blood samples (0.25 mL ∼ 1.0 mL) were collected from the orbital region at 0.25 h, 0.5 h, 1 h, 2 h and 4 h and placed in 1% sodium heparin tubes. 11 These samples were then centrifuged at 14,000 rpm for 10 min to obtain the supernatant. All of the above collected bio-samples were combined and initially stored in −80 °C refrigerator for freezing and preservation. The experiment flow diagram was shown in Figure 1.

Experiment Flow Diagram.
After drying and grinding, the feces (2.0 g) were added to 50% methanol (30.0 mL), and the mixture was ultrasonically vibrated for 30 min to obtain the fecal extract. The fecal extract was concentrated and dried using a rotary evaporator at 50 °C. It was then dissolved with 2 ml of water, centrifuged at 14,000 rpm for 10 min at 4 °C, and the resulting supernatant 1 was collected. The thawed urine (6 mL) was added to methanol (6 mL) to precipitate the protein, vortexed and centrifuged successively as the above procedures to obtain the supernatant 2. Both supernatant 1 and 2 were processed separately on Oasis HLB columns, and the methanol eluate was collected and blown dry with nitrogen gas at 4 °C. The residues were dissolved with 50% methanol (300μL), centrifuged at 14000 rpm for 10 min, and then centrifuged twice to obtain the final supernatants. The thawed plasma (4 mL) was added to methanol (12 mL) to precipitate the protein, vortexed and centrifuged according to the previous procedures and blown dry with nitrogen gas at 4 °C. The residue was dissolved with 50% methanol (100 μL), centrifuged as urine and feces were centrifuged, to obtain the final supernatants. The method for the control group was the same as that for the YQT group. All the final supernatants will eventually have 3μL taken for LC-MS analysis.
Chromatography and Mass Spectrometry Conditions
Chromatography Conditions
A Waters ACQUITY UPLC BEH C18 (2.1 mm × 100 mm,1.7μm) was used at 30 °C with a sample volume of 3 μL. The mobile phases were 0.1% formic acid-water (A) and acetonitrile (B) and the flow rate was 0.35 mL/min, with gradient program was optimized as follows: 3% B (0 min); 3%-10% B (0-15 min); 10%-20% B (15-35 min); 20%–50% B (35-52 min); 50%-70% B (52-55 min); 70%–95% B (55-60 min).
Mass Spectrometry Conditions
The operating parameters were: ESI− mode and ESI+ mode, drying gas (N2) flow rate, 8.0 L/min; Gas Temperature, 350 °C, Nebulizer, 40psi; VCap3500 V; OctopoleRFPeak, 750; Fragmentor, 100 V; Collision energy: 12 V; Scan mass range: 50-1500 (MS), 50-1500 (MS/MS). By spraying a calibration solution encompassing internal reference masses at m/z 121.0509 and 922.0998 in positive mode, as well as m/z 112.9856 and 1033.9881 in negative mode, precise mass measurements were achieved for each peak.
Data Processing
The data were analyzed using the Agilent MassHunter Qualitative analysisi 10.0 software and MetabolitePilot 2.0.4 software to achieve the screening of prototypes and metabolites in rats.
Network Pharmacology Analysis
Screening of Active Ingredients and Disease Targets
SMILES numbers for the blood-absorbed components were collected from the Pubchem (https://pubchem.ncbi.nlm.nih.gov/) and sequentially imported into Swiss Target Prediction (http://www.swisstargetprediction.ch/) to gather corresponding targets for the components, with duplicate targets removed. Using “Fever” as the keyword and specifying “Homo sapiens” as the species, potential disease targets with a “probability > 0” were collected from the GeneCards database (https://www.genecards.org/). Finally, the targets of the active ingredients were merged with the disease targets to obtain the potential targets for YQT in treating fever, and a Venn diagram was plotted to illustrate this.
Construction of Protein-Protein Interaction (PPI) Network and Screening of Key Targets
Upload the potential targets to the STRING 12.0 database, select “Homo sapiens” as the biological species, and obtain the PPI (Protein-Protein Interaction) network. Import the data into Cytoscape 3.7.1 software for visualization analysis. Use the cytoscape2.2 plugin to calculate the Closeness, Betweenness, and Degree values to screen for key targets.
Potential targets were uploaded into the STRING database (version 12.0, https://cn.string-db.org/) and the biological species was selected as “Homo sapiens” to obtain the PPI network. The data were imported into Cytoscape 3.7.1 software for visualization and analysis. The Cytoscape 2.2 plugin was employed to select key targets based on the criterion that their Closeness, Betweenness, and Degree centrality values all exceeded the median values.
Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Enrichment Analysis
The potential targets were imported into the David database (https://david.ncifcrf.gov/), and the biological species was selected as “Homo sapiens”, and the KEGG pathways were enriched and analyzed. Exploring the potential mechanism of action of YQT in the treatment of fever based on the degree of core pathway enrichment.
Molecular Docking
The receptor protein IDs of core targets were retrieved from the UniProt database (https://www.rcsb.org/), followed by downloading their 3D structures in PDB format from the PDB database (https://www.rcsb.org/). Water molecules and small-molecule ligands bound to the corresponding residues were removed. The 2D structures of nine blood-absorbed components were downloaded from the PubChem website and saved in SDF format. Finally, cavity-detection guided blind docking (https://cadd.labshare.cn/cb-dock2/php/index.php) was performed between the prototypes in plasma and core targets. The molecular docking results were visualized and analyzed using PyMOL software.
Results
Data Analyzing Strategy
The In-house-library was imported into the automatic search database of the software (Table S1, Figure S1). The parameters were set in the method editor: analysis time 0- 60 min; retention time deviation ± 0.5 min; mass deviation ± 10 mDa; mass range 50- 1200 m/z; target ions: [M-H]−, [M + H]+, [M + HCOO]−. The “Find by molecular formula” function was used to retrieve the prototypes. The targeted screening was performed by comparing retention time, molecular formula, relative molecular mass and primary and secondary fragment ions to screen prototypes. Meanwhile, the results were further confirmed by comparison with reference substances. The non-targeted screening is based on the “Find by Auto MS/MS” function in the software analysis and MetabolitePilot 2.0.4 software to assist in the identification of metabolites. By comparing the MS spectra of the control group and YQT group, while subtracting the prototypes and endogenous components from plasma, urine and feces, the metabolites were screened in a non-targeted way. Finally, based on the prototypes and metabolites, the metabolic patterns of different types of compounds were summarized in order to refine the metabolic pathways of exogenous components of YQT in rats. It is worth noting that isomers will inevitably appear during the analysis, and this study will differentiate them based on their retention time, Calculated LogP (CLogP) and the degree of response in the MS spectra. The Clog P value is calculated through ChemDraw Professional 22.0. In general, in a reversed-phase liquid chromatography system, compounds with higher values tend to have longer retention times. The data analysis strategy was shown in Figure 2.

A Strategy for in Vivo Metabolism of YQT Combined Targeted and Non-Targeted UPLC-Q-TOF-MS/MS.
Characterization of Prototypes in YQT
According to the LC-MS conditions, the total ion chromatogram (TIC) in positive and negative ion modes were obtained as shown in Figure 3A-F. The detailed MS/MS information of compounds is shown in Table 1. A total of 39 prototypes were detected from drug-containing plasma, urine and feces, including 16 flavonoids, 4 organic acids, 5 lignans, 3 phenylethanoid glycosides, 4 alkaloids, 4 saponins, 1 iridoid, and 2 terpenoids. Nineteen of the compounds were standardized (Figure 3G-H).
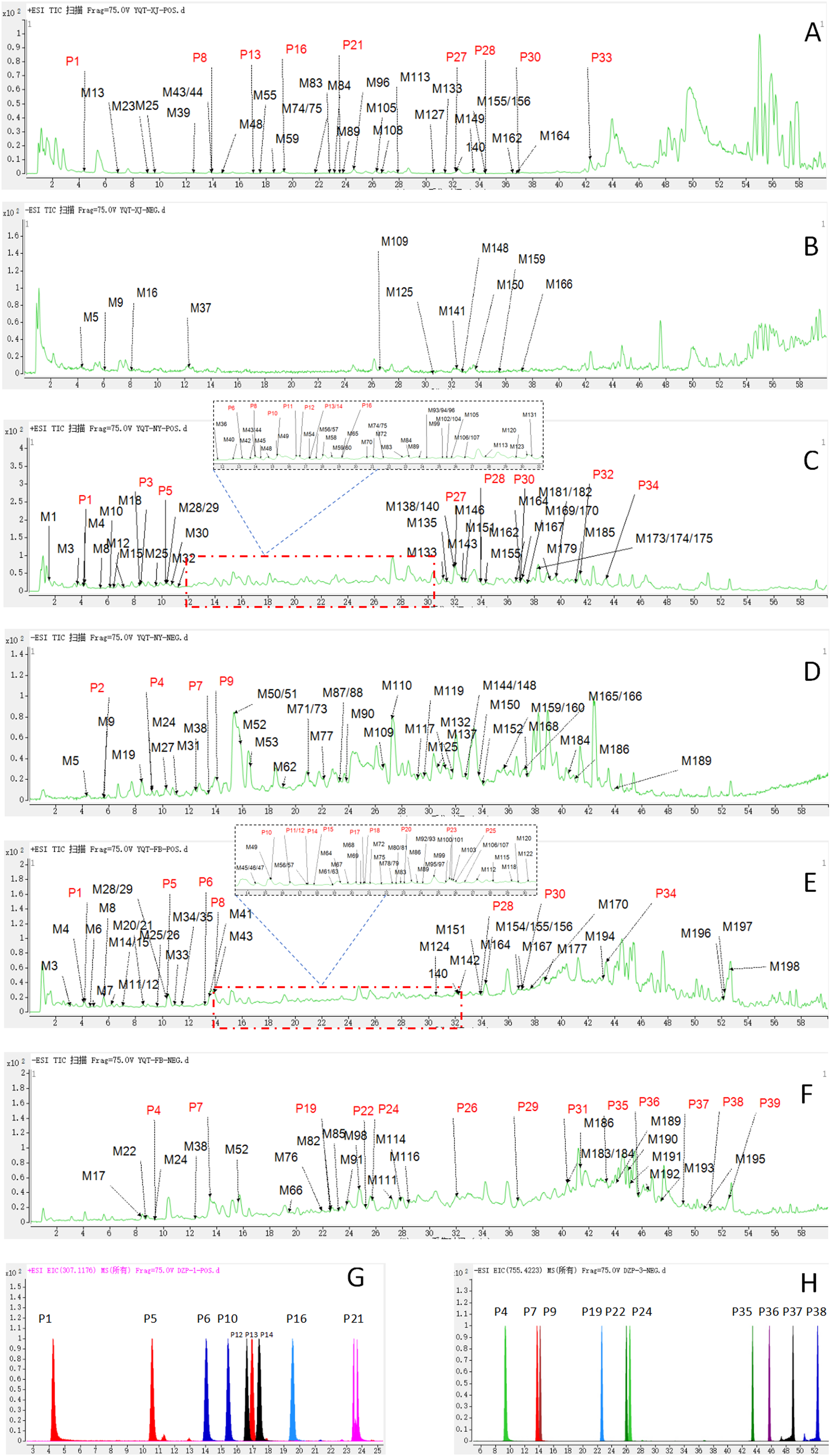
The Total Ion Chromatogram of YQT-Related Prototypes and Metabolites in Bio-Samples of Rats and Reference Standards in Positive (A, Plasma; C, Urine; E, Feces; G, Reference Standards) and Negative (B, Plasma; D, Urine; F, Feces; H, Reference Standards) Ion Modes.
Characterization of Prototypes of YQT in Rat Bio-Samples.

P: plasma, U: urine, F: feces, Xyl-C5H8O4, Glc-C6H10O5, Api-C5H8O4, *Confirmed with reference compounds.
Characterization of Metabolites and Their Classification in YQT
A total of 198 metabolites (40 in plasma, 106 in urine and 117 in feces) were found in drug-containing bio-samples. A total of 187 metabolites including flavonoids, phenylethanol glycosides, organic acids, lignans, saponins, alkaloids, and terpenoids were further identified through MS/MS information and literature (the TIC was outlined in Figure 3A-F and the MS/MS information was shown in Table 2). The phase II metabolites detected in drug-containing plasma mainly involved reactions of glucuronidation and sulfation. In drug-containing urine and feces, phase I metabolites such as hydrogenation, dehydroxylation, demethylation, deglycosylation, and dehydration, as well as phase II metabolites including glucuronidation, sulfation, methylation, and hydroxylation, were mostly observed. Additionally, some compounds also showed acetylation reactions. It is noteworthy that the metabolites after a series of biochemical reactions are different from the prototypes, but their basic skeletons are basically the same and produce identical fragment information. Therefore, this study can classify and summarize the metabolites and metabolic pathways of various compounds based on these characteristic fragments. The representative MS2 spectra of various compounds were shown in Figure 4.

Characterized Fragments of Type of Compounds in MS/MS Spectra.
Characterization of Metabolites of YQT in Rat Bio-Samples.
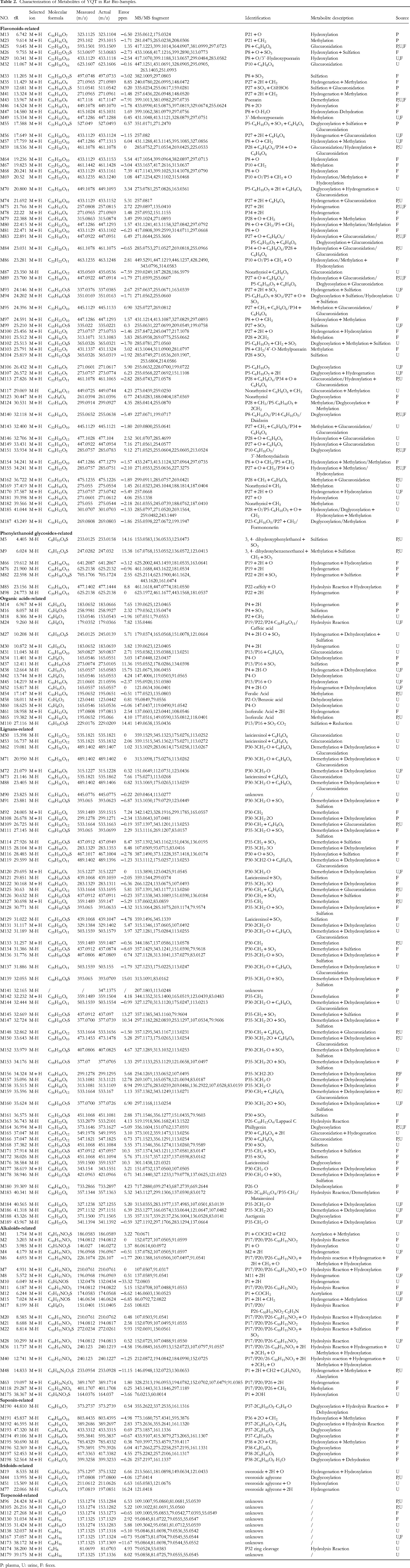
P: plasma, U: urine, F: feces.
Flavonoids-Related Metabolites
The Flavonoids-related metabolites were derived from PLR. Flavonoid compounds are known for their unique Retro Diels-Alder reaction (RDA). In this process, the molecular structure undergoes rearrange, and C4H8O4 (120 Da) serves as the most common characteristic fragment in the RDA reaction. This is accompanied by the shedding of fragments such as glycosyl, CH3, H2O, CO and other ionic fragments.
Flavonoids were hydrogenated to produce dihydroflavonoids. Three metabolites (

Possible Metabolic Pathways of Flavonoids of YQT in Rats.
Phenylethanoid Glycosides and Organic Acids-Related Metabolites
The Phenylethanoid glycosides-related metabolites were derived from FF, with 3, 4- dihydroxyphenylethanol as its basic nucleus, which was linked with two glycosyls. Due to the difference in the connecting sites of caffeoyl groups,
The
The metabolic pathway of caffeic acid could also be divided into two types. First, caffeic acid underwent phase I and phase II reactions. Caffeic acid were converted to
Lignans-Related Metabolites
The lignans-related metabolites were derived from AF and FF. A total of 63 metabolites were detected from the drug-containing plasma, feces and urine, mainly including phase I reactions (demethylated and dehydroxylated) and phase II reactions (glucuronidation and sulfation). Interestingly, the phase II metabolites are almost derived from the phase I metabolites.
5
Due to the identical molecular formula and similar structures of phillygenin (
In the MS2 spectra, Lappaol C (

Possible Metabolic Pathways of Lignans of YQT in Rats.
When discussing the phase II metabolites,
Alkaloids-Related Metabolites
The alkaloids-related metabolites were derived from IR and CT. A total of 22 metabolites were detected from the drug-containing plasma, urine and feces. Its metabolic pathways were phase I metabolism (hydrogenation and oxidation) and, furthermore, phase II metabolism (methylation, hydroxylation, acetylation and alanine reaction).
Saponin-Related Metabolites
The saponin-related metabolites were derived from AR. A total of 9 metabolites were detected from the drug-containing bio-samples. The metabolic pathways were deglycosylation, deresidue, dehydration, hydroxylation, and methylation.
Iridoids and Terpenoid-Related Metabolites
The iridoid-related metabolites were derived from LJF. Combined with the metabolites provided on the MetabolitePilot, 4 metabolites were detected in the urine of rats after oral administration of YQT. Sweroside was first hydrolyzed in rats to remove one molecule of glucose, generating sweroside aglycone (
The terpenoid-related metabolites were derived from MHH, and a total of 10 metabolites were detected. Specifically, the prototypes α-pinene and piperone were detected in plasma and urine, respectively, both as volatile compounds. However, the limited detection capabilities of LC-MS conditions did not permit the identification of additional volatile components. In contrast, our preexperimental study demonstrated that many volatile compounds could be successfully detected using GC-MS (Table S2, Figure S6).
Statistical Analysis of Prototype Drugs and Metabolites in Rats
Pie charts and bar charts can visualize the distribution of prototypes and metabolites after oral administration of YQT in rats (Figure 7A). Heatmaps can intuitively express the pathways and intensities of Phase I and II reactions of various compounds (Figure 7B). The results showed that flavonoids and lignans accounted for the largest proportion, suggesting that these two components may be the main active ingredients. Flavonoids mainly underwent deglycosylation, methylation, hydroxylation, glucuronidation, and sulfation reactions, while lignans mainly underwent demethylation, dehydroxylation, glucuronidation, and sulfation. A large number of lignan metabolites were observed in feces, indicating that the main metabolism of lignan compounds occurred in the intestinal flora of rats. More organic acid metabolites were observed in urine, suggesting that these components were excreted from urine after being absorbed by the kidney. The saponin-related metabolites were exclusively detected in the drug-containing feces. One of the reasons was that the saponins may not have been absorbed into the blood circulation or metabolized by microorganisms in the intestine before being excreted. A large number of microorganisms exist in the intestine, which may have the ability to decompose saponins. Due to the special chemical properties of saponins, such as large molecular weight, low permeability, and low solubility, oral administration has low bioavailability. 28 The other one of the reasons was that YQT consists of a variety of herbs, and the complexity of their preparation process can also lead to a decrease in the content of saponins, which explains why that they cannot be detected in urine and plasma. It is noteworthy that the literature reports glucuronidation and sulfation metabolites of Sweroside, 29 but these were not detected in this study. Additionally, the abundance of iridoid-related metabolites was not high, and only a few compounds were detected. It is speculated that sweroside primarily exerted the efficacy in vivo primarily as the prototype of YQT.

A, Percentage of YQT-Related Prototypes and Metabolites and Distribution in Bio-Samples; B, Heatmap of Metabolic Reaction Distribution and Intensity of Various Compounds.
Network Pharmacology Analysis of YQT for the Treatment of Fever
After removing duplicates, a total of 446 active ingredient targets were obtained from the prototypes screening. GeneCards database yielded 9481 disease targets, which were screened by median using Relevance score as a parameter, resulting in 1184 disease targets. The active ingredient targets were combined with the disease targets to take the intersection, and 112 potential targets were obtained (Figure 8B). These 112 key targets were imported into the STRING database to form a PPI network, which was visualized and analyzed using Cytoscape 3.7.1. The results are shown in Fig. A total of 115 nodes (target proteins) and 1887 edges (protein interactions) were obtained. The key targets were screened according to Closeness, Betweenness and Degree values (Closeness > 0.00507510897627994, Betweenness > 87.2869565217387, Degree > 32.8173913043478). There were 22 key targets for the treatment of fever by YQT, including GAPDH, TNF, AKT1, EGFR, STAT3 and others (Figure 8A). In KEGG enrichment, a total of 163 pathways were enriched, and the top 10 pathways were shown in Figure 8C. The results showed that the treatment of fever with YQT might be associated with EGFR tyrosine kinase inhibitor resistance, prostate cancer, PD-L1 expression and PD-1 checkpoint pathway in cancer and so on.

A, Visualization of the PPI Network for Potential and Key Targets. The Size and Color of the Nodes Represent their Importance. B, Venn Diagram of Targets of Prototypes in YQT and Targets of Fever. C, KEGG Pathway Enrichment Analysis.
Molecular Docking
The top five core targets ranked by degree value for YQT in treating fever were selected. Receptor proteins of these core targets were obtained from the PDB database, and the chemical structures of nine blood-entering components were downloaded from PubChem. A binding energy of less than −5.0 kJ/mol between the compound ligand and the protein receptor indicates good docking performance, while a binding energy below −7.0 kJ/mol suggests significant affinity. 30 The results (Figure 9A) demonstrate that, except for Goitrin, all other components exhibit strong binding activity and stable molecular conformation with the targets. Notably, puerarin, cimifugin, 3'-methoxydaidzein, daidzein, and phillygenin showed significant affinity with TNF, GAPDH, STAT3, and EGFR. Finally, molecular docking was performed using PyMOL software, and the visualization results are presented in the Figure 9B.

A, Heatmap of Binding Energy of Composition and Target Docking. B, Docking Results of TNF with Puerarin (A), GAPDH with Daidzein (B), EGFR with Phillygenin (C), AKT1 with 3’-Methoxydaidzein (D).
Discussion
This study systematically analyzed the metabolic profile of YQT in rat plasma, urine, and feces, and found that compared with single plasma analysis, combined urine and feces characterization significantly expanded the coverage of metabolites. 31 After administration, the drug was first absorbed into the blood circulation of rats. Wang proposed that the prototypes or metabolites of traditional Chinese medicine that are orally ingested and enter the plasma are considered to be the active ingredients that can be absorbed. 32 However, due to the existence of a steady-state regulation within the plasma, not all absorbed drugs will take effect or be metabolized immediately. There were differences in the absorption degrees of various compounds, resulting in few prototypes being detected in the plasma. At the same time, the detection of prototypes in urine can also indirectly reflect the absorption of the formulation by the body. Therefore, most of the prototypes detected in plasma can also be detected in urine. Furthermore, the detection of prototypes in feces may be related to metabolism that is not significantly absorbed, whereby the prototypes are excreted through the intestine for consistency in tense and clarity. 33 Additionally, they may remain in the gastrointestinal tract under the influence of intestinal flora, undergoing transformations that alter their bioavailability, bioactivity, or toxicity, thereby affecting the individual's response to the drug. 34
It is generally believed that the important active ingredients of LJF are caffeoylquinic acid derivatives, which account for a relatively high proportion. Wang found that chlorogenic acid and related derivatives could be detected in the plasma of rats after oral administration of LJF.
35
But in this study, the related compounds were not detected in the bio-samples from rats, possibly due to poor absorption or detection limitations under our conditions.
36
Nevertheless, other organic acid compounds, such as caffeic acid and isoferulic acid, were easily detected with high response values, highlighting their importance as active ingredients in LJF
37
and CR.
38
This may be attributed to the rapid metabolism of caffeoylquinic acid derivatives. In addition, research has demonstrated that caffeic acid had the ability to inhibit the degree of cellular inflammation induced by lipopolysaccharide (LPS). And it is hypothesized that organic acids were important ingredients in the antipyretic effect of YQT. Puerarin (
GAPDH and AKT1 interact with various cellular signaling pathways, influencing cell apoptosis, proliferation, and stress responses. Studies have demonstrated that the downregulation of GAPDH enzyme activity effectively inhibits LPS-induced macrophage activation, leading to the exertion of an anti-inflammatory effect, which in turn contributes to achieving antipyretic outcomes. 52 In addition, another study showed that inhibition of AKT phosphorylation can reduce inflammatory response. 53 TNF, a common target in fever and associated with inflammatory responses, 54 is enriched in multiple metabolic pathways, suggesting that YQT's treatment of fever is closely related to inflammatory pathways. EGFR is related to the inhibition of proliferation, metastasis and apoptosis of tumor cells. 55 YQT for fever treatment was enriched in multiple cancer pathways, and the induction of EGFR protein degradation is inextricably linked to cancer treatment, suggesting that EGFR targets were closely related to the treatment of fever. Interleukin-6 (IL-6) is considered an endogenous mediator of LPS-induced fever. 56 STAT3, as one of the important targets in the mechanism of fever treatment, can inhibit the differentiation of T cells and suppress autoimmune responses by inhibiting the secretion of anti-inflammatory factors such as IL-6 and IL-10, thereby exerting an anti-inflammatory effect. 57 The molecular docking results demonstrated that the blood-absorbed components of YQT exhibited favorable binding activity with key target proteins including GAPDH, TNF, AKT1, EGFR, and STAT3. These findings suggest that YQT exert their antipyretic effects through multi-component, multi-target, and multi-mechanism interactions.
Conclusion
This study aimed to elucidate the chemical components of YQT in vivo, uncover its potential mechanisms, and identify active ingredients for treating fever, thereby providing a basis for a deeper understanding of the material foundation underlying its pharmacological effects. A total of 39 prototypes and 198 metabolites were analyzed for the first time in plasma, urine and feces of rats after oral administration of YQT using UPLC-Q-TOF-MS/MS, and the reasons for the presence of different types of compounds in different bio-samples were analyzed. Furthermore, the mechanism of YQT in treating fever was predicted through network pharmacology analysis based on the blood-absorbed components. A PPI network was constructed to screen for 112 key targets, including GAPDH, TNF, AKT1, EGFR, STAT3, etc KEGG enrichment analysis obtained 163 pathways, which were mostly related to cancer and inflammation. Molecular docking results demonstrated that the blood-absorbed components of YQT exhibited favorable binding activity with the key target proteins. These results can not only clarify the material basis of the pharmacological effect, but also provide a more precise guidance in clinical application, further study the pharmacological activity of YQT and promote its wider clinical application.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251359862 - Supplemental material for Combining UPLC-Q-TOF-MS/MS with Network Pharmacology and Molecular Docking to Explore the Potential Mechanism of Yin-Qiao-Qing-Re Tablets in Treating Fever
Supplemental material, sj-docx-1-npx-10.1177_1934578X251359862 for Combining UPLC-Q-TOF-MS/MS with Network Pharmacology and Molecular Docking to Explore the Potential Mechanism of Yin-Qiao-Qing-Re Tablets in Treating Fever by Zerong Cai, Pei Liu, Wenjun Liu, Mengyu Qian, Liang Cao, Yumei Hu and Zhenzhong Wang in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251359862 - Supplemental material for Combining UPLC-Q-TOF-MS/MS with Network Pharmacology and Molecular Docking to Explore the Potential Mechanism of Yin-Qiao-Qing-Re Tablets in Treating Fever
Supplemental material, sj-docx-2-npx-10.1177_1934578X251359862 for Combining UPLC-Q-TOF-MS/MS with Network Pharmacology and Molecular Docking to Explore the Potential Mechanism of Yin-Qiao-Qing-Re Tablets in Treating Fever by Zerong Cai, Pei Liu, Wenjun Liu, Mengyu Qian, Liang Cao, Yumei Hu and Zhenzhong Wang in Natural Product Communications
Supplemental Material
sj-doc-3-npx-10.1177_1934578X251359862 - Supplemental material for Combining UPLC-Q-TOF-MS/MS with Network Pharmacology and Molecular Docking to Explore the Potential Mechanism of Yin-Qiao-Qing-Re Tablets in Treating Fever
Supplemental material, sj-doc-3-npx-10.1177_1934578X251359862 for Combining UPLC-Q-TOF-MS/MS with Network Pharmacology and Molecular Docking to Explore the Potential Mechanism of Yin-Qiao-Qing-Re Tablets in Treating Fever by Zerong Cai, Pei Liu, Wenjun Liu, Mengyu Qian, Liang Cao, Yumei Hu and Zhenzhong Wang in Natural Product Communications
Footnotes
Ethical Considerations
Ethical approval to report this case was obtained from the Institutional Animal Care and Use Committee of Kanion Pharmaceutical Co., Ltd (Approval No. 2023111420).
Author Contributions
Zerong Cai: Methodology, Investigation, Formal analysis, Writing-original draft, Validation, Visualization. Pei Liu: Investigation, Formal analysis, Writing-editing. Wenjun Liu: Investigation, Resources, Software. Mengyu Qian: Data curation. Liang Cao: Project administration. Yumei Hu: Conceptualization, Validation, Writing-review and editing. Zhenzhong Wang: Conceptualization, Funding acquisition, Supervision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the grants from the Basic Research Program of the Natural Science Fund—Frontier Leading Technology Basic Research Special Project (No. SBK2023050003).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Animal Rights
All procedures in this study were conducted in accordance with the Institutional Animal Care and Use Committee of Kanion Pharmaceutical Co., Ltd.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
