Abstract
Objective
To evaluate the underlying pharmacodynamics and therapeutic mechanism of Shaofuzhuyu Decoction (SFZYD) in treating Endometriosis (EMs).
Methods
Ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS) was employed to identify prototype ingredients of SFZYD in serum samples of EMs model rats. SFZYD was screened by Network Pharmacology for potential bioactive components, targets and key pathways in vivo according to the prototype components in blood. Subsequently, the binding activities of the potential bioactive compounds and key targets were tested by molecular docking techniques.
Results
170 components of SFZYD were characterized in vitro using UPLC-Q-TOF-MS, and a total of 40 prototype ingredients of SFZYD were identified in rat serum. Network pharmacology predicted 90 protein targets and 193 pathways associated with EMs based on these prototypes. Five components of SFZYD were screened as potential biological activities, and four core targets were identified, these components were trans-4-hydroxycinnamic acid, ethyl p-methoxycinnamate, ferulic acid, protoferulic acid, and 3,5-O-dicaffeoylquinic acid, while the identified core targets were ESR1, EGFR, SRC, and MAPK1. Vigorous binding activities between potential bioactive components and the core targets were demonstrated by molecular docking studies.
Conclusion
The present study elucidated that 13 active components in SFZYD act to treat EMs by regulating four core targets, ESR1, EGFR, SRC and MAPK1, through multiple signalling pathways, and have multi-target, multi-component and synergistic effects, which may serve as a reference and basis for the development of new drugs for treating EMs.
Keywords
Introduction
Endometriosis (EMs) is a chronic condition in which tissue similar to the lining inside the uterus, known as the endometrium, grows outside the uterus. This can cause a range of symptoms including pel-vic pain, heavy menstrual bleeding, painful periods, infertility, and fatigue. 1 The exact cause of endometriosis is not fully understood, but it is believed to be influenced by a combination of genetic, hormonal, and immune factors. There is currently no cure for endometriosis, but there are several treatment options available to manage the symptoms and improve quality of life for those affected by the condition. Treatment options for endometriosis may include: Pain medication: Over-the-counter pain relievers such as ibuprofen or naproxen may help to alleviate the pelvic pain and cramping associated with endometriosis; Hormone therapy: This may include hormonal birth control (such as pills, patches, or rings), progestin therapy, gonadotropin-releasing hormone (GnRH) agonists, or danazol to help regulate hormonal imbalances and reduce the growth of endometrial tissue; Surgery: For severe cases of endometriosis, surgery may be recommended to remove endometrial tissue growths (endometriomas) and scar tissue (adhesions). 2 In some cases, a hysterectomy (removal of the uterus) may be recommended as a last resort for women who do not plan to have children. Lifestyle changes: Eating a healthy diet, exercising regularly, and managing stress may help to alleviate symptoms and improve overall well-being for women with EMs. It is important for women with endometriosis to work closely with their healthcare provider to develop a treatment plan that is tailored to their individual needs and preferences. Improving treatment options to enhance efficacy while minimizing recurrence rates is paramount. Several clinical studies have demonstrated that traditional Chinese medicine (TCM) could achieve these goals and is well-tolerated by patients. 3
The Shaofuzhuyu Decoction (SFZYD) formula, originally recorded in “Yilin Gaicuo”, a monograph compiled by the Qing Dynasty Wang Qingren, is considered to be a classic and effective formula for treating gynaecological disorders such as primary dysmenorrhoea, EMs, tubal obstruction infertility, uterine fibroids, ovarian cysts etc, which is composed of Angelica sinensis (Oliv.) Diels (Danggui), Paeonia lactiflora Pall. (Chishao), Ligusticum chuanxiong S.H.Qiu, Y.Q.Zeng, K.Y.Pan, Y.C.Tang & J.M.Xu (Chuanxiong), Trogopterori Faeces (Wulingzhi), Typha angustifolia L. (Puhuang), Corydalis yanhusuo (Y.H.Chou & Chun C.Hsu) W.T.Wang ex Z.Y.Su & C.Y.Wu (Yanhusuo), Commiphora myrrha (Nees) Engl. (Moyao), Foeniculum vulgare Mill. (Xiaohuixiang), Zingiber officinale Roscoe. (Ganjiang), and Cinnamomum cassia (L.) J.Presl (Rougui). It effectively dispels blood stasis and warming meridians, regulates Qi, and relieves pain. Clinically, SFZYD can effectively treat EMs, but the pharmacological basis and mechanism of action remain unclear.4,5
The Chinese herbal formulas have the characteristics of “multi-components, multi-targets and multi-pathways”, which also brings excellent difficulties in revealing the mechanism of action. Combining of serum pharmacochemistry and network pharmacology is considered a promising method for deciphering complex systems.6,7 Most network pharmacology approaches currently obtain molecular information about drugs from public databases to investigate their corresponding targets. However, the presence of specific components in the formulas that do not reach the minimum level of their efficacy drives bias in the results predicted by the databases. Serum pharmacology is a scientific method that employs modern techniques to identify and analyze potential pharmacodynamic components absorbed into the bloodstream and subsequently determine the active ingredient(s) of a formula. 8 Replacing database mining in network pharmacology research with a combined serum pharmacochemical approach to investigate active ingredients and their mechanisms in Chinese herbal formulas has proven a new and effective analytical method.9,10
Therefore, to evaluate the underlying pharmacodynamics and therapeutic mechanism of SFZYD in treating Ems the prototypical components absorbed into the blood were identified using UPLC-Q-TOF-MS analysis, combined with a network pharmacology strategy to predict the target of action in this study.
Results
Results of Ectopic Tissue Volume
Figure 1A displayed the findings on ectopic tissue volume, revealing notable ectopic tissue presence in the uterus of the model rats. In contrast, the the SFZYD group rats exhibited a notable decrease in ectopic tissue compared to the model group.
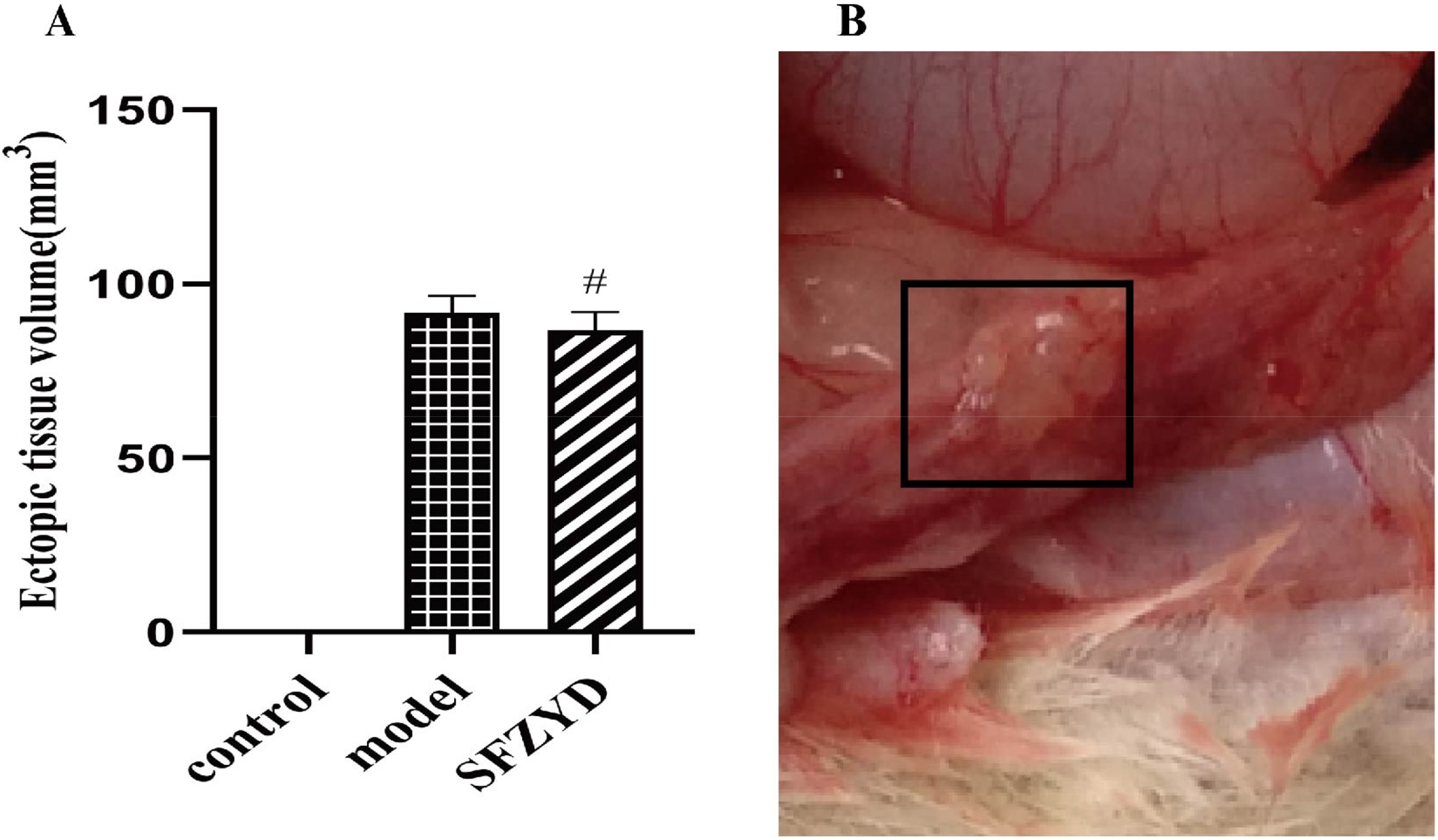
Effect of SFZYD on ectopic tissue volume in EMs rats. (A) The ectopic tissue volume; (B) Ectopic tissue photo in uterus of the model rats. Notes: Values are expressed as the mean ± SD (compared with the mode group: # p < 0.05).
Results of serum Pharmacochemistry Analysis
The Chemical Components of SFZYD
The composition of SFZYD evaluated by HPLC-Q-TOF-MS, in the positive ion mode, the compounds detected in the SFZYD sample spectra were represented mainly by quasi-molecular ions [M + H]+, while in the negative ion mode, the detected compounds were represented mainly by quasi-molecular ions [M-H]−. 170 components were characterized in the positive and negative ion modes (Table S1, Figure 2).
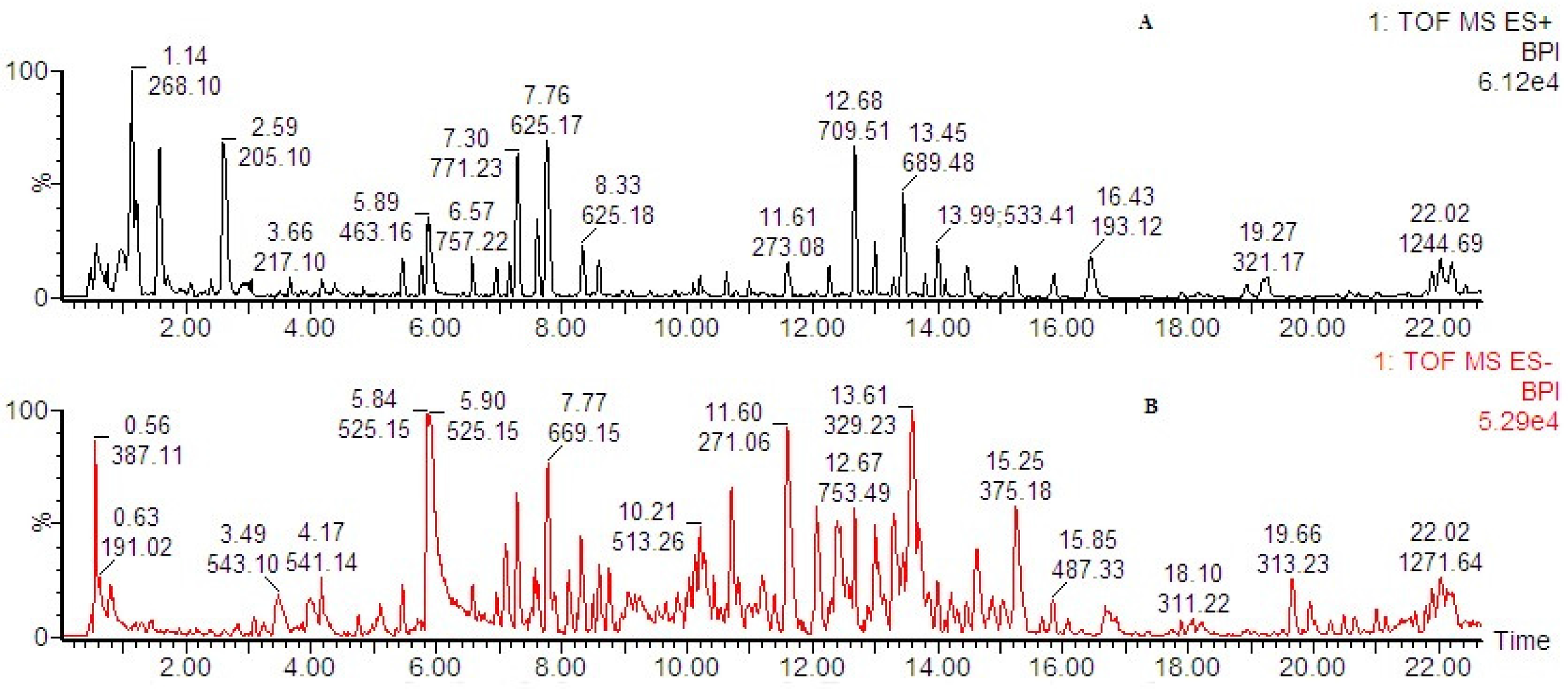
The composition of SFZYD evaluated by HPLC-Q-TOF-MS/MS. (A) The base peak chromatogram in positive ion mode; (B) The base peak chromatogram in negative ion mode.
Prototype Ingredients Identification
A total of 40 prototype ingredients were identified in the serum samples after oral administration of SFZYD in rats (Table S2). Among them, 8 components were derived from Chishao, 8 components from Chuanxiong, 6 components from Danggui, 3 components from Ganjiang, 4 components from Moyao, 4 components from Gepuhuang, 5 components from Rougui, 9 components from Wulingzhi, 7 components from Xiaohuixiang, and 4 components from Yanhusuo (Figure 3A-B).
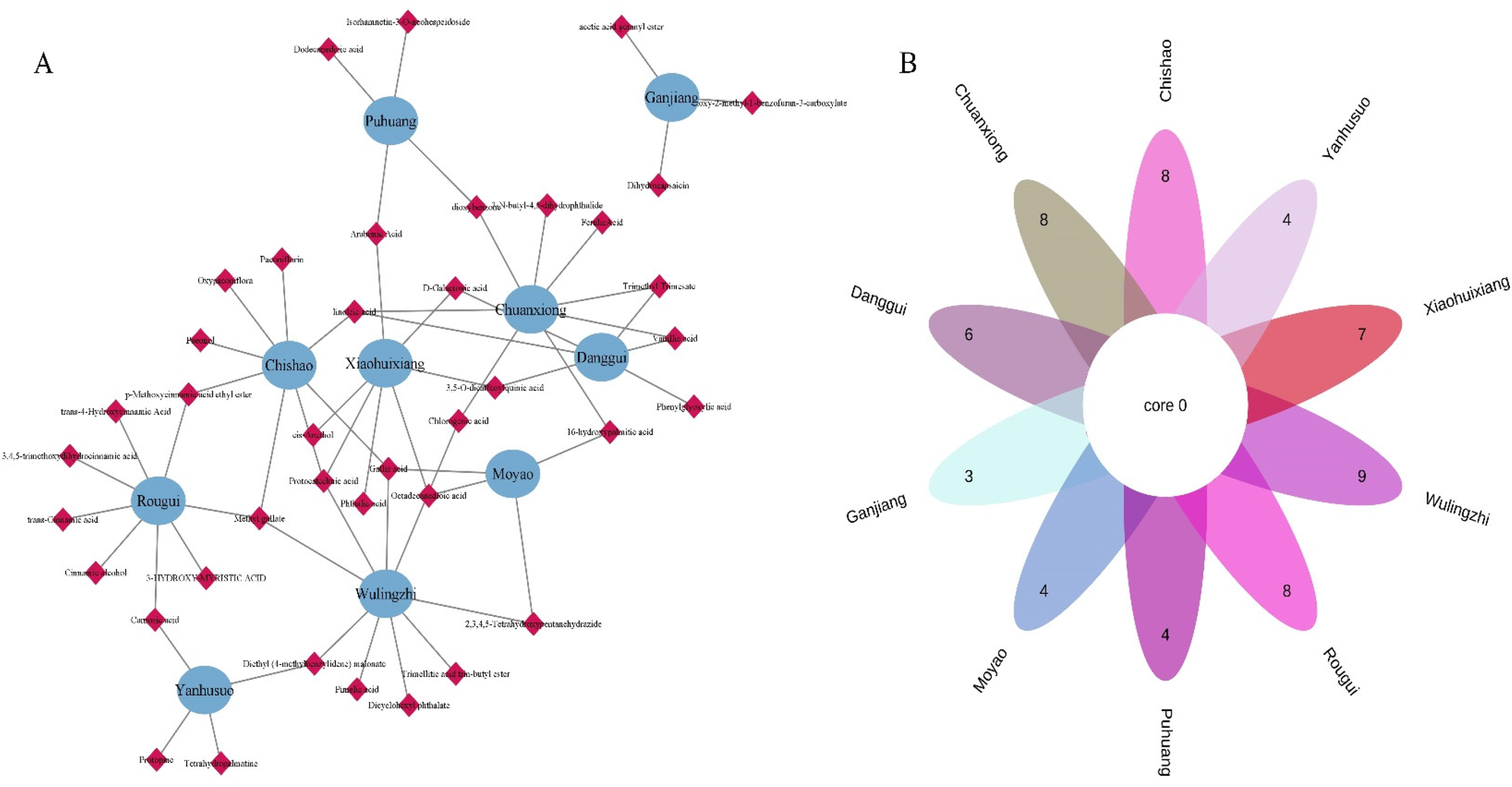
Prototype ingredients in rat serum samples intervened by SFZYD. (A) Network diagram of serum prototype ingredients and SFZYD components; (B) Venn diagram of serum prototype ingredients and SFZYD components.
Results of Network Pharmacology Analysis
The Construction of a Components-Protein-Protein Interaction (C-PPI) Network of Intersecting Target Proteins and Components
A total of 954 SFZYD targets and 4108 EMs-related targets were retrieved form the database. Finally, 303 intersection targets were obtained (Figure 4A). A PPI network of 303 nodes and 6861 edges was formed through the String database (Figure S1). The network was then imported into Cytoscape 3.10.1 software, and the network nodes were optimized based on their Degree values (Degree > 8). This resulted in a network graph consisting of 90 targets with 90 nodes and 515 edges (Figure 4B).

Network pharmacology analysis of SFZYD with endometriosis.(A) Venn diagram of SFZYD potential targets and PF-related genes; (B) The top 90 targets of PPI network analysis; (C) Network diagram of 40 blood prototype ingredients and 90 targets; (D) Network diagram of top 25 nodes targets and their corresponding blood component.
Construction of the Blood Component-Potential Pharmacological Target Network
A network consisting of 40 blood prototype ingredients and 90 targets was constructed, with 130 nodes and 537 edges (Figure 4C). Based on the Degree values, the top 10 blood prototype ingredients in the network were ranked as trans-4-hydroxycinnamic Acid, Isorhamnetin-3-O-neohespeidoside, Ferulic Acid, cis-anethol, Protopine, p-methoxycinnamic acid ethyl ester, 16-hydroxypalmitic Acid, Cinnamic alcohol, Ethyl 5-methoxy-2-methyl-1-benzofuran-3-carboxylate, and 3,5-O-dicaffeoylquinic Acid. These components may play a more critical role in the treatment of endometriosis.
The 90 targets were subjected to network analysis and filtered using the Degree topological parameter. Then, 25 targets with high protein interaction relationships (Degree > 18) were screened. A network was then constructed for these 25 targets and their corresponding blood components, and the cross-targets were sorted based on their Degree values. The four key targets with the highest association were ESR1, EGFR, SRC, and MAPK1, which may serve as main action sites (Figure 4D). Thirteen blood components belonging to 8 herbal medicines were selected based on their high association with these four key targets (Figure 5A). Five overlapping components were then identified from the 13 highly associated blood components, and the top 10 prototype blood ingredients were screened. They were trans-4-hydroxycinnamic Acid, p-methoxycinnamic acid ethyl ester, Ferulic Acid, Protopine, and 3,5-O-dicaffeoylquinic Acid, and were considered to be potential bioactive components for the treatment of endometriosis using SFZYD.

Network diagram of SFZYD with endometriosis. (A) Network diagram of SFZYD, blood components, and key targets; (B) GO enrichment analysis; (C) KEGG pathway enrichment analysis.
GO and KEGG Enrichment Analysis
The GO enrichment analysis results were shown in Figure 5B. The KEGG pathway enrichment analysis obtained 174 signalling pathways (P < 0.01). The signal pathways mainly included TNF signalling pathway, IL-17 signalling pathway, Inflammatory mediator regulation of TRP channels, NOD-like receptor (NLR) signalling pathway, PI3 K/Akt signalling pathway, MAPK signalling pathway, EGFR tyrosine kinase inhibitor resistance, Chemokine signalling pathway, Estrogen signalling pathway, VEGF signalling pathway, mTOR signalling pathway, cAMP signalling pathway, Progesterone-mediated oocyte maturation, NF-κB signalling pathway, TGF-β signalling pathway, Vascular smooth muscle contraction, Steroid hormone biosynthesis, AMPK signalling pathway, Wnt signalling pathway, and Arachidonic acid metabolism (Figure 5C). The above results suggested that SFZYD components in blood regulate multiple signalling pathways simultaneously, and the inflammatory response and the cell proliferation and apoptosis were the pathways that were mainly affected.
Molecular Docking
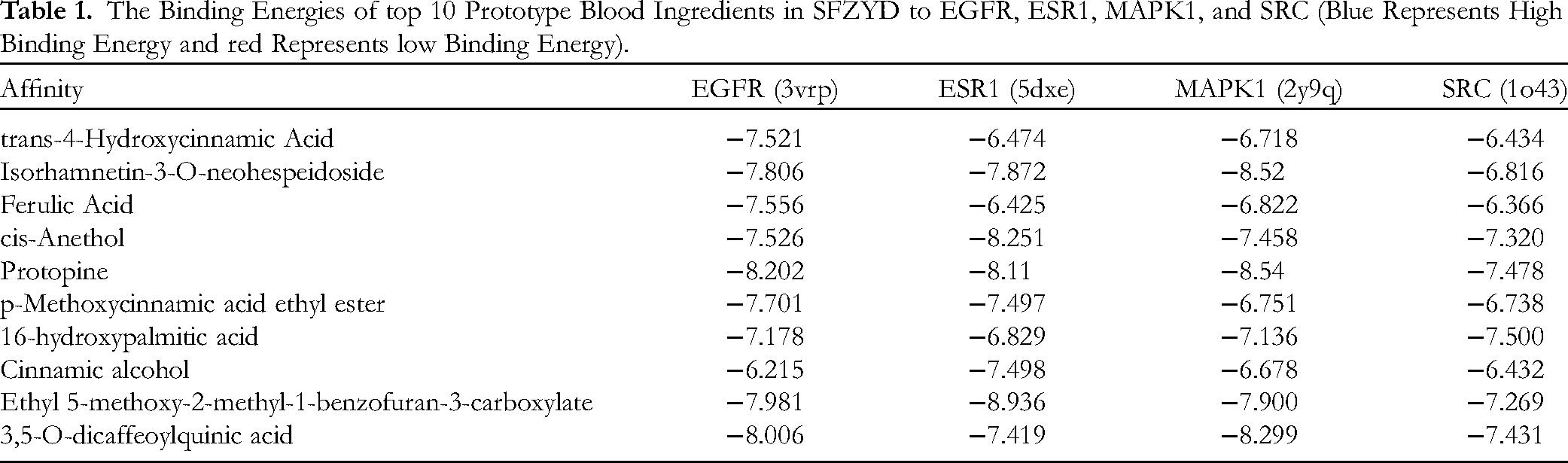
The result showed that the top 10 prototype blood ingredients in SFZYD had good binding abilities with the 4 core targets. The cis-Anethol, Protopine, Ethyl 5-methoxy-2-methyl-1-benzofuran-3-carboxylate, and 3,5-O-dicaffeoylquinic acid had vigorous binding activities with the 4 core targets. The best-performing group regarding binding energy was Ethyl 5-methoxy-2-methyl-1-benzofuran-3-carboxylate × ESR1 (Affinity = -8.936 kcal/mol). The detailed results can be found in Table 1. In order from low to high binding performance, the docking results of the selected top 4 molecules were visualized and validated using PyMOL software: Ethyl 5-methoxy-2-methyl-1-benzofuran-3-carboxylate × ESR1, Protopine × MAPK1, Isorhamnetin-3-O-neohespeidoside × MAPK1, 3,5-O-dicaffeoylquinic acid × MAPK1, the formation of hydrogen bonds between receptor proteins and ligand small molecules was also observed (Figure 6).
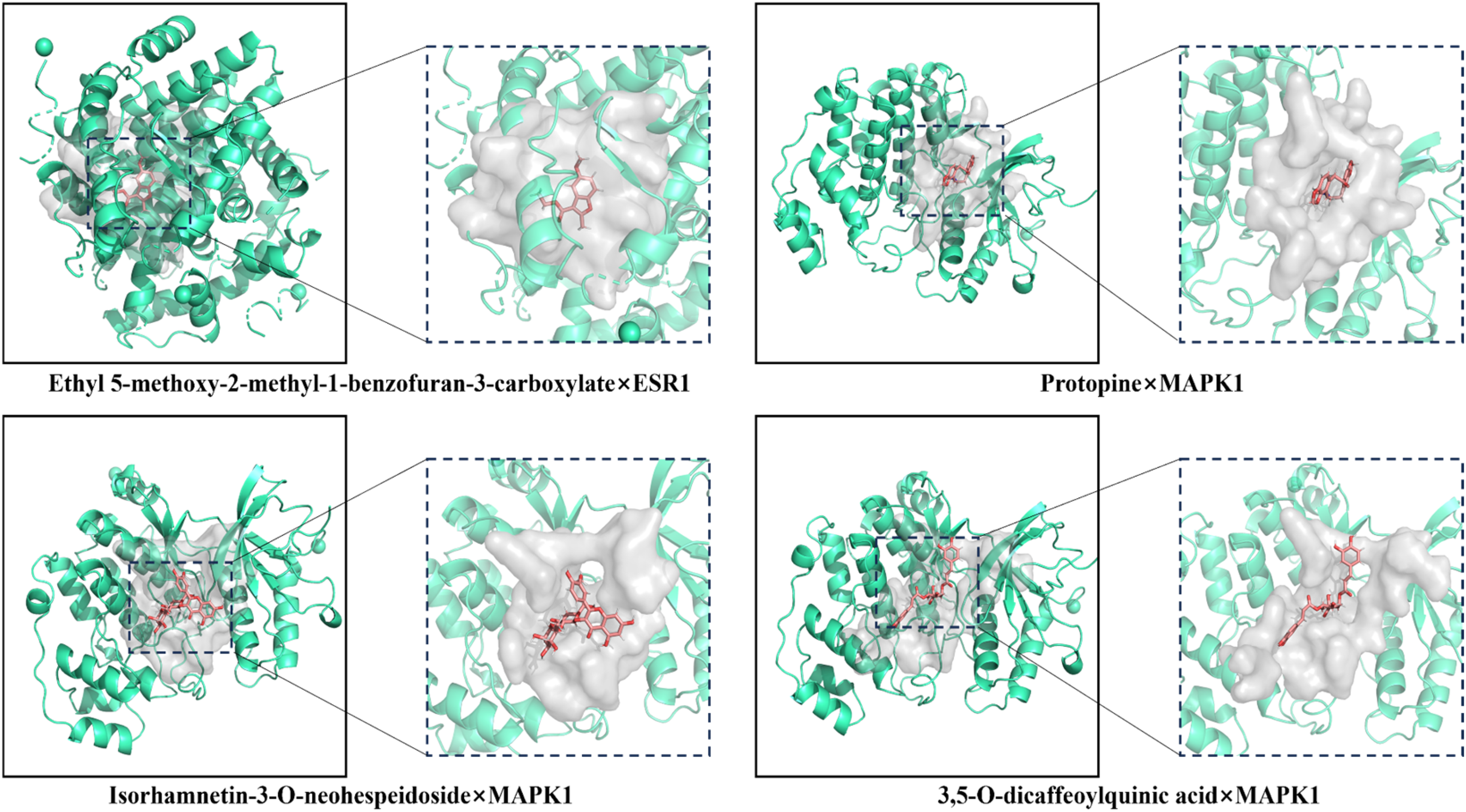
Visualisation of molecular docking results.
The Binding Energies of top 10 Prototype Blood Ingredients in SFZYD to EGFR, ESR1, MAPK1, and SRC (Blue Represents High Binding Energy and red Represents low Binding Energy).
Discussion
According to Traditional Chinese Medicine (TCM), EMS belongs to the blood stasis syndrome and is usually treated by activating blood circulation and removing blood stasis. 11 Several generations of medical practitioners have used SFZYD, has the effect of activating blood circulation, eliminating blood stasis, warming meridian and relieving pain, and is known as “the best formula for regulation of menstruation and prevention of miscarriage”. It is mainly used in the treatment of chronic pelvic inflammatory disease, primary dysmenorrhoea, endometriosis, tubal obstruction infertility, uterine fibroids and other conditions. In order to explore the material basis and mechanism of SFZYD for treating EMs, the present study was conducted using serum pharmacochemistry and network pharmacology methods for prototype ingredients annotation, target prediction, protein association and molecular docking.
40 prototypical ingredients of SFZYD were identified from the serum samples, of which 8 components were from Chishao, 8 components were from Chuanxiong, 6 components were from Danggui, 3 components were from Ganjiang, 4 components were from Moyao, 4 components were from Puhuang, 5 components were from Guangui, 9 components were from Wulingzhi, 7 components were from Xiaohuixiang, 4 components were from Yanhusuo. Xiaohuixiang, Ganjiang and Guangui disperse cold by warming the meridian and reaching the lower Jiao; Yanhusuo and Moyao promote qi and dissipate blood stasis, subdue swellings and relieve pain; Puhuang and Wulingzhi activate blood circulation and promote blood stasis, and dissipate knots and relieve pain; Danggui and Chuanxiong and Chishao are used to activate blood circulation and promote qi circulation and disperse stagnation and regulate menstruation. The whole formula considers both qi and blood, warms and passes both. It embodies the characteristics of multi-ingredients acting together in a Chinese herbal formula.
Current theories of the pathogenesis of EMs include retrograde menstruation, Müllerian remnants, and coelomic metaplasia, but they do not fully explain all the different types of endometriosis. The retrograde menstruation hypothesis is one of the most convincing theories, in which fragments of endometrium travel retrogradely through the fallopian tubes to the pelvis, where they translocated and localized in the peritoneum and abdominal organs and led to chronic inflammation and adhesion formation. 12
Ferulic acid, protopine, acid trans-4-hydroxycinnamic, p-methoxycinnamic acid ethyl ester, and 3,5-O-dicaffeoylquinic acid were screened as possible potentially biologically active components of the SFZYD by using the “Into serum components - Action target - Key pathway” analysis method and the four key targets with the highest association were ESR1, EGFR, SRC, and MAPK1. In endometriotic stromal cells, the expression level of ESR1 was significantly decreased, which in turn altered the mode of action of estrogen and affected a variety of pathological processes in endometriosis, including apoptosis, proliferation, invasion, and inflammation. 13 In clinical samples, ESR1 was found to be able to keep endometriosis patients in an immunosuppressed state by modulating endoplasmic reticulum stress-related gene expression. 14 Angiogenesis may be a key process in the pathogenesis of endometriosis, and the EGFR may be a potentially valuable marker for establishing genetic susceptibility to endometriosis, at least in Chinese women. 15 Abnormal SRC expression may induce apoptosis, pyroptosis and endoplasmic reticulum stress signalling in EMs lesions. 16 The MAPK1 expression is significantly elevated in the endometrium of patients with endometriosis in vivo. 17 Molecular docking showed that the top 10 prototype blood ingredients, including the above five potentially biologically active components, had good binding ability to the four key targets, validating the predicted mechanism of action. It was suggested that these active ingredients were synergistic and could work together to produce an effect. This synergy may be an essential factor in the efficacy of SFZYD in treating EM. Ferulic acid is a common active ingredient in herbal medicine. To date, ferulic acid has significant roles in inflammation, oxidative stress, vascular endothelial damage, fibrosis and platelet aggregation. 18 It has also been shown that ferulic acid, in combination with chasteberry glycoside and tetrahydropalmatine, inhibits EMs through MMP/TIMP signalling, and in combination with chuanxiongzine and tetrahydropalmatine exerts anti-proliferative effects in EMs through modulation of the Notch pathway.19,20 Furthermore, trans-4-hydroxycinnamic acid from the lees of Bokbunja wine was found to have potent anticoagulant and antiplatelet effects. 21 3,5-O-dicaffeoylquinic acid has been suggested as a potential inflammatory pain reliever. 22 3,5-dicaffeoylquinic acidis is the primary ingredient of SiMiaoYongAn Decoction, which reduces myocardial inflammation by inhibiting of autophagy and apoptosis. 23 Ethyl p-methoxycinnamate (ethyl-p-methoxycinnamate) in Kaempferia galanga has been shown to inhibit inflammation by inhibiting IL-1 and TNF-α. 24 The main active ingredient in yanhusuo are alkaloids, including protoxanthin, protopine and apocynin alkaloids. 25
The TNF signalling pathway, IL-17 signalling pathway, NLR signalling pathway, MAPK signalling pathway, the PI3 K/Akt signalling pathway and inflammatory mediator regulation of TRP channels were identified as potential mechanistic pathways through correlation analysis of EMs-related protein targets of prototypical serum ingredients. The TNF signalling pathway, IL-17 signalling pathway, PI3 K/Akt signalling, pathway MAPK signalling pathway and the regulation of TRP channels by inflammatory mediators are closely related to inflammation, and the crosstalk between MAPK signalling pathway, PI3 K/Akt signalling pathway and NLR signalling pathway regulate apoptosis and transplantation. Many TCM herbs have been shown to treat EMs through the above signalling pathways. Imperatorin can inhibit the secretion of TNF-α and IL-6 by inhibiting the activation of the PI3 K/Akt/NF-κB pathway, thus acting as an anti-endometriosis agent. 26 Aberrant activation of MAPK during migration, implantation, invasion of pelvic structures, proliferation and apoptosis leads to the formation of endometriosis and worsens the condition of patients with endometriosis. 27 The traditional Chinese medicine Danshen (Salvia miltiorrhiza Bge) activates blood circulation, relieves pain, calms the heart and the mind, etc Tanshinone IIA is the active ingredient in Danshen. Studies have shown that Tanshinone IIA can regulate the adhesion, invasion and angiogenesis of ectopic endothelium by blocking the activation of the PI3 K/Akt/mTOR signalling pathway, thereby inhibiting the formation of ectopic lesions. 28 Hyperestrogenic production is a prominent endocrine feature of patients with EMs, and hormone therapy aims to reduce endogenous ovarian estrogen production. There are two subtypes of estrogen receptors (ERs), estrogen receptor α and estrogen receptor β (ERα and ERβ), which are encoded by the estrogen receptor 1 (ESR1) and 2 (ESR2) genes, respectively. 29 These observations prompted consideration of the feasibility of MAPK/ERK and PI3 K/AKT pathway-specific inhibitors for treating EMs. 30
The strength of this study is that by revealing the material basis and mechanism of SFZYD for the treatment of EMs, which provides a reference and basis for the subsequent development of new drugs for the treatment of EMs. However, the interactions between the active ingredients in the results intervene on the efficacy of the drug, and the effects of the interactions between the core targets on the disease process are not clear remains a limitation of this study. Therefore, in vivo/in vitro experiments are needed to further validate the efficacy of the active ingredients with the core targets for the treatment of EMs.
Conclusions
In summary, five components of SFZYD were screened as potential biological activities, these components were trans-4-hydroxycinnamic acid, ethyl p-methoxycinnamate, ferulic acid, protofer-ulic acid, and 3,5-O-dicaffeoylquinic acid, and ESR1, EGFR, SRC, and MAPK1 were identified as core targets in treating EMs. It plays a role in the treatment of EMs by regulating pathways such as TNF signalling pathway, IL-17 signalling pathway, NOD-like receptor signalling pathway, MAPK signalling pathway, Inflammatory mediator regulation of TRP channels, PI3K-Akt signalling pathway. It has the characteristics of multiple targets, multiple components, and synergistic effects in the treatment of EMs. This study provides a reference and basis for the subsequent development of new drugs for the treatment of EMs.
Materials and Methods
Materials and Reagents
HPLC grade acetonitrile and methanol were purchased from Fisher Scientific Corporation (Waltham, USA), while formic acid and leucine enkephalin were purchased from Sigma–Aldrich (St Louis, MO, USA). Pure distilled water used for LC-MS analysis was purchased from Watsons Water (Guangzhou, China). The Shaofuzhuyu Decoction used were produced by Beijing Tongrentang Pharmacy Harbin Branch (Harbin, China) and were authenticated by Prof. Sun Huifeng, Department of Biomedicine, Heilongjiang University of Traditional Chinese Medicine. The specimens of these ten drugs were stored in National Key Laboratory of Innovation in Integration of Traditional Chinese Medicine and Western Medicine, labelled SFZYD-20190514-001, SFZYD-20190514-002, SFZYD-20190514-003, SFZYD-20190514-004, SFZYD-20190514-005, SFZYD-20190514-006, SFZYD-20190514-007, SFZYD-20190514-008, SFZYD-20190514-009, SFZYD-20190514-010, respectively.
Experimental Animals
SPF Wistar rats (female, 200-220 g) (Permit No. SCXK2014-004) were acclimated to a constant and humid environment for one week prior to experimental processing with the support of the Drug Safety Evaluation Center of Heilongjiang University of Chinese Medicine (Harbin, China). The construction of the rat model of EMs using a combination of autologous transplantation and ice water immersion has been described in a previous study. 31 Roughly modelling method, the rats were anesthetized and ligated to the right uterus, the middle of the uterus removed, then cut into two pieces and sewn into the left abdominal wall and uterus, respectively. Meanwhile, the rats were placed in an ice-water mixture of 0–1 °C daily for 8–10 min for two weeks before surgery. The procedure was replicated in the third week following the surgery. Following the completion of the model, the medication treatment was given over 4 weeks. The experiment was approved under the guidance of the Helsinki Declaration by the Ethics Committee of Heilongjiang University of Chinese Medicine (Approval No.2019042501).
SFZYD Sample Preparation
According to Danggui, Chishao, Chuanxiong, Wulingzhi, Puhuang, Yanhusuo, Moyao, Xiaohuixiang, Ganjiang, and Rougui in a proportion of 3: 2: 2: 2: 3: 1: 2: 1.5: 3: 3, 984 g of drugs for SFZYD were weighed and dissolved in 10-fold water to extract twice for 40 min each time. SFZYD lyophilized powder was obtained by freeze-drying with a yield of 30.92%.
The content of gallic acid, oxypaeoniflora, paeoniflorin, isorhamnetin-3-O-neohespeidoside, ferulic acid, typhaneoside, senkyunolide-I, naringenin and their fingerprints were considered to be the quality indicators of the extracts. The mean contents of the above components in 10 batches of lyophilized powder were 6.920, 2.131, 1.565, 0.905, 0.792, 0.318, 0.577, and 0.574, respectively. The fingerprint similarity of 10 batches of SFZYD is more significant than 0.9. The samples prepared are consistent and controllable.
Animal Administration and Collection of serum Samples
The EMs model rats were randomly divided into the model group and the SFZYD group (n = 10). A sham surgery group was also established as a control group. Rats in the SFZYD group were given SFZYD freeze-dried powder by gavage at a dose of 2.826 g/kg, and other groups were given distilled water by gavage at a dose of 10 ml/kg for 4 weeks. The dose of SFZYD freeze-dried powder was double the clinical equivalent dose. After 60 min of the last administration, the rats were anaesthetised with 2% pentobarbital sodium and 5 ml of blood was collected via the hepatic portal vein and centrifuged at 3000 rpm/min at 4 °C for 15 min after standing for 30 min to obtain serum. After excising the ectopic tissue, measurements of its length, width, and height were taken using a vernier calliper, followed by multiplying the three indices by 0.52 to determine the volume. The urine samples of rats were collected and stored at −80 °C.
Serum Pharmacochemistry Analysis
Preparation of Analytical Samples
0.5 g of SFZYD powder was weighed into a stoppered conical flask, and 10 mL of 50% methanol was added, weighed, and ultrasonicated for 30 min. Remove from the flask, cool, make up for the mass loss with solvent, and then weighed, centrifuged at 3000 r/min for 10 min, and 2 μL of the supernatant was filtered through a 0.22 μm membrane, and then used in the UPLC.
1 ml of blank control serum and drug-containing serum were mixed with 10-fold volume of methanol, then centrifuged at 3000 rpm/min for 15 min at 4 °C, and the supernatant was dried with nitrogen. The residue was re-dissolved with 200 μL of methanol, centrifuged for 10 min, and then filtered through 0.22 μm membrane for UPLC-MS analysis.
UPLC-Q-TOF-MS/MS Conditions for the Identification of SFZYD Compounds In Vitro and In Vivo
A Waters Acquity UPLC system combined with a Waters ESI-Q-TOF/MS controlled by Masslynx 4.1 with an Acquity UPLCTM Phenomenex column (1.7μm, 2.1 mm × 50 mm, Waters, USA) was used to acquire data of SFZYD samples at 30 °C. The 0.1% formic acid-acetonitrile was used as mobile phase A, and the 0.1% formic acid-water was used as mobile phase B. The flow rate of the mobile phase was 0.4 mL/min with a gradient of 0–3 min at 2–8% A, 3–8 min at 8–20% A, 8–12 min at 20–30% A, 12–18 min at 30–40% A, 18–24 min at 40–70% A, 24–27 min at 70–100% A, 27–30 min at 100% A, and 30–30.1 min at 100–2% A.
In positive ion mode, the capillary voltage was 3.0 kV, and the cone voltage was 40 V, whereas in negative ion mode, the capillary and cone voltages were 2.4 kV and 30 V, respectively. In both ion modes, the solvation gas flow rate was 800 L/h, the source temperature was 110 °C, the solvation temperature was 300 °C, and the mass scanning range was from m/z 100 to 1500 Da. The low energy setting was 10–30 eV, and the high energy setting was 30–50 eV when acquiring MS/MS fragments in centre mode. MS/MS fragments were acquired in centre mode with low energy settings of 10–30 eV and high energy settings of 30–50 eV.
Data Processing
Identification of Drug Components in Vitro
The Masslynx V4.1 workstation was used to analyze the chromatographic peaks of the SFZYD sample. The primary mass spectra obtained the retention time and accurate molecular weight of each compound. The Elemental Composition tool in Masslynx was used to analyze the elemental composition and calculate the degree of unsaturation. Potential elemental compositions were determined using the mass-to-charge ratio from the mass spectrometry detection, and possible chemical structures were deduced by analyzing the secondary ion fragments of the chromatographic peaks. These were compared with the relevant literature and the Human Metabolome Database (HMDB) (https://hmdb.ca/) to confirm the molecular structure of the target compound.
Identification of Drug Prototype Ingredients In Serum
A baseline background was established using the “Strips” tool on the Masslynx V4.1 workstation by utilizing a blank serum sample spectrum. The resulting chromatogram was generated by subtracting the blank serum from the drug-containing serum, which was subsequently confirmed manually before further investigation. This process identifies chromatographic peaks in the drug-containing serum absent in the blank serum, indicating potential SFZYD components in the blood. The retention time, precise molecular weight and fragmentation ions of the identified migrating components in the blood were compared to relevant information on SFZYD's chemical composition profile. The compounds that corresponded in both datasets were regarded as the prototype ingredients of SFZYD that access the bloodstream.
Network Pharmacology Analysis
Collection of Targets of Prototype Ingredients In Serum and EMs
The SwissTargetPrediction database (http://www.swisstargetprediction.ch/) was used to predict the target proteins of the components entering the bloodstream in SFZYD. To procure disease-associated target proteins, the OMIM (https://www.ncbi.nlm.nih.gov/omim/) and GeneCards (http://www.genecards.org) databases were individually examined for “Endometriosis”. These findings were merged, duplicates were excluded, resulting in a directory of target proteins linked to endometriosis. The proteins under consideration were standardized by utilizing the UniProt database (https://www.uniprot.org). The Venny2.1 tool (https://bioinfogp.cnb.csic.es/tools/venny/index.html) was employed to illustrate and obtain the intersection of components entering the bloodstream in SFZYD and endometriosis, which could potentially serve as targets for treating endometriosis with SFZYD.
Protein-Protein Interaction (PPI) Network Construction and Screening of Core Targets and Potential Bioactive Components
The PPI analysis of overlapping targets was performed using the STRING database (https://string-db.org/). The corresponding “tsv” files were obtained and imported into Cytoscape 3.10.0 software to analyse and visualise the complex PPI network. In this study, the Degree of each node was calculated using Cytoscape software to select the core target genes based on their degree values. The “Network Analyzer” function in Cytoscape 3.10.0 software (https://cytoscape.org) was used to construct and analyze the interaction target network of the components in the serum. The nodes represented the chemical components of the drug and potential target proteins, while the edges represented the connections between SFZYD components and potential targets. The SFZYD components were sorted based on their Degree values. Potential bioactive components were selected accordingly.
Signal Pathways and Functional Enrichment Analysis
The gene ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis was performed using the Metascape database (http://metascape.org/gp/index.html). A significance level of p < 0.01 was set in the Metascape database for GO and KEGG analyses. The results of the GO analysis and KEGG enrichment analysis were visualized using bar graphs and bubble plots, respectively, using the bioinformatics online platform (http://www.bioinformatics.com.cn).
Molecular Docking Verification Mechanisms
The key components and core target proteins were input as ligands and protein receptors into the DockThor website (https://dockthor.lncc.br/v2/) to obtain information on binding energy and other relevant parameters for the corresponding combinations. The four combinations with the lowest molecular docking binding energy were selected as the optimal docking results. The PyMOL 2.6 software was used to represent the optimal docking results visually.
Statistical Analysis
Data analysis and image acquisition were performed using GraphPad Prism version 8.0.2 (GraphPad Software, CA, USA). All data were expressed as mean ± standard deviation. One-way analysis of variance (ANOVA) was performed to analyze the differences in the measured data. p < 0.05 was considered statistically significant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241274177 - Supplemental material for Identifying Potential Bioactive Components and Targets of Shaofuzhuyu Decoction in Treating Endometriosis Using serum Pharmacochemistry and Network Pharmacology
Supplemental material, sj-docx-1-npx-10.1177_1934578X241274177 for Identifying Potential Bioactive Components and Targets of Shaofuzhuyu Decoction in Treating Endometriosis Using serum Pharmacochemistry and Network Pharmacology by Qiyao Li, Jing Liu, Lin Ding, Dongxia Yang, Chengyu Piao, Yang Yu and Xiuhong Wu in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241274177 - Supplemental material for Identifying Potential Bioactive Components and Targets of Shaofuzhuyu Decoction in Treating Endometriosis Using serum Pharmacochemistry and Network Pharmacology
Supplemental material, sj-docx-2-npx-10.1177_1934578X241274177 for Identifying Potential Bioactive Components and Targets of Shaofuzhuyu Decoction in Treating Endometriosis Using serum Pharmacochemistry and Network Pharmacology by Qiyao Li, Jing Liu, Lin Ding, Dongxia Yang, Chengyu Piao, Yang Yu and Xiuhong Wu in Natural Product Communications
Footnotes
Author Contributions
Q.L.: Validation, Formal analysis, Data curation, Software, Investigation. J.L.: Writing – original draft, Software, Visualization, Formal analysis, Funding acquisition. L.D.: Writing – review & editing, Visualization, Data curation. D.Y.: Supervision, Methodology. C.P.: Software, Validation. Y.Y.: Software, Methodology. X.W.: Conceptualization, Funding acquisition, Methodology, Resources, Writing – review & editing, Project administration. All authors have read and approved the final manuscript.
Data Availability
The data used to support the findings of this study are available from the corresponding authors upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Heilongjiang University of Chinese Medicine (Approval No.2019042501).
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 81573877), Natural Science Foundation of Heilongjiang Province of China (grant numbers JQ2023H003), Heilongjiang University of Chinese Medicine graduate innovative research project (grant numbers 2023yjscx002).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the guidance of the Helsinki Declaration by the Ethics Committee of Heilongjiang University of Chinese Medicine.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
