Abstract
Background
Postmenopausal osteoporosis (PMOP) is characterized by low bone mass and increased bone fragility in postmenopausal women. Shen Gu An capsule (SGA) has been clinically used to treat bone diseases, especially PMOP with kidney Yang deficiency.
Objectives
We aimed to study the effect of SGA on the treatment of PMOP.
Methodology
The key candidate targets and pathways of SGA for treating PMOP were predicted by Network pharmacology analysis. Then, in vivo validation using bilaterally ovariectomized C57BL/6 mice was performed, and the bone quality was analyzed by Micro-CT and histological analysis.
Results
In this study, we obtained 113 active compounds and 82 targets via the database of Traditional Chinese Medicine Systems Pharmacology. Subsequently, compound-target and protein−protein interaction networks were constructed. Then GO and KEGG analysis revealed that NF-κB signaling pathway might be the underlying mechanism as its well-known function in osteoclastogenesis. Furthermore, the in vivo experiments using ovariectomized mice demonstrated that SGA could decrease the number of osteoclasts and preserve bone mass through inhibiting the NF-κB signaling pathway.
Conclusions
SGA could prevent postmenopausal osteoporosis in ovariectomized mice through inhibiting NF-κB signaling pathway. The finding of this study provides more evidence for the clinical application of SGA to treat osteoporosis.
Introduction
Postmenopausal osteoporosis (PMOP) is a systemic metabolic disease with low bone mass and subtle structural degeneration of bone tissue in postmenopausal women, leading to reduced bone strength, and easy to fracture.1,2 According to the current studies, PMOP is mainly associated with the reduction of estrogen synthesis by postmenopausal ovaries.3,4 With the aggravation of population aging, osteoporosis has seriously threatened the health of middle-aged and elderly people, especially women.
The main goals of the treatment of PMOP are to prevent fractures, maintain or increase bone mineral density (BMD), improve physical function, and improve the quality of life of patients. 5 At present, the clinical prevention and treatment of PMOP is basically to consume adequate vitamin D and calcium, and use menopausal hormone therapy and other anti-osteoporosis drugs.6,7 However, menopausal hormone therapy is easy to produce some side effects, such as vaginal bleeding. 8 Other anti-osteoporosis drugs should also be evaluated for contraindications and side effects.9,10
NF-κB signaling pathway plays essential roles in multiple pathological processes. 11 It has been shown to play vital role in bone homeostasis, as the mice with NF-κB1 and NF-κB2 deletion displayed severe osteopetrosis owing to the blocking of osteoclastogenesis. 12 Upon activation of the canonical NF-κB pathway, p65 forms heterodimer with p50 to transcriptionally activate the downstream target genes. NF-κB ligand (RANKL), interleukin-1 (IL-1), TNFα, endotoxins, and oxygen radicals, etc have been identified as prominent activators of NF-κB signaling pathway. Consistently, inhibition of the RANKL signaling by deleting RANKL diminishes NF-κB activity, leading to osteopetrosis in mice. 13 On the other hand, the lack of RANKL decoy receptor, osteoprotegerin (OPG), resulting in increased number of osteoclasts and osteoporosis in mice. 14
Traditional Chinese medicine (TCM) is used to treat various diseases because of its high safety and clinical efficacy.15,16 Shen Gu An capsule (SGA) is composed of Ba Ji Tian (Radix Morindae Officinalis), Xian Mao (Rhizoma Curculiginis), Fu Zi (Radix Aconiti Lateralis Preparata), Dan Shen (Radix Salviae Miltiorrhizae). SGA has been clinically used to treat osteoporosis, bone fracture, and spinal cord injury in patients with kidney Yang deficiency. Ba Ji Tian is the dry root of the Radix Morindae Officinalis that has anthraquinones, iridoid components, oligosaccharides. 17 It can reinforce Yang qi, enhance muscle and bone, and improve blood circulation, so it is widely used to treat diseases with kidney Yang deficiency. 18 Xian Mao is a perennial herb can be used in the treatment of four limb weakness, lumbar and knee arthritis, and to strengthen the tendons and bones. 19 Fu Zi is commonly used for the treatment of rheumatoid arthritis, tumors, edema, etc.20,21 Dan Shen, a dried rhizome, is a drug with extensive protective effects against cardiovascular and cerebrovascular diseases, also widely used in the treatment of osteoporosis. 22 It can stimulate blood circulation, eliminate blood stasis, relieve pain, and protect blood vessels. 23 However, the mechanism of SGA for the treatment of PMOP is very complex and has not been investigated up to now.
In this study, network pharmacology was applied to screen the active ingredients of SGA. Then, through enrichment analysis and network construction, we predicated the mechanism of SGA for treating PMOP. Finally, the effect and underlying mechanism of SGA were validated using the ovariectomized mice.
Materials and Methods
Preparation of SGA
The compositions of SGA was shown in Table 1. The extraction process of SGA was performed as previously described. 17 Generally, the herbs of SGA were soaked in double-distilled water for 30 min, and then boiled, concentrated to 1 g/ml, then divided and stored in the refrigerator at 4°C for further use. The dosage of mice was converted by body surface area and was converted by an adult dosage of adult person per day.
The Compositions of SGA.

Identification of Active Ingredients and Targets in SGA
The active ingredients and targets of individual herbs (Radix Morindae Officinalis, Rhizoma Curculiginis, Radix Aconiti Lateralis Preparata, Radix Salviae Miltiorrhizae) in SGA (Supplementary Table 1) were mined from the Traditional Chinese Medicine Systems Pharmacology (TCMSP) database (http://ibts.hkbu.edu.hk/LSP/tcmsp.php). The screening criteria were set as drug-like (DL) ≥ .18, and oral bioavailability (OB) ≥ 30%. 24
PMOP Targets Screening
The PMOP-associated target genes were collected from GeneCards database (https://www.genecards.org/) and OMIM database (https://www.omim.org/). Through inputting “Postmenopausal osteoporosis”, a total of 1284 PMOP-related targets were obtained. A Venn diagram (generated online, https://bioinfogp.cnb.csic.es/tools/venny/index.html) was drawn to show the overlapping genes between SGA and PMOP groups. These identified overlapping genes were termed as potential therapeutic targets.
Gene Ontology (GO) and KEGG Pathway Enrichment Analysis
The overlapping genes were subjected to GO analysis using DAVID database (https://david.ncifcrf.gov/home.jsp). KEGG database was used to identify the enriched signaling pathways (Supplementary Table 2).
Construction of Compound-Target and Protein-Protein Interaction (PPI) Networks
The herbs of SGA, active ingredients and their targets were imported into Cytoscape3.9.1 software to establish a compound-target network. And the overlapping targets were analyzed by String database (https://string-db.org/, version11.5) to build the PPI network.
Immunofluorescence Staining
The RAW264.7 cells were seeded in 24-well plates. Then the cells were induced to osteoclasts with RANKL (50 ng/mL) alone, or with SGA extract (.5 μg/ml) for three days. The cells were fixed with 4% paraformaldehyde for 20 min. After blocking for 1 h, cells were incubated with primary antibody against p-p65 (1:200) at 4 °C overnight. Then the fluorescent secondary antibody was added for 1 h. Images were captured with a fluorescence microscope (Olympus).
Ovariectomy (OVX) Model and Treatment
Twelve-week-old female C57BL/6J mice were randomly divided into three groups (n = 10 in each group). Mice were subjected to bilateral ovariectomy, or just extracted equal surrounding fatty tissues of ovaries. After three days post-surgery, SGA aqueous solution was orally administered to the mice at a dosage of .75 g/kg per day. The mice in the sham and OVX group were given .9% normal saline. All mice were sacrificed at 8 weeks after oral intervention. All experiment protocols were reviewed and approved by the Animal Experimental Ethical Inspection of The First Affiliated Hospital of Guangzhou University of Chinese Medicine (approval No. TCMF1-2022037).
MicroCT Analysis
The spinal samples obtained from each group were analyzed by a micro-CT scanner (SkyScan, Bruker, Germany). The parameters of bone microstructure including BMD, bone volume fraction (BV/TV), trabecular thickness (Tb. Th), trabecular number (Tb. N), and trabecular separation (Tb. Sp) were calculated as previously described. 25
TRAP Staining
The femur samples were fixed and decalcified with EDTA. Tartrate-resistant acid phosphatase (TRAP) staining was performed according to the manufacturer's instructions (Beyotime, China). Images of osteoclasts were captured with Olympus IX-73 microscope.
Immunohistochemistry (IHC) Staining
The femur samples were decalcified with EDTA, sectioned for histological examination.
The IHC analysis was performed according to our published protocol. 26 Generally, the sections were incubated in primary antibodies of phosphorylated p65 (p-p65) (1:100, Santa Cruz Biotechnology), Runx2 (1:100, Abcam), OCN (1:100, Santa Cruz Biotechnology), NFATC1 (1:100, Bioss) overnight at 4 °C. Then sections were incubated in horseradish peroxidase-conjugated secondary antibodies and diaminobenzidine (DAB) solution to detect positive staining. Image J software was used to quantify positive staining.
Statistical Analysis
Statistical analysis was carried out using SPSS 21.0 software. The normally distributed data was analyzed with the student's t-test. P < .05 was considered statistically different.
Results
Active Compounds of SGA
The bioactive chemicals and targets of SGA were selected by TCMSP database (Supplementary Table 1). Eighty-two common targets between PMOP and SGA were identified using Venn diagram (Figure 1). Then the PPI network construction were established to reveal the relationships of these targets (Figure 2).
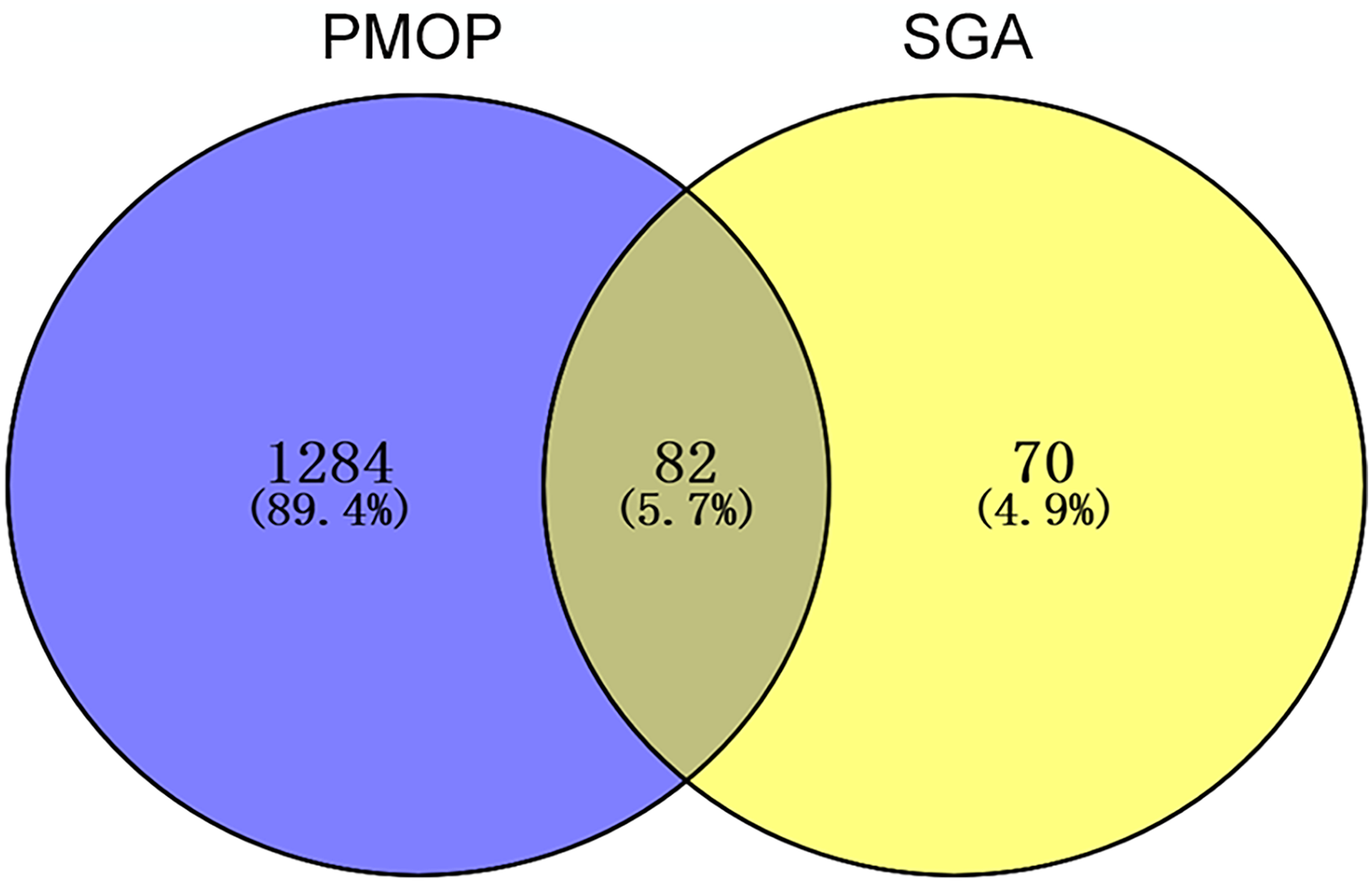
Venn diagram showing the intersection SGA and PMOP.

Protein-protein interaction network showing the relationship between the targets of SGA and PMOP.
Compound-Target Network Construction of SGA
Then, to elucidate the specific mechanism of how SGA influences PMOP, a compound-target network containing 113 compounds and 82 targets was created using the Cytoscape platform (Figure 3).

The compound-target network showing the combined mechanism of SGA. XM: Xian Mao; DS: Dan Shen; FZ: Fu Zi; BJT: Ba Ji Tian.
GO Analysis of SGA
The target proteins were analyzed by GO analysis, and the Top 20 enriched GO terms were shown in Figure 4. The top five molecular functions included response to hormone, oxygen levels, organic cyclic compound, lipid and xenobiotic stimulus.
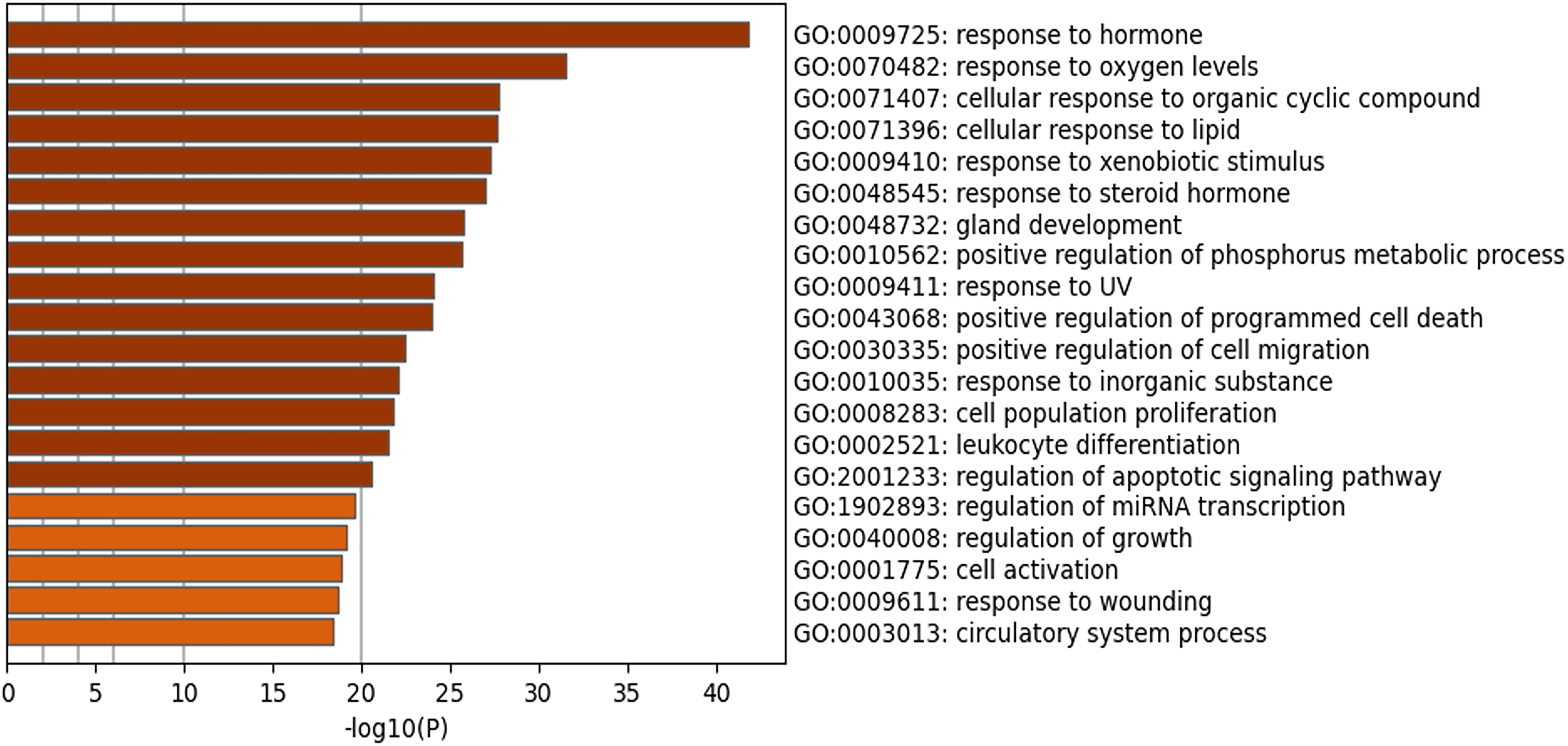
Top 20 enriched GO terms of the target proteins.
KEGG Analysis of SGA
In addition, the screened targets were subjected to KEGG analysis, and the Top 20 enriched KEGG terms were shown in Figure 5. Most of these pathways have been demonstrated to be involved in osteoporosis, such as PI3K-Akt, MAPK, NF-κB, calcium signaling pathways, etc.

Top 20 enriched KEGG terms of the target proteins.
SGA Inhibited NF-κB Signaling Pathway
Among the enriched KEGG pathways, the activation of NF-κB signaling pathway was well known in promoting osteoclastogenesis and inhibiting osteogenesis, so we chose it for further validation. In vitro, the RAW264.7 cells were induced to osteoclast differentiation using RANKL alone or with SGA extract. The immunofluorescence staining showed that the levels of phosphorylated p65 (p-p65) were significantly decreased by SGA during osteoclastogenesis (Figure 6).
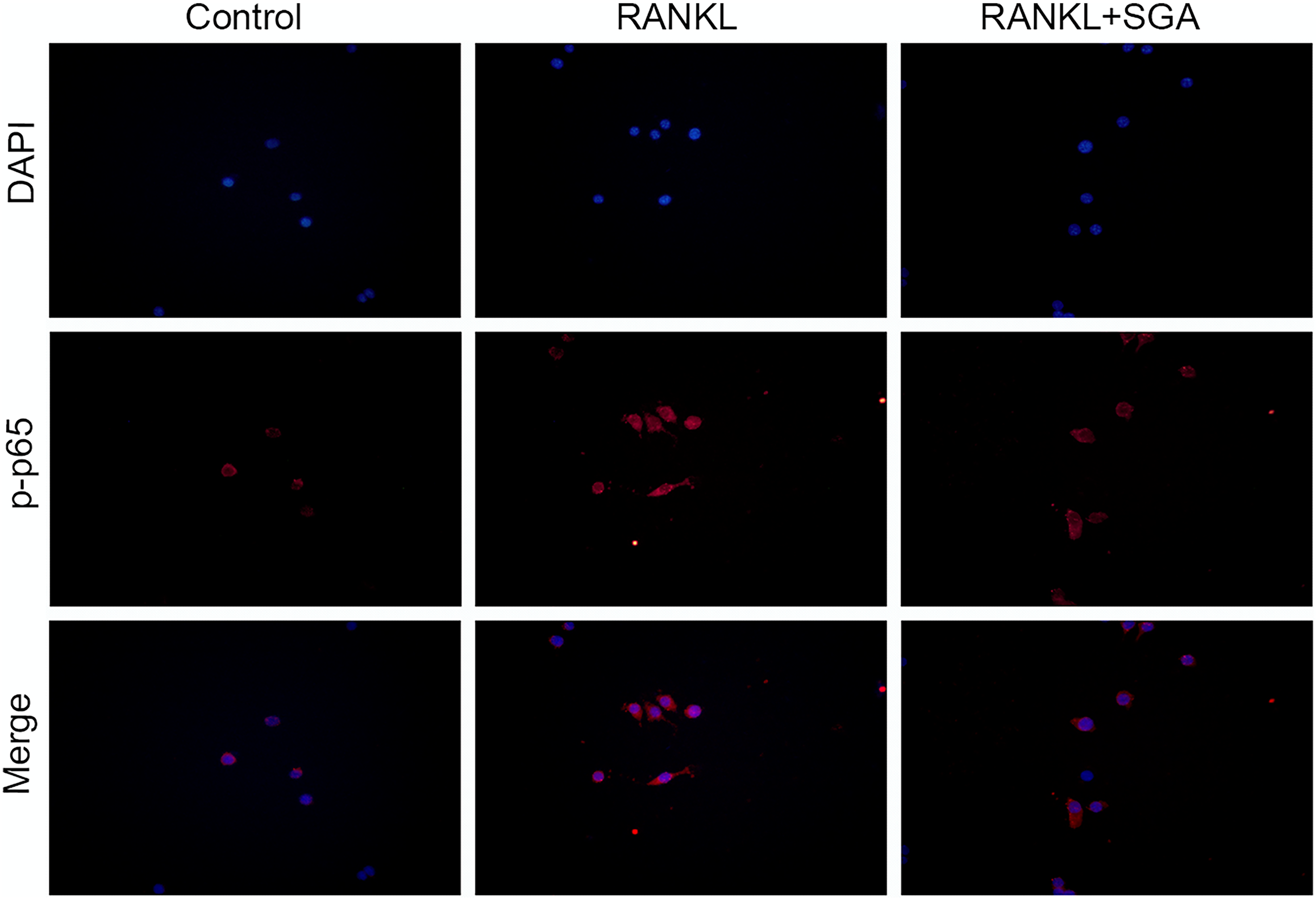
SGA inhibited the activation of NF-κB signaling pathway by RANKL during osteoclastogenesis. RAW264.7 cells were induced to osteoclast differentiation using RANKL (50 ng/mL) alone, or with SGA extract (.5μg/ml). Three days later, the levels of phosphorylated p65 (p-p65) were detected by immunofluorescence staining with specific antibody.
SGA Decreased the Number of Osteoclasts and Preserved Bone Mass in OVX Mice
Finally, the predicted mechanism based on network pharmacology was validated in vivo using OVX mice treated with SGA for 8 weeks. The microCT analysis showed that SGA could effectively alleviated bone loss observed in the OVX mice (Figure 7). The bone microstructure parameters including the BMD, Tb.N, BV/TV, and Tb.Sp were significantly improved by SGA treatment. Furthermore, the TRAP staining showed SGA significantly decreased the number of osteoclasts, which means bone absorption was inhibited by SGA. The representative images of IHC staining and quantitative analysis showed that the levels of p-p65 and Nuclear factor of activated T cells 1 (NFATc1) were increased in the OVX mice, but Osteocalcin (OCN) and Runt-related transcription factor 2 (Runx2) were decreased. However, the levels of these genes were all restored after SGA treatment (Figure 8), indicating that SGA could prevent OVX-induced bone loss through inhibiting NF-κB signaling.
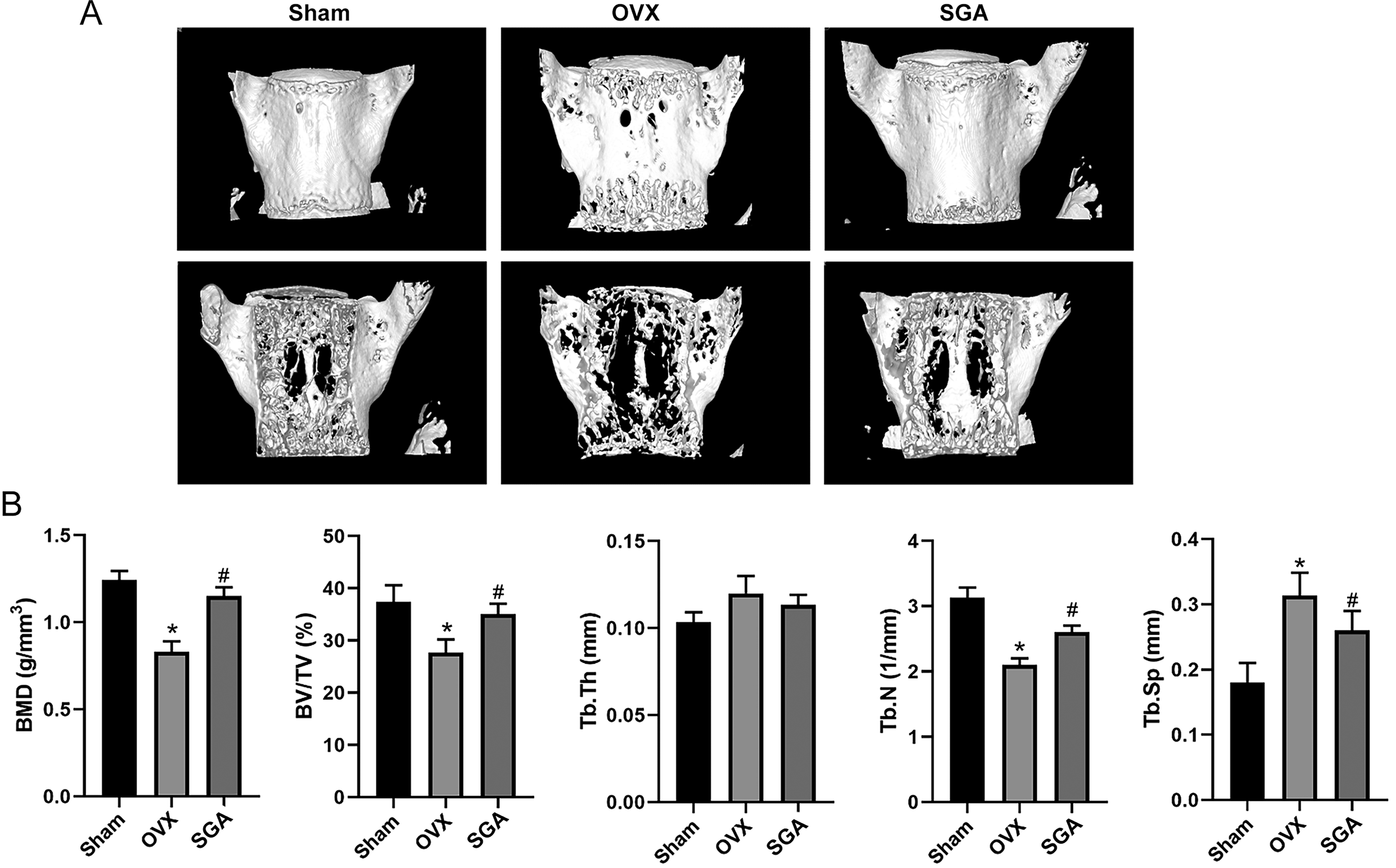
SGA preserved bone mass in OVX mice. (A) Representative 3D images of microCT analysis of each group. (B) Quantification of microstructural parameters of microCT analysis including BMD, BV/TV, Tb.Th, Tb.N and Tb.Sp in each group. Data are presented as mean ± SD, n = 5. *P < .05, compared with the Sham group; #P < .05, compared with the OVX group.

TRAP staining and immunohistochemistry detection of p-p65, Runx2, OCN, NFATC1. Representative TRAP staining images to detect osteoclasts, and IHC staining images to detect p-p65, Runx2, OCN, and NFATC1. Scale bars: 1000 µm. Data are presented as mean ± SD, n = 3. *P < .05, compared with the Sham group; #P < .05, compared with the OVX group.
Discussion
PMOP has grown rapidly due to the increase in the aging population. Chinese medicine has been largely used to protect against osteoporosis in China with fewer side effects. In the present study, we have explored the molecular mechanism of SGA through network pharmacology prediction and animal experiment validation. We found that SGA could inhibit osteoclast formation through NF-κB signaling pathway, and preserve bone mass in OVX mice.
There are four herbs in SGA including Radix Morindae Officinalis, Rhizoma Curculiginis, Radix Aconiti Lateralis Preparata, and Radix Salviae Miltiorrhizae. Radix Morindae Officinalis, Rhizoma Curculiginis, and Radix Aconiti Lateralis Preparata have long been used as tonics for nourishing kidney in China and northeast Asia, which can strengthen bone and enhance immunofunction in the treatment of osteoporosis, spinal cord injury and rheumatoid arthritis.27–31 Radix Salviae Miltiorrhizae has been used to improve the perfusion of ischemic myocardium and to enhance blood circulation, as well as bone formation. 32 In this study, one hundred and thirteen active compounds, and eighty-two common targets between PMOP and SGA were identified using TCMSP database. The GO and KEGG analysis predicted that NF-κB signaling pathway might be the potential underlying mechanism to explain the effect of SGA on PMOP prevention. NF-κB signaling pathway has been well known in promoting osteoclastogenesis and inhibiting osteogenesis, which has been proved by numerous studies.33–35 NF-κB1/2 double knockout mice displayed failure of tooth eruption and osteopetrosis due to absence of osteoclasts.12,36 RANKL KO mice also showed similar defect in osteoclastogenesis. 37 NFATc1 is a downstream effector of RANKL, and mice lacking NFATc1 also developed osteopetrosis owing to defects in osteoclast differentiation. 38 Runx2 is a central transcription factor governing osteoblast differentiation. Mice with a homozygous mutation in Runx2 showed a complete lack of ossification. 39 OCN, specifically produced by osteoblasts, is the most abundant non-collagenous protein in bone. Consistent with these previous findings, our study demonstrated that NF-κB signaling and NFATc1 were significantly inhibited by SGA, while the levels of Runx2 and OCN in bone tissue were increased.
The active compounds identified in herbs have been used to treat various diseases and specify signal transduction crosstalk mechanisms.40,41 For the herbs used in this study, some active compounds can explain their effects on bone formation and resorption to a great extent. For instance, Chaodong Zhang et al have found that Morinda officinalis polysaccharides (MOP) can ameliorate bone growth by regulating gut microbiota and oxidative stress. 42 Curculigoside is a naturally phenolic glycoside compound identified in Rhizoma Curculiginis, has been shown to mitigate osteoclastogenesis through activating Nrf2 while inhibiting NF-κB signaling pathway. 43 A novel polysaccharide COP70-1, purified from the rhizomes of Rhizoma Curculiginis has been found to stimulate osteogenic differentiation by activating the BMP and Wnt signaling pathways. 44 And our previous study has demonstrated that Salvianolic acid B, a major bioactive component of Salvia miltiorrhiza, could promotes osteogenesis by activating ERK signaling pathway. 45 However, we should realize that there are hundreds of compounds in a single herb, and a single compound may have several targets, some of the compounds may even have contrary effects. Although our study revealed that NF-κB signaling pathway was the main underlying mechanism for SGA to preserve bone mass, the exact compounds account for this biological action were not defined, which is the limitation of this study. For the future studies, how to construct an efficient network to elucidate the effects of herb or herbal formula is still a great challenge and also an urgent necessity.
Conclusion
Taken together, our study demonstrated that SGA could efficiently inhibit osteoclast formation through NF-κB signaling pathway as predicted by network pharmacology and validated by the OVX model of mice, which provides more evidence for the clinical application of SGA to treat osteoporosis.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241297039 - Supplemental material for Shen Gu An Capsule Inhibition of Postmenopausal Osteoporosis in Ovariectomized Mice Using Network Pharmacology-Based Mechanism Prediction and Pharmacological Validation
Supplemental material, sj-docx-1-npx-10.1177_1934578X241297039 for Shen Gu An Capsule Inhibition of Postmenopausal Osteoporosis in Ovariectomized Mice Using Network Pharmacology-Based Mechanism Prediction and Pharmacological Validation by Meilin Du, Shiguo Yuan, Jiayang Wang, Qiangqiang Zhao, Baofei Ye, Haiyan Wu, Liangliang Xu and Yu Hou in Natural Product Communications
Footnotes
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Authors’ Contributions
YH and LLX: conceived, designed, supervised, and commented on all the drafts of this paper. MLD, SGY, QQZ: conducted the overall experiments, wrote the main manuscript. BFY, JYW and HYW: participated in the data collection, analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
All animal experiments were approved by the Ethical Committee of the First Affiliated Hospital of Guangzhou University of Chinese Medicine (approval number: TCMF1-2022037).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Hainan Province Clinical Medical Center ([2021] No. 276), and National Natural Science Foundation of China (NSFC No. 82360941).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Institutional Animal Care guidelines of the First Affiliated Hospital of Guangzhou University of Chinese Medicine.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
