Abstract
Objective
Rheumatoid arthritis (RA) is a chronic inflammatory autoimmune disorder for which current therapeutic options are often insufficient. This study aimed to evaluate the therapeutic potential of hesperidin (HSP), in suppressing complete Freund's adjuvant (CFA)-induced RA in Wistar albino rats, through modulation of key pro-inflammatory markers (TNF-α, IL-6, IL-1β, NF-κB, and COX-2), supported by in silico docking and pharmacokinetic profiling.
Methods
Forty-eight female Wistar albino rats were randomized into six groups (n = 8). Group 1 received normal saline; Group 2 was induced with RA via 0.1 ml CFA and left untreated; Group 3 received CFA + naproxen (8 mg/kg). Groups 4, 5, and 6 received hesperidin at 100, 200, and 400 mg/kg, respectively, following RA induction. Serum pro-inflammatory markers were quantified using standard biochemical assays. Molecular docking was performed to assess HSP binding affinity toward COX-2, TNF-α, IL-1β, and IL-6, while ADMET profiling was carried out to predict pharmacokinetic and toxicity attributes.
Results
In vivo results revealed that RA induction significantly elevated TNF-α, IL-6, IL-1β, and NF-κB levels (p < .05) across the treated groups. Whereas, hesperidin treatment produced a dose-dependent and significant (p < .05) reduction in these markers, restoring them to levels comparable to naproxen-treated rats. In silico docking analysis revealed that hesperidin has multi-target binding capability inhibitor to COX-2, TNF-α, IL-1β, and IL-6. Protein-ligand binding was predominantly surface-oriented except for COX-2, which showed interior pocket binding. ADMET predictions indicated that HSP is a P-glycoprotein substrate with low gastrointestinal absorption, does not cross the blood–brain barrier, shows no cytochrome P450 enzyme subunit inhibition, and lacks carcinogenic or hepatotoxic potential.
Conclusion
Combined in vivo and in silico findings demonstrate that hesperidin exhibits strong anti-inflammatory effects by inhibiting key mediators of RA pathogenesis, alongside a favorable safety pharmacokinetic profile.

Keywords
Background
Rheumatoid arthritis (RA) is a systemic, autoimmune disease that causes joint stiffness, deformity, restricted motion, and irreversible functional loss to the joints. 1 A key pathological feature of RA is synovial inflammation, which is characterized by the development of pannuses, the proliferation of histiocytes, the infiltration of inflammatory cells, the expression of cell surface adhesion molecules, and variations in the levels of protease and cytokines. 2 Over 20 million people worldwide are affected by RA each year, with middle-aged people being the main demography affected. 3 In industrialized nations, the prevalence rate ranges from 0.5% to 1%, with women experiencing a 2.5- to 3-fold increase in prevalence compared to men. 3 The expected survival time for RA patients is shortened by three to ten years due to complications like neurological and cardiovascular diseases. 3
Glucocorticoids, biological agents, disease-modifying antirheumatic drugs, and nonsteroidal anti-inflammatory drugs are still the mainstays of treatment for RA to date. 4 These medications block immune components and inflammatory mediators, to delay the course of disease.
Nevertheless, the majority of medications only show meaningful therapeutic benefits when taken frequently and at high doses. Although dosage and side effects are closely related, some studies indicate that using biologics or combining medications can result in better therapeutic effects. 4
Nowadays, biomedical and pharmacological researchers have been employing computational power to expedite the laborious processes of drug discoveries to reduce the large number of experimental animals used in research design. 5 Computer-aided drug design, also known as In-silico design, is applied to optimize drug profiles in terms of absorption, distribution, metabolism, excretion, and toxicity in the fields of biomedicine and pharmacology. 6 It also serves to expedite hit/lead molecule detection and prevent adverse drug reactions. 6 The In silico approach in drug design is capable of analyzing mechanisms of action of potential drug candidates thereby switching from the conventional method of searching for specific target proteins during drug design to a more thorough evaluation using molecular docking. 6
Complete Fraud's adjuvant (CFA)-induced arthritis appears to be the most dependable 7 and scientifically supported AIA model in rats.8,9 Experimental models of autoimmune illnesses, both acute and chronic, such as RA, e.t.c are frequently reproduced using CFA. 10 When juxtaposed with IFA and pristine, CFA-induced arthritis produces more severe and systemic arthritogenic effects.9,11,12 CFA is said to have a higher activating power for the formation of antibodies compared to other adjuvants. 10 Additionally, it elicits sensory responses, which could help researchers find novel drugs to treat symptoms associated with arthritic conditions.
This study investigated the potential of hesperidin in suppressing Complete Freund's adjuvant (CFA)-induced rheumatoid arthritis (RA) in rats by targeting important inflammatory pathways, including TNF-α, IL-6, IL-1B, NF-κB, and COX-2. A significant novelty of this study lies in its simultaneous targeting of multiple inflammatory pathways, which may offer a more effective treatment strategy than therapies focusing on a single pathway. This comprehensive approach aims to improve clinical outcomes by addressing the complex nature of RA's pathophysiology. Additionally, the study employs both In vivo and In silico approaches. The In vivo component involves CFA-induced RA in female albino rats, providing a biologically relevant model for testing hesperidin. Due to ethical limitations in human experimentation, In silico analyses were also conducted, integrating human biochemical data with drug and disease databases. 12 This allowed for the identification of specific target proteins involved in RA therapy, using principles like protein-drug interaction studies, molecular docking, and ADMET profiling.
Most importantly, this study's relevance to RA drug discovery is crucial, given that current therapies of RA often come with substantial side effects and fail to address the root causes of inflammation. Hesperidin, a natural compound found in peels of citrus fruits such as oranges and tangelos, presents a promising alternative, offering the potential for safer and, a more accessible treatments, particularly for older patients with comorbidities.
Finally, the use of the CFA-induced arthritis model further strengthens the study's relevance, as it mimics the inflammatory processes seen in human RA. By identifying the molecular mechanisms behind hesperidin's effects, this study could make a substantial contribution to the development of targeted, natural therapies for RA.
Materials and Methods
Materials and Instrument Details Used for in Vivo Study
Materials and equipment employed in the present study include an electronic weighing balance (G and G Electronic China), a spectrophotometer (UV-Vis (Hitachi Japan), U-2900), a centrifuge (Gallen Lamp USA), latex gloves (Gallen Kamp, USA), sterile needles (Danigold, China), a sterile specimen bottles (Rumex), a pH meter, a rat cage, a deep freezer, an incubator, a Hb pipette, a micropipette and cotton wool. All instruments and materials were standardized and calibrated prior to use to ensure accuracy and reproducibility of results.
Chemicals and Reagents
Hesperidin with the purity of 95% and CFA were procured from Bristol Scientific Limited, Nigeria. The standard rheumatoid arthritis drug (Naproxen) was purchased from Octovia Pharmacy in Ebonyi, Nigeria. Elabscience® ELISA rat test kits were purchased from Wuhan Elabscience Biotechnology Limited, China. Every other reagent and chemical used was bought from licensed chemical shops and was of diagnostic standard. All other reagents and chemicals were obtained from licensed chemical suppliers and were of diagnostic grade.
Methods Used for in Vivo Study
Ethical Approval Statement
This study was conducted in accordance with internationally accepted guidelines for the care and use of laboratory animals, as outlined in the NIH Guide for the Care and Use of Laboratory Animals (NIH Publication No. 80-23, revised 1996). Ethical approval was granted by the Department of Biochemistry Research Ethics Committee, acting on behalf of the Core Institutional Animal Care and Use Committee (IACUC) of Ebonyi State University, under protocol number EBSU/BCH/ET/23/001, issued on 30th March 2023. The reporting of this study conforms to ARRIVE 2.0 guidelines (Animal Research: Reporting of In Vivo Experiments). 13
Procurement and Housing of Experimental Animals
This study utilized only female Wistar albino rats to minimize variability from sex-specific hormonal differences, as female rats are more susceptible to CFA-induced arthritis, aligning with findings from previous literatures.14,15 Female wistar albino rats, used for this study, were purchased from the Department of Veterinary Medicine animal house at the University of Nigeria, Nsukka, Enugu, Nigeria. Upon arrival, the rats were housed in metal cages at the Animal House of the Department of Biochemistry, Ebonyi State University, Abakaliki. The animal house is well-ventilated and maintained under optimal laboratory conditions, including a 12-h light/dark cycle and controlled room temperature. Prior to the commencement of the study, the rats were allowed a 7-day acclimatization period. During this acclimatization period, the rats received unlimited access to clean drinking water and were provided with standard rodent feed (Vital Feed®, Grand Cereals Ltd, Jos, Nigeria) ad libitum.
Rheumatoid Arthritis Induction in Albino Rats
CFA was kept in a deep freezer and protected from sunlight. The rats were anesthetized using isoflurane for 5 min at a dose of 3% by the drop-jar method. 16 One dose of subcutaneous injection of 0.1 ml of 10 mg/mL CFA into the right hand footpad of the female rats resulted in adjuvant arthritis. Indicators of inflammations such as redness, edema, hyperalgesia, and allodynia were localized in the region of the injected paw as indicators of RA. 17
Acute Toxicity of Hesperidin
This study adopted the median lethal dosage (LD50) of HSP at 4837.5 mg kg−1 as reported by Li et al. 18 in their study of both male and female rat models.
Experimental Design and Animals Grouping
Forty-eight (48) female albino rats of Wistar strain of body weights between 150 and 200 g were used. The rats were divided into the following groups, each housing 8 rats: Rats in group 1 received normal saline and feeds only. Group 2 was induced RA by a single subcutaneous injection of 0.1 ml of CFA in rats without treatment. Group 3 was induced RA with CFA and treated with a standard RA drug, naproxen, at a dose of 8 mg kg−1 body weight (b.w). 19 Group 4 was induced RA using CFA and treated with hesperidin at 100 mg kg−1 b.w. Group 5 was induced RA using CFA and treated with Hesperidin at 200 mg kg−1 b.w. Group 6 was induced RA with CFA and treated with Hesperidin at 400 mg kg−1 b.w.
Anesthetic Protocol Prior to Euthanasia
At the end of the 28-day treatment, the rats were anesthetized using the drop-jar method with approximately 1 mL of liquid isoflurane (equivalent to 3% vapor concentration) administered via a gauze pad in a sealed chamber for 5 min. 20 Following confirmation of deep anesthesia via loss of pedal reflex by each rats, euthanasia was carried out through cardiac puncture, which also served as the method for terminal blood collection. 21 Blood samples were collected directly from the heart and transferred into plain sample tubes. The sera were separated and used for the assay of TNF-α, NF-κB, and IL-6 levels. This procedure ensured humane termination and high-quality serum recovery for cytokine analysis.
In Vivo Determination of serum Cytokines
Measurement of serum TNF-α, NF-k beta, IL-6, and C-reactive proteins was determined using specific Elabscience® ELISA rat test kits following the manufacturer's guidelines.22,23 Assays were performed in duplicates to enhance reliability of measurements.
Histopathological Investigation
Ankle joint tissues were collected, fixed in 10% neutral buffered formalin, dehydrated through graded alcohols, cleared in xylene, and embedded in paraffin. Sections (5 μm) were stained with hematoxylin and eosin (H&E). Synovial hyperplasia, inflammatory cell infiltration, and cartilage erosion were scored semi-quantitatively. A vehicle control group received normal saline. Rats were randomly assigned to groups, and histopathological evaluation was performed blinded to treatment allocation. 24
Materials and Methods Used for in-Silico Analysis
Software and Web servers used for
In silico
analysis
The software used for this study are Discovery Studio 2020, UCSF Chimera, AutoDock Vina Plugin PyRx, and ChemSketch. The web servers used are PuChem, MedChemExpress, RCSB Protein Databank, Drugbank, pKCSM, and SwissADME.
Retrieval of Ligands and Target Proteins
The ligands used were hesperidin (drug candidate) (PubChem CID:10621) and standard inhibitors of the target proteins, including naproxen, which is the standard drug that targets cyclooxygenase II (COX 2), anakinra, which targets IL-1b, and tocilizumab, which targets IL-6. They were all retrieved from the drug bank, and a compound with MedChemExpress database catalog number HY-112275 (CAS:444287-49-4) was used to target TNF-α. Except for hesperidin, whose 3D crystal structure was obtained from the PubChem database, the standard inhibitors were drawn using ChemSketch, imported into UCSF Chimera, and saved in PDB format before docking. The target proteins were retrieved by their identification numbers from Protein Databank resources.
Preparation of Target Proteins
The target proteins were designed using UCSF Chimera software. The 3-D crystal structures of COX-2, IL-1β, and IL-6 were retrieved by their PDB IDs: COX-2: PDB ID 5F19; IL-1β: PDB ID 4G6J; IL-6: PDB ID 1ALU; while the structure for TNF-α was obtained through its MedChemExpress catalog number (HY-112275, CAS: 444287-49-4) as outlined by Agu et al. 25 Except for hesperidin, whose structure was obtained from the PubChem database, the standard inhibitors were designed in ChemSketch, imported into UCSF Chimera, and saved in PDB format for docking studies. Non-standard ligands were simply selected and removed by deletion. The proteins were then minimized using default settings, with the addition of hydrogen bonds and charges via Gastigar. After minimization, the proteins were saved in PDB format, ready for molecular docking.
Molecular Docking Protocol
Molecular docking was performed following the protocols described by Trott et al. 26 with modifications using the AutoDock Vina Plugin in PyRx. The target proteins were individually loaded into PyRx and converted into macromolecule files. The ligands, including standard inhibitors/drugs and hesperidin, were imported in chemical table format (CT) into PyRx.
Ligands for docking were prepared by energy minimization with the addition of hydrogen atoms and partial charges using the Gasteiger. Following this, the ligands were converted into the PDBQT format, which includes information about the atom types, partial charges, and rotatable bonds. The molecular docking process was carried out using the Vina Wizard, with the grid box centered and sized according to the active sites of the target proteins as follows: COX-2: Centered at X (25.5739), Y (60.5207), Z (7.4013) with dimensions X (60.5207), Y (78.8037), Z (64.8366), TNF-α: Centered at X (21.9746), Y (59.6013), Z (42.0435) with dimensions X (59.6013), Y (46.0177), Z (42.0435), IL-1β: Centered at X (75.8870), Y (47.4546), Z (57.0796) with dimensions X (47.4546), Y (50.9885), Z (47.2200), IL-6: Centered at X (82.3601), Y (44.7719), Z (−36.5667) with dimensions X (44.7719), Y (53.6007), Z (35.7100).The docking runs were performed with an exhaustiveness setting to ensure thorough sampling of the binding poses. The docking poses with a root mean square deviation (RMSD) ≤ 1 Å were considered for analysis.
Hesperidin's binding affinity was evaluated by comparing its docking pose to that of the standard inhibitors, specifically focusing on the position where it binds at the same site as the reference inhibitor. The interaction profiles of the protein-ligand complexes were visualized using Discovery Studio 2020. This allowed for the identification of key amino acid residues involved in the binding and the types of interactions (eg, hydrogen bonds, hydrophobic interactions, ionic bonds) that contribute to the stability of the ligand-protein complexes.
ADMET studies
ADMET profiling of hesperidin was performed as adopted by Daoud et al. 27 using the pkCSM web server (http://biosig.unimelb.edu.au/pkcsm/prediction). Briefly, the Canonical SMILE of hesperidin obtained from the PubChem database was inserted into the workspaces of the respective web servers. The prediction command was clicked, and the predicted values (physicochemical properties in terms of the Lipinski rule of five, pharmacokinetic properties, and toxicity profile) were retrieved after 10 min.
Statistical Analysis
All data are expressed as mean ± standard deviation (SD). Data were analyzed using one-way analysis of variance (ANOVA), followed by Dunnett's multiple comparison post hoc test to evaluate differences between treated groups and the control. Statistical significance was defined as p < .05. All statistical analyses were performed using GraphPad Prism 8 software (GraphPad Software, California, USA) to ensure accurate data processing and visualization.
Results of in Vivo Studies
Impact of HSP on serum TNF-α, NF-κB, and IL-6 in CFA-Induced RA Wistar Albino Rats
The concentrations of TNF-α, NF-κB, and IL-6 were markedly increased in Wistar albino rats following induction with CFA (Figures 1–3; p < .05). Compared with the RA control group, treatment with hesperidin (HSP) at various doses significantly reduced these elevated serum cytokine levels (p < .05). However, as shown in Figures 1– 3, the group receiving normal saline exhibited cytokine levels similar to the RA group treated with 400 mg/kg b.w. of HSP (Figure 4).

Effects of HSP on TNF-α-level of CFA-induced RA Rats. Data are expressed as mean ± SD (n = 6). *The mean data obtained from various rat groups were compared with the R.A group using Dunnet's multiple comparisons test, with notable differences at p < .05. IL-6, Interleukin-6; NS, Normal saline; RA, Rheumatoid arthritis; HSP, Hesperidin.

Effects of HSP on NF-kB-level of CFA-induced RA in rats. Data are expressed as mean ± SD (n = 6). *The mean data obtained from various rat groups were compared with the R.A group using Dunnet's multiple comparisons test, with notable differences at p < .05. IL-6, Interleukin-6; NS, Normal saline; RA, Rheumatoid arthritis; HSP. Hesperidin.

Effects of HSP on IL-6-level of CFA-induced RA in rats. Data are expressed as mean ± SD (n = 6). *The mean data obtained from various rat groups were compared with the R.A group using Dunnet's multiple comparisons test, with notable differences at p < .05. IL-6, Interleukin-6; NS, Normal saline; RA, Rheumatoid arthritis; HSP, Hesperidin.

Molecular docking protocol of hesperidin and standard ligands with their respective target proteins. Hesperidin demonstrated exceptional ability to competitively bind to other target proteins (COX-2, TNF-alpha, IL-1 beta and IL-6).
Results of in Silico Studies
Comparative Binding Affinities of Hesperidin and Standard Ligands/Drugs to Their Target Proteins
Hesperidin exhibited strong binding affinities to the target proteins: −9.3 kcal/mol (COX-2), −7.1 kcal/mol (TNF-α), −7.9 kcal/mol (IL-1β), and −7.7 kcal/mol (IL-6), comparable to the binding affinities of standard inhibitors (−7.5 kcal/mol for COX-2, −7.1 kcal/mol for TNF-α, −7.9 kcal/mol for IL-1β, and −10.1 kcal/mol for IL-6) (Figure 5). Both hesperidin and standard inhibitors interact with their respective target proteins using similar types of bonds, as revealed by 3D (Figures 6–9), and 2D interaction analyses (Figures 10–13). Hesperidin exhibits multi-target inhibitory actions with COX-2, TNF-α, IL-1β, and IL-6, whereas the standard drugs exhibit selective inhibition for their respective targets.

Comparative docking scores of HSP and standard ligands/drugs, showing their respective binding affinities to target proteins.
Docking Pose of the Protein-Ligand Binding
Docking analyses revealed that hesperidin binds competitively to the same pocket as the standard inhibitors. Interestingly, only COX-2 exhibited an interior binding pocket interaction, while TNF-α, IL-1β, and IL-6 demonstrated surface binding interactions (Figures 6–9).

Display of 3D structure of the nonselective docking pose of COX2-hesperidin within the same binding pocket as naproxen (standard inhibitor/drug). Green is the standard inhibitor (Naproxen).

Display of the 3D structure of the nonselective docking pose of TNF-α-hesperidin within the same binding pocket as standard inhibitor. Green is the standard inhibitor (CAS: 444287-49-4).

Display of 3D structure of the non-selective docking pose of IL-Iβ-hesperidin within the same binding pocket as standard inhibitor/drug. Green is the standard inhibitor (Anakinra).

Display of the 3D structure of the nonselective docking pose of IL-6- hesperidin within the same binding pocket as standard inhibitor/drug. Green is the standard inhibitor (Toxicilimab).
Protein-Ligand Interactions
Results from the visualization of the 2D interactions revealed that both hesperidin and other standard inhibitors use similar kinds of bonds to interact with different amino acid residues within their binding pockets (Figures 10–13).

Display of 2D structure showing the comparison in molecular interaction forces of Naproxen (A) and Hesperidin (B) within their respective COX2 binding pockets.

Display of 2D structure showing the comparison in molecular interaction forces of Naproxen (standard inhibitor) (A) and Hesperidin (B) ligands within TNF-α binding pockets.

Display of 2D structure showing the comparison in molecular interaction forces of Anakinra (A) and Hesperidin (B) ligand within IL-1β binding pocket.
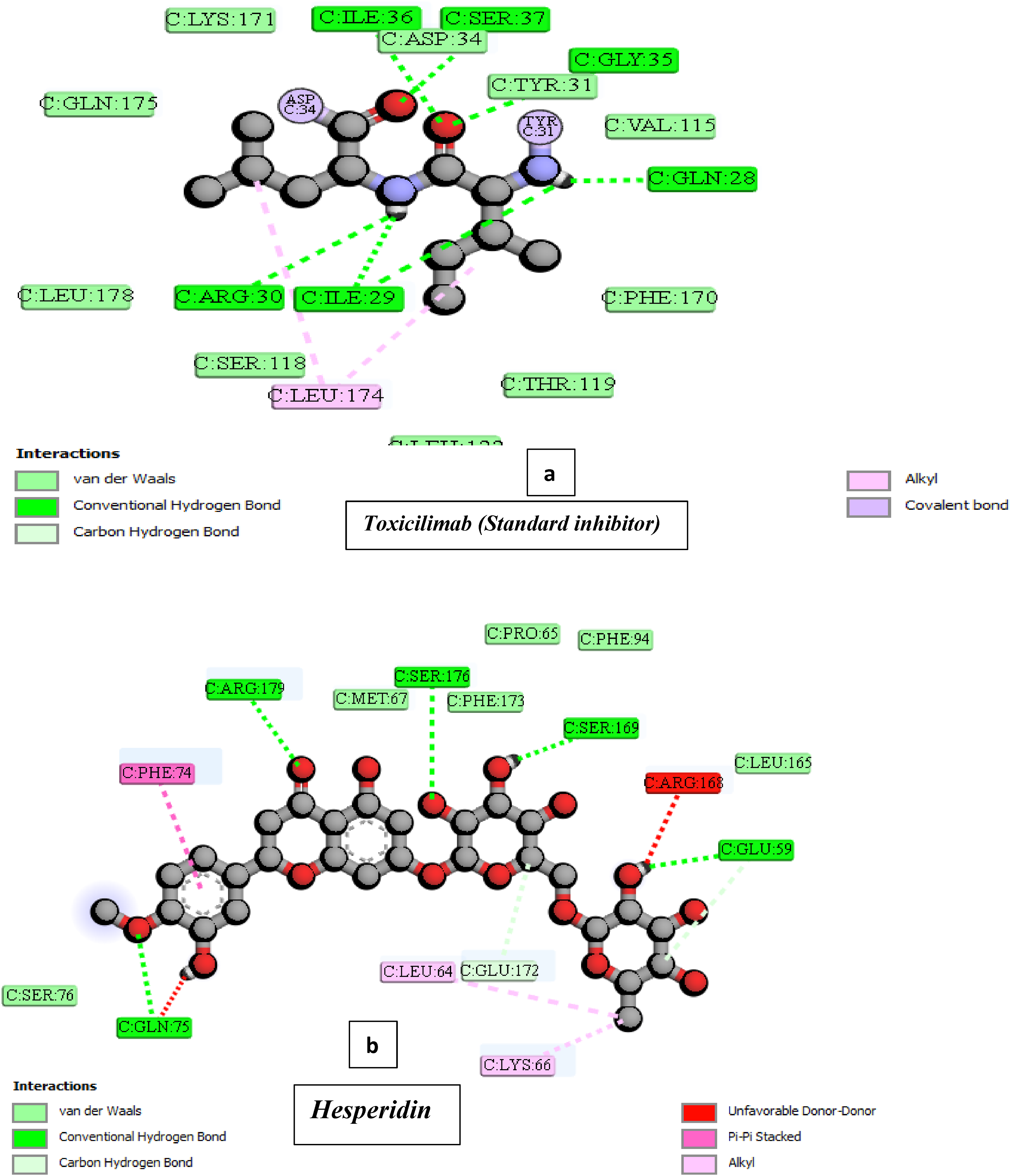
Display of 2D structure showing the comparison in molecular interaction forces of Toxicilimab (A) and Hesperidin (B) ligands within IL-6 binding pocket.
ADMET Profiling of Hesperidin
Physicochemical Properties (Lipinski Rule of Five)
In Table 1, hesperidin shows several deviations from Lipinski's Rule of Five, which predicts oral bioavailability. With a high molecular weight of 610.56 Da, 15 hydrogen bond acceptors, 8 hydrogen bond donors, and a large TPSA of 234.29 Å, 2 the molecule exceeds multiple thresholds, resulting in three RO5 violations. Although its LogP value of 2.6 suggests a moderate balance between hydrophilicity and lipophilicity, the high polarity and extensive hydrogen bonding capacity limit passive diffusion across biological membranes. These physicochemical features collectively imply that hesperidin may have poor oral absorption and low systemic bioavailability unless formulated or modified to enhance permeability.
Druglike Properties of Hesperidin.
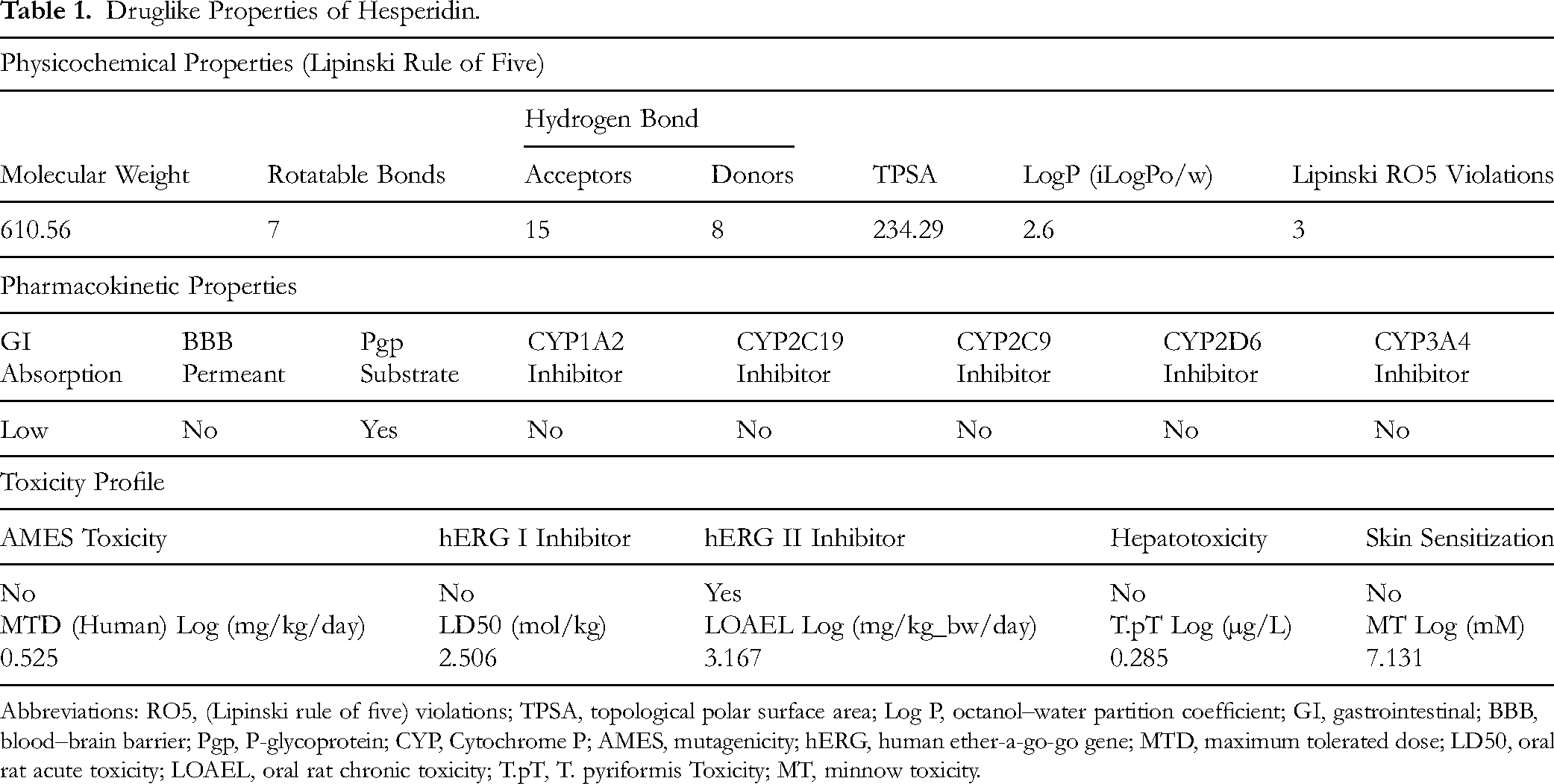
Abbreviations: RO5, (Lipinski rule of five) violations; TPSA, topological polar surface area; Log P, octanol–water partition coefficient; GI, gastrointestinal; BBB, blood–brain barrier; Pgp, P-glycoprotein; CYP, Cytochrome P; AMES, mutagenicity; hERG, human ether-a-go-go gene; MTD, maximum tolerated dose; LD50, oral rat acute toxicity; LOAEL, oral rat chronic toxicity; T.pT, T. pyriformis Toxicity; MT, minnow toxicity.
Pharmacokinetic Properties
The pharmacokinetic profile indicates that hesperidin has low gastrointestinal (GI) absorption, consistent with its high polarity and RO5 violations. It is not blood–brain barrier permeant, confirming its limited ability to cross lipid-rich barriers. Hesperidin is identified as a P-glycoprotein (Pgp) substrate, meaning it can be actively effluxed out of cells, further reducing its absorption and intracellular accumulation. Additionally, hesperidin does not inhibit major CYP450 enzymes (CYP1A2, CYP2C19, CYP2C9, CYP2D6, CYP3A4), suggesting that it is unlikely to cause significant drug–drug interactions via CYP-mediated metabolism (Table 1). These findings highlight hesperidin's limited systemic exposure but a favorable metabolic safety profile.
Toxicity Profile
The toxicity assessment suggests that hesperidin is relatively safe with minimal toxicological concerns. It is non-mutagenic (negative AMES test), and it does not inhibit the major cardiac ion channel hERG I, though a positive hERG II inhibition indicates a mild potential for cardiotoxicity at higher concentrations. Importantly, the compound shows no hepatotoxicity and no skin sensitization risk, indicating a good safety margin for topical and systemic exposure. The maximum tolerated dose (MTD) in humans (0.525 Log mg/kg/day) and the LOAEL (3.167 Log mg/kg/day) suggest a wide therapeutic window, while the LD50 value (2.506 mol/kg) denotes low acute toxicity. The T. pyriformis toxicity (0.285 Log µg/L) and minnow toxicity (7.131 Log mM) further indicate low environmental toxicity (Table 1).
Discussion
Rheumatoid arthritis (RA) is a chronic autoimmune condition that causes joint inflammation and damage to bone and cartilage. 28 Current treatments options often involve synthetic drugs like NSAIDs and DMARDs, which can lead to adverse side effects. 28 In light of these challenges, this study explored the potential of hesperidin administration, a naturally occurring polyphenol found in the peels of citrus fruits such as sweet oranges and tangelos,29,30 to treat RA using a Wistar albino rat model. Hesperidin is metabolized in vivo to hesperetin and its conjugates (sulfate or glucuronide), as well as low-molecular-weight phenolic acids, which are the circulating molecules potentially exerting the reported effects. 31 Hesperidin has been previously reported to possess antioxidant, anti-inflammatory and anti-cancer properties with a strong safety profile.32,33
RA was induced in rat models using complete Freund's adjuvant (CFA), which is well-established as a reliable model for investigating the disease's pathophysiology. CFA-induced RA was reported for its severity and systemic effects,9,10 making it a valuable tool for studying the disease's sensory responses and evaluating potential therapeutic interventions. Only female rats were used in this study due to the higher prevalence of RA in females and their increased susceptibility to CFA-induced arthritis, which better models the human disease. 13
Figures 1–3 of this study showed that the concentrations of TNF-α, NF-Kb, and, IL-6 were significantly (P < .05) increased, following the induction of RA in Wistar albino rats using the CFA. However, upon treatment with HSP with the dosages of 100, 200, and 400 mgkg−1 b.w, the concentration of these cytokines was considerably (P < .05) decreased below the levels observed in the RA group (untreated group) (Figures 1–3). Intriguingly, the group which was administered 400 mg kg−1 b.w. of HSP revealed a comparable outcome with the group that received normal saline (Figures 1–3).
TNF-α, IL-6, and NF-κB are recognized as primary contributors to the long-term inflammation observed in the joints during rheumatoid arthritis (RA).34,35 This chronic inflammation can lead to erosive alterations in cartilage and bone. Studies involving rat models has provided valuable insights into these mechanisms.34,35 Among these cytokines, TNF-α is particularly well-known for its role in modulating inflammatory responses. It plays a crucial part in the pathophysiology of autoimmune and inflammatory diseases, including RA. 36 Similarly, IL-6 and NF-κB are also significant in driving inflammation in this condition. 36
This study highlighted significant findings regarding the effects of hesperidin on serum cytokine levels in rheumatoid arthritis (RA) rats. Administration of graded doses of hesperidin led to a marked decrease in serum concentrations of TNF-α, NF-κB, and IL-6, with a notable (p < .05) reduction observed at a dose of 400 mg/kg. Treatment with HSP resulted in cytokine levels that were statistically comparable to those group that were administered naproxen. These findings suggest that hesperidin may exert a therapeutic effect in RA by suppressing the proliferation of inflammatory cytokines in synovial cells, which is critical to the disease's pathophysiology. The data, illustrated in Figures 1–3, reinforced the notion that hesperidin can modulate immune responses, making it a potential candidate for RA treatment.
In a similar study, 37 employed collagen-induced arthritic rats and administered hesperidin at a dose of 160 mg/kg body weight for 20 days following RA induction. Histological examinations, metabolic markers, and clinical scoring were employed to evaluate hesperidin's anti-rheumatoid effects. The results revealed significant anti-arthritic potential, with hesperidin causing a substantial reduction (P < .05) in serum inflammatory cytokines such as TNF-α, NF-Kb, and IL-6. These cytokines are linked to inflammation, as monocyte activation in arthritic rats leads to the production of TNF-α, IL-1, and IL-6, which contribute to immunological reactions and increased matrix metalloproteinases and acute-phase proteins38,39 Similarly, in other studies, hesperidin therapy decreased TNF-α and IL-6 in rats with induced arthritis, 40 and reduced serum inflammatory biomarkers when administered at doses of 40 and 80 mg/kg over 45 days. 41 These findings support the hypothesis of this study that hesperidin exerts its anti-arthritic effects by regulating pro-inflammatory biomarkers, aligning with the results presented in Figures 1–3 of this study.
The In-silico study aimed to assess the binding affinities of hesperidin compared to established rheumatoid arthritis (RA) treatments such as naproxen, anakinra, and toxicilimab. By docking these compounds with target proteins like COX-2, IL-1β, IL-6, and TNF-α, this study sought to clarify hesperidin's molecular mechanisms against RA and predict its efficacy and safety relative to standard drugs. A key metric in this analysis is binding energy, which reflects the strength of interaction between the receptor proteins and the ligands or drugs Figure 5. Generally, a more negative binding energy indicates a more favorable binding interaction, suggesting that the ligand is more likely to effectively inhibit the target protein. Information on binding energies of ligands with target proteins is crucial for understanding the potential therapeutic role of hesperidin in managing RA, as it can help identify its effectiveness and safety compared to conventional options. 42
The docking binding energy outcome as presented in Figure 5 revealed the binding affinities of hesperidin (HSP) and standard inhibitors to their target proteins. HSP (−9.3 Kcal/mol) shows stronger binding to cyclooxygenase-2 (COX-2) than naproxen (−7.5 Kcal/mol), suggesting HSP may be a more effective COX-2 inhibitor. For IL-1beta (−7.9 Kcal/mol) and TNF-alpha (−7.1 Kcal/mol), the binding affinities of standard inhibitors are similar to HSP. However, the standard inhibitor for IL-6 (−10.1 Kcal/mol) has a higher affinity than HSP (−7.7 Kcal/mol). The notion that higher binding energies correlate with better target inhibition reinforces the potential of HSP as a competitive inhibitor when it occupies the same docking sites as these standard drugs. 43
The findings from Figure 5 also suggest that HSP exhibits a stronger binding affinity to COX-2 compared to naproxen, positioning it as a potentially more effective therapeutic agent for rheumatoid arthritis (RA). While naproxen is widely used, the superior binding characteristics of HSP could enhance its efficacy in managing RA symptoms. This present study highlights the importance of exploring HSP as a new inhibitor that would offer improved treatment options when compared to conventional drugs currently used for RA treatments.
The docking analysis (Figures 6– 9) reveals that hesperidin acts as a non-selective inhibitor for target proteins like COX2, TNF-α, IL-1B, and IL-6, competitively binding to their pockets. COX2 uniquely shows binding within its pocket, while other proteins interact with hesperidin on the surface. Two-dimensional interaction analysis (Figures 10–13) identifies key binding forces, including hydrogen bonds, van der Waals interactions, pi-bonds, unfavorable donor interactions, and salt bridges. Hydrogen bonds and van der Waals interactions are the main stabilizing forces for both hesperidin and other ligands. The study highlights the potential for targeting cell surface receptors and ligands in drug development, with soluble ligands or extracellular receptor domains improving drug access to specific cellular functions. 44
The ADMET and drug-like properties study (Table 1) shows that hesperidin violated three of Lipinski's Rule of Five, suggesting potential bioavailability issues. These violations include a molecular weight of 610.56 (instead of ≤500), 15 H-bond acceptors (instead of ≤10), and 8 H-bond donors (instead of ≤5). Despite these, hesperidin demonstrated a potent COX-2 inhibitor and inhibits pro-inflammatory cytokines like TNF-alpha, IL-6, and IL-1β. The In-silico analysis deepens understanding of hesperidin's pharmacokinetic properties, essential for RA drug development. This study elucidated the significance of evaluating drug-likeness properties and illustrates how different computational tools can impact In vivo pharmacokinetics and pharmacodynamics. 45
Hesperidin (HSP) exhibited low gastrointestinal (GI) absorption, which violates Lipinski's criteria (Table 1), suggesting that its oral bioavailability is limited. To improve this, lead optimization of HSP's structure is recommended. This process in drug discovery refines lead compounds to enhance safety, absorption, metabolism, distribution, and therapeutic efficacy. Lead optimization can also improve pharmacokinetic profiles and binding affinities, which could improve HSP become a potential drug candidate for rheumatoid arthritis (RA).
The findings regarding hesperidin (HSP) as a P-glycoprotein substrate as shown in Table 1 is quite interesting. Its low absorption rate in the gastrointestinal tract and inability to cross the blood-brain barrier suggest that while it may not have central nervous system effects, it can still exert significant pharmacological actions through other mechanisms. Furthermore, clinical evidence supports HSP's efficacy in improving cognitive functions and alleviating symptoms of depression, highlighting its potential as a nutraceutical for central nervous system disorders. 46 Thus, while it may not impact the CNS directly, HSP still contributes to neuroprotection and overall health. Furthermore, hesperidin's interaction with P-glycoprotein could indeed influence its therapeutic profile. Since P-glycoprotein plays a crucial role in drug absorption and efflux, understanding HSP's behavior as a substrate could help optimize its use in combination therapies. The fact that it does not induce or inhibit cytochrome P-450 enzymes subunits reduces the likelihood of metabolic interactions, which is advantageous for its safety profile. Moreover, HSP's inhibition of the hERG II channel without being hepatotoxic or carcinogenic adds to its potential as a therapeutic agent. This balance of effects makes it a promising candidate for further research, especially in the context of drug-drug interactions and its potential use in various clinical settings.
The Cytochrome P450 (CYP450) family of enzymes plays a pivotal role in drug metabolism and the processing of xenobiotics, as highlighted by Manikandan and Nagini. 47 When drug-drug interactions alter CYP450 functions, they can lead to adverse reactions or ineffective treatments. Identifying these interactions is vital for developing new drug candidates, particularly given their influence on clinical efficacy. In this study, the focus on the inhibition profile of hesperidin (HSP) concerning key CYP450 isoforms (CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4) revealed that HSP does not inhibit any of these enzyme's subunits. This finding suggests that HSP would have a favorable safety profile as a therapeutic candidate for RA, as it is less likely to interfere with the metabolism of concurrently administered medications.
Summarily, the in vivo findings of this study, showing hesperidin's potential to reduce inflammatory cytokines such as TNF-α, IL-6, and NF-κB in CFA-induced RA female rats, align with the in silico molecular docking analysis, highlighting hesperidin's therapeutic potential. In-vivo, hesperidin, at doses of 100, 200, and 400 mg/kg, effectively reduced cytokine levels, with the highest dose yielding results comparable to the standard anti-inflammatory drug naproxen. These findings are supported by the In-silico docking analysis, where hesperidin showed strong binding affinities to key inflammatory targets such as COX-2, IL-1β, and TNF-α, with a higher binding affinity to COX-2 than naproxen. The In-silico results also suggest that hesperidin competes with standard inhibitors for the same binding sites, highlighting its potential as a competitive inhibitor. However, while the In-vivo data support its anti-inflammatory effects, hesperidin's molecular weight and violations of Lipinski's Rule of Five in the In-silico ADMET analysis indicate potential bioavailability challenges. Despite these issues, the pharmacokinetic properties could be improved with structural modifications to enhance its solubility and absorption, further supporting hesperidin's candidacy as a promising RA treatment. These combined In-vivo and In-silico findings illustrate hesperidin's potential to inhibit key inflammatory pathways in RA while providing insight into areas for improving its drug-likeness for clinical application.
Limitations of the Study
Despite the promising findings of this study, several limitations should be acknowledged. First, the study exclusively employed female Wistar albino rats, which, while modeling the higher prevalence of RA in females, may limit the generalizability of the findings to males. Second, the oral bioavailability of hesperidin was not directly measured in vivo; given the in-silico ADMET analysis showing violations of Lipinski's Rule of Five, the actual systemic absorption may differ from predicted pharmacokinetics. Third, the study focused primarily on pro-inflammatory cytokines (TNF-α, IL-6, NF-κB) and did not explore other mediators of RA pathogenesis, such as IL-17, IL-1β, or matrix metalloproteinases, which could provide further understanding of hesperidin's anti-arthritic effects. Fourth, the in-silico docking studies, while informative, represent predictive models of binding interactions and do not fully account for in vivo metabolic transformations, protein dynamics, or cellular uptake, which could influence hesperidin's efficacy. Future studies addressing these limitations, including pharmacokinetic optimization, dose-response evaluation, and broader mechanistic investigations, are warranted to fully validate hesperidin as a candidate for RA treatment.
Conclusion
In conclusion, this study has demonstrated hesperidin's potential as an anti-inflammatory treatment for rheumatoid arthritis (RA). In-vivo, hesperidin reduced cytokine levels (TNF-α, IL-6, NF-κB) in CFA-induced RA rats, with the highest dose showing effects similar to naproxen, the standard drug for RA treatment. In-silico docking analysis revealed strong binding to inflammatory targets like COX-2, IL-1β, and TNF-α, with hesperidin binding more strongly to COX-2 than naproxen. Despite potential bioavailability limitations due to molecular weight and Lipinski's violations, lead optimization could improve HSP's pharmacokinetics, making hesperidin a promising, multi-target RA therapeutic candidate, highlighting the novelty of this study.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251413007 - Supplemental material for Hesperidin Suppresses Complete Freund's Adjuvant (CFA)-Induced Rheumatoid Arthritis (RA) in Wistar Albino Rats by Targeting TNF-α/IL-6/IL-1β and NF-κB/COX-2 Pathways
Supplemental material, sj-docx-1-npx-10.1177_1934578X251413007 for Hesperidin Suppresses Complete Freund's Adjuvant (CFA)-Induced Rheumatoid Arthritis (RA) in Wistar Albino Rats by Targeting TNF-α/IL-6/IL-1β and NF-κB/COX-2 Pathways by Celestine O Ogbu, Peter Chinedu Agu, Daniel E Uti, Kizito Akachukwu Agu, Samuel Agada, Joseph U Odo, Christian Emeka Offor, Mathias J Okpe, Nkiru N Ezeani, Obasi U Orji, Ilemobayo Victor Fasogbon, Angela Musyoka Mumbua, Ibrahim Babangida Abubakar, Rosemary Duruiheoma and Patrick Maduabuchi Aja in Natural Product Communications
Footnotes
List of Abbreviations
Acknowledgements
We most heartily acknowledge the Department of Biochemistry, Ebonyi State University Abakaliki, Nigeria, Federal University of Health Sciences Otukpo, Nigeria and Department of Biochemistry, Faculty of Biomedical Sciences, Kampala International University, Western Campus, Uganda for the insightful encouragements and technical supports throughout this study period.
Ethical Approval Statement
All procedures in this study were carried out in compliance with protocols approved by the Department of Biochemistry Research Ethics Committee, acting on behalf of the Institutional Ethics Review Committee of Ebonyi State University. Ethical approval was granted under license number/code EBSU/BCH/ET/23/001, issued on March 30, 2023.
Authors’ Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
