Abstract
N-acetylglucosamine (GlcNAc) is a versatile monosaccharide with broad applications in medicine, food, and cosmetics due to its antimicrobial, antioxidant, and anti-tumor properties. Efficient synthesis methods for GlcNAc remain a prominent research focus. O-GlcNAc modification, prevalent in nuclear and cytoplasmic proteins, is of particular interest, especially regarding the catalytic mechanism of the O-GlcNAcase (OGA) enzyme. Enzymatic processes using chitin as a substrate, yield GlcNAc-based oligosaccharides, offering diverse health benefits including antioxidant, anti-inflammatory, anti-tumor, and antimicrobial effects. This review examines GlcNAc preparation methods, O-GlcNAc modification mechanisms, and the applications of GlcNAc-based oligosaccharides in various fields.
Keywords
Introduction
Chitin, the most abundant nitrogen-containing biopolymer on Earth, yields its monomer, N-acetylglucosamine (GlcNAc), a compound of significant application value across various domains and a vital intermediate in chitin's conversion to other organic nitrogen compounds. GlcNAc (

Chemical structure of GlcNAc and its derivatives.
Tomonaga et al randomly selected 120 healthy subjects without arthritis and evaluated cartilage metabolism by analyzing markers of type II collagen degradation (C2C) and type II collagen synthesis (PIICP), as well as the ratio of type II collagen degradation to type II collagen synthesis (C2C/PIICP). The observation results indicate that oral administration of GlcNAc at a dose of 500 milligrams per day has a cartilage protective effect on subjects without arthritis. This effect is achieved by improving cartilage metabolism (reducing type II collagen degradation) without causing significant adverse effects. 9 Gueniche et al evaluated the effect of GS on skin aging in vitro through different immunohistochemical assays, histology, and clinical studies using biopsy. Real time quantitative PCR was used to evaluate the regulation of selected skin physiological markers through skin biopsy of 8 healthy women over 50 years old who consumed 250 mg of GS once a day for 8 weeks. After 8 weeks of oral administration of GS, significant increases in mRNA expression of vimentin, fibronectin, disaccharides, xylose transferase, hyaluronic acid synthase, type I and III collagen, bone morphogenetic protein-1, and core proteoglycans were observed (all p ≤ 0.05). Therefore, this experiment indicates that GS has a positive effect on age-related epidermal and dermal markers. 10
Chitosan, a biopolymer formed by binding GlcN through β-glycosidic linkages, offers a versatile platform for derivatives including polysaccharides, oligosaccharides, and monosaccharides. These derivatives exhibit various therapeutic activities spanning immune modulation, anti-tumor properties, support for osteoarthritis treatment, wound healing, tissue engineering, drug delivery, and regenerative medicine. 11
In recent years, the terminal hydrolysis product of chitosan, GlcN, has emerged as a compelling food supplement and cosmetic biomaterial. Moreover, N-acetylglucosamine possesses a naturally sweet taste, rendering it a viable alternative to certain sugar additives.
The role of GlcNAc derivatives in contributing to sustainability in agriculture and the food industry is a topic of increasing interest and importance. GlcNAc derivatives can be used to develop biopesticides or biocontrol agents. For example, chitinases derived from GlcNAc can degrade chitin in insect exoskeletons, making them effective against pests like beetles or caterpillars. This reduces reliance on chemical pesticides, which can be harmful to the environment. 12 Meanwhile, the use of green pesticides also ensures the food safety of agricultural and sideline products.
Preparations
In general, three strategies exist for the chemical production of GlcNAc. The first involves employing the Haber-Bosch process, which yields large quantities. 13 The second strategy entails screening and modifying microorganisms to ferment GlcNAc. 14 The third strategy revolves around directly degrading nitrogen-containing biomasses like chitin to release GlcNAc units. 15 Research has focused on enzymes and chemical catalysts for chitin degradation into GlcNAc. Compared to the first strategy, the latter two are more appealing as they enable GlcNAc production from renewable resources. Moreover, the development of environmentally friendly technologies for efficient and selective GlcNAc production is crucial for fully leveraging the potential of nitrogen-containing biomasses.
Preparations by Chemical Means
Presently, the industrial synthesis of nitrogen-containing chemicals heavily relies on the Haber-Bosch process. This method involves incorporating nitrogen functionalities onto nitrogen-free substrates, such as glucose, through the Haber process and subsequently converting them into GlcNAc. However, this approach often generates significant amounts of by-products and consumes substantial energy, rendering it environmentally taxing. Hence, there is an urgent imperative to develop a green and sustainable method for GlcNAc synthesis. This has sparked increasing interest in producing these organic nitrogen compounds from biomass. Chitin, the most abundant nitrogen-containing biopolymer on Earth, serves as a prime candidate for conversion into GlcNAc, a topic garnering significant attention in the synthesis of nitrogen-containing compounds. In recent years, various chemical methods have emerged for GlcNAc production from biomass. Compared to enzymatic approaches, chemical methods typically offer advantages such as lower cost, simplicity of operation, and higher efficiency. Scientists have identified numerous factors influencing the chemical production process of GlcNAc, including substrates, solvents, catalysts, reaction temperature, and duration.
Producing pure GlcNAc from chitin poses several challenges, including achieving high yield and selectivity while minimizing the consumption of reagents and additives. Directly converting chitin to GlcNAc remains difficult due to the rate-determining depolymerization of the crystalline chitin region and the formation of undesirable byproducts. To address these hurdles, it is essential to establish effective catalytic systems for chitin depolymerization. Additionally, methods such as ball mill grinding can be utilized to decrease crystal size and disrupt hydrogen-bond networks, thereby enhancing chitin reactivity.
Chitosan stands out as the predominant substrate utilized in the chemical preparation of GlcNAc. The industrial processing of shrimp generates substantial quantities of inedible by-products, which, if left unchecked, can detrimentally impact the environment and human health. Notably, these shrimp industrial by-products contain a significant chitin content, 16 offering a valuable resource for the production of nitrogen-containing platform compounds like GlcNAc through specific chemical reactions. 17 Efficient methods for converting chitosan into NAG involve establishing effective catalytic systems for chitosan degradation, employing ball milling to diminish crystal size and disrupt hydrogen bonding networks to heighten chitosan reactivity, and utilizing multifunctional solid acid catalysts for highly selective conversion of chitosan into NAG or glucosamine.
During chitin degradation, acids ionize in water, yielding hydrogen ions that subsequently combine with water to form unstable hydronium ions. These hydronium ions then engage with the β-1,4 glycosidic bond of chitin, causing the conversion of a hydrogen ion into the oxygen atom within the glycosidic bond. This process facilitates the cleavage of the C-O bond. Under conditions of heightened acidity or increased loading, chitin can undergo direct hydrolysis to yield GlcN. Typically, an acetylation process involving acetic anhydride is necessary to convert GlcN back into GlcNAc.
The simplicity and efficiency of the chitosan liquid acid hydrolysis process is demonstrated when the treatment of acid wastewater is not taken into account, as well as the support of further expensive corrosion-resistant equipment.
18
Apart from inorganic acids, various inorganic salts have been investigated for their potential in chitin degradation to GlcNAc. Gözaydın et al
19
conducted a study on the dissolution and depolymerization of chitin in Acidic Molten Salt Hydrate (AMSH) systems (
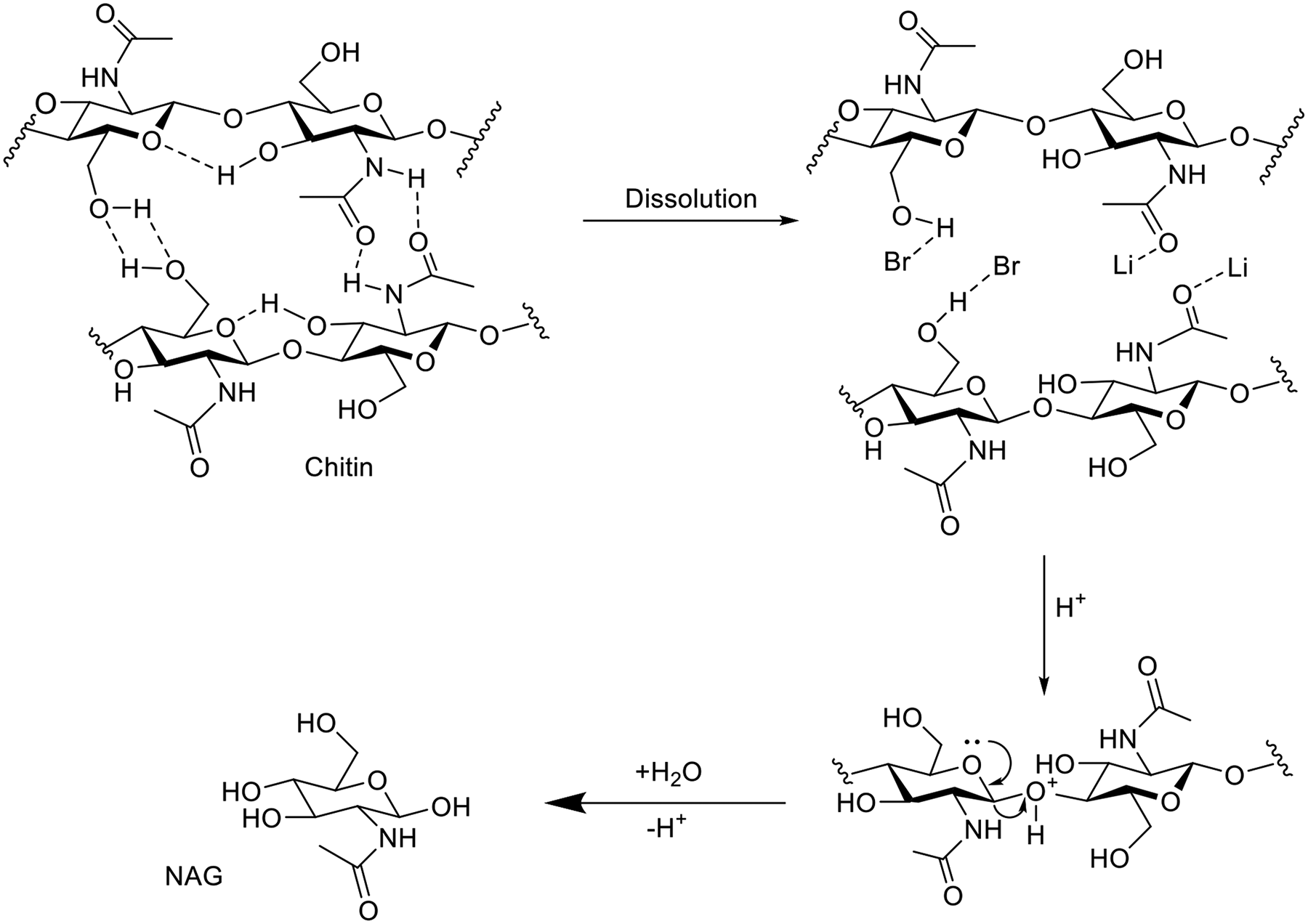
Dissolution and depolymerization of chitin in Acidic Molten Salt Hydrate (AMSH). Adopted from Reference, 19 under the copyright of the Royal Society of Chemistry.
Preparation by Biological Means
Microbial fermentation typically employs glucose as the substrate and amino acids as the nitrogen source to produce GlcNAc. Although this process can be conducted under mild conditions, fewer harmful by-products; however, it is comparatively less efficient. GlcNAc produced through enzymatic methods is characterized by its safety, low cost in terms of raw materials, and ready availability. Due to the inherent resistance of the chitin structure, the key of this approach often necessitates extensive pretreatment processes to achieve high yields of GlcNAc.
Various chemical methods and auxiliary physical treatments 21 have been employed to reduce the highly crystalline structure of chitin and achieve successful depolymerization. Enzymatic hydrolysis presents distinct advantages in overcoming challenges such as low product quality, low yield, and environmental concerns. If the use cost of enzymatic method can be reduced, it will greatly promote its application in industry. 14 Therefore, researchers have turned their focus to inducing microorganisms to produce chitinases using chitin-rich by-products, driven by considerations of environmental sustainability and cost reduction in enzyme production.
Firstly, glucose undergoes phosphorylation to form Glucose-6-Phosphate (G-6-P) catalyzed by Glucokinase (GK), encoded by the glcK gene, within the glycolytic pathway. Subsequently, Phosphoglucose Isomerase (PGI), encoded by the PGI gene, converts G-6-P to Fructose-6-Phosphate (F-6-P). F-6-P is then transformed into Glucosamine-6-Phosphate (GlcN-6-P) by Glucosamine Phosphate Synthase (GlmS), encoded by the GlmS gene, utilizing glutamine as the amino donor.
13
This conversion constitutes the rate-limiting step in the GlcN synthesis pathway and is subject to feedback inhibition by the final product GlcN. GlcN-6-P can undergo acetylation to produce N-acetylglucosamine-6-Phosphate (GlcNAc-6-P) via the action of Glucosamine N-acetyltransferase-1 (GNA1), encoded by the GNA1 gene22,23 (

Metabolic engineering pathways of GlcNAc. GlcNAc: acetylglucosamine; Glc-6-P: glucosamine-6-phosphate; Fru-6-P: fructose-6-phosphate; GlcNAc-6-P: acetylglucosamine-6-phosphate; ptsG: gene encoding glucose phosphate transporter; HMP: hexose monophosphate pathway; galP: galactosidase permease gene; glcK: glucokinase gene; pgi: phosphoglucose isomerase gene; glmS: glucosamine synthase gene; gamA, nagB: GlcN-6-P deaminase gene; manX: mannose phosphate transporter gene; gna1: acetylglucosamine synthase gene; nagA: 6-phospho-N-acetylglucos-aminedeacetylase; nagE: acetylglucosamine phosphate transporter gene; ywpJ: phosphatase gene. nagP: phosphotransferase system GlcNAc specific protein gene; zwf: Glucose-6-phosphate dehydrogenase gene. glmM: GlcN-1-P mutase; glmU: uridine acyltransferase.
Research on microbial fermentation for GlcNAc production primarily emphasizes three aspects: microbial screening, microbial metabolic control, and fermentation process optimization. Of these, microbial metabolic control poses the greatest challenge. For instance, in the metabolic pathway of GlcNAc in Bacillus subtilis, metabolic control typically entails enhancing the synthesis pathway of GlcNAc, inhibiting the degradative pathways of GlcNAc, and facilitating the transport system for recombinant GlcNAc. 27
The production of GlcNAc using metabolically engineered bacteria typically involves utilizing glucose as the substrate and amino acids as the nitrogen source. Before 2010, most studies employed Escherichia coli as the host organism, achieving GlcNAc productivity of up to 118.78 g/L under optimized conditions. However, due to the endotoxins produced by E. coli during fermentation and its susceptibility to bacteriophage attacks, GlcNAc produced using this method often fails to attain Generally Recognized as Safe (GRAS) status, limiting its application in the nutrition and pharmaceutical industries. In recent years, research has predominantly focused on constructing genetically engineered bacteria, such as Bacillus subtilis and Corynebacterium glutamicum, as expression hosts for fermentative GlcNAc production. These bacteria are favored for their ease of cultivation, lack of spore production, and resistance to bacteriophage infection. Under optimized conditions, engineered B. subtilis and C. glutamicum can achieve GlcNAc productivities of 35.77 g/L and 83 g/L, respectively. Additionally, some researchers have explored the use of Saccharomyces cerevisiae for GlcNAc fermentation; however, the product yield is low, and the fermentation process is prolonged. Under optimized conditions, GlcNAc productivity after 135 h of fermentation is only 3.0 g/L. 28
The metabolic pathways of GlcNAc have been extensively explored in most strains, yet they remain incompletely understood. The challenge in metabolic control lies in gaining a clear understanding of the bottleneck or limiting steps in these pathways, identifying relevant genes, and comprehending the mechanisms that regulate the expression of these genes. It is anticipated that with the advancement of gene editing technologies, the pace of constructing engineered bacteria with the desired metabolic pathways will significantly accelerate. Additionally, aside from microorganisms, fermentation conditions and methods play a significant role in GlcNAc production. Factors influencing GlcNAc yield encompass inoculum cell density, lactose induction time, glucose feeding rate, medium composition and dissolved oxygen levels, among others. 29 Existing fermentation methods can be categorized into batch fermentation, fed-batch fermentation, and staged fermentation, with fed-batch fermentation being the most commonly utilized approach.
Chitinases comprise a broad category of enzymes capable of breaking down glycosidic bonds linking GlcNAc monomers, encompassing exochitinases and endochitinases. Exochitinases further divide into chitobiosidases and β-N-acetylglucosaminidases based on their action on chitin. Chitinase specifically targets chitin, a biopolymer composed of beta - (1→4) - linked N-acetylglucosamine (GlcNAc) units. This specificity enables chitinase to effectively degrade chitin into GlcNAc monomers and oligosaccharides. These enzymes can be acquired through various pathways, being naturally produced by microorganisms, plants, animals, and even viruses. 30 Bacteria primarily produce chitinases to degrade chitin, utilizing it as a source of carbon and nitrogen to meet their nutritional requirements for growth and reproduction. 31 Of note, microbial chitinases are extensively employed in chitin production for GlcNAc, owing to their abundance, high activity, accessibility, and potential for large-scale production. 32
The technical route for enzymatic production of GlcNAc primarily comprises three steps: (1) screening for high-producing chitinase strains. Colloidal chitin commonly serves as the carbon source for sreening high chitinase-producing strains, with enzyme activity typically measured using the dinitrosalicylic acid method. Strain identification involves morphological observations, such as colony and cell morphology, along with DNA sequence analysis, including 16S rDNA sequence analysis. (2) The production of chitinase is influenced by various factors, including fermentation temperature and duration, substrate, and pH, inoculum size, inoculum age, fermentation volume, agitation speed, and additives. (3) Optimization of the enzymatic hydrolysis process. Commonly used methods for optimizing culture medium formulations and fermentation conditions encompass Plackett-Burman design, gradient optimization experiments, response surface methodology, single-factor experiments, and orthogonal experimental design. 33 After fermentation, a purification process is necessitated to obtain relatively pure chitinase, typically employing techniques such as ammonium sulfate precipitation, dialysis, and chromatography. SDS-PAGE can be employed to assess the purity and molecular weight of chitinase. 34 The CRISPR/Cas system is a precise gene editing tool that can be used to modify the genome of microorganisms to optimize metabolic pathways. In GlcNAc production, the CRISPR/Cas system can be used to knock out or overexpress related enzyme genes, optimize key enzymes for glucosamine synthesis, such as the gene for N-acetyltransferase (GlcNAc convertase), thereby improving production efficiency.15,24,35
Some chitinases exhibit broad substrate specificity, functioning not only as chitinases or chitosanases to degrade chitin into oligosaccharides but also further hydrolyzing the oligosaccharides into GlcNAc. Tran et al
36
purified chitinase TKU048 from Streptomyces sp. TKU048, which displayed a specific activity of up to 45.668 U/mg and could hydrolyze 73.64% of β-chitin into GlcNAc within 96 h under conditions of 50 °C and pH 5.0. Zhang et al
37
isolated chitinase CmChi1 from the chitinolytic fungus SYBC-H1, which, after purification, exhibited a specific activity of 15.3 U/mg and completely hydrolyzed 98% of colloidal chitin into GlcNAc within 24 h. Pradeep et al
38
purified chitinase Ch147 from Streptomyces sp. CS147. Under conditions of 50 °C and pH 11.0, the reaction yielded 1.058 g/L of GlcNAc within 24 h. Koji Matsuoka, et al
39
found an improved preparation of O - (2-acetamido- 3, 4, 6-tri-O-acetyl-2-deoxy-β-D-glucopyranosyl)-(1→4)-2-acetamido-13,6-tri-O-acetyl-2-deoxy-
Compared to other GlcNAc production methods, biological methods generally provide benefits such as operating under mild reaction conditions and yielding high amounts of target products, they often entail longer processing times and complex operating procedures. Moreover, the cost associated with screening microorganisms and producing enzymes is substantial. Future research efforts should focus on deepening our understanding of the synergistic degradation mechanisms involving multiple enzymes, thereby establishing the theoretical groundwork for the efficient conversion of chitin into GlcNAc.
O-GlcNAc Modification
The O-linked N-acetyl Glucosamine (O-GlcNAc) glycosylation modification stands as a critical post-translational alteration of proteins, where a single N-acetylglucosamine (GlcNAc) forms an O-glycosidic bond with the hydroxyl group of Serine (Ser) or Threonine (Thr) residues within proteins. GlcNAc is the actual sugar that gets attached to proteins in the O-GlcNAcylation process, making it a fundamental component of this post-translational modification. This modification is closely associated with numerous diseases, and the emergence of O-GlcNAc perturbation as a novel therapeutic avenue underscores the paramount importance of identifying and quantifying protein sets modified by O-GlcNAc. O-GlcNAc stands as one of the primary chemical codes utilized in cellular signal transduction. Its discovery in mammalian cells dates back to 1984, and since then, it has been identified on thousands of proteins within the nuclei, cytoplasm, and mitochondria across various species.40–43 Given its presence on proteins within these cellular compartments and its biosynthetic connection to glycolysis, amino acid synthesis, nucleotide levels, and fatty acid levels, O-GlcNAc modification is commonly acknowledged as a nutrient sensor.
The intersection between O-GlcNAc and phosphorylation signaling pathways was first proposed with the discovery of O-GlcNAc on serine and threonine residues, also serving as phosphorylation sites. 44 This finding spurred numerous investigations aimed at exploring the crosstalk between these two modifications. The interface of O-GlcNAc and phosphorylation is a crucial pillar of cellular signal transduction orchestrated by these modifications, particularly because many sites of O-GlcNAc modification overlap with phosphorylation sites. Like phosphorylation, O-GlcNAc is a dynamic modification that can rapidly cycle on protein substrates. 45 However, in contrast to phosphorylation, which is regulated by over 500 kinases and phosphatases catalyzing the addition and removal of phosphate groups on thousands of substrates, O-GlcNAc is solely regulated by two enzymes acting on thousands of substrates: O-GlcNAc Transferase (OGT) for installation and O-GlcNAcase (OGA) for removal of O-GlcNAc from proteins. Early studies have demonstrated the crucial importance of OGT and OGA for organismal development, providing evidence for the functional significance of O-GlcNAc cycling. OGT is essential for normal human neurodevelopment, and its loss in mice leads to embryonic lethality. In mouse embryos, loss of OGA results in neonatal death and developmental delay. Additionally, conditional loss of OGT in many cell types leads to aging and apoptosis. Numerous functional consequences of protein O-GlcNAcylation have been described, including changes in protein-protein interactions, subcellular localization, enzyme activity, and protein stability. Dysregulation and abnormal levels of O-GlcNAc have been associated with various diseases,46,47 including diabetes, 48 cancer, 49 immune disorders, 50 and neurodegenerative diseases, 51 sparking efforts to target O-GlcNAc modification as a potential therapeutic intervention.
O-GlcNAc modification has emerged as a significant regulator of phosphorylation in cellular signal transduction pathways. Beyond its interplay with protein substrate phosphorylation, it also influences kinase activity. This regulatory mechanism through O-GlcNAc modification potentially allows cells to allocate resources across different pathways based on nutrient availability, thereby finely tuning signaling pathways through phosphorylation and other modifications. As techniques for studying and manipulating O-GlcNAc on both a system-wide and individual protein level converge, further investigations into the role of O-GlcNAc on kinases and the enzymes responsible for its installation will provide valuable insights into the regulatory mechanisms cells employ to integrate these pathways. Specifically, the impact of O-GlcNAcylation on the STE, TK, and TKL kinase families remains to be fully elucidated. Given the pervasive nature of O-GlcNAc in cells, a deeper understanding of its effects on kinases promises significant discoveries in cellular regulation and dysregulation across various biological contexts, spanning normal physiology to disease states.
Detection and quantification of O-GlcNAc modification within the proteome pose considerable challenges. 41 Its discovery in 1984, amidst measurements of radiolabeled sugars in the nuclear-cytoplasmic space, occurred decades after the identification of other PTMs like phosphorylation on the same proteins. This delayed recognition of O-GlcNAc may be attributed to its elusive nature; changes in O-GlcNAc levels typically do not alter the migration of glycoproteins during gel electrophoresis, and O-GlcNAc quickly degrades on enzymes upon cell disruption or lysis. Furthermore, O-GlcNAc's chemical instability leads to ion suppression of glycopeptides in Mass Spectrometry (MS) analysis, 52 and direct fragmentation of peptide glycans during ionization in the mass spectrometer, ultimately enhancing the detection limit for MS analysis of O-GlcNAc. Recent years have witnessed significant progress in chemical glycoproteomics, accelerating the identification of global protein modification sites.53,54 Recent reviews have emphasized these advancements, particularly highlighting the strides made in large-scale analysis of O-GlcNAc, characterized by extensive complex glycoproteomic studies. 55
O-GlcNAc modification plays a crucial role in regulating various cellular functions across different tissue types. In neuronal tissues, O-GlcNAc modification is involved in synaptic plasticity, learning, and memory formation. It affects the activity and stability of signaling proteins, such as those involved in neurodegenerative diseases like Alzheimer's disease, where aberrant O-GlcNAcylation can contribute to tau hyperphosphorylation and neurofibrillary tangle formation. O-GlcNAc modification affects cell cycle progression and apoptosis in cancer cells. It can modulate the function of tumor suppressors and oncogenes, influencing cancer cell proliferation and survival. In muscle tissues, O-GlcNAc modification impacts muscle growth, insulin sensitivity, and metabolism. It affects the function of proteins involved in muscle contraction and metabolism. 56
Drawing the O-GlcNAc-modified proteome presents some unique challenges in traditional bottom-up DDA proteomics, not only due to challenges in site localization when using collision-induced MS2 fragment techniques but also due to limitations in sample enrichment.57,58 Various strategies for O-GlcNAc enrichment exist, each with its advantages and limitations. O-GlcNAc metabolic labeling techniques have been successful in multiple iterations, providing an excellent platform for studying various issues, but may not be easily applicable to samples generated outside of laboratory tissue cultures.57,58 Lectin weak-affinity chromatography, as a cost-effective method, has been widely used for the enrichment of naturally occurring O-GlcNAc peptides and has generated the largest dataset of O-GlcNAc-modified proteomes to date.59,60 However, it is limited by sample input requirements when dealing with scarce samples.59,60 Anti-O-GlcNAc antibodies hold promise as a sensitive and specific method for the enrichment of naturally occurring peptides, capable of mapping over 1000 unique sites with only 4 milligrams of input material, 61 but may be too expensive for some studies. 61 Antibody-based methods may have limitations due to the specificity of antigen-binding sites; however, polyclonal antibody mixtures have demonstrated significant coverage across multiple PTM spaces.61,62
Aberrant O-GlcNAcylation is linked to various cancer types and biological processes relevant to cancer.63,64 Consequently, a logical progression involves conducting global quantitative site mapping of O-GlcNAcylation in naturally occurring tissues of tumor types exhibiting aberrant O-GlcNAcylation. Similar to other Post-Translational Modification (PTM) spaces, mapping O-GlcNAcylation across different cancer types may yield biomarkers for diagnostics and insights into signaling mechanisms for therapeutic intervention. Moreover, natural peptide O-GlcNAc enrichment methods can be integrated with sequential PTM enrichment strategies to elucidate signaling interactions between PTM spaces. Beyond identification and abundance, understanding the functional impact of O-GlcNAc modification on target proteins remains paramount. One cancer-related phenomenon influenced by O-GlcNAcylation is the Warburg effect, a characteristic of cancer cells.65,66 This effect is characterized by elevated glycolysis and glucose uptake rates, leading to preferential lactate production even in the presence of oxygen. The heightened glucose consumption serves as a carbon source for biosynthetic processes, supporting uncontrolled cell proliferation. Key regulators of this effect include glycolytic enzymes such as Phosphoglycerate Kinase 1 (PGK1) and Pyruvate Kinase M2 (PKM2).
The current technology for identifying and quantifying O-GlcNAc sites on proteins is limited. Post-translational mutagenesis has been proposed as a method to overcome some limitations of protein ligation for studying site-specific O-GlcNAc modifications. Precision mapping of the human O-GalNAc glycoproteome through SimpleCell technology has shown promise for the identification of native O-GlcNAc modified peptides. Recent developments in analytical methods for disease-specific protein O-GlcNAcylation have also been reported. 67 Furthermore, advancements in mass spectrometry-based techniques have shown potential for the precise characterization and quantification of O-GlcNAc modifications on proteins, offering new insights into the regulatory mechanisms of this post-translational modification. 56
GlcNAc-Based Oligosaccharides
In recent years, significant attention has been directed towards low-molecular-weight oligosaccharides based on GlcNAc, acquired through chitin degradation or biosynthesis, with their molecular weight serving as an indicator. Among these, N-acetyl chitooligosaccharides (N-acetyl COS), also termed fully acetylated chitooligosaccharides, stand out as a prominent example of GlcNAc-based oligosaccharides linked by β-1,4 glycosidic bonds containing GlcNAc units.68,69 Various effective physical, chemical, and enzymatic methods have been employed for chitin degradation to yield N-acetyl COS. 70 These oligosaccharides possess outstanding biological activities, including antimicrobial, antitumor, and hypoglycemic properties, coupled with water solubility, temperature stability, and pH stability. Particularly, N-acetyl COS with a Degree of Polymerization (DP) of 2–6 has garnered significant attention recently and has found wide applications in agriculture, food, and pharmaceutical industries.71,72 Apart from N-acetyl COS, lacto-N-triose II (GlcNAcβ1-3Lactose, LNT2) stands as another notable GlcNAc-based oligosaccharide. Human Milk Oligosaccharides (HMOs) exhibit diverse biological activities crucial for infant health and development, including enhancing gut microbiota, promoting systemic immune system development, and reducing pathogenic infections.35,73 Many HMOs, such as lacto-N-neotetraose (galβ1-4GlcNAcβ1-3Lactose, LNnT) and lacto-N-tetraose (galβ1-3GlcNAcβ1-3Lactose, LNT), can be synthesized by extending LNT2 with other oligosaccharides or monomers. 74 LNT2 can be generated through whole-cell biotransformation or synthesized using N-acetyl COS as the donor via β-N-acetylhexosaminidase.74,75 Serving as the backbone precursor of most HMOs,76,77 LNT2 exhibits diverse biological activities, including prebiotic, antimicrobial, and immunomodulatory properties.
The extensive utilization of these two GlcNAc-based oligosaccharides can be attributed to their remarkable biological activities, encompassing antioxidant, anti-inflammatory, anti-tumor, and antimicrobial properties, alongside immunomodulatory, plant elicitor, and prebiotic effects.70,78 Nonetheless, in comparison to another chitin-derived polysaccharide, chitosan oligosaccharides (chitosan), the reported biological activities of these two GlcNAc-based oligosaccharides are relatively less explored, primarily due to the greater challenges associated with large-scale production. Compared with other biomolecule compounds, these two glcnac based oligosaccharides have lower molecular weight and better solubility, and have more beneficial health effects, including antioxidant, anti-inflammatory, anti-tumor, and antibacterial activities. 70 N-acetyl cos has the effect of inhibiting cellular oxidative stress and can be used as an antioxidant component. Through indirect and direct means, low molecular weight N-acetyl cos may have potential free radical scavenging effects by inhibiting and preventing biomolecule damage caused by free radicals in living cells. 79 Inflammation constitutes a pivotal response of the host to various stimuli, including physical injury, microbial invasion, ultraviolet radiation, and immune reactions that can precipitate various diseases.78,80,81 N-acetyl COS demonstrates anti-inflammatory activity under specific conditions; it can mitigate the production of nitric oxide (NO) and Prostaglandin E2 (PGE2) by downregulating the activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), thereby suppressing Interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α, consequently manifesting anti-inflammatory effects. Additionally, it attenuates Lipopolysaccharide (LPS)-induced phosphorylation of Mitogen-Activated Protein Kinases (MAPKs) in BV-2 microglial cells. 82
The immune system plays a pivotal role in defending against foreign pathogens, and the appropriate use of immunomodulators can bolster the host's defense response. 83 Following intraperitoneal injection of 50 mg/kg of N-acetyl COSs (with degrees of polymerization ranging from 4-7) for 3 h, male BALB/c mice exhibited an augmentation in the number of Peritoneal Exudate Cells (PEC). Polymorphonuclear leukocytes (PMNs), which constitute the majority of PEC, are presumed to harbor receptors for N-acetyl glucosamine residues on their cell surface. 84 By binding to these receptors, N-acetyl COSs can activate PMNs, thereby eliciting an immune-regulatory response in these cells. Subsequent investigations have demonstrated that N-acetyl hexosamine possesses the capacity to combat infections caused by Candida albicans, Mycobacterium avium, and Pseudomonas aeruginosa, consequently enhancing the activity of splenic Natural Killer (NK) cells and fostering the production of inflammatory cytokines.85,86
Cancer, poses a significant global health challenge with its high mortality rates. 87 Since the 1980s, extensive research has shed light on the anti-tumor properties of N-acetyl chitosan, particularly its ability to markedly impede the growth of murine transplantable sarcoma 180 and MM-46 solid tumors, demonstrating notable tumor growth inhibition rates. 84 Investigations have revealed a correlation between the anti-tumor efficacy of N-acetyl COSs and their Degrees of Polymerization (DPs). Specifically, N-acetyl chitosan has been validated to suppress the growth of methyl a solid tumor transplanted in BALB/c mice, while N-acetyl chitosan did not elicit a similar effect. 88 The anti-tumor mechanisms of N-acetyl COSs are purportedly associated with their capacity to enhance T-cell proliferation by stimulating lymphocyte cytokines. Essentially, it is believed that N-acetyl COSs bolster acquired immunity by expediting T-cell maturation, thereby augmenting cytotoxicity and sustaining T-cell functionality throughout the anti-tumor process. 89
While advancements have been made in both the production and biological functionality of these two varieties of GlcNAc oligosaccharides (
Biological Activities of COS and GlcNAc. 11 .

Conclusion
GlcNAc, a versatile monosaccharide with extensive applications in food, medicine, and cosmetics, is attracting increasing attention as a crucial component of various functional polysaccharides and oligosaccharides. The utilization of bioprocesses for GlcNAc production represents a promising avenue. Presently, microbial synthesis of GlcNAc can be modulated not only through conventional methods such as gene knockout and regulation but also by exploring potential metabolic network regulation tools. Studies have demonstrated that effective metabolic network regulation tools play a pivotal role in reallocating metabolic flux, making them optimal for enhancing metabolite yield and productivity.
O-GlcNAc, a multifaceted Post-Translational Modification (PTM), exerts significant regulatory control over numerous cellular signaling processes by modifying thousands of proteins across diverse tissue types. Notably, advancements in technology have accelerated the ability to accurately map the O-GlcNAc-modified proteome at a site-specific level. Various strategies for O-GlcNAc enrichment exist, each presenting distinct advantages and limitations. The ETD data acquisition strategy stands out as a hallmark technique for site localization of HexNAc residues. The combination of a product-triggered approach, employing both HCD and ETD fragmentation methods, yields valuable insights into putative HexNAc-modified peptides. Furthermore, HCD fragmentation may generate an “O-GlcNAc fingerprint” through the dissociation of HexNAc oxonium ions, providing crucial stereochemical resolution for probing O-GlcNAcylation beyond cytoplasmic and nuclear proteins.
GlcNAc, a versatile monosaccharide with wide-ranging applications in food, medicine, and cosmetics, garners increasing attention as a vital component of functional polysaccharides and oligosaccharides. Microbial synthesis of GlcNAc shows promise, regulated through traditional gene knockout and metabolic network tools. Significant advancements in mapping O-GlcNAc-modified proteome have accelerated, aided by diverse enrichment strategies. GlcNAc-based oligosaccharides offer diverse health benefits and find widespread use in food, agriculture, and pharmaceutical industries. Chitinases emerge as preferred enzymes for GlcNAc oligosaccharide production due to their mild conditions and sustainability. Further research on chitinases suitable for industrial purposes remains crucial.
Footnotes
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
This article does not contain any studies with human or animal participants, ethical approval was not required
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the High-level Talents Special Program of Zhejiang, Lishui KRD Project, Lishui Public Welfare Project, (grant number 2022R52036, 2021FSYYZZ08, 2022ZDYF24, 2022GYX69).
Data Availability
The authors will supply the relevant data in response to reasonable requests.
