Abstract
Context
A prescription medication called Anchang Decoction (ACD) has anti-inflammatory properties.
Objective
To evaluate the mechanism of ACD against inflammation.
Materials and methods
The “pharmacodynamic constituents - potential targets” and “protein interaction networks” were mapped using Cytoscape software and the STRING database, respectively. The degree of binding between important pharmacodynamic components of ACD and possible targets was then examined using molecular docking analysis using Autodock Vina and PyMOL software, and GO and KEGG pathway enrichment analysis using the David database and the Weishengxin online tool. These findings were eventually confirmed in vitro.
Results
After the intersection of the two, 539 inflammatory targets and 217 related targets, 34 main active components, and 42 potential anti-inflammatory targets were obtained. These include AKT1, MAPK14, SRC, EGFR, GSK3B, MMP9, MMP2, PTGS2, SYK, ESR1, and MMP2 and GO enrichment results. These three key targets are chosen as downstream validation targets for experimental verification. Furthermore, the colonic tissue and mucosa of ACD group were undamaged in comparison to the model group, and there was no sign of inflammatory cell infiltration. According to CCK-8 data, treatment with 20% ACD drug-containing serum resulted in a significant increase in RAW264.7 cell viability (P < 0.05) when compared to the normal serum group; Serum of ACD-containing medication may considerably lower the NO content of macrophage inflammation and prevent the production of inflammatory markers like TNF-α and IL-6 (P < 0.05); The expression levels of AKT1, MAPK14, and SRC proteins were considerably reduced by ACD in RAW264.7 macrophage inflammation, according to Western Blot data (P < 0.05).
Discussion and conclusions
ACD exerts anti-inflammatory effects through multi-component interaction with the target, and the mechanism may involve the inhibition of the release of inflammatory cytokines by AKT, MAPK and non-receptor tyrosine kinase signaling pathways. Here, the molecular mechanism of ACD against inflammation was partially clarified and experimentally validated, offering theoretical evidence for more effective clinical application.
Introduction
Not only is inflammation a vital protective mechanism of the body, but it also serves as a foundation for numerous clinical disorders. 1 First of all, inflammation is the body's protective mechanism that works to eliminate foreign objects, boost tissue repair, and activate the immune system. 2 However, prolonged inflammation causes the body to release an excessive amount of inflammatory factors, which can cause uncontrollable inflammation and its highly negative outcomes, including cancer, septic shock, autoimmune illnesses, and even endangerment of life.3,4
The anti-inflammatory properties of Traditional Chinese Medicine formulations are distinguished by their multi-component, multi-pathway, and multi-target attributes.5–7 Professor Shi Huang's experience prescription for diarrheal irritable bowel syndrome (IBS-D) is ACD, which is created by combining and deleting Tongxie formulae. Generally, it is a frequently prescribed medication for diarrheal irritable bowel syndrome. Its actions include balancing the liver and spleen, tonifying the spleen and softening the liver, eliminating moisture, and reducing diarrhea.8–10 It is noteworthy that while long-term clinical experience with ACD demonstrates its precise therapeutic efficacy, its exact mechanism of action remains unclear. Prior studies have demonstrated that prescribing ACD to individuals with diarrhea-predominant irritable bowel syndrome (IBS-D) can significantly reduce their symptoms of abdominal pain. Visceral pain, which can similarly be controlled by ACD, is strongly correlated with the gene expressions of PPKγ, ERK1, mGluR5, and NR2B in the pain-sensitized central dorsal root ganglia and spinal cord dorsal horn. Furthermore, there is a steady relationship between visceral sensitivity, the level of intestinal inflammation, and the expression of these genes.11–13 ACD can reduce the expression of intestinal inflammatory factors and visceral hypersensitivity, to improve clinical symptoms. 14 Meanwhile, network pharmacology, a rapidly developing drug development paradigm, plays a crucial role in investigating the connection between the holistic illness concept of traditional Chinese medicine and the active ingredients in herbs. For example, Tan et al 15 and Guo et al 16 used network pharmacology and molecular docking methods to analyze the mechanism of action of TXACD and TPACD decoction in ISB-D and colorectal cancer, respectively. Thus, to predict the anti-inflammatory components and targets of ACD in the study, network pharmacology and molecular docking were employed. Additionally, preliminary molecular biology experiments were used to confirm the anti-inflammatory efficacy of ACD, laying the groundwork for future investigations into the anti-inflammatory mechanism of ACD.
Results
Screening of Pharmacodynamic Components and Target of ACD
A total of 182 pharmacodynamic components of ACD were retrieved by TCMSP database search, and 161 were acquired after reaggregation, based on OB ≥ 30% and DL ≥ 0.18. The target of pharmacodynamic components is screened in the SwissTargetPrediction with “Homo sapiens” set as the species screening criterion. Compounds lacking an effective target are eliminated, and the screening condition is set at Probability ≥ 0.2. 32 pharmacodynamic components of ACD from PubChem (https://pubchem.ncbi.nlm.nih.gov/) were found after duplications were removed and summaries were produced. Of these, 217 possible related targets of interaction were found, all of which were represented in Table 1.
the Active Ingredients of ACD.

Inflammation Targets Intersection
The inflammation-related targets were searched in the GeneCards and OMIM databases with the inflammation-related genes, and after the repetition was removed and summarized, 539 target genes were screened with a correlation score ≥ 5 as the screening condition. 42 targets for drug-disease co-prediction were found after mapping 217 targets of the pharmacodynamic components of ACD and inflammatory targets acquired in 2.1 using Venny 2.1.0 software (Figure 1). Namely, the anti-inflammatory components and potential targets of ACD: NR1H3, AR, ESR1, ALOX15, EGFR, F2, ALOX5, MPO, GSK3B, SRC, MMP13, MMP3, MMP9, MMP2, CXCR1, AKT1, ABCB1, GPR35, SYK, PIK3CG, ARG1, TTR, APP, MMP12, PTGS2, CFTR, TERT, PLG, PTGES, VDR, NOS2, NR1H2, HIF1A, NR3C1, PTGS1, TACR1, CCR1, MAPK14, LGALS3, F3, IL2 and PPARA.

Venn diagram of common targets of ACD compounds against inflammation.
PPI Network Construction and Screening Results of Key Targets
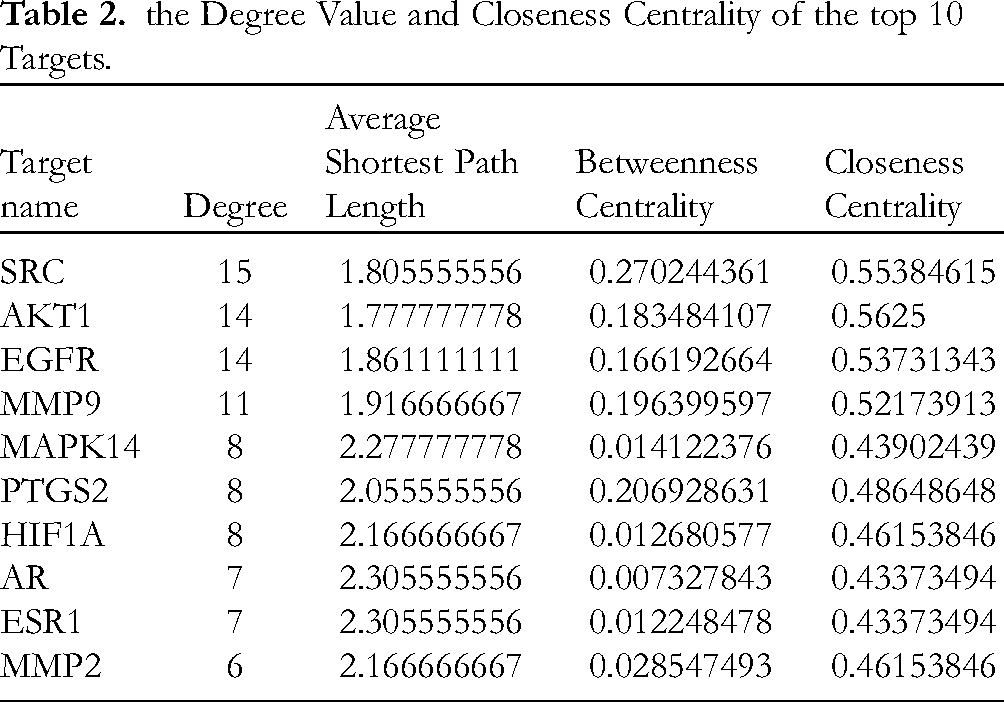
To perform protein interaction, the 42 intersection targets (Table 2) checked by the STRING database were entered. The pharmacodynamic components were chosen and supplied to the software (v3.9.1) for processing based on “high confidence (0.700)”. The protein interaction diagram (Figure 2) with a total of 37 nodes and 90 edges was constructed after deleting solitary targets. The degree and criticality of a target are ascertained by counting the number of line segments that connect to it. The top 10 genes with the highest Degree were selected as key targets: SRC, AKT1, EGFR, MMP9, MAPK14, PTGS2, HIF1A, AR, ESR1, and MMP2. The results are shown in Table 2.
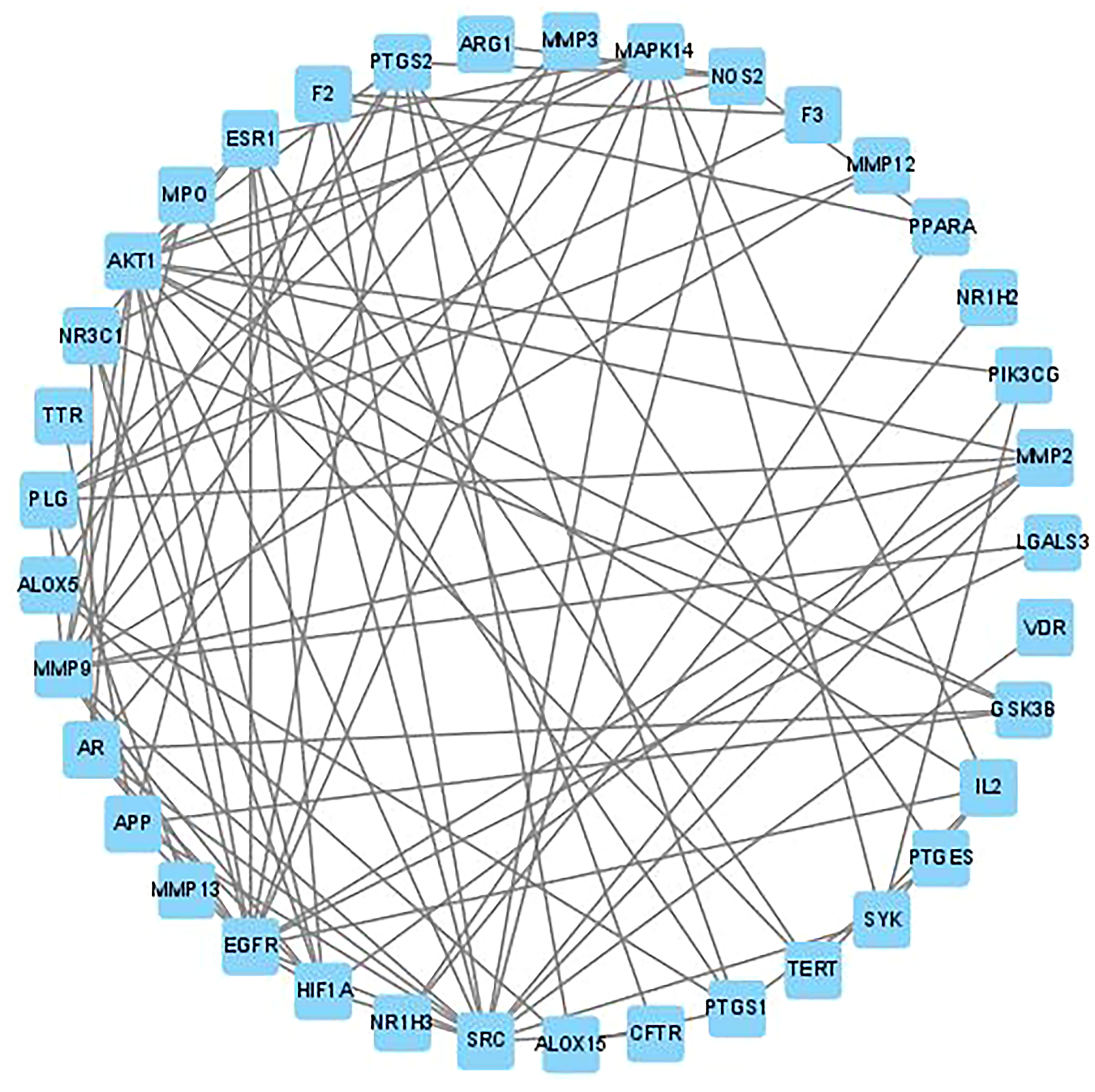
Protein-protein interaction network graph: top 37 core targets with the largest degree values.
the Degree Value and Closeness Centrality of the top 10 Targets.
“Pharmacodynamic Ingredient-Target” Network Analysis
We created a network diagram that displays “drug-pharmacodynamic ingredients-target” by inputting the ID numbers of ACD, pharmacodynamic components, and associated inflammatory target proteins into Cytoscape software (v3.9.1). Figure 3 displays a total of 7 nodes and 19 edges once isolated targets are removed, confirming that ACD modulates many targets via 32 active components to produce its anti-inflammatory effects. Furthermore, Degree value is commonly recognized as a measure of the active components that indicate the effectiveness of the medication. Kaempferol, luteolin, quercetin, isorhamnetin, α-spinasterol, chrysoeriol, stigmasterol, stigmasterol, β-sitosterol, and stigmasterol glucoside are the top 10 major active components with Degree values.

Network diagram of ACD for anti–inflammation “drugingredient–target”. Orange circles, light green quadrilaterals and a yellow square represent target proteins, active ingredients and ACD, respectively.
GO Functional Enrichment and KEGG Signaling Pathway Analysis
Biological enrichment analysis was performed on 42 target genes of ACD associated with inflammation by querying the DAVID database. The screening conditions were set as P < 0.05 and FDR < 0.05. The findings are displayed in Figures 4 to Figure 5. A total of 335 GO functional enrichment results were found, encompassing 64 chemical activities, 246 biological processes, and 25 cell components. Following the selection of the top 10 GO functional enrichment P value items, a bubble map was created on the Weishengxin platform. Similarly, bubble maps of the top 20 channels were created using the Weishengxin platform based on P-value ranking after 69 KEGG channels were eliminated using the DAVID database (Figure 4). 10 core signaling pathways, including the relaxin signaling pathway, the Kaposi sarcoma-associated herpes virus infection, the cancer pathway, endocrine resistance, platelet activation, proteoglycan in cancer, IL-17, prostate cancer, the C-type lectin receptor, and arachidonic acid metabolism, were primarily involved in the predicted therapeutic effects of ACD powder on inflammation. The KEGG signaling pathway association diagram of ACD for the treatment of inflammation was created using Cytoscape 3.9.1 software and the Weishengxin platform (Figure 5). AKT1, MAPK14, SRC, EGFR, GSK3B, MMP9, MMP2, PTGS2, SYK, ESR1 and other targets were the targets of multiple pathways (Table 3).
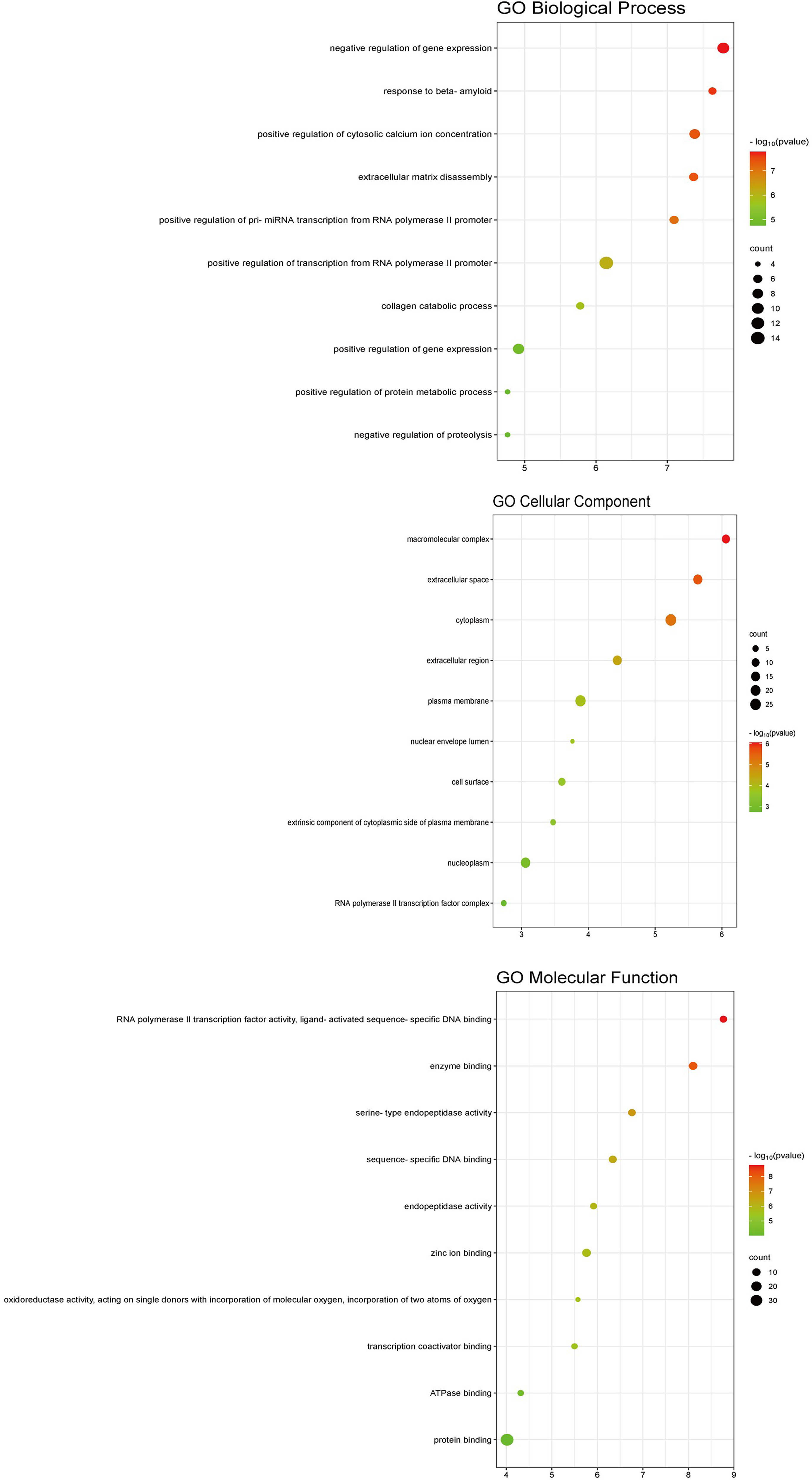
The results of go function enrichment analysis. Bubble diagram of biological process, cellular component and molecular function. The Y axis of the bubble diagram represents the processes, the X axis represents the ratio of genes, the area of the bubble represents the number of genes enriched in this pathway, and the color of the bubble reflects the size of the P-value. The more significant the P-value, the higher the confidence that the gene is enriched in the process.
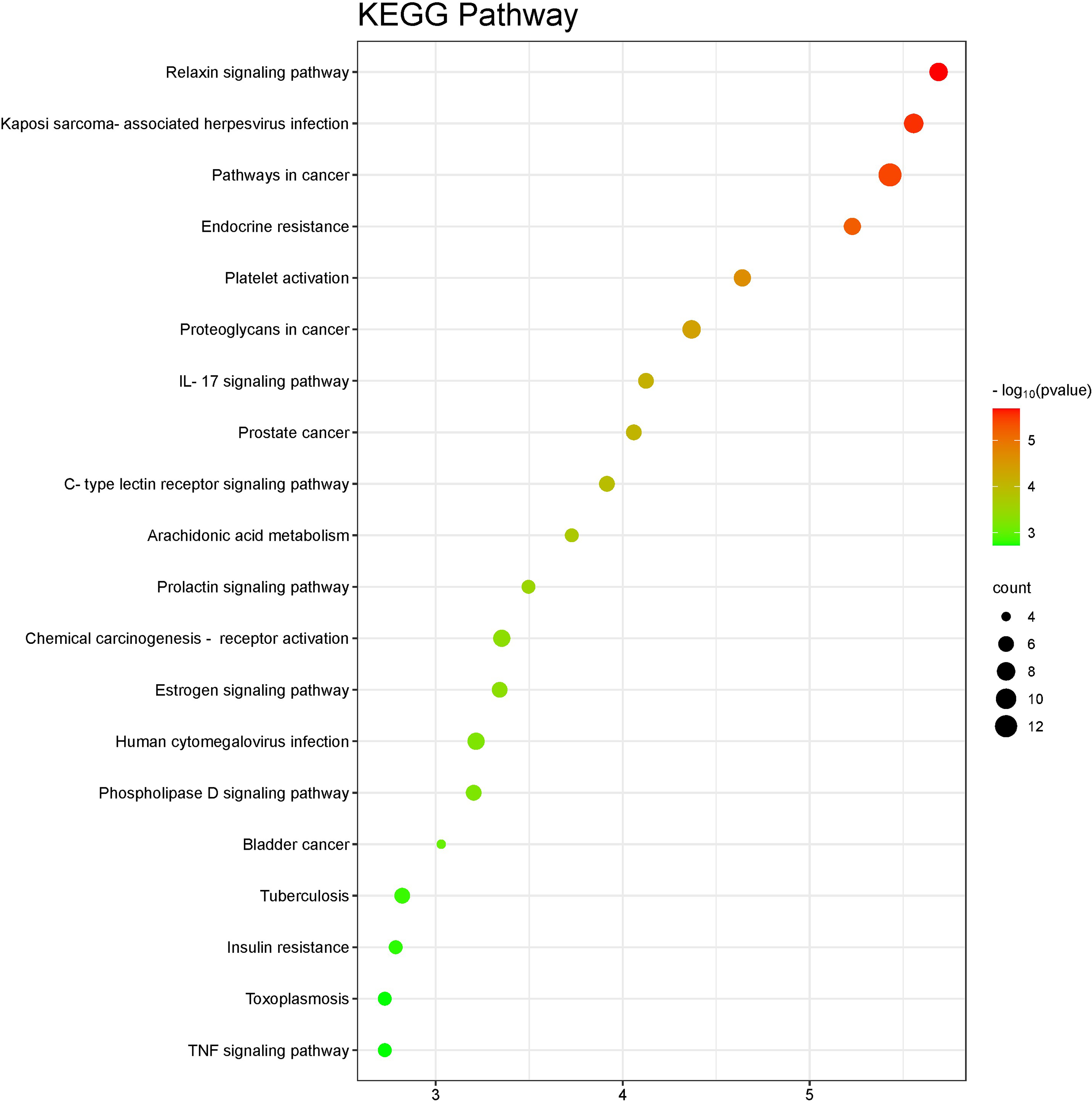
The results of KEGG pathway enrichment analysis. In the picture above, the Y axis of the bubble diagram represents the name of pathways, the X axis represents the ratio of genes, the area of the bubble represents the number of genes enriched in this pathway, and the color of the bubble reflects the size of the P-value. The more significant the P-value, the higher the confidence that the gene is enriched in the signaling pathway. Network diagram of ACD for anti-inflammation"ingredient-target-pathway". Orange rectangles, yellow quadrilaterals and green ovals represent pathways, target proteins and active ingredients in the next diagram, respectively.
The Edge Count of the top 10 targets.

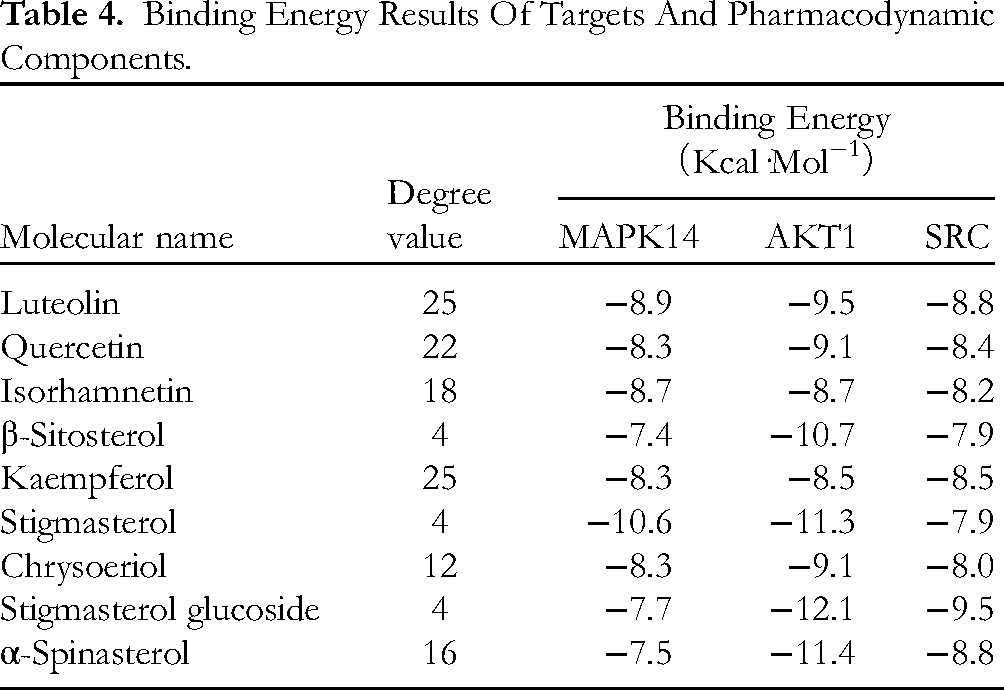
Molecular Docking
Molecular docking was used to estimate the binding ability between the bioactive components of ACD and the predicted core target proteins. The core targets MAPK14 (PDB ID: 6sfo), AKT1 (PDB ID: 6hhg) and SRC (PDB ID: 1fmk) were selected as the protein receptors based on the highest degree value in the PPI network. Accordingly, the major active components include kaempferol, luteolin, quercetin, isorhamnetin, α-spinasterol, chrysoeriol, stigmasterol, β-sitosterol and stigmasterol glucoside. Table 4 lists the binding energy of molecular docking in which the binding energy is lower than −7.0 kcal·mol-1 indicating that the binding condition is better. The better binding groups were luteolin-MAPK14, luteolin-AKT1, luteolin-SRC, quercetin-MAPK14, quercetin-AKT1, quercetin-SRC, isorhamnetin-MAPK14, isorhamnetin-AKT1, isorhamnetin-SRC, kaempferol-MAPK14, kaempferol-AKT1, kaempferol-SRC, stigasterol-MAPK14, stigasterol-AKT1, stigasterol-SRC, chrysoeriol-MAPK14, chrysoeriol-AKT1, chrysoeriol-SRC, stigmasterol glucoside-MAPK14, stigmasterol glucoside-AKT1, stigmasterol glucoside-SRC, α-spinasterol-AKT1, α-spinasterol-SRC and β-sitosterol-MAPK14, and visualized some of the groups with better results (see Figure 6).

The result of molecular docking of MAPK14 (PDB ID:6sfo), AKT1 (PDB ID: 6hhg) and SRC (PDB ID: 1fmk) with main active components of ACD. The ligands are shown as an orange rod-like structure, the receptor proteins are green, the residues that interact with the ligands are marked in purple, and the yellow dashed line indicates hydrogen bonds between the receptor and the ligand. In addition, several diagrams do not show hydrogen bonds between the molecules, but the minimum binding energy is small enough to show a strong binding tendency, they are also listed and shown.
Binding Energy Results Of Targets And Pharmacodynamic Components.
Effect of ACD on the Activity of RAW2647
When ACD drug-containing serum was added to RAW264.7 cells at several dosages (2.5%, 5%, 10%, 20%, and 40%) for 24 h, as compared to the negative control group, RAW264.7 macrophage activity was increased in a dose-dependent manner, as illustrated in Figure 7. However, cell differentiation can be achieved at 40% concentrations of the drug-containing serum of ACD (Figure 8). To conduct the following studies, drug-containing serum at concentrations of 5%, 10%, and 20% were chosen as low, medium, and high dosages to rule out the impact of higher concentrations (40%) on cell state.
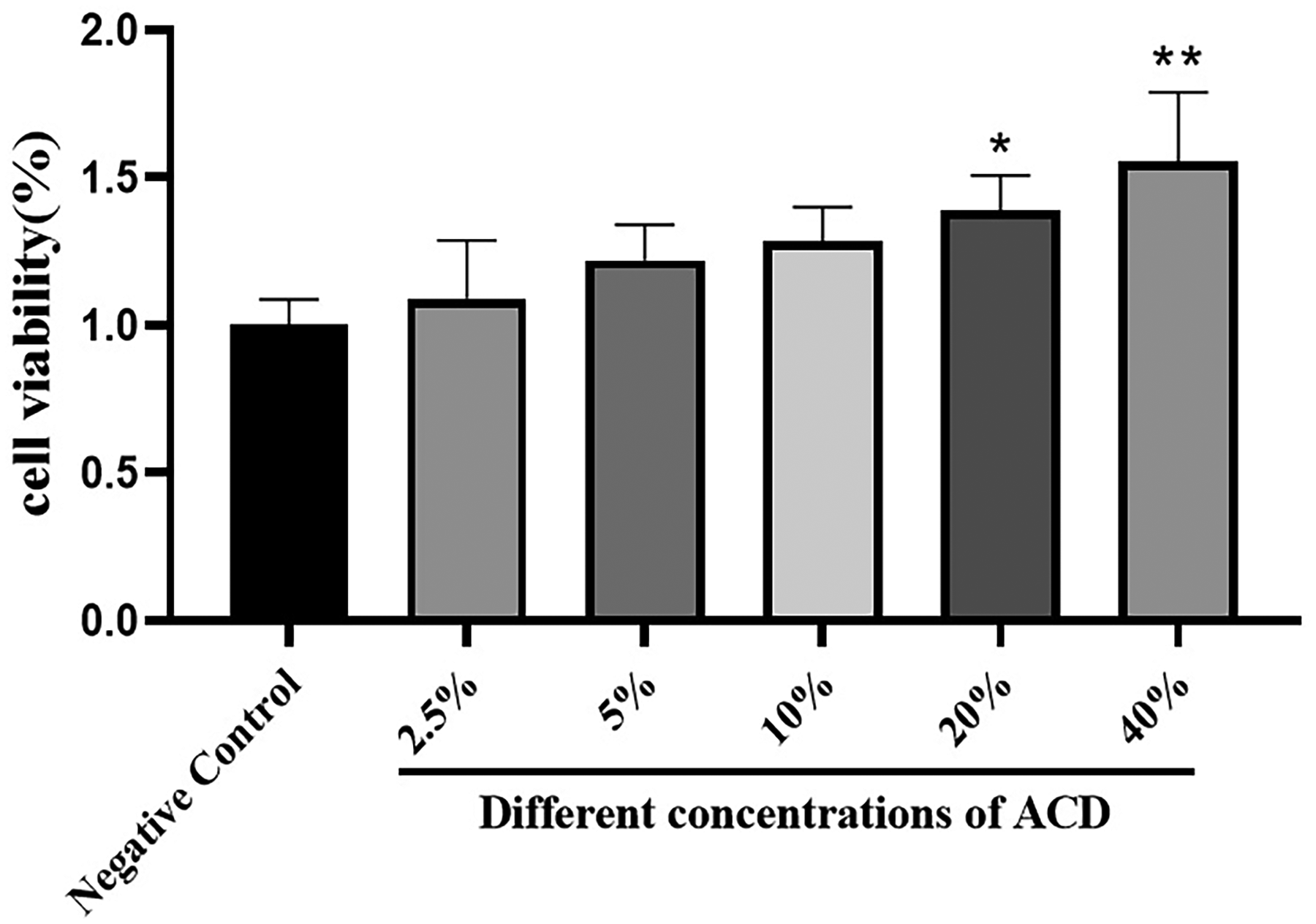
Effect of ACD on macrophage activity of RAW264.7. The results of three separate trials were provided as the mean ± standard deviation (x ± s), n = 3. Compared with the negative control group, *P < 0.05, **P < 0.01.

Cell state comparison (x40).
ACD Attenuated LPS-Induced Pro-Inflammatory Responses in RAW 2647
According to network pharmacology studies, the anti-inflammatory molecular mechanism of ACD may involve lowering certain pro-inflammatory cytokines or signaling molecules. The Griess test was utilized to measure the amount of NO in the cell supernatant of each group, and the enzyme-linked immunosorbent assay was employed to determine the relative levels of TNF-α and IL-6 protein production to corroborate the anti-inflammatory impact of ACD. The model group's relative NO levels were substantially higher (P < 0.001) than those of the normal group (Figure 9). Meanwhile, there was a significant increase (P < 0.001) in the relative levels of TNF-α and IL-6 of the model group (Figure 10A, B). Furthermore, there was a substantial drop in the levels of NO, IL-6, and TNF-α in the drug-containing serum group at different concentrations (5%, 10%, and 20%) as compared to the negative control group. These findings suggested that by lowering the concentration of pro-inflammatory cytokines and signaling molecules, ACD can mitigate inflammation brought on by lipopolysaccharide (LPS).

Effects of different doses of ACD on no contents in the supernatant. The results of three separate trials were provided as the mean ± standard deviation (x ± s), n = 3. Compared with the Control group, *P < 0.05, **P < 0.01, ***P < 0.005, ****P < 0.001.

Effects of different doses of ACD on TNF-α (A) and IL-6 (B) contents in the supernatant. The results of three separate trials were provided as the mean ± standard deviation (x ± s), n = 3. Compared with the model group, **P < 0.01, ***P < 0.005, ****P < 0.001.
ACD Decreased the Relative Expression Levels of AKT1, MAPK14 and SRC Target Proteins in RAW 264.7 Induced by LPS
The relative expression levels of AKT1, MAPK14 and SRC target proteins in RAW 264.7 stimulated by LPS were further detected by Western blot analysis, which confirmed the interaction of pharmacodynamic components on important target proteins identified by network pharmacological screening. Macrophages of the model group exhibited significantly higher relative levels of AKT1, MAPK14 and SRC proteins as compared to the control group (P < 0.01, P < 0.001, P < 0.005); Compared with the negative control group, the relative expression levels of AKT1, MAPK14 and SRC target proteins in macrophages in the ACD drug-containing serum group were all dramatically down-regulated (P < 0.05, P < 0.01, P < 0.05) (Figure 11).

Immunoblotting(A) of major targets and signaling pathways in RAW264.7 in each group. ACD inhibits the relative expressions of AKT1(B), MAPK14(C) and SRC(D) proteins. The results of three separate trials were provided as the mean ± standard deviation (x ± s), n = 3. Compared with the control group, *P < 0.05, **P < 0.01, ***P < 0.005, ****P < 0.001; Compared with the negative control group, #P < 0.05, ##P < 0.01.
the Histopathological Changes of Colon in Each Group After ACD Treatment
Under a microscope, the colon tissue and mucosa of the rats in the control group exhibited no necrosis or infiltration of inflammatory cells. The connective tissue of the submucosa was loose, and there was a tiny degree of inflammatory cell infiltration in the lamina propria in the model group. In the positive group, inflammatory cell infiltration was occasionally observed in the lamina propria, and the submucosa connective tissue was loose. In the ACD group, the mucosa and colonic tissue were unharmed, and there was no sign of inflammatory cell infiltration (Figure 12).

He staining of colon tissues in each group.
Discussion
The safety of traditional Chinese medicine has received increasing attention as it has become more widely used. The toxic and side effects of traditional Chinese medicine primarily refer to the damage caused by traditional Chinese medicine to cells, tissues, and organs of the body to varying degrees after it enters the human body, resulting in a greater negative impact on the physiological function of the body. In serious cases, it may interfere with or even destroy the physiological system of the body, and pose a direct threat to the life safety of the human body In this regard, to ensure the safety of drug use to the maximum extent, it is necessary to strictly examine the types of picking and regions of traditional Chinese medicine, and at the same time, select correct processing technology to ensure the rationality of drug compatibility, strengthen dose and usage control, and carry out basic education in traditional Chinese medicine. Only in this manner can the occurrence of harmful and adverse effects of traditional Chinese medicine be significantly minimized. 17 ACD is a frequent prescription for the clinical treatment of IBS-D, and its medication composition is based on traditional Chinese medicine. The compatibility and formulation meet national drug standards, ensuring safety and efficacy.
The pharmacological action of ACD is uncertain due to its complex nature. Thus, network pharmacology and molecular docking technology, by the holistic idea of traditional Chinese medicine, were used to examine the mechanism of ACD composition on cell inflammation, and the regulatory network of the anti-inflammatory action of ACD was established. This study used network pharmacology and molecular docking technology to investigate the anti-inflammatory mechanism of compound Chinese medicine ACD in vitro. 161 active components of ACD were identified using oral bioavailability and drug-like screening criteria. The “medicine-component-target” network analysis was used to screen 32 active ingredients and 42 inflammatory treatment targets. The network analysis of “key active ingredients-core target-pathway” revealed that the primary active elements of ACD for treating inflammation were kaempferol, luteolin, quercetin, isorhamnetin, α-spinasterol, chrysoeriol, stigmasterol, β-sitosterol, stigmasterol glucoside, etc Studies show that kaempferol and β-sitosterol have anti-inflammatory, antioxidant, and anti-tumor properties.18,19 The role of Luteolin in regulating inflammation can be realized by regulating the expression level of non-coding RNA, such as inhibiting the expression of miR-142-3p, thereby reducing the apoptosis level of LPS-induced alveolar macrophages, improving the inflammatory environment of the body, and alleviating the inflammatory state. 20 Luteolin and quercetin decrease COX2 expression via the NF-κB signaling pathway, thereby reducing inflammation.21,22 Isorhamnetin is a quercetin metabolite with antioxidant and anti-inflammatory effects. Studies have demonstrated that isorhamnetin pretreatment can dramatically decrease inflammation and oxidative stress generated by lipopolysaccharides, hence reducing ALI.23,24 It can inhibit the inflammatory response of RAW264.7 macrophages caused by lipopolysaccharide, reduce nitric oxide (NO) and prostaglandin E2 production by the cells, and diminish the gene expression levels of IL-6, IL-1β, and TNF-α. 25 Plant sterols β-sitosterol and stigmasterol have anti-inflammatory, antioxidant, and immune regulation properties. They inhibit the expression of interleukin-2 and interleukin-10, as well as the proliferation of helper and toxic T cells, to reduce inflammation. 26 β-sitosterol has therapeutic effects on chronic inflammatory illnesses like colitis and asthma. Its mechanism involves targeting anti-pathogenic microorganisms or inhibiting ovalbumin via modulating dendritic cells.27,28 A PPI network diagram was used to identify the primary targets of ACD in the treatment of inflammation: SRC, AKT1, EGFR, MMP9, MAPK14, PTGS2, HIF1A, AR, ESR1, MMP2, and so on. PPI, GO functional enrichment, and KEGG signaling pathway analysis all interact, with mitogen-activated protein kinase 14 being the top three important targets. TCM treatment targets multiple targets, including MAPK14, protein kinase B (AKT1), and non-receptor tyrosine kinase (SRC). AKT1, a member of the Ser/Thr protein kinase family, is essential for signal transduction pathways and basic physiological processes such as cell proliferation, survival, metabolism, and angiogenesis. In addition, AKT1 also participates in the migration and invasion of cancer cells and plays an important role in cancer progression and metastasis.29,30 AKT1 is one of the subtypes of AKT, and it has been found that the cytokine chemokine ligand 3 (CCL3) can activate AKT and mediate the inflammation of prostate epithelial cells. 31 In addition, AKT1 can gradually aggravate inflammation in vivo by promoting a large number of inflammatory cytokines and immune-active factors. 32 p38 mitogen-activated protein kinase is an important member of the MAPK family. 33 p38 MAPK, also known as MAPK14, is allosteric through dual-phosphorylation activation of MAPKK3 and MAPKK6, 34 thereby altering the kinase domain (N and C-terminal arrangement) and improving enzyme activity. 35 p38 MAPK can promote the aggregation and activation of white blood cells, regulate the activity of transcription factors and the synthesis of cytokines, and thus mediate the occurrence and development of inflammatory response. 36 Studies have confirmed that the activation of p38 MAPK signaling pathway can produce a variety of inflammatory factors, such as IL-1β, IL-6, TNF-α, etc, and their expression levels are positively correlated with the severity of inflammatory response. 37 Xin et al 38 constructed myocardial damage models of sepsis in vivo and in vitro and found that inhibiting the activation of NF-κB and p38 MAPK signaling pathways could protect against myocardial damage in sepsis. Non-receptor protein tyrosine kinase (SRC) is involved in signaling pathways that control a variety of biological activities, such as cell proliferation, migration, extracellular matrix adhesion, and tumorigenesis. 39 It can recruit MyD88 and activate the MAPK and NF-κB signaling pathways, leading to the secretion of inflammatory cytokines and mediators like TNF-α, IL-1β, IL-6, COX-2, and NO, causing inflammation. 40 Studies have shown 41 that the expression of SRC protein is up-regulated in patients with perianal abscesses. The SRC protein family is a family of proteins with tyrosine-protein kinase activity, which is widely present in vascular endothelial cells as an important molecule connecting signal pathways inside and outside cells. SRC protein kinase can participate in the infection of pathogenic microorganisms through various mechanisms. The above targets are closely related to the occurrence and development of inflammation, so it can be seen that ACD can perform a therapeutic role in inflammation through multiple targets. Subsequently, the enrichment of the KEGG signaling pathway further shows that ACD-induced inflammation is treated via Relaxin signaling pathway, Kaposi sarcoma-associated herpesvirus infection, Pathways in Cancer, Endocrine resistance, Platelet activation, Proteoglycans in cancer, the IL-17 signaling pathway, Prostate cancer, C-type lectin receptor signaling pathway and so forth.
The molecular docking results showed that the average binding energy of the 10 main active components of ACD and the 3 core targets was −8.97kJ/mol. The highest binding capacity was AKT1 and Stigmasterol glucoside, with binding energy −12.1 kJ/mol. The lowest binding capacity was MAPK14 and beta-sitosterol, with a binding energy of −7.4kJ/mol. Meanwhile, the molecular docking results indicated that most of the active components formed hydrogen bonds with core targets, such as the formation of hydrogen bonds between Luteolin and the 156th arginine residue of SRC protein, and between the 362nd glutamine residue and the 397th asparagine residue; the formation of hydrogen bonds between Quercetin and the 79th glutamine residue and the 271st valine residue of AKT1 protein; the formation of hydrogen bonds between Chrysoeriol and the 199th histidine residue of MAPK14 protein; which helps to change the conformation of the protein and stabilize the binding state of the active ingredient and the core target protein to exert the pharmacodynamic function. These results suggest that ACD exerts its multiple pharmacological effects in a complex and stable manner, which may be the key anti-inflammatory link of ACD.
This study found that kaempferol, luteolin, quercetin, isorhamnetin, α-spinasterol, chrysoeriol, stigmasterol, β-sitosterol and stigmasterol glucoside, which are the main active ingredients of ACD, may further regulate the relaxation signaling pathway of herpes Kapo sarcoma virus by binding to core proteins such as AKT1, MAPK14 and SRC. Endocrine resistance platelets activate the proteoglycan IL-17 signaling pathway in cancer, such as the metabolism of C-type lectin arachidonic acid in prostate cancer, thereby regulating transcription factor activity and cytokine synthesis and inhibiting the apoptosis of inflammatory cells. HE staining revealed that ACD improved colon tissue inflammation and reduced intestinal inflammation in IBS-D rats. The results of an in vitro investigation revealed that the expression level of AKT1, MAPK14 and SRC in ACD-containing serum could be greatly lowered, indicating that ACD had a greater anti-inflammatory impact in vitro. In addition, it should be pointed out that this study still has some limitations: 1. Although network pharmacology is based on database and analysis software, due to the current technological development, the database is not perfect, the reliability of the provided data and analysis results needs to be further verified, and the criteria for screening ingredients needs to be optimized; 2. Although network pharmacology can explain the compatibility of TCM compounds, most studies are limited to the qualitative analysis of the “ingredient-target-disease” network, and ignore the influence of each component; 3. Some active ingredients screened by network pharmacology in this study are highly similar, such as quercetin and kaempferol, etc The explanation of this phenomenon still needs to be further explored. 4. In this study, cell experiments were used to verify the prediction results of network pharmacology, but in vivo, efficacy verification in animal experiments was lacking, so further verification of the accuracy of the research results may be needed.
This study used molecular docking technology to study the active ingredients in ACD, revealing a correlation between traditional Chinese medicine compounds and inflammation. Results from HE staining and cell experiments supported network pharmacology predictions and provided a reference for future research. As a consequence, the anti-inflammatory activity of ACD is the result of a direct or indirect synergistic effect of multiple compounds, targets, and pathways, and cell studies confirmed the trustworthiness of the systematic pharmacological analysis results in this work. Future research will look at the micro-synergies of numerous substances in ACD, as well as the relationship between their effects and metabolic systems.
Conclusion
In conclusion, this study investigated the anti-inflammatory mechanism of ACD using network pharmacology and molecular docking verification, validated the key anti-inflammatory targets of ACD-containing blood using molecular biology experiments, and first discussed the anti-inflammatory mechanism of ACD. However, one limitation of this study is the lack of animal experiments on the anti-inflammatory effects of inflammation ACD. Subsequent research will look at the impact of inflammatory ACD in vivo. In a word, this work provides a preliminary reference for the mechanism of action of traditional Chinese medicine in the prevention of inflammatory disorders, as well as a novel notion for clinical use of the anti-inflammatory efficacy of ACD.
Methods
Identification of Pharmacodynamic Components and Target Proteins of ACD
The primary source for the pharmacodynamic elements of ACD was the PubChem website (https://pubchem.ncbi.nlm.nih.gov/). Chaihu (Radix Bupleuri), Baishao (Radix Paeoniae Alba), Baizhu (Atractylodes Macrocephala Koidz), Chenpi (Citrus Reticulata), Fangfeng (Saposhnikoviae Radix), Chuanxiong (Ligusticum Wallichi), Xiangfu (Cyperi Rhizoma), Zhiqiao (Fructus Aurantii), Gancao (Licorice) pharmacodynamic components were sourced from the TCMSP database (https://old.tcmsp-e.com/tcmsp.php) and subsequently filtered using the criteria of “oral bioavailability ≥ 0.30, drug similarity ≥ 0.18”. To predict the target protein, the Smiles numbers or 2D structures for each of the tested pharmacodynamic components were also collected from the PubChem website. These data were then loaded into the SwissTargetPrediction website (http://www.swisstargetprediction.ch/). Duplicate components and targets of non-Homo sapiens were deleted, and the most likely targets in the pharmacodynamic components were screened with Probability ≥ 0.2.
Component-Disease Common Target Acquisition
Using “inflammation” as the search keyword, the OMIM (https://omim.org/) and GeneCards (http://www.genecards.org/) databases were searched for disease-related targets, and once duplicate items were eliminated, inflammation-related proteins were found. The potential component target of ACD was intersected with the target of inflammation using the online tool Venny 2.1.0 (https://bioinfogp.cnb.csic.es/tools/venny/index.html) to create a Venny map of disease targets in both databases. The potential target genes of diseases were the common targets of the map.
Network Construction for Protein-Protein Interactions (PPI)
The STRING database (https://cn.string-db.org/) was used to import the predicted probable target proteins of common diseases. To achieve PPI network analysis, the “minimum required interaction score” was set to a “high confidence (0.700)”. The data results in TSV format were uploaded to Cytoscape (v3.9.1) software to generate the network diagram, and key genes in the network were screened according to the Degree values for further investigation.
“Component - Target - Disease” Interaction Network
To create the “component-target-disease” network diagram, the pharmacodynamic components relating to the important target proteins and ACD were acquired and imported into Cytoscape (v3.9.1) software.
GO Functional Enrichment and KEGG Signaling Pathway Analysis
Using the DAVID database (https://david.ncifcrf.gov/), intersection targets were subjected to GO functional enrichment and KEGG pathway enrichment analysis. The findings were evaluated with P < 0.05 and FDR <0.05 as crucial values. The acquired data were then examined using the Weishengxin web application (https://www.bioinformatics.com.cn) to create maps of the KEGG signal pathway and GO functional enrichment. Lastly, Cytoscape (V3.9.1) was used to create the “target-pathway” network map.
Molecular Docking
To confirm the validity of the “component-target-disease” interaction network, molecular docking was carried out between the top three disease genes MAPK14 (PDB ID:6sfo), AKT1 (PDB ID: 6hhg) and SRC (PDB ID: 1fmk) and the top nine pharmacodynamic components (Luteolin, Quercetin, Isorhamnetin, β-Sitosterol, Kaempferol, Stigmasterol, Chrysoeriol, Stigmasterol glucoside, α-Spinasterol). In addition, the PDB database (https://www.rcsb.org/) and the UniProt database (https://www.uniprot.org/) provided the pdb format structure of predicted proteins. The chemical 3D structural formula of pharmacodynamic components was collected from the TCMSP and PubChem databases. The binding energy was found and the molecular docking between pharmacodynamic compounds and anticipated proteins was predicted using Autodock Vina (v 1.1.2) software. The information on binding sites (hydrogen bond, spatial position, etc) for visualization was then handled by PyMOL2.2.0 program. It is generally accepted that an active component has good binding activity with the protein if its binding energy is less than −5.
Preparation of Drug-Containing Serum
ACD granules, composed of Radix Bupleuri 24 g, Radix Paeoniae Alba 12 g, Pericarpium Citri Reticulatae 10 g, Rhizoma Atractylodis Macrocephalae 10 g, Saposhnikoviae Radix 10 g, Ligusticum Wallichi 10 g, Cyperi Rhizoma 10 g, Fructus Aurantii 10 g, Prepared Licorice 8 g (Jiangyin Tianjiang Pharmaceutical Co., LD, batch number 2210194301 2210029301 2208103301 2201147301 21100823 2209124301 2212184301 2204038301 2208343301). Granule preparation method: Boil the above 9 drugs twice, add 8 times the amount each time, cook for 1 h each time, and filter; The two filtered liquids were combined and concentrated to 1:1 under atmospheric pressure (each mL was equivalent to 1 g of the original medicinal material) with a specific gravity of about 1.08; The concentrated liquid was doubled with 95% ethanol, stirred while adding, stood for 24 h, filtered, and then concentrated to 1:5 (each mL equivalent to 5 g of the original medicinal material) after ethanol recovery in the filtrate, with a specific gravity of about 1.3; The extract was mixed with 2.5 times the amount of dextrin, made of soft material with 95% ethanol as wetting agent, screened into wet particles through 12-mesh galvanized iron wire, and finally dried at about 60. 24 male SD rats with SPF grade, body weight 180–200 g, animal production license No. SCXK (Hunan) 2019-0004, were purchased from Hunan Silaike Jingda Laboratory Animal Co., LD, and were raised in the standardized animal room of the Laboratory Animal Center of Guangxi University of Chinese Medicine with the room temperature controlled at 23 °C∼25 °C. Ordinary feed (batch No. 201810030) feeding, no restriction on drinking water. This experiment was approved by the Experimental Animal Ethics Committee of Guangxi University of Chinese Medicine, and the animal experiment ethics number is DW20230620-119. The daily dosage for the rat was determined to be 10.83 g/kg of the original medicinal material, and 1.083 g/mL of the original medicinal material was prepared with conventional pure water and stored in the refrigerator at 4 °C for later use, by the conversion ratio of human and animal body surface area42. The rats were randomly divided into two groups: the drug-containing serum group and the negative control group. The negative control group received the same volume of intragastric administration of normal saline once a day for ten days, while the drug-containing serum group received ACD by intragastric administration. At 1 h after the last intragastric administration, rats were anesthetized by intraperitoneal injection of 1% pentobarbital sodium (40 mg/kg) and blood was taken from the abdominal aorta. After plasma resting at 4°C for 2 h, then, serum was isolated by centrifugation at 3500 r/min for 10 min. For subsequent experiments, the complement in the serum was inactivated in a water bath at 56°C for 30 min, the 0.22 μm microporous filter membrane was used for bacteria removal and filtration, and the serum was stored in a low-temperature refrigerator at −20°C.
Grouping and Modeling
Mouse macrophage RAW 264.7 cells (number: 05MWLEBITO) were purchased from Wuhan Punosai Life Technology Co., LTD and cultured in DMEM high sugar medium with 10% FBS, 100U/ml penicillin, and 100μl/ml streptomycin, which were at 37°C in humidified incubator containing 5% dioxide carbon. Experimental procedures were conducted when the cells reached 80–90% confluence in the logarithmic phase of growth. To screen the appropriate modeling concentration, the CCK-8 assay was performed to measure cell viability in different concentrations of ACD (2.5%, 5%, 10%, 20% and 40%). The number of cells (1 × 104 cells/well) were inoculated into a 96-well plate, and the group intervention was conducted according to the grouping method as mentioned above, and a blank group without cells was set up, with 5 multiple wells in each group. After 24 h of culturing, CCK-8 reagent was added at 10% medium volume in each well and incubated at 37°C for 1.5 h. After that, a microplate reader was used to determine optical density (OD) at 450 nm. The cells were split into six groups for the ensuing experiments: 10% FBS was given to the control group, 10% rat serum + LPS was given to the model group, 10% rat serum was given to the negative control group, 10% rat serum was given to the ACD low-dose group, 10% rat serum was given to the ACD medium-dose group, 10% rat serum was given to the ACD high-dose group, and 10% rat serum was given to the ACD low-dose group. Using the random number table method, forty clean male SD rats were split into four groups: ACD, Western medicine, normal, and model. Each group consisted of ten rats. In addition to the normal group, the model group, positive group and ACD group were treated with capsaicin gavage combined with binding and tail clip stress to replicate IBS-D model for 2 consecutive weeks. After the successful evaluation of IBS-D model, the western medicine group was given 15 mg/kg Piverium bromide tablet by intragastric administration, while the ACD group was given ACD by intragastric administration of 6.2 g/kg once a day for 2 weeks.
Detection of Nitric Oxide (NO) and Inflammatory Cytokines
The cells in the logarithmic stage were inoculated with 2 × 105 cells/well on 24-well plates. After cell culture for 24 h, the cells were pretreated with 10% FBS (normal group) and different concentrations (5%, 10% and 20%) of ACD drug-containing serum (experimental group) for 1 h. The cells in the model group were not treated, and the final concentration of LPS was added to each well with 4 μg/mL. In the control group, only the same volume of complete medium was added, and the supernatant of each pore cell was collected after culture for 24 h. After centrifugation at 12,000 r/min for 15 min, the supernatant was discarded and stored at −20°C. The Griess method was used to detect NO secretion levels in cell supernatants of each group, and enzyme-linked immunosorbent assay was used to detect TNF-α and IL-6 protein secretion levels in cell supernatants of each group.
Western Blot Analysis
The cells in the logarithmic stage were inoculated with 5 × 105 cells/well on 6-well plates. The cells were pretreated with 10% rat serum (negative control group) and 10% ACD drug-containing serum (experimental group) for 1 h, the model group was not treated, and the final concentration of 4 μg/mL LPS was added to each well, while the control group was only added with the same amount of complete medium, cultured at 37°C and 5%CO2 for 24 h, and the total protein was collected. Then, the protein concentration of each group was determined by the BCA protein quantitative method. After SDS-polyacrylamide gel electrophoresis and transfer, the membrane was sealed with a rapid sealing solution for 15 min. According to the specification, AKT1 (1:5000 dilution), MAPK14 (1:1000 dilution) and SRC (1:500 dilution) were added to the primary antibody, incubated overnight at 4°C, washed 3 times with TBST for 5 min each time, added to the secondary antibody, incubated at room temperature without light for 1 h, rinsed with TBST 3 times for 5 min each time, and developed. Eventually, the gray values of each strip were scanned by the gel imaging system and processed by Image J software to calculate the relative expression of the target proteins.
HE-Stained
After being fixed in a 4% paraformaldehyde solution, the colonic mucosa tissues were translucent, embedded in paraffin wax, and dehydrated with alcohol before being cut. Following dewaxing, the sections were sealed, dehydrated, and dyed using the HE process before being examined and captured on camera under a microscope.
Statistical Analysis
The statistical program GraphPadPrism 8 was used to process the data, which was all displayed as mean ± standard deviation (x ± s). The LSD-t test was used for pairwise comparison, the Dunnett test was used for multiple comparisons, and one-way analysis of variance was used for multi-group comparison. P < 0.05 was deemed statistically significant at that point.
Footnotes
Author Contribution
The experiments were designed and conceived by WT, SH and WZ, performed by QQ and WF, and analyzed by LZ and XY. In addition to LZ and XY, all authors contributed reagents and materials. and analysis tools to this study. WT and WZ equally contributed to the article. All authors have read and approved the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was performed in line with the principles of the Declaration of Helsinki. The protocol was approved by the Committee on the Ethics Committee of Guangxi University of Chinese Medicine (Ethics Number: DW20230620-119).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82060845) and the Innovation Project of Guangxi Graduate Education of GXUCM (YCSY2023039).
Innovation Project of Guangxi Graduate Education of GXUCM, National Natural Science Foundation of China (grant number YCSY2023039, 82060845).
Statement of Animal Rights
All experimental procedures were approved by the Ethics Committee of Guangxi University of Traditional Chinese Medicine (Ethics Number: DW20230620-119).
