Abstract
Objective
Immune thrombocytopenia (ITP) is an autoimmune hemorrhagic disease. The immune imbalance of T cells is an essential pathological mechanism of ITP, more specific, the balance of Effector T cells (Teff)/Regulatory T cell (Treg) maintains T cell immune homeostasis. Dendritic cells (DC), the largest antigen-presenting cell, interfere with T cell differentiation pathways and exert immune regulatory functions. Our team created and applied the “ NingXueShengBan” (NXSB) decoction. According to our prior studies, we found that NXSB decoction could restore Th17/Treg balance in ITP mice by regulating protein expression in Notch pathway. Therefore, we further research the mechanism of NXSB decoction that regulates ITP immune homeostasis through affecting the interaction between DC and T cells.
Methods
In this study, we collected peripheral blood from health donors and ITP patients and extracted DC and CD4 + T cells then grouped in control group, model group and NXSB group. Moreover, we established a co-culture model simulating the immune environment in ITP patient to further investigate whether NXSB had therapeutic effects by the interaction between DC and CD4 + T cells.
Results
In the current study, we found that NXSB decoction potently regulated interaction between DC and T cells by inhibiting DC development and function, specifically, the differentiation of CD4+ T cells was inhibited after NXSB decoction intervened. Our data showed that NXSB decoction inhibited Toll-like receptor 4 (TLR-4), reduced the expression of Nuclear factor kappa-B (NF-κB) and led to the reduction of downstream inflammatory factors, thereby inhibiting the inflammatory response. More importantly, NXSB decoction reduced the DC surface expression of activation and maturation markers. Moreover, we detected that the decreased expression of the phosphorylated Phosphatidylinositol 3-kinase (PI3 K), Protein kinase B (Akt), Mammalian target of rapamycin (mTOR), and their downstream effectors indicated, we found that NXSB decoction could inhibit PI3 K/Akt/mTOR signaling pathways significantly.
Conclusions
Collectively, our data suggested that NXSB decoction ameliorates ITP-induced immune imbalance via its anti-inflammation and immunosuppressive by regulating TLR-4/ NF-κB and PI3 K/Akt/mTOR signaling pathways.
Introduction
Immune thrombocytopenia (ITP) is currently defined as an acquired autoimmune disorder, the incidence of ITP in the general population is estimated to be between 2 and 5 cases per 100,000 people (2-5, per 100,000). 1 ITP is characterized by decreased platelet count in peripheral blood (<100 × 109/L) and impaired maturation of bone marrow and megakaryocytes. Clinically, it can be observed mucous membrane bleeding, skin bleeding, and severe cases can cause gastrointestinal or even intracranial hemorrhage or even life-threatening. 2
The conventional view is that ITP is mainly caused by humoral immunity dysfunction, in recent years, many studies focused on cellular immunity dysregulation, in which, the dysregulation of CD4+ T cell homeostasis played an important role in the pathogenesis of ITP, and related studies showed that disturbed T cell subsets are involved in ITP.3–5 In addition, DC, the most powerful and specialized Antigen presenting cell (APC), involved in T cell function mainly by transforming from immature to mature states, which receiving external antigenic stimulation, providing three types of stimulatory signals to activate the initial T cells, and leading to the differentiation of T cells into different subsets. 6 CD4+ T cells activated by DC can proliferate and differentiate into Treg and Teff. 7 In ITP patients, platelet autoreactive CD4+ effector T cells are excessively activated to decrease apoptosis, whereas CD4+ CD25+ Foxp3+ Tregs are numerically and functionally impaired. 8 Cumulative research has consistently demonstrated the dysregulation in Th1/Th2 and Th17/Treg among ITP patients.9,10 Furthermore, DC subgroups imbalance has been found to lead to the increased differentiation of CD4+ T cells into Th1 and Th17 cells. 10 The reason for this may be attributed to activated DCs played a crucial role in the body's immune regulation by secreting various cytokines. These include interleukin,7–12 IL-23, IL-6, IL-2, Tumor Necrosis Factor (TNF)-α, and IL-18. 11 TLR4 serves as pattern recognition receptor (PRR) in DC and a receptor of Lipopolysaccharide (LPS), and LPS can combine with it to activate a TLR4/NF-κB signaling pathway. 12 It is most closely related to the immunological maturation of DCs. Numerous studies demonstrated that TLR4 is abnormal in ITP patient, even the presence of TLR4 variants is associated with ITP pathophysiology.13–15 mTOR has been identified as a family member of phosphatidylinositol kinase-like kinases and is known to regulate NF-κB activation in a Akt-dependent manner. 16 Previous studies showed that PI3 K/Akt/mTOR signaling pathway participates in the T cell differentiation process by affecting downstream pathways, and its high expression may be closely related to Treg formation and Teff function inhibition, thereby affecting the immune function of T cells. 17
NXSB decoction, is derived from the XiJiaoDiHuang decoction, a classical traditional Chinese medicine formula, is effective in treating bleeding disorders clinical therapy and suggested new therapeutic potentials for NXSB decoction. In our previous study, we demonstrated that NXSB decoction can increase and maintain the platelet level by restoring T-cell imbalance and modulating pro-inflammatory cytokines in ITP mice. 18 However, the previous study only established a correlation between T cell subsets and the therapeutic effect of NXSB decoction in treating ITP mice; it did not prove evidence of the involvement of DC-T correlation in NXSB decoction's efficacy against ITP. To further clarify the role of DC and T cells in treating ITP by NXSB decoction, we conducted experiments in vitro by collecting peripheral blood from ITP patients and normal donors. This allowed us to examine the therapeutic effects of NXSB decoction-altered cellular mechanism.
Methods
Animals
Male SPF grade SD rats (age: 8 weeks, weight: 200 ± 20 g) were purchased from BEIJING HFK BIOSCIENCE CO. and fed in the Experimental Animals Center of Tianjin Medical University. The experiment was carried out after adaptive feeding for 7 days and conducted in accordance with the Guide for the Care and Use of Laboratory Animals (revised in 1996) of the National Institutes of Health of China and approved by the Animal Management an Ethics Committee of Tianjin Medical University.
Patient Material
Healthy volunteers (n = 20) and ITP patients (n = 40) were recruited from the outpatient hematology clinic of the Tianjin Medical University General Hospital (Tianjin, China) with approval by the Ethics Committee of Tianjin Medical University General Hospital. (IRB2020-KY-189) and informed consent in accordance with the Declaration of Helsinki. The diagnosis was according to the diagnostic criteria of ITP. In additional, ITP peripheral blood samples were collected from 40 ITP patients hospitalized in the Department of Hematology, Tianjin Medical University General Hospital from September 2021 to January 2022, including 19 males and 21 females, aged between 18 and 50 years (median age 25 years old). All patients had a platelet count of less than 30 × 109/L. 20 healthy volunteers, including 12 women and 8 men, aged between 20 to 50 years old (median age 20 years old), peripheral blood platelet count is 125–245 × 109/L (median 185 × 109/L). With informed consent, 10 ml fasting venous blood was obtained from every subject.
Cells Sorting and Culture
In the study, the CD4 + T cells and CD14 + T cells from Peripheral Blood Mononucleo Cyte (PBMC) were selected as research object. They were isolated by CD4 or CD14 immunomagnetic beads according with the manipulation instruction, respectively. CD4 + T cells and CD4 + T cells were cultured in medium which including RPMI-1640 with 10% fetal calf serum, 2 mM L-glutamine, penicillin and streptomycin (100 U/ml). On the one hand, CD4 + T cells were activated in the presence of anti-CD3 mAb (10 μg/ml), anti-CD28 mAb (2 μg/ml) in a 37 °C incubator at 95% humidity and 5% CO2. On the other hand, CD14 + T cells were cultured in the presence of rhGM-CSF (100 ng/ml) and rhIL-4 (50 ng/ml) in a 37 °C incubator at 95% humidity and 5% CO2 for 5 days to induce to dendritic cells.
Preparation of NXSB Decoction
NXSB decoction decoction was composed of cornu bubali, radix rehmanniae, radix paeoniae alba, Paeonia suffruticosa Andr, Ligustrum lucidum Ait, Eclipta prostrata, angelica sinensis, and raw radix astragalis, and its granules (20 g/kit) were purchased from Kang Ren Tang. According to our team prior research, NXSB decoction granules was dissolved with distilled water, making the final drug concentration to 200 g/L.
Preparation of Drug Serum
The rats were divided into control group and experimental group, and they were given intragastric administration of distilled water and NXSB decoction, respectively. According to the conversion of human-rat body surface area, the equivalent dose method, the dose of intragastric administration was 2 g/(kg·d). The rats were intervened with NXSB decoction at 8.00 am and pm for 3 days in experimental group, when the rats were given distilled water in control group. After intragastric administration, the rats were anesthetized, meanwhile, the whole blood were collected by abdominal aortic method, stood for 1.5 h, centrifuged 10 min at 3000 rpm. The upper layer of the serum was collected as control serum and drug serum, inactivated by sterilizing filter and thermostatic water bath and stored at −20°C.
Study Design
The study set three group: the Control group, the ITP group and the NXSB group. The cells in the control group were isolated in the healthy honors peripheral blood. The cells in the ITP group and NXSB group were isolated by the ITP patients peripheral blood. In the NXSB group, the 20% drug serum substituted the 10% fetal calf serum in RPMI-1640 medium, when the control serum was applied in the control group(The relevant data on drug concentration gradient experiment were in the supplementary data). The cells were intervened for 72 h. The DC cells loaded platelet antigen were induced, specifically, the autologous platelets and CD14+ cells at ratio 100:1 were cultured in the above-mentioned medium and sequentially stimulated with 1 μg/ml LPS for 1 day to induce to the mature DC cells loaded platelet antigen. The mature DC cells loaded platelet antigen co-cultured with CD4 + T cells at Transwell (ratio 1:10), and CD4 + T cells were set the upper well when the DC cells were set the lower well.
Flow Cytometry
Cells were collected by centrifugation at 300 g for 10 min and washed by PBS three times. Then cells were suspended by 100 μl Phosphate buffered saline (PBS), according to the experimental requirement, negative tube and single stain tube were reserved and stained with the appropriate mAbs or isotype matched Igs (negative controls) for 30 min at 4°C away from the light. After staining, the cells were fixed in 1 ml 4% w/v paraformaldehyde for 30 min. After the cells were washed three times, the prepared samples were detected by flow cytometry. The regents: CD4, CD25, Foxp3, CD80, CD83, HLA-DR
Western Blot
The total cells protein was extracted by previously prepared protein lysis solution including RIPA, PMSF and phosphatase inhibitor at 100:1:1. All processes should be conducted on the ice. The bicinchoninic acid (BCA) kit was used for protein quantitative analysis, and the protein concentration of each sample was calculated by enzyme standardization instrument at the wavelength of 562 nm. Then each sample protein was quantified and subjected to SDS-PAGE electrophoresis. After protein were transferred to PVDF membranes, following membranes were blocked with Tween 20 (TBST) containing 5% skim milk for 1 h. The membranes were washed by TBST and incubated with various primary antibodies overnight at 4 °C. After washed three times in TBST, membranes were incubated with secondary antibody at room temperature for 1 h and washed. The membranes were immersed in the configured luminescent solution for 2–3 min and put into a chemiluminescent gel imaging system. For quantifying protein expression, all blots were scanned, and the signal densities were measured using NIH ImageJ software. The regents: TLR4, NF-κB, PI3 K, p-PI3 K, Akt, p-Akt, mTOR, p-mTOR. Above techniques were studies from BioTechniques (https://doi.org/10.2144/btn-2022-0034)
ELISA
By using multiple ELISA kit to quantify the cytokine secretion levels in culture supernatants. According to the reagent manufacturer's instructions, at the end, absorbance was measured with an enzyme marker. The regents: IL-2, IL-6, IL-10, IL-12, IL-17, Tumor Necrosis Factor α (TNF-α), Interferon-γ (IFN-γ), Transforming growth factor-β(TGF-β). Above techniques were studies from BioTechniques (https://doi.org/10.2144/000113475)
Statistical Analysis
The statistical analysis was completed by using GraphPad Prism 8.0 software, starting with a normality test and chi-squared test. The T-test was used to compare data between two groups. A one-way or two-way analysis of variance (ANOVA) was used to compare data between multiple groups. The data of the experiment were presented as mean ± standard (x̄±SD), P < 0.05 means the difference was statistically significant.
Results
NXSB Decoction Inhibited DC Maturation Through the TLR4/NF-κB Pathway
The results of Western blot showed that the expression of TLR4 and NF-κB was up-regulated in the ITP group, and the difference was statistically significant (P<0.05). The above two proteins were down-regulated in the NXSB decoction group, compared with the ITP group (P<0.05), implying the activation of the TLR4/NF-κB pathway in DC and ITP patients is abnormal in this pathway (Figure 1A). The Flow cytometry results showed that the expression of DC costimulatory molecules and maturation markers, CD80, CD83 and HLA-DR were significantly up-regulated after LPS stimulation (P<0.01), suggesting that LPS successfully stimulated the maturation of the DC. After NXSB decoction intervention, compared with the ITP group, the expression of DC surface costimulatory molecules CD80 decreased significantly (P<0.05); compared with the ITP group, the expression of DC surface maturation markers CD83 and HLA-DR decreased significantly (P<0.01) (Figure 1B). It was suggested that NXSB decoction could inhibit DC maturation. We used ELISA to detect the effect of NXSB on cytokines secreted by DC and found that the levels of TNF-α, IL-6 and IL-12 in the supernatant of the ITP group tended to increase compared with the control group (P<0.01). After the intervention of NXSB decoction, compared with the ITP group, the levels of TNF-α, IL-6 and IL-12 in the cell supernatant decreased significantly (P<0.01) (Figure 1C).

NXSB Decoction Modulated T Cell Function and Differentiation Via the PI3K-AKT-mTOR Pathway
We examined the effects of NXSB decoction on PI3 K/Akt/mTOR and phosphorylated proteins by Western Blot. It is very clear p-PI3 K is regulated by administration of NXSB but the PI3 K, as it the expression level remained same in each group. Similarly, the p-AKT expression is being regulated by herbal formula but not the AKT itself and same goes for the p-mTOR expression. Thus, the difference of Akt was not statistically significant, the ratio of p-PI3 K/PI3 K was higher (P < 0.05), p-mTOR/mTOR was higher (P < 0.05), and p-Akt/Akt ratio was statistically increasing in ITP group. In the NXSB decoction group, compared with the ITP group, p-PI3 K/PI3 K, p-Akt/Akt, p-mTOR/mTOR protein levels were reduced (P < 0.05), the differences in the levels of PI3 K, Akt and mTOR, were not statistically significant (Figure 2A). We conducted flow cytometry of T cells in each group and found that the proportion of Teff cells was significantly increased, the proportion of Treg was significantly decreased, and the Teff/Treg ratio was higher in the ITP group compared with the control group (P < 0.05). Compared with the ITP group, the proportion of Teff cells was reduced, the proportion of Treg cells was increased, and the Teff/Treg ratio was reduced in the NXSB decoction group (P < 0.05) (Figure 2B). We further examined the effect of NXSB on cytokine secretion by CD4 + T cells. Compared with the control group, the levels of pro-inflammatory factors IL-2, IL-6, IL-17, IFN-γ and TNF-α secreted by Teff cells in the ITP group were increased, and the levels of anti-inflammatory factors TGF-β and IL-10 secreted by Treg cells were decreased, and the differences were statistically significant (P < 0.05). The levels of IL-2, IL-6, IL-17, IFN-γ, TNF-α and IL-10 were significantly different (P < 0.01). Compared with the ITP group, the levels of IL-2, IFN-γ, TNF-α, IL-6 and IL-17 in the NXSB decoction group decreased, and the levels of TGF-β and IL-10 increased, and the differences were statistically significant (P < 0.05). IL-2, IFN-γ, TNF-α, IL-17, TGF-β and IL-10 were significantly different (P < 0.01) (Figure 2C).

The Effect of NXSBT in ITP Immune Microenvironment
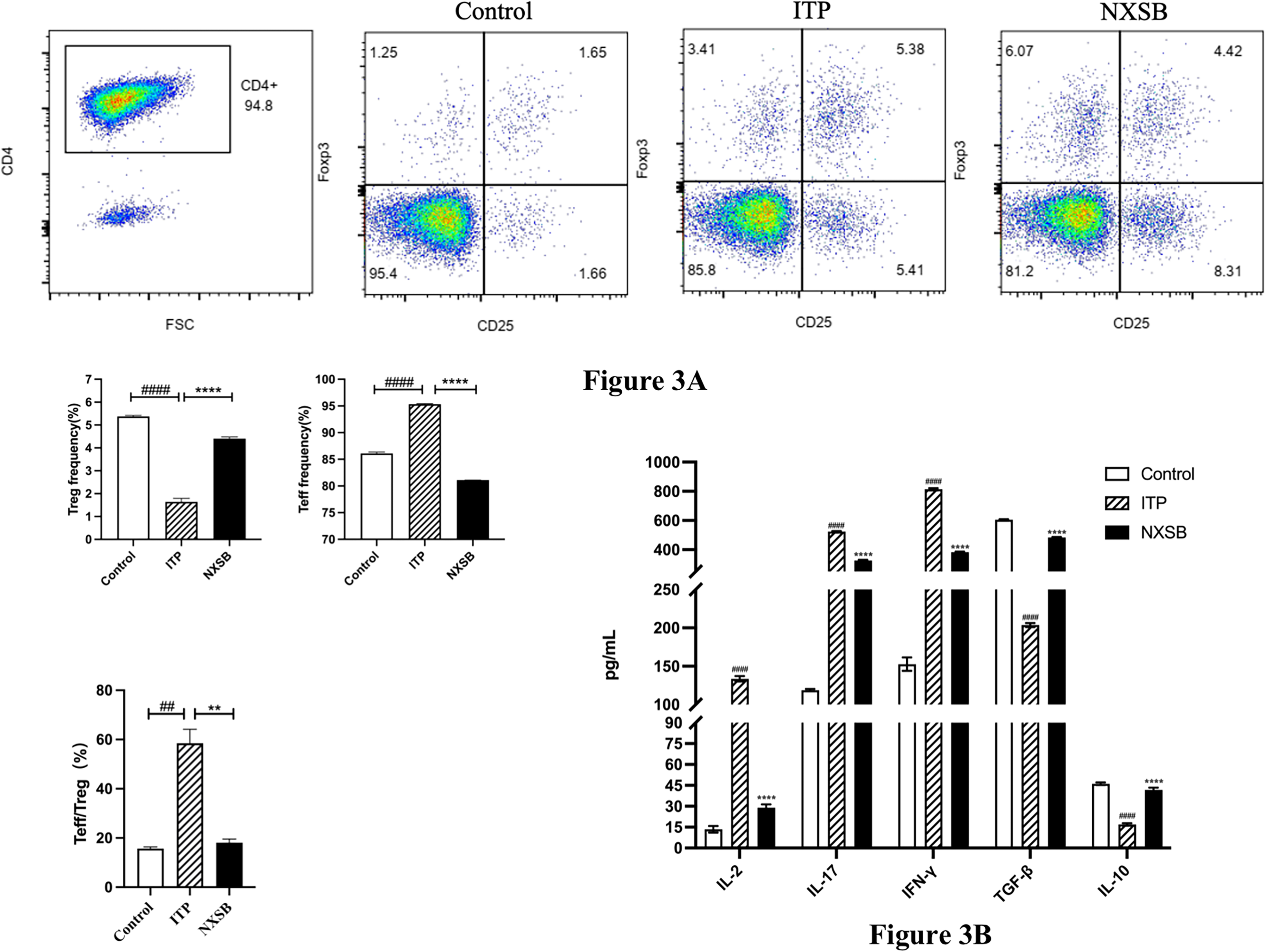
Through co-culture, the CD4 + T cells were stimulated by DC cells loaded platelet antigen. Furthermore, CD4 + T cells subset was detected by flow cytometry, the ratio of Teff/Treg increased in the model group compared with the control group, when the NXSB decoction made the ratio of Teff/Treg decrease obviously (Figure 3A). As for the function of CD4 + T cells subset, ELISA was applied to test the level of cytokines (Figure 3B). The pro-inflammatory factors, IL-2, IL-17 and IFN-γ, were increased in the ITP model group compared with the control group, in the meantime the level of pro-inflammatory factors showed decline in the NXSB decoction group compared with the ITP model group. In contrast, the anti-inflammatory factors, IL-10 and TGF-β, showed the increase trend in the NXSB decoction group compared with the ITP group, which suggesting that NXSB decoction had the effect of anti-inflammatory to the CD4 + T cells.
Discussion
Recently, cellular immunity dysregulation associated with ITP mechanism has been widely studied, more and more studies focused on T cell immune disorders, including the decrease of Tregs, the imbalance of Th1/Th2 cells, 19 and the increase of Th17, 20 Th22 and follicular helper T cells (Tfh) in ITP patients. 21 Recent studies found that DC, as the professional APC that initiates potent adaptive immune responses, plays an important role in the T cell differentiate mechanism. Immature DC affects not only the generation of Treg but also induces the immune tolerance effects of Treg and down-regulates the immune response together with Treg. 7 However, mature DC induces the apoptosis of platelets, activates specific T cells to differentiate into different subsets and causes immune balance disorder, eventually leading to the occurrence of ITP. 22 Previous studies revelated that immature DCs are activated upon recognizing pathogen-associated or damage-associated molecular patterns (PAMPs or DAMPs) via PRR.23,24 PAMPs are mostly derived from some bacteria, like Lipopolysaccharide (LPS). 25 TLRs, as one important subgroup in PRRs, are stimulated to initiate a signaling cascade resulting in activation of transcription factors, including NF-κB. 26 Henning et al found that DC played an emerging role in hepatic inflammation, 27 and DC have also recently emerged as important mediators in noninfectious chronic fibro-inflammatory conditions. 28 However, despite their activated phenotype, DC can have a protective role in acute pancreatitis by limiting sterile inflammation. 29 NF-κB is known to promote the expression of pro-inflammatory cytokines, which further stimulates an immune response. 30 Based on these studies, we hypothesized that the TLR/ NF-κB might play critical role with DC in ITP.
We performed experiments to prove this hypothesis, and our results supported it. We created an LPS-induced DC maturation cellular model in vitro and strictly set the control, ITP and NXSB decoction groups, and collected cellular samples 24 h later. First, we detected the levels of TLR4 and NF-κB treated with NXSB decoction were significantly decreased. Second, we detected the DC maturation markers (CD83, HLA-DR), and these markers were significantly increased after LPS stimulation, indicating that the LPS-induced DC maturation model was successfully established. Notably, these markers were obviously decreased after NXSB decoction treatment. It implied that NXSB decoction inhibited of dendritic cell maturation. The mechanism may be related to the inhibition of TLR-4/NF-κB pathway. According to prior findings, we further detected increased amount of pro-inflammatory cytokines secreted by the LPS-stimulated DCs, such as TNF-α, IL-6 and IL-12. 31 Not surprised to us, we found that these above cytokines were downregulated in the NXSB decoction group compared with ITP group. Taken together, NXSB decoction inhibited the DC maturation further reduced the levels of inflammatory factors IL-6, IL-12 and TNF-α through the TLR-4/NF-κB pathway. Thereby maintaining DC in an immature state may be an important mechanism of NXSB decoction for treating ITP.
Abnormal T cell homeostasis, including the deficiency of Treg cells and excessive activation of Teff cells, plays a critical role in the pathogenesis of ITP. Teff and Treg are two T cell subsets with opposite functions that proliferate and differentiate after naive T cells are activated. Teff and Treg antagonize each other to maintain the immune balance of the body. Chronic inflammation and probably autoimmune disorders would result from persistent Teff activation that is uncontrolled by Tregs. 32 Han et al demonstrated that decitabine, a hypomethylating drug, increased platelet counts in ITP by restoring T cell homeostasis through regulation of Tregs with increasing their numbers and immunosuppressive functions. 33 This finding is inspiring to our study indication regulation role of NXSB decoction in ITP induced T cell homeostasis, and we subsequently confirmed the results using flow cytometry analysis. We sought to explore which pathway promoting pathophysiological processes in ITP patients are affected by NXSB decoction. Involvement of PI3 K/Akt and TNF-α-mediated activation of NF-κB has been suggested in the apoptotic process of various types of cells.34–36 Wang et al demonstrated that platelet autophagy was diminished in ITP patients and was regulated by the PI3 K/AKT/mTOR pathway. 37 Therefore, we detected the PI3 K/AKT/mTOR pathway protein expression by Western blot, including the levels of PI3 K, p-PI3 K, Akt, p-Akt, mTOR, p-mTOR. Our studies indicated that the PI3 K/AKT/mTOR signaling pathway is abnormally activated in ITP group T cells. NXSB decoction significantly suppressed phosphorylation of PI3 K and mTOR. It indicated that suppression of PI3 K leads to inhibition in its downstream activation of Akt and mTOR, which induced the differentiation of T cells towards Treg and reduced the pro-inflammatory cytokines secretion, meanwhile inhibiting Teff differentiation. Moreover, inhibiting the PI3 K/Akt/mTOR signaling pathway reduced the differentiation of T cell to Th1, Th2 and Th17, and promoted the generation of Treg cells. 17 Activated T cells have been shown to play important roles in ITP. 38 In several studies, CD4+ T cell differentiation abnormally, specifically Teff/Treg imbalance, was found in ITP patients. Thus, correcting the Teff/Treg imbalance became an important entry point for the treatment of ITP. In this study, we used the flow cytometry experiment to investigate whether NXSB decoction could regulate the immune imbalance in ITP patients. The results showed that NXSB decoction could enhance the ratio of Teff/Treg and inhibit the proliferation of Teff. It has been suggested that restoring immune tolerance by reversing Treg damage and suppressing Th1 and Th17 subsets may be the key to sustain treatment of ITP.39,40 Pro-inflammatory cytokines secreted by Teff, IL-2, IL-6, IL-17, IFN-γ and TNF-α, have attracted much attention due to their important roles in inflammation-related diseases. 41 Tregs have an anti-inflammatory role by releasing anti-inflammatory cytokines like IL-10 and TGF-β. 42 In a previous study of acute and chronic ITP children, a significant elevation in the expression of these pro-inflammatory cytokines (IFN-γ, TNF-α, IL-4 and IL-6) and a significant reduction in Treg dysfunction was observed. 39 In addition, Ning Hong Guo et al found that the level of anti-inflammatory cytokine (IL-10) in the ITP patients were significantly upregulated after the administration of resveratrol. 43 In our study, we examined the levels of major pro-inflammatory cytokines between different groups and found that IL-2, IFN-γ, TNF-α, IL-6 and IL-17 were significantly decreased in the NXSB decoction group compared with the ITP group. Furthermore, the anti-inflammatory cytokines TGF-β and IL-10 were markedly upregulated after NXSB decoction treatment. In parallel to data obtained in other studies, our study indicated that NXSB decoction was beneficial to suppress the inflammatory response and restore the immunosuppressive function of Treg in the ITP occurrence.
T cell proliferation and differentiation requires the participation of DCs. After DC was stimulated by external antigens, began to transform from immature state to mature state, and provided three kinds of stimulus signals to activate the initial T cells, so that the T cells continue to proliferate and differentiate in different subsets. In our study, DC cells were loaded platelet antigen and co-cultured with CD4+ T cells, and the process of ITP immune microenvironment was simulated in vitro. In our study, the co-culture model in vitro simulates the process of DCs maturation, stimulating from antigens and antigen presenting, and the mature DCs induce CD4 + T cells to excessively proliferate and differentiate in different directions. In ITP, the interaction between DCs and Tregs has little been investigated although decreased number/function of Tregs as well as altered DCs have been described. A previous study found that in ITP, freshly isolated circulating DCs show decreased ability of antigen presenting after in vitro maturation with LPS. This finding suggested the mature DCs reduced the ability of converting T cells into Tregs. 44 Therefore, we focused on the level of CD4+ T cells proliferation and differentiation in ITP. We found that CD4+ T cells were excessive proliferation in ITP patients. These data are in agreement with the previous experiment data that CD4+ T cell single culture intervened by NXSB decoction, that demonstrates the NXSB decoction strongly inhibited in vitro cell proliferation CD4+ T cells. According to the prior experiments, we would like to detect whether NXSB decoction intervened the DCs maturation then influenced the T cells differentiation. Our data indicated that NXSB decoction increased the number of Treg, decreased the secretion of pro-inflammatory factors, such as IL-2, IL-17, and IFN-γ, and increased the secretion of anti-inflammatory factors TGF-β and IL-10.
Taken together, our results indicate that NXSB decoction affecting the PI3 K/Akt/mTOR and TLR4/NF-κB pathway may be a new and safe therapeutic agent for treatment of ITP.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241276960 - Supplemental material for NingXueShengBan Decoction Regulates Immune Homeostasis by PI3 K/Akt/mTOR and TLR4/NF-κB Signaling Pathways in Immune Thrombocytopenia Patients
Supplemental material, sj-docx-1-npx-10.1177_1934578X241276960 for NingXueShengBan Decoction Regulates Immune Homeostasis by PI3 K/Akt/mTOR and TLR4/NF-κB Signaling Pathways in Immune Thrombocytopenia Patients by Haijin Wang, Mengxiao Wang, Wuxia Yang, Runfeng Ni, Lin Ma, Dan Wang, Chao Yan, Yuhong Wu, Baoshan Liu and Aidi Wang in Natural Product Communications
Supplemental Material
sj-tiff-2-npx-10.1177_1934578X241276960 - Supplemental material for NingXueShengBan Decoction Regulates Immune Homeostasis by PI3 K/Akt/mTOR and TLR4/NF-κB Signaling Pathways in Immune Thrombocytopenia Patients
Supplemental material, sj-tiff-2-npx-10.1177_1934578X241276960 for NingXueShengBan Decoction Regulates Immune Homeostasis by PI3 K/Akt/mTOR and TLR4/NF-κB Signaling Pathways in Immune Thrombocytopenia Patients by Haijin Wang, Mengxiao Wang, Wuxia Yang, Runfeng Ni, Lin Ma, Dan Wang, Chao Yan, Yuhong Wu, Baoshan Liu and Aidi Wang in Natural Product Communications
Supplemental Material
sj-tiff-3-npx-10.1177_1934578X241276960 - Supplemental material for NingXueShengBan Decoction Regulates Immune Homeostasis by PI3 K/Akt/mTOR and TLR4/NF-κB Signaling Pathways in Immune Thrombocytopenia Patients
Supplemental material, sj-tiff-3-npx-10.1177_1934578X241276960 for NingXueShengBan Decoction Regulates Immune Homeostasis by PI3 K/Akt/mTOR and TLR4/NF-κB Signaling Pathways in Immune Thrombocytopenia Patients by Haijin Wang, Mengxiao Wang, Wuxia Yang, Runfeng Ni, Lin Ma, Dan Wang, Chao Yan, Yuhong Wu, Baoshan Liu and Aidi Wang in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
All animal experiments in this study were approved and supervised by the Ethics Committee of Tianjin Medical University General Hospital (approval no. IRB2020-KY-189).
Funding
The current study received grants from the National Natural Science Foundation of China (82004334 and 81373847 to WAD,81973636 to LBS)
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the by the Ethics Committee of Tianjin Medical University General Hospital (approval no. IRB2020-KY-189). Experiments were carried out in compliance with the ARRIVE guidelines. The experiments were conducted according to the principles of the Declaration of Helsinki. All methods were carried out in accordance with relevant guidelines and regulations in the declarations section.
Statement of Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
