Abstract
The traditional Chinese medicine (TCM) formula Ento-PB containing Periplaneta americana (Linnaeus) (Blattidae) and Taraxacum mongolicum Hand.-Mazz. (Compositae) has great potential for treating inflammation. This study explored the effects of Ento-PB on ulcerative colitis (UC). The UC model was induced with 2,4,6-trinitrobenzene sulfonic acid (TNBS) by enema. Male Sprague–Dawley rats (n = 32) were divided into four groups: (1) control group that received 2.5 mL/kg normal saline, (2) TNBS group that received 2.5 mL/kg normal saline, (3) Ento-PB low-dose group that received 100 mg/kg Ento-PB, and (4) Ento-PB high-dose group that received 200 mg/kg Ento-PB. Rats were administered drugs via enema for 14 days after modeling. The disease activity index (DAI), colon mucosa damage index (CMDI), histopathological score (HS), levels of interleukin-8 (IL-8), IL-10, IL-17, tumor necrosis factor-α (TNF-α), and C-reactive protein (CRP) in serum, contents of IL-2, myeloperoxidase (MPO), transforming growth factor-β1 (TGF-β1), and epidermal growth factor (EGF) in the colon, and abundance of Bifidobacterium, Lactobacillus, Enterococcus, Bacteroides, and Escherichia coli were assessed. Ento-PB administration showed a significant reduction in DAI, CMDI, and HS, contents of IL-2, IL-8, IL-17, TNF-α, CRP, and MPO, and a significant increase in the levels of IL-10, TGF-β1, and EGF. Compared with the TNBS-administered group, the abundance of Bifidobacterium, Lactobacillus, Enterococcus, and E. coli decreased, while an obvious increase in the proportion of Bacteroides was found in the Ento-PB-administered groups. Ento-PB alleviated inflammation in UC by regulating the equilibrium of Th1/Th17/Treg cytokines and recovering the imbalance between the gut microbiota. Applying Ento-PB in treating UC could be suggested.
Introduction
Limited to the colon and rectum, ulcerative colitis (UC) is a chronic nonspecific inflammation characterized by repeated relapse and remission. The morbidity of UC has increased rapidly worldwide, which makes UC one of the severe diseases that influences people's health.1–3 Edema, hyperemia, erosion, ulcers in the colon mucosa, diarrhea, bloody feces, and abdominal pain are observed in UC patients. 4 In addition, the pathogenesis of UC is unclear, and it is believed that abnormal immune responses, gut microbiota, mucosal barrier disorder, and other factors contribute to the incidence of UC. 5 Currently, nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, and immunosuppressant drugs are popular treatments for UC. However, these drugs are limited in clinical application because of their side effects, such as allergies, anti-antibody reactions, and infections. 6 Therefore, investigating alternative approaches is important for UC patients.
When used to treat a variety of diseases for 3000 years, traditional Chinese medicine (TCM) is one of the sources of complementary and alternative therapies and has fewer adverse effects. 7 The use of TCM formulas that contain 2 or more traditional Chinese herbs is the main form of TCM treatment. In addition, previous studies have proven that an increasing number of TCM formulas exert certain therapeutic effects on UC in animal models, such as Huangqin decoction, Ping Weisan decoction, and Gegen Qinlian decoction.8–10
The TCM formula Ento-PB contains two Chinese herbs: Periplaneta americana (Linnaeus.) (Blattidae) and Taraxacum mongolicum Hand.-Mazz. (Compositae). The ratio between P. americana and T. mongolicum in Ento-PB is 7:3. According to the Compendium of Materia Medica, P. americana can promote blood circulation and regulate gastrointestinal function, while T. mongolicum can dissipate swelling and facilitate detoxification. 11 In addition, current research showed that P americana could attenuate inflammation in a rat paw edema model induced by carrageenan 12 and that T mongolicum could suppress inflammation in the lung induced by lipopolysaccharide. 13 This evidence implies that the TCM formula Ento-PB may be a promising approach for treating UC, but this hypothesis needs more research.
Therefore, the purpose of this study was aimed to explore whether the TCM formula Ento-PB could attenuate 2,4,6-trinitrobenzene sulfonic acid (TNBS)-induced UC in rats and to preliminarily investigate its potential mechanism.
Materials and Methods
Chemicals and Reagents
P. americana was purchased from Good Doctor Pharmaceutical Group Co. Ltd, (Chengdu, Sichuan, China) in 2018 and was identified by Dr Zi-Zhong Yang from Dali University. T. mongolicum was bought in Jinbei medicinal materials markets (Dali, Yunnan, China) in 2018 and was identified by Dr Miao He from Dali University. The voucher specimens were deposited in the sample collection room of the Yunnan provincial key laboratory of entomological biopharmaceutical R&D. TNBS was purchased from Sigma-Aldrich (Shanghai, China), chloral hydrate from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China), rat enzyme-linked immunosorbent assay kits for IL-2, IL-8, IL-10, IL-17, tumor necrosis factor-α (TNF-α), C-reactive protein (CRP), transforming growth factor-β1 (TGF-β1), and epidermal growth factor (EGF) from Jiancheng Bioengineering Institute (Nanjing, China), HPLC-grade methanol from Fisher Scientific (Waltham, MA, USA), and HPLC-grade formic acid from Shanghai Aladdin Bio-Chem Technology Co., Ltd (Shanghai, China). Pure water (18.2 MW) for HPLC analysis was obtained from a water purification system purchased from Merck Millipore (Darmstadt, Germany). Caffeic acid, uracil, hypoxanthine, adenosine, and inosine were purchased from the National Institutes for Food and Drug Control (Beijing, China), uridine from Sigma-Aldrich Corporation (Saint Louis, USA), caftaric acid from Solarbio (Beijing, China), cichoric acid from Chengdu Biopurify Phytochemical Ltd (Sichuan, China), a stool DNA kit from Tiangen Biotech Co., Ltd (Beijing, China), the forward and reverse primers for gut microbiota from Sangon Biotech (Shanghai) Co., Ltd (Shanghai, China), 2 × Taq PCR master mix, 2 × RealStar power SYBR mixture UNG (with ROX), DNA marker, DNA gel extraction kit, and plasmid miniprep kit from GenStar Biosolutions Co., Ltd (Beijing, China), and Escherichia coli DH5α competent cells and pMD®19-T vector cloning kits from Takara Bio Inc. (Shiga Ken, Japan).
Drugs Preparation
The TCM formula Ento-PB was prepared according to the following method. First, 0.7 kg P americana and 0.3 kg T mongolicum were dried and extracted with 10 L water, three times (2 h per time) at 95°C. After filtration, the filtrate was evaporated under reduced pressure to obtain an extract with a density of 1.15 g/mL. Finally, 60% ethanol was added and, after 24 h, the precipitate was removed by filtration and the filtrate was evaporated under reduced pressure to obtain the TCM formula Ento-PB.
HPLC Analysis of the Chemical Composition of Ento-PB
An Agilent 1260 (Agilent Technologies) equipped with a diode array detector was utilized to analyze the chemical composition of Ento-PB. A Sepax HP-C18 column (250 mm × 4.6 mm, 5 μm, 120 Å; Sepax Technologies) was used for the analysis at 25°C. The flow rate, detection wavelength, and injection volume were 1 mL/min, 254 nm, and 10 µL, respectively. The samples of Ento-PB (20 mg/mL) and mixed standard compounds of uracil, hypoxanthine, uridine, adenosine, inosine, caftaric acid, caffeic acid, and cichoric acid (all 0.125 mg/mL) were dissolved in pure water and filtered through a 0.22 μm membrane filter. Solution A (methanol) and solution B (water with 0.1% formic acid [v/v]) were utilized as the mobile phases. The elution gradient was as follows: t = 0 min, eluent A (0%), eluent B (100%); t = 10 min, eluent A (0%), eluent B (100%); t = 12 min, eluent A (8%), eluent B (92%); t = 14 min, eluent A (8.8%), eluent B (91.2%); t = 19 min, eluent A (8.8%), eluent B (91.2%); t = 22 min, eluent A (10%), eluent B (90%); t = 27 min, eluent A (35%), eluent B (65%), t = 37 min, eluent A (35%), eluent B (65%); t = 60 min, eluent A (95%), eluent B (5%). In addition, the contents of compounds were analyzed by the external standard method. The calibration curve (y = ax + b) was plotted based on the peak areas (y) versus the concentrations of compounds (x).
Animals
Male Sprague–Dawley rats (n = 32, 200 ± 20 g, Certificate No. SCXK [Xiang]-2015-030) were purchased from Hunan SJA Laboratory Animals Co., Ltd (Hunan, China). All rats were housed in an air-conditioned room (temperature 25 ± 2°C, 50% humidity, 12 h light/dark cycle) and were free to chow and water. The animal experiment was carried out after rats were acclimatized to the laboratory environment for 1 week. This protocol was approved by the Animal Ethics Committee of Dali University (No. 2018-0514).
Grouping and Administration
Thirty-two rats were divided into 4 groups randomly: the control group, TNBS group, and Ento-PB low- and high-dose groups. The TNBS-induced colitis model was established by the method described by Morris. 14 Briefly, rats were fasted but free to drink water for 1 day. Then, the rats were anesthetized by chloral hydrate (0.3 mL/100 g), and a soft tube equipped with a 1 mL syringe was inserted into the rat's anus for approximately 8.0 cm. TNBS (75 mg/kg body weight) was dissolved in 30% ethanol, and then 0.6 mL TNBS ethanol solution was instilled into the colon via a soft tube. To prevent leakage of TNBS, animals were maintained in a head downward position for 10 s. The control group received an equivalent volume of normal saline. On the eighth day after modeling, the control and TNBS groups received normal saline at a dose of 2.5 mL/kg. The Ento-PB low- and high-dose groups received 100 and 200 mg/kg Ento-PB, respectively. All agents were administered by enema for 14 days.
Evaluation of the Viscera Index
The rats were anesthetized with chloral hydrate (3 mL/kg) and sacrificed 24 h after the last administration. The thymuses, livers, spleens, and colons of rats were dissected and weighed. The viscera index was calculated by the following formula: viscera index = (viscera weight)/(rat weight).
Evaluations of DAI, Colon Mucosa Damage Index (CMDI), and HS
Stool consistency, blood in feces and body weight of rats were recorded to assess disease activity index (DAI) on the 1st, 4th, 7th, 10th, 14th, and 21st days according to the method described by Hamamoto 15 (Table 1). After the last administration, rats were sacrificed, and the colons were dissected, weighed, cut along the mesentery, and cleaned with normal saline. Colon length and width were measured. Then, the colons were examined and scored by combining the criteria of Ekström 16 and Luk. 17 The criterion is shown in Table 2. Colon tissues from rats were fixed in 10% formalin, dehydrated with ethanol, embedded in paraffin, cut into 3 to 5 μm sections and dyed with hematoxylin and eosin. Then, the samples were observed using a microscope and scored by combining the methods of Hamamoto 15 and Luk. 17 The method is shown in Table 3.
The Standard for DAI Evaluation.
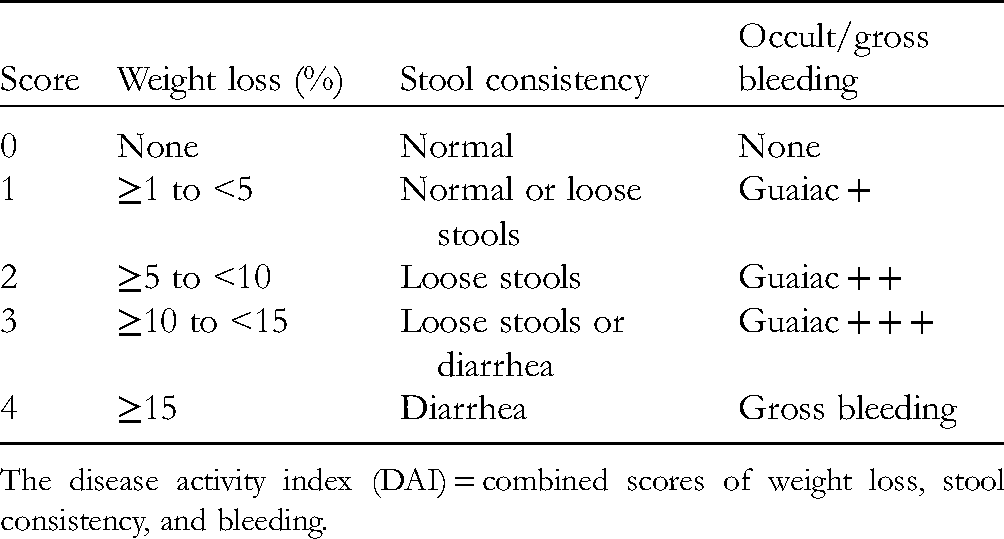
The disease activity index (DAI) = combined scores of weight loss, stool consistency, and bleeding.
The Standard for CMDI Evaluation.

Abbreviation: CMDI, colon mucosa damage index.
The Standard for HS Evaluation.
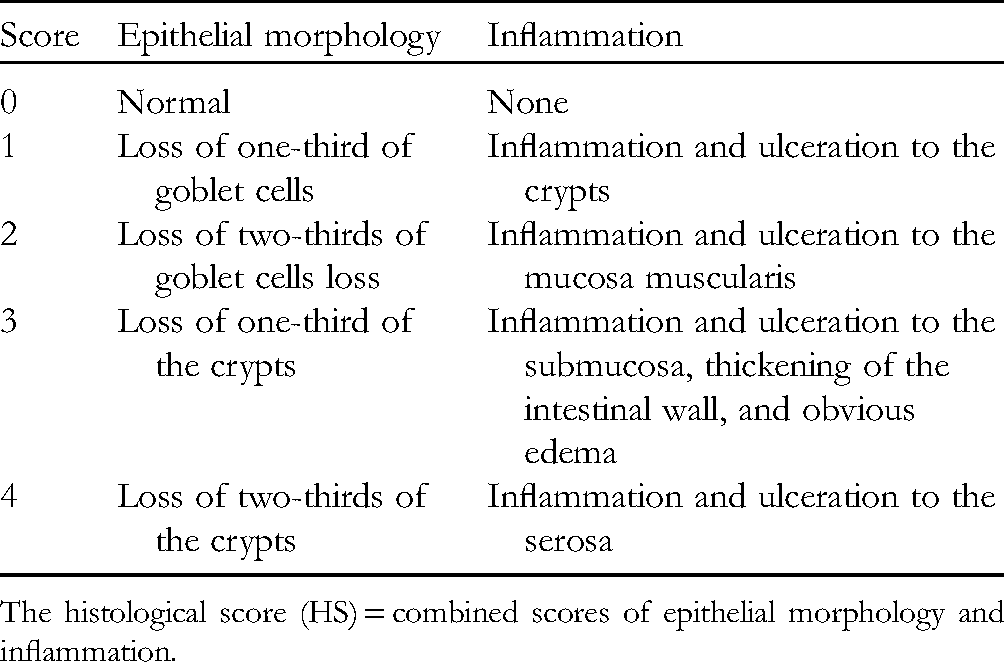
The histological score (HS) = combined scores of epithelial morphology and inflammation.
Evaluation of Inflammatory Cytokines
Blood samples of rats were taken from the abdominal aorta and centrifuged. Subsequently, supernatants were collected for the detection of the levels of IL-8, IL-10, IL-17, CRP, and TNF-α by ELISA kits. Colons were homogenized with normal saline and centrifuged, and then the supernatants were collected to measure the levels of myeloperoxidase (MPO), TGF-β1, EGF, and IL-2 in the colon via ELISA kits.
Gut Microbiota Analysis
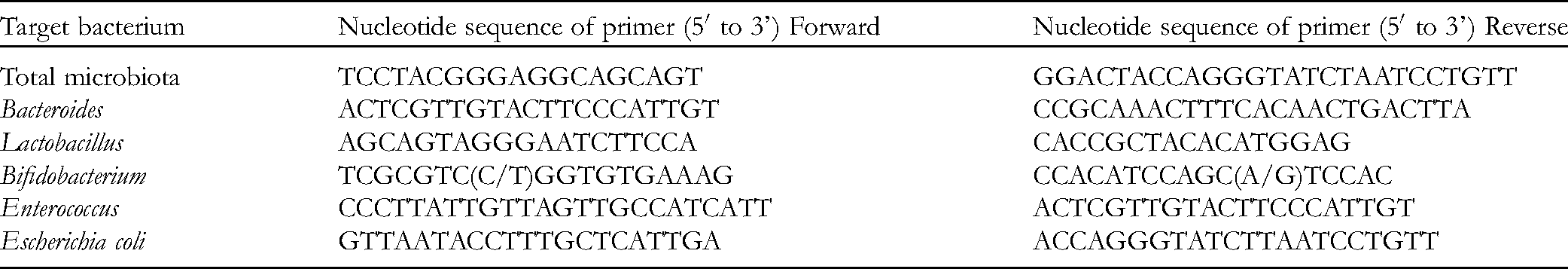
Feces were collected after rats were sacrificed. The DNA of the feces was extracted by stool DNA kits and quantified through a NanoPhotometer (Implen GmbH). Then, the 16S rDNA V3 to V4 region of the ribosomal RNA was amplified using the primers as shown in Table 4 via an ABI StepOnePlus real-time PCR system (Life Technologies). Amplicons were purified using a DNA gel extraction kit. The purified amplicons were connected with plasmid by a pMD®19-T vector cloning kit and then transferred to E coli DH5α competent cells. After blue–white plaque selection, the plasmids were extracted with a plasmid miniprep kit and identified by Sangon Biotech (Shanghai) Co., Ltd. The plasmids were quantified through a NanoPhotometer and diluted to a series of solutions with different concentrations to make the standard curves. Finally, the contents of the gut microbiota were quantified using an ABI StepOnePlus real-time PCR system.
Sequences of Primers Used for Quantitative Real-Time PCR (qRT-PCR).
Statistical Analysis
The data were analyzed using SPSS 17.0 (IBM), and all values are expressed as the mean ± SD. The data were analyzed by one-way analysis of variance (ANOVA). A P value < .05 was considered statistically significant.
Results
HPLC Analysis of the Chemical Composition of Ento-PB
After HPLC analysis of the chemical composition of Ento-PB, 8 compounds were identified in Ento-PB. They were uracil, hypoxanthine, uridine, adenosine, inosine, caftaric acid, caffeic acid, and cichoric acid. Based on the literature3, 18, the chemical constituents of P americana are uracil, hypoxanthine, uridine, adenosine, and inosine, and of T mongolicum are caftaric acid, caffeic acid, and cichoric acid; their contents are shown in Table 5.
Determination of Compound Contents in Ento-PB (n = 3).

Effects of Ento-PB on Viscera index
As shown in Figure 1, in comparison with the control group, the TNBS group significantly increased colon and liver indices (P < .01) and significantly decreased thymus and spleen indices (all P < .01). However, the Ento-PB high-dose group showed an upward trend in the thymus index (P < .05) and spleen index (P < .01) and a decrease in the colon and liver indices (all P < .01) compared with the TNBS group. In addition, the Ento-PB low-dose group showed the same trend of viscera index as the Ento-PB high-dose group, but only the downregulation of liver index was found to be significantly different (P < .05) when compared with the TNBS group.

Effects of Ento-PB (high dose: 200 mg/kg; low dose: 100 mg/kg) on visceral indices  : (A) colon index, (B) liver index, (C) thymus index, and (D) spleen index.
: (A) colon index, (B) liver index, (C) thymus index, and (D) spleen index.
Effects of Ento-PB on DAI
After modeling, the rats exhibited anorexia, weight loss, diarrhea and bloody stools. As shown in Figure 2, the TNBS group showed an obvious increase in DAI compared with the control group (P < .01) on the first day after modeling, suggesting the success of colitis induction. Interestingly, with Ento-PB treatment, the DAI showed a decreasing trend in comparison to the TNBS group. A significant decrease in the DAI (P < .01 or P < .05) was observed at 10 d, 14 d, and 21 d between the Ento-PB-treated groups and the TNBS group, which indicated that Ento-PB could attenuate inflammation in UC rats.

Effects of Ento-PB (high dose: 200 mg/kg; low dose: 100 mg/kg) on DAI.
Effects of Ento-PB on the Colon Length/Width Ratio and CMDI
As shown in Figure 3, severe ulcers were found in the TNBS-administered group, whereas in the Ento-PB-administered groups this was ameliorated in a dose-dependent manner. In addition, compared with the control group, the TNBS group displayed a great downward trend in colon length and colon length/width ratio (P < .01) and showed a strong increase in CMDI (P < .01). However, the Ento-PB-treated groups had significantly increased colon length (P < .01) and showed an obvious reduction in CMDI (P < .01) compared to the TNBS group. Additionally, compared with the TNBS-treated group, the colon length/width ratio in the Ento-PB high-dose group increased remarkably (P < .01), but no significant difference was found in the Ento-PB low-dose group.

Effects of Ento-PB (high dose: 200 mg/kg; low dose: 100 mg/kg) on macroscopic changes in the colon, colon index, colon length, colon length/width ratio and CMDI  : (A) macroscopic changes in the colon, (B) CMDI, (C) colon length, and (D) colon length/width ratio.
: (A) macroscopic changes in the colon, (B) CMDI, (C) colon length, and (D) colon length/width ratio.
Effects of Ento-PB on Colon HS
As shown in Figure 4, under a microscope, the colon in the control group displayed intact mucosal epithelium without inflammatory cell infiltration. However, the colons in the TNBS group showed severe inflammatory cell infiltration and great loss of goblet cells and crypts. Interestingly, with the increase in Ento-PB dose, inflammatory cell infiltration, and the loss of goblet cells and crypts were obviously improved. In addition, histopathological score (HS) in the TNBS group increased significantly (P < .01) compared to that in the control group. However, a significant decrease in HS (P < .01 or .05) was found in the Ento-PB-treated groups compared with the TNBS group.

Effects of Ento-PB (high dose: 200 mg/kg; low dose: 100 mg/kg) on microscopic changes (H&E, × 200) in the colon and HS of the colon  : (A) microscopic changes in the colon and (B) HS of the colon.
: (A) microscopic changes in the colon and (B) HS of the colon.
Effects of Ento-PB on serum Levels of IL-8, IL-10, IL-17, CRP, and TNF-α
As shown in Figure 5, compared with the control group, the serum levels of IL-8, IL-17, CRP, and TNF-α increased significantly (P < .01), and the production of IL-10 in serum showed a downward trend (P < .01) in the TNBS group. However, compared with the TNBS group, the Ento-PB-administered groups exhibited remarkable decreases in the expression of IL-8, IL-17, and TNF-α (both P < .01) and showed obvious increases in the level of IL-10 (P < .01). In addition, the Ento-PB high-dose group markedly downregulated CRP (P < .01) in comparison with the TNBS group, but no significant difference was observed in the Ento-PB low-dose group.

Effects of Ento-PB (high dose: 200 mg/kg; low dose: 100 mg/kg) on the levels of IL-8, IL-17, IL-10, CRP, and TNF-α in serum  : (A) IL-8, (B) IL-17, (C) IL-10, (D) CRP, and (E) TNF-α.
: (A) IL-8, (B) IL-17, (C) IL-10, (D) CRP, and (E) TNF-α.
Effects of Ento-PB on the Levels of IL-2, MPO, TGF-β1, and EGF in the Colon
As shown in Figure 6, compared with the control group, the TNBS group showed an obvious decrease in the levels of TGF-β1 and EGF (P < .01) and a great increase in the expression of MPO and IL-2 (P < .01). However, compared with the TNBS group, the Ento-PB-treated groups showed not only a striking decrease in the levels of MPO and IL-2 (both P < .01), but also an increase in the expression of EGF (P < .01). In addition, compared with that in the TNBS-treated group, the expression of TGF-β1 in the Ento-PB high-dose group was notably elevated (P < .05), while no significant difference was indicated in the Ento-PB low-dose group.

Effects of Ento-PB (high dose: 200 mg/kg; low dose: 100 mg/kg) on the levels of TGF-β1, MPO, EGF, and IL-2 in the colon  : (A) TGF-β1, (B) MPO, (C) EGF, and (D) IL-2.
: (A) TGF-β1, (B) MPO, (C) EGF, and (D) IL-2.
Effects of PB on gut Microbiota Analysis
As shown in Figure 7, the population of total microbiota showed no significant difference among the groups. Compared with the control group, the abundance of E. coli increased significantly (P < .05), while an obvious reduction in the proportion of Bacteroides (P < .05) was found in the TNBS-treated group. In addition, although the abundances of Bifidobacterium, Lactobacillus, and Enterococcus in the TNBS-treated group increased, no significant difference was detected between the TNBS group and the control group. However, the trend of the microbiota was reversed in rats treated with Ento-PB. Compared with the TNBS group, all Ento-PB dose groups had reduced proportions of E. coli, Bifidobacterium, Lactobacillus, and Enterococcus and increased abundances of Bacteroides. The change in Bifidobacterium was found to be significant (P < .05) in all Ento-PB-administered groups.

Effects of PB (high dose: 200 mg/kg; low dose: 100 mg/kg) on gut microbiota  .
.
Discussion
With increasing morbidity, UC is a chronic and relapsing inflammatory disease of the intestinal tract. There is still no effective treatment for UC patients, which underscores the need for alternative treatments. 19 Currently, TCM have been developed and suggested as complementary and alternative treatments for many diseases, including UC. 20 Herein, we demonstrated that the TCM formula Ento-PB could ameliorate UC by regulating the balance of Th1/Th17/Treg cytokines and improving intestinal microbiota disorders.
Based on the multiple ingredients, TCM have been used to treat many complex diseases successfully. According to the HPLC analysis, uracil, hypoxanthine, uridine, adenosine, inosine, caftaric acid, caffeic acid, and cichoric acid were identified in Ento-PB. Some of the compounds have been reported to have activities against UC or inflammation. Caffeic acid could improve mucosal histopathology, balance biochemical indices, and inhibit the recruitment of macrophages, which slowed the development of UC. 21 Cichoric acid significantly decreased the degradation of IκBα and the expression of IL-1β, TNF-α, COX-2, and PGE2 in MSU-stimulated THP-M cells through the NF-κB signaling pathway and the NLRP3 inflammasome. 22 Inosine increased in colon microbiota-derived metabolites when barley leaf extract was given to UC mice, and it was found that administrating inosine could exert a protective effect in DSS-induced colitis, similar to that of barley leaf extract. These suggested that Ento-PB has the potential to treat UC via multiple ingredients and pathways. 23
In TCM, UC is called “xie xie” and “li ji”, which means severe diarrhea, loose stool, and abdominal pain. Based on traditional Chinese medical theories, UC is related to the spleen, liver, kidney, and lung. Specifically, the downregulation of spleen function is the main factor that causes excessive dampness in UC patients. 24 In addition, it is well known that the spleen and thymus are important organs in modulating the immune response of our body. In addition, CRP is an acute-phase protein produced by liver tissue, and it has been observed that CRP rises sharply in the period of inflammation. 25 In this study, the Ento-PB high-dose group reversed the reduction in the spleen and thymus indices, and the increase in CRP in the TNBS group, indicating that Ento-PB might play an important role in recovering the function of the spleen, thymus, and liver.
The TNBS-induced UC model was used in this study. TNBS is a hapten that can cause immunological responses when it combines with macromolecular substances. In addition, ethanol can also do harm to the mucosa. Therefore, the use of TNBS and ethanol contributes to serious inflammation in the colon. Moreover, this model is simple, and the histology and distribution of inflammation in this UC model are similar to the clinical manifestations of UC.14,26 In this research, it was found that Ento-PB could attenuate inflammatory cell infiltration, ulcers, and edema in the colon of UC rats. Additionally, increased DAI, CMDI, and HS induced by TNBS were significantly reduced after administration of Ento-PB, which suggested that it could promote the proliferation of epithelial cells and repair tissue injury in UC.
As a crucial part of the body, the gut microbiota interacts with the immune system of the host and is involved in the etiology of UC. 27 The proportion of beneficial bacteria and pathogenic bacteria is associated with the incidence and development of UC. Beneficial bacteria, such as Bacteroides, Bifidobacterium, and Lactobacillus, can provide a variety of advantages for the host in nutrition, metabolism, and the immune system.28–30 However, pathogenic bacteria such as E. coli, Enterococcus faecalis, and Helicobacter hepaticus cause different types of colitis in IL-10-deficient mice. 31 In addition, intestinal flora can produce a variety of substances, such as lactate, short-chain fatty acids, and inorganic compounds, by fermenting carbohydrates. Among the substances, some provide a variety of benefits for the host's health, while others harm the intestinal tract of the host. Lactate contributors (eg Bifidobacterium, Lactobacillus, and Enterococcus) can produce lactic acid, which will reduce the pH in the gut, change the abundance and distribution of the gut flora and exacerbate the inflammation of the intestinal tract in disease situations. 32 In this study, the population of lactate producers, such as Bifidobacterium, Lactobacillus, and Enterococcus, increased in the TNBS group compared with the control group. At the same time, it was also found that the abundance of E. coli was elevated, while a downregulated trend was found in the proportion of Bacteroides after the administration of TNBS. Contrary to the TNBS group, the Ento-PB-treated groups showed the opposite result of the population among the gut microbiota, which highlighted the effect of Ento-PB in regulating the proportion of intestinal microbiota in UC.
Under normal physiological conditions, there is an equilibrium between the intestinal microbiota and immune system, but homeostasis is disrupted in disease situations. In UC, changes in the intestinal microbiota affect the differentiation and cytokine expression of T cells, including Th1, Th17, and Treg cells, which cause excessive tissue inflammation. 33 Th1 cells trigger classical macrophages and regulate the immune response through the expression of IL-2, TNF-α, and IFN-γ. 34 Th17 cells can secrete proinflammatory cytokines such as IL-17, which contribute to the recruitment of neutrophils together with the expression of MPO. 35 As a result of neutrophil infiltration, IL-8 increases and EGF decreases, which slows wound healing and aggravates the inflammatory response in UC.36,37 In contrast, Treg cells can produce IL-10 and TGF-β1 to suppress the proliferation of Th cells that can express proinflammatory cytokines and thus exert a vital effect in reducing inflammation in UC. 38 In this work, it was observed that TNBS-administered rats diminished the levels of IL-10, TGF-β1, and EGF while elevating the expression of IL-2, IL-8, IL-17, TNF-α, and MPO in comparison with the control group. Conversely, totally different results of the above cytokines were found in the rats treated with Ento-PB, which showed that Ento-PB played a beneficial role in regulating the balance of Th1/Th17/Treg cytokines.
In the current study, it was shown that Ento-PB exerted a therapeutic effect in UC by modulating Th1/Th17/Treg cytokines. However, further investigations are needed to study the change in the number of Th1/Th17/Treg cells in the immune organs, which accurately reflects the effect of Ento-PB on inflammation in UC. In addition, although the abundance of gut microbiota was improved by the Ento-PB treatment when compared with the TNBS group, there were some opposite trends reported in the changes of Bifidobacterium, Lactobacillus, and Bacteroides in UC.39,40 This may be related to the multicomponent and multitarget effects of TCM. Thus, follow-up studies are necessary to demonstrate clearly the correlations between intestinal flora and Ento-PB in UC. Moreover, based on the multiple components and targets, TCM treat diseases in a holistic way, so it was not clear whether the curative effects originate from active compounds. Therefore, research should focus on the active compounds of Ento-PB and how they exert effects in UC in the future.
Conclusion
According to our findings, the TCM formula Ento-PB could attenuate TNBS-induced inflammation in UC rats, and its potential mechanism might be related to regulating the balance of Th1/Th17/Treg cytokines and recovering the proportion of gut microbiota. These findings indicated that Ento-PB could become a potential alternative drug for UC in the future.
Footnotes
Acknowledgments
We thank American Journal Experts for the help in English language editing service.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by theNational Natural Science Fund of China under grant (Nos. 82060780, 81860742, and 81860765), Yunnan Fundamental Research Projects (Nos. 2019FH001-003, 202001AT070021), Innovative Team of Dali University for Digital Development of Medicinal Insects and Arachnids Resources (No. ZKLX2020303), Special Fund for Traditional Chinese Medicine Pieces of Department of Industry and Information Technology of Yunnan Province (No. 2019-YG-067), and the Special Program of Science and Technology of Yunnan Province (No. 202002AA100007).
Ethical Approval
All experimental procedures involving the animals were conducted in accordance with the Animal Ethics Committee of Dali University (Approval number: 2018-0514).
Informed Consent
There are no human subjects in this article, and therefore, informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
