Abstract
Introduction
Oxidative stress and inflammation are closely linked phenomena that occur normally in the human body. Both conditions are required for regulatory and protective effects. The immune system responds in a complex manner to harmful stimuli, such as infections, injuries, and damaged cells, by inducing the production of both free radicals and pro-inflammatory cytokines. 1 After eliminating harmful substances, a healthy body can neutralize free radicals and inflammation by producing antioxidants, repairing damaged tissues, regenerating cells, and reverting the affected area to a state of normal function that maintains body balance. Prolonged exposure to oxidative stress and chronic inflammation can cause significant cellular damage, including protein and DNA damage. Damage to cellular components can lead to several diseases, including cancer, diabetes, vascular system problems, and arthritis. 2 The World Health Organization ranks chronic diseases, including chronic inflammation, as one of the highest threats to human health. 3 A healthy lifestyle and dietary or therapeutic supplements can align with the natural processes of the body, neutralize oxidative stress, and mitigate inflammation.
Seaweeds have been used as a powerful functional food and medicinal source.
4
They contain a diverse range of bioactive substances, including polyphenols, polysaccharides, fatty acids, sterols, peptides, and pigments, which exhibit antioxidant and anti-inflammatory properties.
5
Materials and Methods
Collection and Preparation
Estimation of Total Phenolic Content
The Folin–Ciocalteu method was used for estimating total phenolic content. 12 A mixture of 1 mL of 0.1 mg/mL algal extract, 1 mL 95% ethanol, 5 mL distilled water, and 0.5 mL 50% Folin-Ciocalteu reagent was prepared for each extract. The mixture was incubated at 21∼ 23 °C for 5 min, followed by adding 1 mL of 5% Na2CO3 solution. The solution was then mixed and incubated for 1 h in the dark. Subsequently, absorbance was measured at 700 nm using a microplate reader (BioTek, Synergy, UT, USA). Different concentrations of gallic acid (10, 20, 40, 60, 80, 100 µg/mL) were used to generate a standard curve, and the results were expressed as gallic acid equivalents (mg GAE/g).
HPLC Analysis
The composition of unknown total phenolic content of the algal extracts was analyzed using HPLC (Agilent 1100, USA). Agilent ChemStation software was used to obtain and analyze the chromatograms. The components were identified by comparison of the retention time of the unknown peak with those of the reference standards; a mixture of phenolic compounds and flavonoids were used separately as the reference standards. A binary mixture of methanol/water (50:50, v/v) adjusted to pH 2.8 using phosphoric acid was used as the mobile phase at an isocratic flow rate of 1.0 mL/min. The detector was set at a range of 200 ∼ 300 nm with the mobile phase. The analysis was performed by the Regional Center of Mycology and Biotechnology (Al-Azhar University, Egypt). 13 The results were expressed as µg/g dry weight.
Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
Chemical analyses of the algal extracts were performed using GC-MS (ISQ LT GC-MS Model, Thermo Scientific, USA). The analysis was performed by the Regional Center of Mycology and Biotechnology (Al-Azhar University, Egypt). Turbomass software was used to handle the mass spectra and chromatograms. The contents of the extracts were identified using linked library (NIST and Wiley Registry eighth Edition) comparisons. This instrument (30 m length; 0.25 mm internal diameter; 0.25 film thickness) used a DB5-MS and a capillary column filled with cross-linked 5% diphenyl and 95% dimethylpolysiloxane. An electron ionization system was employed for GC/MS detection. It was ionized with a 70 eV energy. The carrier was made up almost entirely of helium gas. It had a 1 ml/min constant flow rate and a 1 ml injection volume (spitless mode; injector temperature, 20 °C; ion-source temperature, 300 °C). The oven was at a set temperature of 40 °C (isothermal for 3 min). It increased at a rate of 5 °C/min until it reached 280 °C, after which it increased at a rate of 7.5 °C/min until it reached 290 °C. A 9-min isothermal at 300 °C marked the end of this phase. At 70 eV, mass spectra were recorded. Additionally, 0.5-s scan intervals and components between 45 and 450 Da were collected. The GC ran for 63 min in total. Mass spectra and chromatograms were processed using a TurboMass, and the relative percentage sum of each factor was calculated by multiplying its average peak area by the total number of areas.
Cell Culture
Vero and RAW264.7 cell lines were obtained from American type culture collection (ATCC, Rockville, MD, USA). Vero cells were cultured in RPMI medium (Gibco, NY, USA) supplemented with 1% antibiotic solution, sodium pyruvate (110 mg/mL), and 10% FBS. RAW264.7 cells were grown in DMEM (Gibco) supplemented with 10% FBS, 1% antibiotic solution, and sodium pyruvate (110 mg/mL). The cells were sub-cultured every 2 d. An RPMI medium containing 10% FBS, 25 ng/mL granulocyte-macrophage colony-stimulating factor (GM-CSF) were used for bone marrow-derived dendritic cells (BMDCs). The culture medium was replaced every day for 6 d and examined daily. All cells were maintained in an incubator supplemented with 5% CO2 atmosphere at 37 °C.
Cytotoxicity Assay
A standard MTT assay was used to test the extracts cytotoxicity on Vero normal cell line and BMDCs. 14 Briefly, MTT solution (50 μl; 2 mg/mL stock) was added to the cells and further incubated for 3 h. Optical density was measured at 540 nm using a microplate reader (BioTek, Synergy, UT, USA).
Measurement of Antioxidant Activity Against AAPH or H2O2
A colorimetric 3-(4,5-dimethyl-2,5 thiazolyl)-2,5 diphenyltetrazolium bromide (MTT, Sigma, St. Louis, MO, USA) assay was performed to estimate the antioxidant protective effect of extracts at different concentrations on the Vero cell line against AAPH and H2O2-induced oxidative stress. 15 Vero cells were seeded in 96-well plates at a density of 1 × 105 cells/well and incubated for 24 h. After 24 h, the cells were treated with 10 µL of different concentrations of each algal extract, incubated for 1 h, and then treated with 10 µL of 200 mM AAPH or 10 µL of 400 mM H2O2 and incubated for 24 h. After incubation, MTT (50 µL; 2 mg/mL) was added to each well to reach a total volume of 250 μL. After 3 h of incubation, the supernatants were aspirated and the formazan crystals in each well were re-dissolved in DMSO. The amount of purple formazan was determined by measuring absorbance at 540 nm using a microplate reader. The optical density of the formazan generated in the control cells was considered as 100% cell viability.
Measurement of NO Production in RAW264.7 Cells
RAW264.7 cells were incubated in a 24-well plate with a concentration of 1 × 105 cells/mL at 37 °C. After 24 h, the cells were treated with 25 µL of each extract at different concentrations, incubated for 1 h, and stimulated with LPS (1 µg/mL). Subsequently, the cells were incubated for 18 h. After incubation, the amount of NO produced in the culture medium was quantified using the Griess reagent. In brief, 100 µL of cell culture medium was mixed with 100 µL of Griess reagent. Later, the mixture was kept at 21∼ 23 °C for 10 min, and the absorbance at 540 nm was detected using a microplate reader. The samples were compared to the control (DMSO-treated only) and LPS (DMSO + LPS-treated only). 16
Cytokine Measurement
BMDCs were seeded at a concentration of 1 × 105 cells/mL in 48-well plates, and incubated at 37 °C. After 1 h, the cells were treated with the extracts at different concentrations for 1 h before stimulating with LPS (1 µg/mL). The cells were incubated for 18 h. After incubation, the amount of cytokine produced in the culture medium was quantified using ELISA (BD PharMingen, CA, USA) according to the manufacturer's instructions.
17
The rate of inhibitory activity (%) of the extracts was calculated for each cytokine using the following formula:
Statistical Analysis
All the results were expressed as means ± standard deviation of three independent values. One-way analysis of variance, followed by Dunnett's post-hoc test, was performed to evaluate the statistical significance of the differences between the control and experimental groups, where values with
Results
Composition of Bioactive Compounds
Seaweeds extracts are known to contain several important bioactive compounds. Phenolic content is one of the characteristic features of seaweeds for its wide range of bioactivities. Therefore, it is an important aspect to estimate the total phenolic content of the studied extracts. In the present study, the two species were found to contain considerable amounts of total phenolics (Table 1). Moreover, HPLC analysis was performed to identify the main components of the phenolic content. The HPLC analysis of the two studied extracts revealed the presence of 19 main compounds, of which 10 were common (Table 2). The
Total Phenolic Content of the Studied Seaweeds in mg Gallic Acid Equivalent (GAE)/g.

List of Phenolic Compounds Identified Using HPLC. The Results Were Expressed as µg/g Dry Weight.

aRT indicates the retention time. Shaded rows are for common compounds in the two extracts.
In order to discover more about the extracts’ bioactive compounds, GC-MS analysis was carried out. The GC-MS chromatogram of the extracts yielded 22 main compounds, of which nine were common (Figure 1, Table 3). These compounds were mainly fatty acids and their derivatives along with diterpenes and steroidal molecules.

GC-MS chromatograms of
Chemical Composition Identified Using GC-MS Analysis.

Shaded rows are for common compounds in the extracts.
The analysis of chemical composition revealed the presence of diverse bioactive compounds within the two extracts, which indicates the potential role of the extracts in possessing bioactivities and encourages the further study of the extracts.
Cytotoxicity
Before proceeding to study the biological activity, it is important to check the toxicity of the two extracts on

Cytotoxicity test of the extracts on Vero cells (A) and BMDCs (B). The cells were pretreated with the extracts at different concentrations (μg/mL) for 24 h. MTT assay was used to determine cell viability (%). Control; no extract. *
Antioxidant Activity
The seaweeds of the Red Sea are known for its developed antioxidant system, represented by the production of bioactive compounds, to enable the living in harsh conditions of salinity and temperature. The extracts of those seaweeds can be used as antioxidants for therapeutic purposes. In order to test this possibility, the protective effects of the current seaweeds’ extracts were tested against AAPH- and H2O2-induced oxidative stress in Vero cell line model (Figure 3, A and B). The results showed that the Vero cell viability % decreased significantly upon the treatment with AAPH or H2O2 compared with untreated control cells, which indicated a damaging effect of oxidative stress on cells (
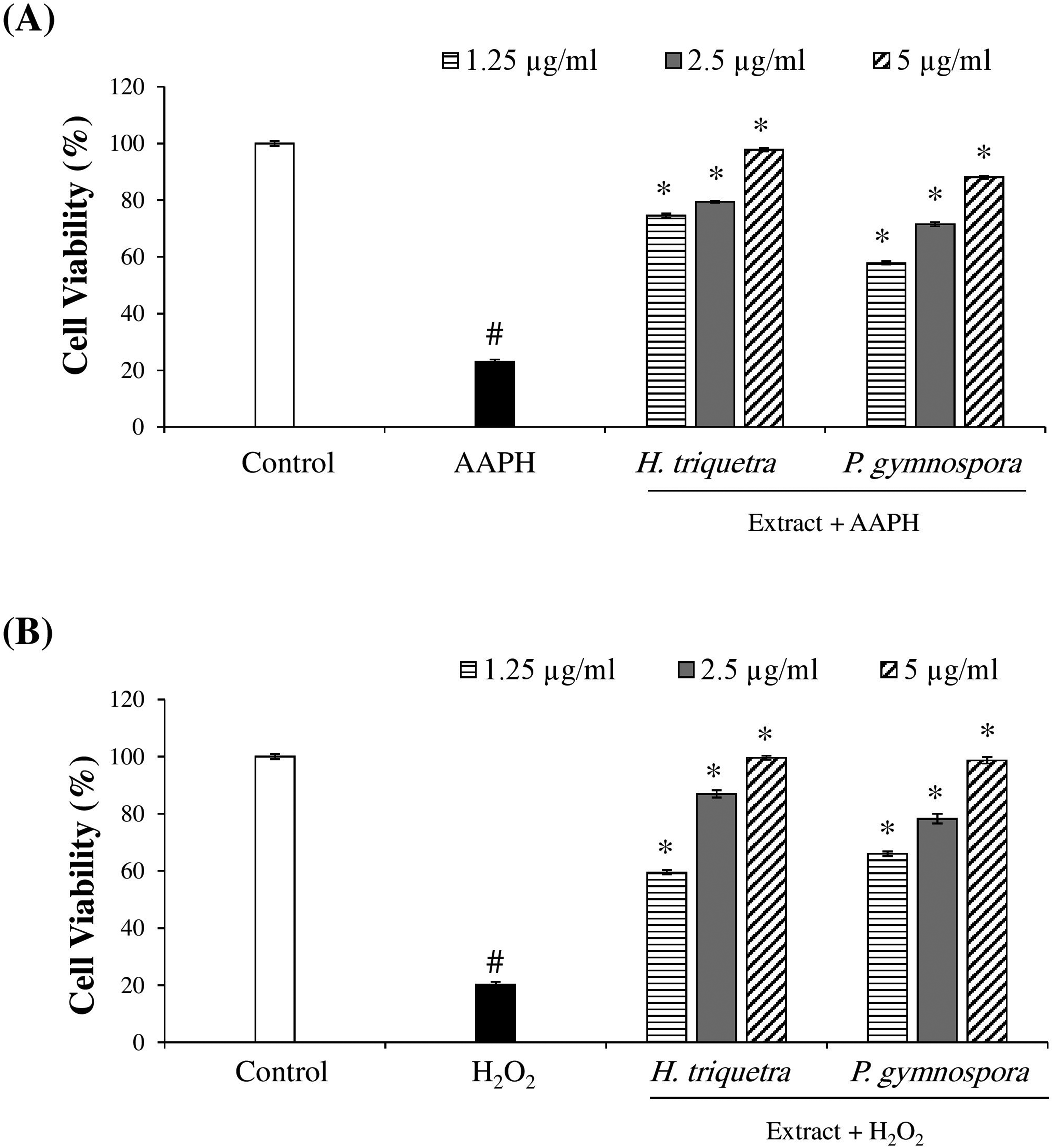
Antioxidant effect of the extracts against AAPH- (A) or H2O2- (B) induced oxidative stress in vero cells. The cells were pretreated with the extracts at different concentrations (μg/mL) for 1 h, followed by stimulation by AAPH (A) or H2O2 (B) for 24 h. Control includes the untreated Vero cells. Cell viability (%) was measured using MTT assay. #
Anti-Inflammatory Activity
As the current studied extracts showed a cellular protective effect against oxidative stress, it was encouraging to study their anti-inflammatory effect

Effect of the extracts on NO production in LPS-stimulated RAW264.7 cells. The cells were pretreated with the extracts at different concentrations (μg/mL) for 1 h, followed by stimulation with LPS for 18 h. Control includes untreated RAW264.7 cells. NO production was tested in the culture medium using the Griess reagent. *
To further confirm the anti-inflammatory activity, the extracts were tested for their inhibitory activity for cytokine production in LPS-stimulated BMDCs. In the current study, the inhibitory activity (%) of the extracts significantly increased in a dose-dependent manner (Figure 5 A, B, and C). The 50% inhibitory concentration (IC50) values of
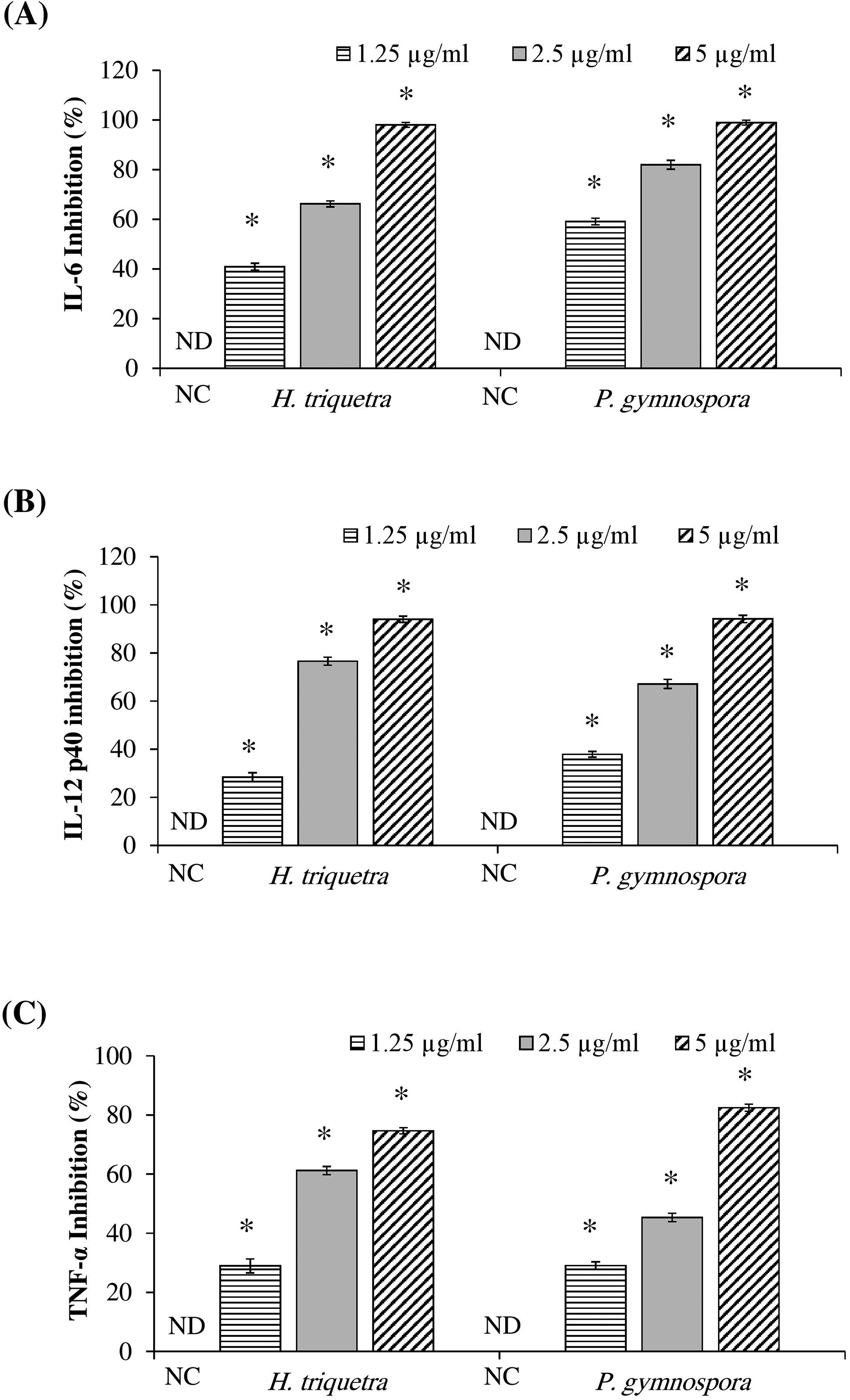
Inhibition activity (%) of the extracts against IL-6 (A), IL-12 p40 (B), and TNF-α (C) in LPS-stimulated BMDCs. The cells were pretreated with the extracts at different concentrations (μg/mL) for 1 h, followed by stimulation with LPS for 18 h. The cytokines were measured in the culture medium using ELISA. The inhibitory activity (%) of the extracts were calculated. NC; negative control (LPS only stimulated BMDCs without extract). ND; not detected. *
IC50 Values (µg/mL) of the Inhibitory Activity of Cytokine Production of the Extracts.
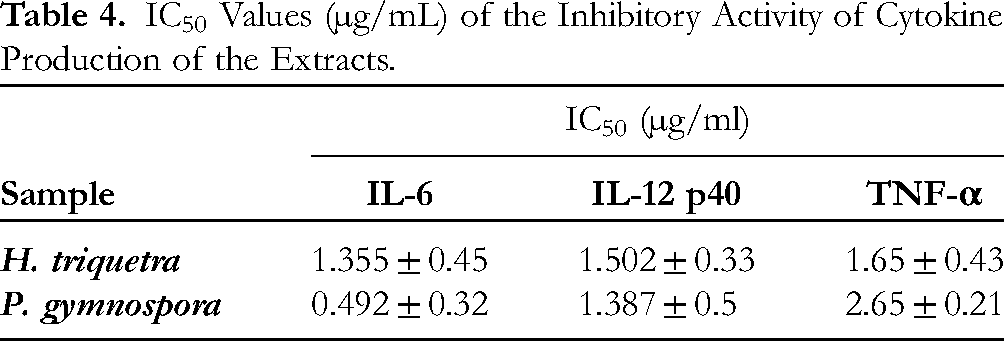
Discussion
This study introduced
To adapt and survive in the Red Sea environment, seaweeds have developed defense and competitive strategies that have resulted in the synthesis of significant bioproducts. In the current study, the analysis of the extracts showed valuable bioactive compounds. One of the most important components in the extracts that was detected is the phenolic content. Phenols are among the most important bioactive compounds produced by seaweed. The key role of phenolic compounds in seaweeds is to strengthen their defense mechanisms against oxidative stress. 20
The HPLC analysis of the phenolic content for the two extracts detected the presence of 19 main compounds. The detected compounds belong to phenolics, flavonoids, carotenoids, and monoterpenes. These compounds have been reported to have a broad range of bioactivities, including antioxidant, anti-inflammatory, antiviral, antimicrobial, anticancer, antiallergic, hepatoprotective, and antidiabetic.20,21 The multifunctional antioxidant effects of phenolic compounds are significantly associated with the phenolic rings that act as electron traps to scavenge free radicals. Flavonoids, carotenoids, and terpenoids have received considerable attention because of their potential antiviral, antiallergic, antiplatelet, anti-inflammatory, anticancer, and antioxidant effects. 21
Moreover, the GC-MS analysis of the two extracts noticed the presence of valuable fatty acids and their derivatives along with diterpenes and steroidal molecules. The fatty acid composition was found to be more diverse than that reported by. 9 The differences in chemical composition could be due to several factors, including the sampling site, season, temperature, current, and biotic conditions. The solvent used and the extraction procedure also affect the chemical composition profile and concentration of the compounds. 22
In the food and pharmaceutical industries, cytotoxicity assays are widely used on normal cells in order to screen the constituents for unwanted cytotoxic effects before investing in their development. 23 The absence of cytotoxic effect of the two extracts against the tested normal Vero cells suggested that the extracts are safe for further bioactivity exploration.
Free radicals are produced in human cells via endogenous processes and cause extensive oxidative damage. Much attention has recently been focused on the participation of active oxygen and free radicals in aging and diseases, such as cardiac disease, inflammation, arthritis, immune system damages, diabetes, and cancer. 24 Marine algae are usually exposed to different combinations of light intensities and oxygen concentrations that promote the development of free radicals and other oxidizing agents 25 ; however, the absence of oxidative damage in the structural components of seaweeds and their resistance to oxidation during storage suggest that their cells have antioxidant defense systems. 26 According to the present results, the significant increase in cell viability % in extract-pretreated Vero cells exposed to oxidative stress demonstrates the protective effects of the two seaweed extracts. The dose-dependent increase in Vero cell viability % highlights the potential of these extracts in mitigating oxidative damage, which is implicated in various diseases. This finding aligns with the previous studies that reported antioxidant activity based on DPPH-scavenging ability.11,27
NO is a gaseous signaling mediator and regulator of inflammatory responses. The murine macrophage RAW264.7 cell line is an excellent model for evaluating the anti-inflammatory activity of natural products and screening their inhibitory activity on NO production. 28 In the current study, the dose-dependent reduction in NO production in LPS-stimulated RAW264.7 cells indicates the anti-inflammatory potential of the two seaweed extracts.
For more confirmation of the anti-inflammatory activity, the inhibition of cytokines production in BMDCs model was screened. BMDCs, which are a part of the immune cells, play a crucial role in regulating immune responses. The cells are involved in the production of several cytokines, including interleukin 6 (IL-6), interleukin 12 p40 (IL-12 p40), and tumor necrosis factor alpha (TNF-α). These cytokines act as pro-inflammatory mediators that regulate immune response and inflammation process. 29 Prolonged inflammation and uncontrolled cytokine production can cause chronic inflammation, leading to several health complications. 30 In the current study, the significant inhibition of cytokines production in LPS-stimulated BMDCs supports the role of these extracts in suppressing pro-inflammatory mediators, which is crucial in preventing chronic inflammation and related health complications. The presence of phenols, flavonoids, carotenoids, and fatty acids in the two extracts likely contributes to their anti-inflammatory activity, as observed in previous studies on brown seaweeds.31,32
While the current study shows the ability of
Conclusion
Our study indicated that the seaweeds of Red Sea can be a valuable source of bioactive compounds which exhibit antioxidant and anti-inflammatory activities. This study is one of the first reports indicating the antioxidant and anti-inflammatory effects of
Footnotes
Author Contributions
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by Brain Pool program funded by the Ministry of Science and ICT through the National Research Foundation of Korea (RS-2023-00221435) and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (RS-2023-00270936)
.
Ethical Approval
This article does not contain any studies with human or animal subjects.
Informed Consent
This article does not contain any studies with human or animal subjects.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
