Abstract
Objective
This study aimed to investigate the interventional effects and potential underlying mechanisms of Xuanfei Pingchuan Decoction on ovalbumin (OVA)-induced allergic asthma (AA) in mice.
Methods
Liquid chromatography-mass spectrometry/mass spectrometry (LC-MS/MS) was used to analyze and identify the constituents of Xuanfei Pingchuan Decoction. An allergic asthma (AA) mouse model was induced by using OVA combined with aluminum hydroxide. The resistance index (RI) in the airway was analyzed with an AniRes2005 animal pulmonary function analyzer. H&E staining was performed to evaluate pathological changes in the lung tissue. Apoptotic cells in lung tissue were assessed via TUNEL staining. The percentage of eosinophils (EOS) in bronchoalveolar lavage fluid (BALF) and the ratio of Th17 to Treg cells in the splenic tissue and peripheral blood were determined by flow cytometry. The levels of IL-6, IL-17A, and IL-10 in the BALF were determined by ELISA. The mRNA expression of RORγt and Foxp3 in lung tissue was measured with RT‒qPCR. Biochemical kits were used to measure the levels of total IgE, sIgE (specific IgE), and IgG1 in the serum.
Results
A total of 340 active constituents were identified from Xuanfei Pingchuan Decoction by LC-MS/MS analysis. Xuanfei Pingchuan Decoction reduced the RI; alleviated histopathological damage; decreased the apoptosis rate; reduced the proportion of EOS; down-regulated the mRNA levels of IL-6, IL-17A, and RORγt; up-regulated the mRNA levels of IL-10 and Foxp3; reduced the levels of IgE, sIgE, and IgG1; and regulated the Th17/Treg balance in AA mice.
Conclusions
Xuanfei Pingchuan Decoction improved airway inflammation in OVA-induced AA mice, and the underlying mechanism may be related to the regulation of Th17/Treg immune imbalance.
Introduction
Allergic asthma (AA) is a chronic inflammatory disease of the airways that is characterized by the infiltration of various inflammatory cell types, such as eosinophils (EOS), mast cells, lymphocytes, and neutrophils.1,2 The interaction of these cells and the inflammatory mediators they release leads to airway hyperresponsiveness (AHR), which triggers recurrent episodes of wheezing, shortness of breath, chest tightness, or coughing. Approximately 300 million people worldwide currently suffer from asthma,3,4 and its morbidity and mortality rates have been increasing for the past 20 years. 5 Children are at high risk for AA, which can cause recurrent episodes of wheezing and coughing that can extend into adulthood with improper intervention. 6 However, there are currently no specific drugs available for the treatment of asthma. Corticosteroids, antihistamines, and leukotriene receptor antagonist have been used clinically for asthma treatment, but their prolonged use can cause significant adverse reactions.7–10 It found that after 18 months of treatment with a high dose (770 μg daily) of budesonide, 9% had experienced more hyperactivity and aggressiveness, suggesting that inhalation corticosteroid treatment can impact children's behavior. 11 Data suggest that montelukast is effective in the treatment of childhood asthma. Still, associated neuropsychiatric adverse effects are also prevalent, such as sleep disturbances, agitation, pain, and hyperactivity. 12 Ketotifen showed limited improvement of lung function in children with cough variant asthma, and the recurrence rate of the disease is high after Ketotifen treatment. 13 Therefore, there is an urgent need to develop alternative drugs that can be used to control or treat AA effectively. The pathogenesis of AA is extremely complex and mainly involves airway inflammation, immune deficiency, airway remodeling, and AHR, but its pathogenesis is still not fully understood. 14
Given its diverse pathogenic mechanisms, traditional Chinese medicine presents potential advantages in the prevention and treatment of AA due to its “multicomponent and multitarget” nature.15,16 Studies have demonstrated that Yanghe Pingchuan Granule improves airway inflammation by inhibiting autophagy via miRNA328-3p/high mobility group box 1 (HMGB1)/mediated Toll-like receptor 4 (TLR4)/nuclear factor κB (NF-κB) pathways in rat models of asthma. 17 Ding Chuan Decoction (DCD) can alleviate OVA-induced allergic asthma and DCD may have a therapeutic effect by regulating intestinal flora and polyamine metabolism through its effects on aldehyde dehydrogenase 2 (ALDH2) and amine oxidase copper-containing 3 (AOC3). 18 Xuanfei Pingchuan Decoction, from “Shesheng Zhongmiao Formula”, is a traditional Chinese medicine preparation consisting of 14 Chinese herbs (Ma Huang, Xing Ren, Kuan Donghua, Huang Qin, Sheng Shigao, Ting Lizi, Sang Baipi, Su Zi, Di Long, Zhi Qiao, Gua Loupi, Fu Ling, Zhe Beimu, Gan Cao) that protects the lung, dispels phlegm, calms asthma and lowers the pressure in the lung. It is used clinically to treat bronchial asthma with remarkable efficacy. AA is also a type of bronchial asthma, but the mechanism by which Xuanfei Pingchuan Decoction helps treat AA has not been reported. Various studies have confirmed that abnormal immune mechanisms are closely related to the development of AA. For a long time, the main immune mechanism for the development of asthma is the Th1/Th2 imbalance. 19 However, In recent years, an increasing body of research has demonstrated that Th17/Treg immune imbalance also plays an important role in the pathogenesis of AA.20–22 Th17 cells mainly secrete cytokines such as IL-17, which are involved in inflammatory reactions and immune responses. Retinoic acid receptor-related orphan receptor gamma t (RORγt) is a specific cellular transcription factor for Th17 cells and promotes the expansion of Th17 cells. The simultaneous action of IL-6 and TGF-β activates the signal transducer and activator of transcription 3 (STAT3), which in turn induces the expression of RORγt and ultimately promotes the differentiation of CD4+ T cells into Th17 cells. Treg cells mainly play an immunosuppressive role and can inhibit excessive immune responses and inflammatory responses. Treg cells and their secretion of a variety of immune regulatory factors, such as TGF-β, IL-10, IL-35, etc, inhibit CD4+ T cell activation and proliferation and play an important role in inducing the direction of their differentiation. IL-10 is the most powerful immune regulatory factor among them, which can act together with TGF-β, and thus contribute to the continuous differentiation of initial CD4+ T cells towards Treg cells. Forkhead box protein P3 (Foxp3) is a signature molecule of Treg cells. Studies have shown that Foxp3 can block the expression of inflammatory factors, ultimately prompting the differentiation of initial CD4+ T cells to Treg cells and inhibiting the differentiation of Th17 cells. 23 This process maintains the inflammatory response within a controlled range and reduces the degree of damage to the organism. Therefore, by intervening in the Th17/Treg balance, immune diseases such as asthma can be effectively treated. However, it is not clear whether Xuanfei Pingchuan Decoction can treat AA by regulating Th17/Treg balance.
In this study, we established a mouse model of AA via ovalbumin (OVA) treatment to investigate the intervention effects of Xuanfei Pingchuan Decoction on AA mice and its impact on Th17/Treg immune imbalance and to provide new ideas for the potential application of Xuanfei Pingchuan Decoction as a therapeutic drug for AA.
Materials and Methods
Reagents
The ingredients in Xuanfei Pingchuan Decoction are as follows: Ephedra Herb, 5 g; Prunus Dulcis, 5 g; Farfarae Flos, 6 g; Scutellaria Baicalensis Georgi, 6 g; Gypsum Fibrosum, 15 g; Semen Descurainiae, 6 g; Cortex Mori Radicis, 10 g; Perillae Fructus, 10 g; Pheretima, 6 g; Fructus Aurantii, 6 g; Trichosanthis Pericarpium, 6 g; Poria Cocos, 9 g; Fritillariae Thunbergii Bulbus, 6 g; and Glycyrrhizae Radixet Rhizoma, 3 g. The clinical dose of 99 g of raw herbs/70 kg of body weight is 1.41 g/kg. Based on the conversion factor (×9.1), a medium dose of Xuanfei Pingchuan Decoction is 12.83 g/kg.
Liquid Chromatography-Mass Spectrometry/Mass Spectrometry (LC-MS/MS)
The chemical composition of Xuanfei Pingchuan Decoction was analyzed and identified by LC-MS/MS. In this experiment, a U3000 high-performance liquid chromatograph (Thermo Fisher Scientific, USA) coupled with a Q Exactive mass spectrometer (Thermo Fisher Scientific, USA) was used for the separation and identification of the constituents contained in Xuanfei Pingchuan Decoction. Xuanfei Pingchuan Decoction (2 mL) was placed in a 3 kD ultrafiltration tube and centrifuged at 12,000 rcf for 10 min at 4 °C, and the supernatant was analyzed by LC-MS.
Chromatographic conditions: Waters HSS T3 column (2.1 mm × 100 mm, 1.7 μm), mobile phase A was 0.1% formic acid, and mobile phase B was 100% methanol. The gradient elution conditions were as follows: 0-1 min, 2% B; 1-5.5 min, 2%-100% B; 5.5-14 min, 100% B; 14-14.1 min, 100%-2% B; and 14.1-16 min, 2% B. The column temperature was set at 40 °C, the injection volume was set at 2 μm, and the flow rate was set at 0.3 mL/min.
Mass spectrometry conditions: electrospray ion source (ESI), positive and negative ion mode data acquisition. Data were processed using MS-DIAL 4.70 software (RIKEN Tokyo, Japan).
Animals
Thirty-six female BALB/c mice (SPF grade, age 8-12 weeks, body weight 18-22 g) were purchased from Chengdu Dashuo Biological Co. The mice were housed in an environment with a temperature of 20-25 °C, relative humidity of 50%-65%, and a 12 h light and dark cycle, and had free access to food and drinking water. This study was approved by the Ethics Committee of Zunyi Medical And Pharmaceutical College (ZMPC-2301-001). The study adhered to the ARRIVE guidelines.
Allergic Asthma (AA) Model
After one week of adaptive feeding, the mice were randomly divided into 6 groups, each with 6 mice: the control group, AA model group (OVA), budesonide group (BUDE), Xuanfei Pingchuan Decoction low-dose group (XFPC-L), Xuanfei Pingchuan Decoction medium-dose group (XFPC-M), and Xuanfei Pingchuan Decoction high-dose group (XFPC-H). The AA model was established via a sensitization period and an excitation period. Mice were intraperitoneally injected with 200 μL of sensitizing solution (100 μg of OVA + 200 μg of aluminum hydroxide dry powder +200 μL of PBS) on the first day and the fourteenth day. On the 21st to 27th days, 5 mL of 1% OVA solution was nebulized continuously for 30 min each time, once a day for 7 d.
Grouping and Drug Administration
The drug was administered by gavage 30 min before each nebulization treatment. The XFPC-L, XFPC-M, and XFPC-H groups were given Xuanfei Pingchuan Decoction at concentrations of 6.42 g/kg, 12.83 g/kg, and 25.66 g/kg, respectively, by gavage; the BUDE group was treated with 5 ml of budesonide suspension for 30 min. The control group and model (OVA) group were given 10 ml/kg of saline by gavage. After the last intervention, the mice were fasted for 24 h, and bronchoalveolar lavage fluid (BALF) was collected.
Lung Function
The resistance index (RI) in the airway was determined using an AniRes 2005 animal lung function analyzer (Bestlab, Beijing, China). Mice were anesthetized with 1% sodium pentobarbital (50 mg/kg) by intraperitoneal injection and immobilized, the skin on the neck was cut, and the trachea was exposed. Mice were extubated and connected to a mouse ventilator (MiniVent Ventilator, Harvard Apparatus). The respiratory frequency was set at 90 breaths/min with a tidal volume of 5 mL/kg, the baseline was adjusted with PBS for the first time, and the basal value of airway resistance was recorded for 1 min; subsequently, atomized acetylcholine (ACh) was applied at mass concentrations of 0 µg/kg, 0.25 μg/kg, 0.5 μg/kg, and 1 μg/kg (1 min each); the RI (cm H2O·s·mL) was recorded after 3 min.
H&E Staining
Lung tissues were routinely dehydrated, embedded in paraffin, sectioned at a thickness of 5 μm, and stained with H&E. Finally, the sections were placed under a microscope to observe histopathological changes in the lung tissue.
TUNEL Staining
Lung tissue sections were dewaxed, dehydrated, and incubated with TUNEL stain (Elabscience, E-CK-A320) at room temperature for 1 h. The sections were then sealed with an anti-fluorescent bursting agent, and the apoptotic cells were observed under a confocal microscope. The percentage of apoptotic cells was quantified by using Image-Pro Plus software.
Flow Cytometry
The percentage of EOS in bronchoalveolar lavage fluid (BALF) and the ratio of Th17 to Treg cells in the splenic tissue and peripheral vein blood were determined by flow cytometry.
BALF (0.7 mL) was collected and centrifuged at 3000 r/min for 10 min. The supernatant was discarded, 100 μL of the antibody mixture (0.5 μL of the CCR3 antibody, 0.5 μL of the CD170 antibody, and 0.5 μL of the CD11c antibody) was added, and the mixture was incubated at 4 °C for 30 min. Then, 1 mL of PBS-2% FBS was added to terminate the reaction, the mixture was centrifuged, the supernatant was discarded, and 500 μL of PBS-2% FBS was added to resuspend the cells. The percentage of EOS was determined on a flow cytometer.
Splenic tissue was ground and digested to obtain a single-cell suspension and then incubated with cell stimulation in a CO2 incubator at 37 °C for 4 h. After stimulation, staining was performed using the corresponding antibody markers, and after rupture of the membrane and nucleus, intranuclear staining was continued by adding 280 µL of PBS for resuspension and detection on the machine. The percentages of Th17 and Treg cells and the Th17/Treg ratio were determined using FlowJo software.
Peripheral vein blood (3 mL) was collected upon fasting and centrifuged at 3000 r/min for 30 min, after which the plasma was collected. Peripheral blood mononuclear cells (PBMCs) were isolated by the Ficoll density gradient centrifugation method. Flow cytometry was performed to detect Th17 cells (the CD4 antibody and IL-17A antibody were used as markers) and Treg cells (the CD4 antibody, CD25 antibody, and Foxp3 antibody were used as markers), and the Th17/Treg ratio was calculated.
ELISA
The levels of IL-6 (ML Bio, ml063159), IL-17A (ML Bio, ml037864), and IL-10 in BALF (ML Bio, ml037888) were measured by ELISA. The experimental procedure was carried out in strict accordance with the instructions provided in the kits. Absorbance was read at 450 nm, the standard curve was plotted, and the concentrations of IL-6, IL-17A, and IL-10 in the BALF were calculated.
RT–qPCR
Total RNA was extracted from 50 mg of lung tissue by adding 1 mL of TRIzol, and the RNA concentration was measured with a UV spectrophotometer. The RNA was reverse transcribed into cDNA with a TaKaRa kit, and the mRNA levels of RORγt and Foxp3 were measured via PCR according to the instructions of the SYBR fluorescence real-time quantitative kit. The amplification and lysis curves were plotted, and the results were analyzed by 2−△△Ct values. β-actin was used as the internal control. The primer sequences were as follows: RORγt-Forward Primer: 5′-CCTGGATGTGTAAAGAATG-3′, RORγt-Reverse Primer: 5′-TAGACTGCTAACGAATCTG-3′, 150 bp; Foxp3-Forward Primer: 5′-ACCTATGCCACCCTTATCC-3′, Foxp3-Reverse Primer: 5′-GCTCCTCTTCTTGCGAAAC-3′, 234 bp; β-actin-Forward Primer: 5′-AGGTCGGAGTCAACGGATTTG-3′; and β-actin-Reverse Primer: 5′-TGTAAACCATGTAGTTGAGGTCA-3′, 197 bp.
Biochemical Indices
The levels of total IgE (Shanghai Xinyu Biotechnology Co., Ltd, XYEA545Po), specific IgE (s-IgE) (Shanghai Huabang Biotechnology Co., Ltd, HBP31231R), and IgG1 (Shanghai Chuntest Biotechnology Co., Ltd, CS10843) in the serum were measured in strict accordance with the instructions provided in the kits.
Statistical Analysis
The experimental data are expressed as the mean ± standard deviation (SD), and SPSS 20.0 software was used to statistically analyze all the data. One-way ANOVA was used for comparisons between multiple groups, and P < .05 indicated a significant difference.
Results
LC-MS/MS Analysis of Xuanfei Pingchuan Decoction
A total of 340 active constituents were identified from Xuanfei Pingchuan Decoction by LC-MS/MS analysis (Figure 1). The representative composition was Lopinavir, (2S,3R,4R,5R,6S)-2-[(4-hydroxy-6-{[(4S,6R,7S,8R,9S,13S,16S)-6-hydroxy-7,9,13-trimethyl-6-[(3R)-3-methyl-4-{[(2R,3R,4S,5S,6R)-34,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}butyl]-5-oxapentacyclo[10.8.0.0,.0,.0,]icosan-16-yl]oxy}-2-(hydroxymethyl)-5-{[34,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxan-3-yl)oxy]-6-methyloxane-34,5-triol, 3_,5_,15_-O-triacetoxy-7_-14_-O-dibenzoyloxymyrsinol, Lycopene, OXONITINE, and Leucinostatin_B2 observed in leucinostatin A/B standard dereplicator annotation. The chemical structures of the major or key molecules present in Xuanfei Pingchuan Decoction are availablefor viewing and for download in the Supplemental Material.
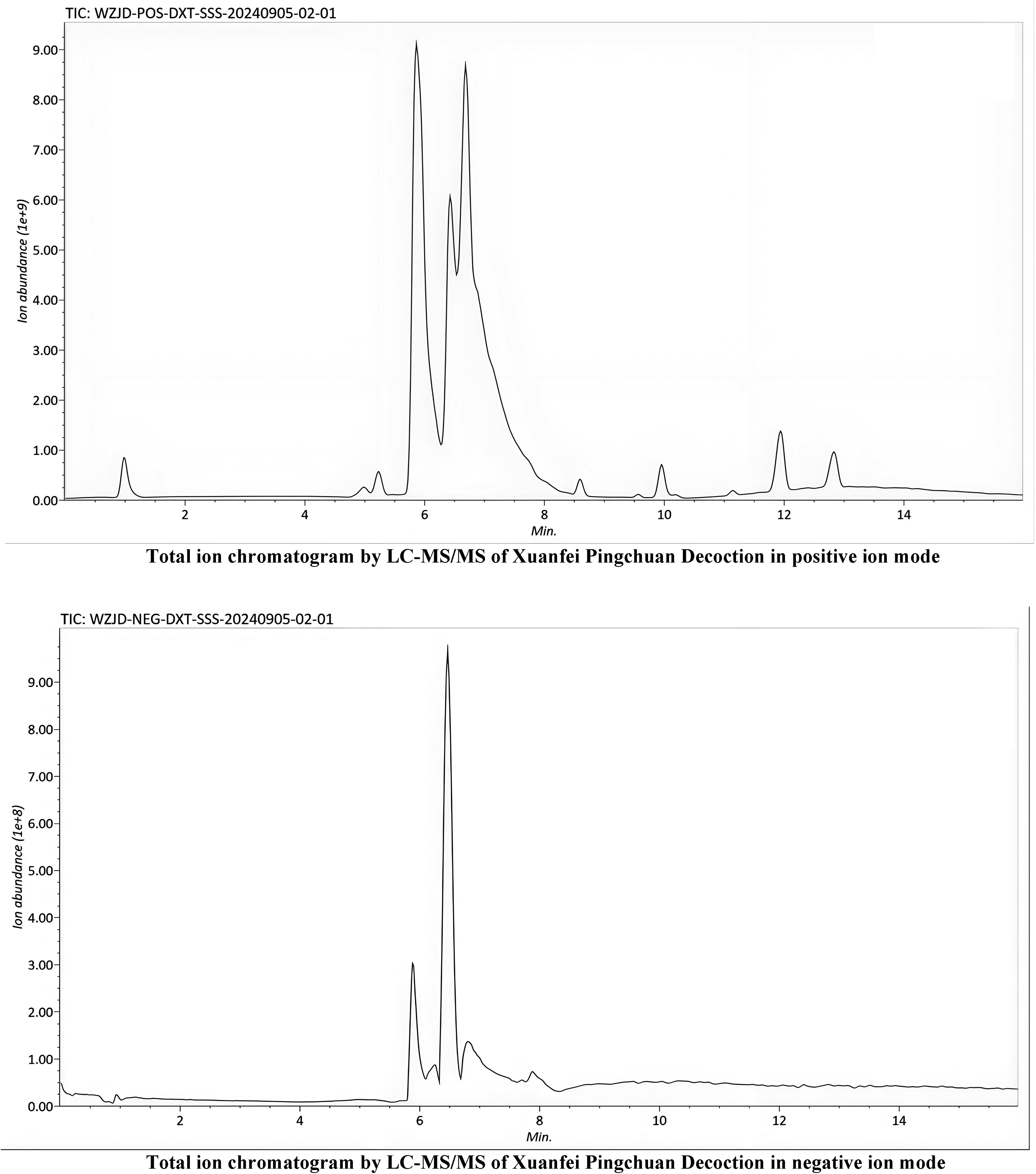
LC-MS/MS analysis of Xuanfei Pingchuan Decoction. Total ion chromatogram in positive and negative ion modes of Xuanfei Pingchuan Decoction.
Effect of Xuanfei Pingchuan Decoction on Lung Function and Pathological Changes of Lung Tissues in AA Mice
Changes in airway responsiveness in each group of mice were assessed with an AniRes 2005 animal lung function analyzer. The results (Figure 2A) showed that the RI in the OVA group increased significantly with increasing ACh concentrations. Compared with that in the control group, the RI in the OVA group was significantly greater when the concentration of ACh was 1.0 µg/kg. The RIs in the XFPC-H and BUDE groups decreased to some extent. H&E staining (Figure 2B) was performed to evaluate the pathological changes in the lung tissues of AA mice. No obvious pathological changes were observed in the lung tissues of the control group. In the OVA group, there was inflammatory infiltration around the lung tissue, bronchi, and blood vessels. Xuanfei Pingchuan Decoction and budesonide significantly reduced the degree of inflammation. Inflammatory infiltration causes the apoptosis of lung tissue cells, leading to structural and functional damage. TUNEL staining (Figure 2C) was used to examine the effect of Xuanfei Pingchuan Decoction on the percentage of apoptotic cells in the lung tissues of mice with AA. The results showed that the rate of apoptosis was significantly greater in the OVA group than in the control group and was decreased by Xuanfei Pingchuan Decoction and budesonide. These results showed that Xuanfei Pingchuan Decoction had a protective effect on t lung tissues from AA mice.
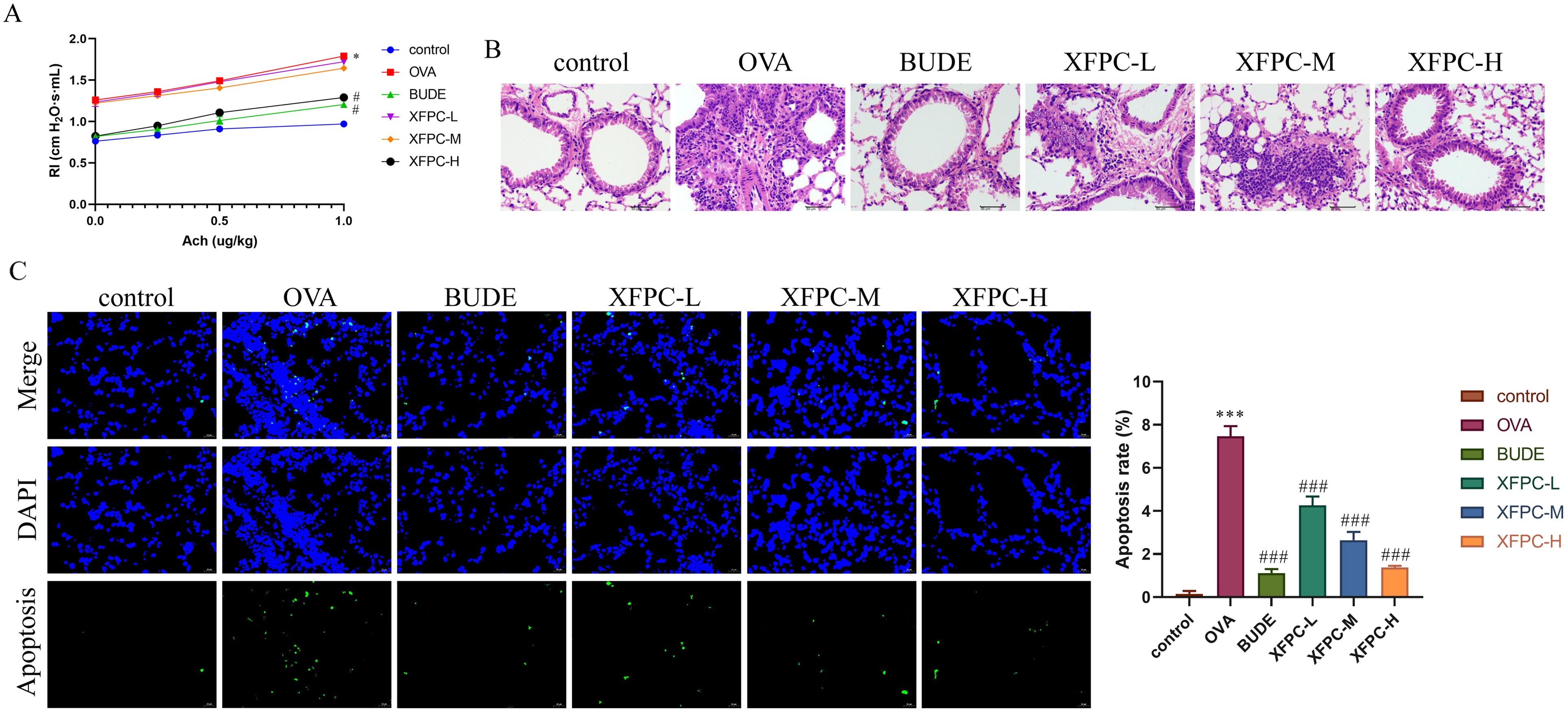
Effect of Xuanfei Pingchuan Decoction on lung function and pathological changes of lung tissues in AA mice. (A) Resistance index (RI) in the airway. (B) H&E staining was performed to detect histopathological changes in lung tissue. Original magnification: ×400. Scale bar = 50 μm. (C) TUNEL staining was performed to determine the percentage of apoptotic cells. Blue fluorescence: DAPI. Green fluorescence: apoptotic cells. Original magnification: ×400. Scale bar = 50 μm. The data are presented as the mean ± SD, n = 6 per group. *P < .05; ***P < .001 versus the control group; #P < .05; ###P < .001 versus the OVA group.
Effect of Xuanfei Pingchuan Decoction on the Percentage of EOS in the BALF of AA Mice
EOS was labeled with CCR3, CD170, and CD11c antibodies. CCR3 + CD170+ and CD11c- cell percentages were significantly greater in the OVA group than in the control group. CCR3 + CD170+ and CD11c- cell percentages were significantly decreased after Xuanfei Pingchuan Decoction and budesonide treatment (Figure 3).
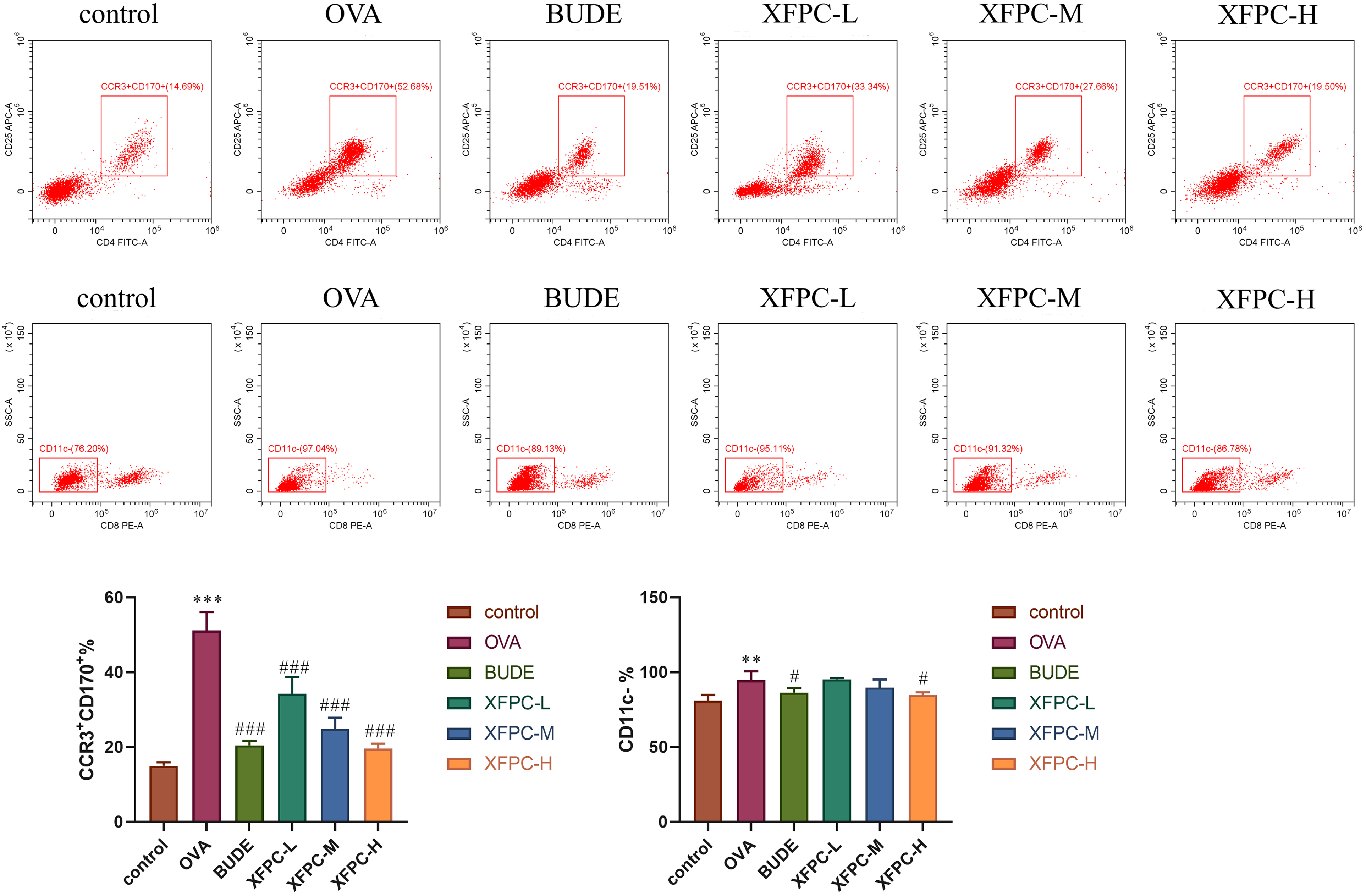
Effect of Xuanfei Pingchuan Decoction on the percentage of EOS in the BALF of AA mice. The percentage of EOS was determined by flow cytometry. The data are presented as the mean ± SD, n = 6 per group. **P < .01; ***P < .001 versus the control group; #P < .05; ###P < .001 versus the OVA group.
Effects of Xuanfei Pingchuan Decoction on Inflammatory Factors in AA Mice
Compared with those in the control group, the levels of IL-6 and IL-17A were significantly increased and the level of IL-10 was significantly reduced in the OVA group. These changes in inflammatory factors levels were reversed by Xuanfei Pingchuan Decoction and budesonide treatment (Figure 4A). RORγt mRNA was up-regulated and Foxp3 mRNA was down-regulated in the OVA group; after Xuanfei Pingchuan Decoction and budesonide treatment, RORγt mRNA was down-regulated and Foxp3 mRNA was up-regulated (Figure 4B).
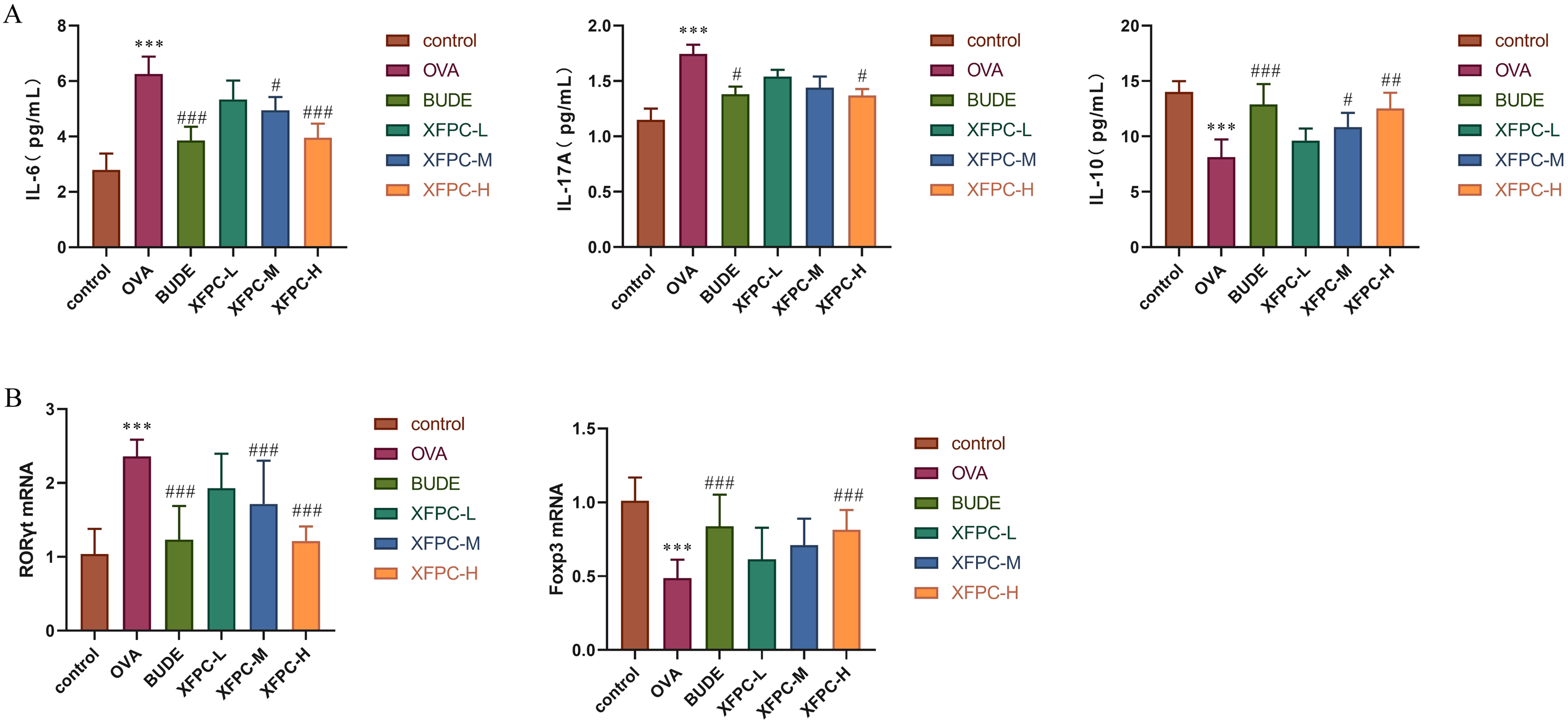
Effects of Xuanfei Pingchuan Decoction on inflammatory factors in AA mice. (A) ELISAs for IL-6, IL-17A and IL-10. (B) RT‒qPCR analysis of the mRNA levels of RORγt and Foxp3. The data are presented as the mean ± SD, n = 6 per group. ***P < .001 versus the control group; #P < .05; ##P < .01; ###P < .001 versus the OVA group.
Effects of Xuanfei Pingchuan Decoction on IgE and IgG1 in AA Mice
The results showed that the levels of IgE, s-IgE, and IgG1 were significantly greater in the OVA group than in the control group. Xuanfei Pingchuan Decoction and budesonide treatment significantly decreased the levels of IgE, s-IgE, and IgG1 (Figure 5).

Effects of Xuanfei Pingchuan Decoction on IgE and IgG1 levels in AA mice. The levels of IgE, s-IgE and IgG1 were detected using a biochemical kit. The data are presented as the mean ± SD, n = 6 per group. *P < .05 versus the control group; #P < .05 versus the OVA group.
Effect of Xuanfei Pingchuan Decoction on the Th17/Treg Balance in AA Mice
Th17/Treg immune imbalance plays an important role in the pathogenesis of asthma, and the levels of Th17 cells and Treg cells in the splenic tissue and peripheral blood of mice with AA were measured by flow cytometry. The results showed that the proportion of Th17 cells was significantly greater and the proportion of Treg cells was significantly lower in the OVA group than in the control group. Xuanfei Pingchuan Decoction and budesonide treatment significantly decreased the proportion of Th17 cells and increased the proportion of Treg cells. This result indicated that Xuanfei Pingchuan Decoction could improve the immune imbalance of Th17/Treg cells in mice with allergic asthma (Figure 6).
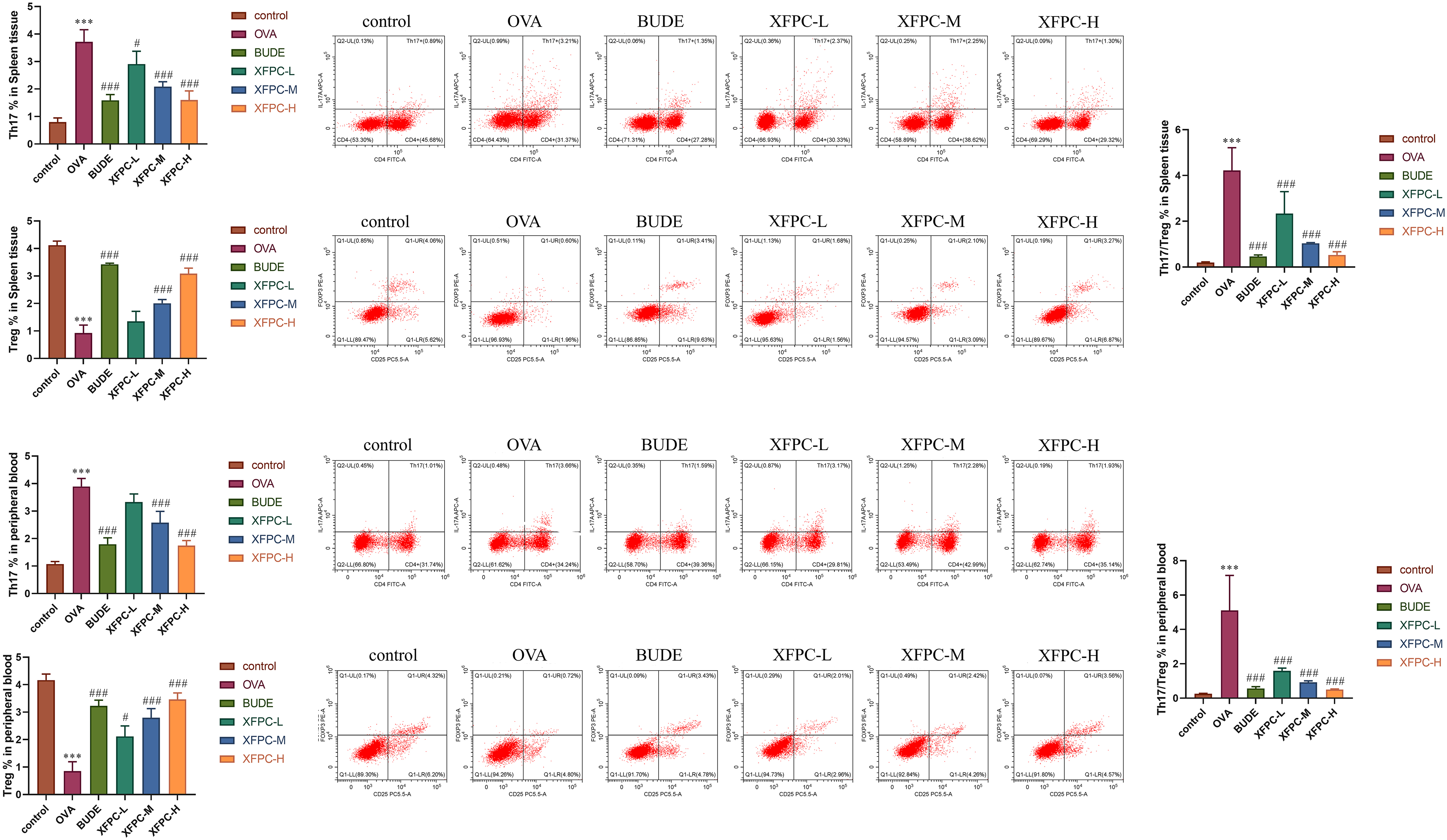
Effect of Xuanfei Pingchuan Decoction on the Th17/Treg balance in AA mice. The percentages of Th17 and Treg cells in the splenic tissue and peripheral vein blood were determined by flow cytometry. The data are presented as the mean ± SD, n = 6 per group. ***P < .001 versus the control group; #P < .05; ###P < .001 versus the OVA group.
Discussion
During the development of AA, immune function is disrupted, and a large number of inflammatory cells, cytokines, and inflammatory mediators are released outside the media, directly damaging the airway epithelium and causing increased lung airway reactivity, leading to the formation and development of chronic airway inflammation, and controlling airway inflammation can effectively treat allergic asthma.24,25 In recent years, with continuous research on TCM, its clinical use has been gradually expanded, and its application value in the treatment of asthma has been confirmed.26–28 Clinically, the identification of pediatric asthma is mostly based on febrile asthma. Chinese medicine has achieved satisfactory efficacy in the treatment of bronchial asthma in pediatrics. It revealed that modified Liu-Wei-Di-Huang-Wan could regulate the immune responses and improve pulmonary function in Der p 2-induced asthmatic mice. 29 Xuanfei Pingchuan Decoction protects the lung, calms asthma, resolves phlegm, and relieves cough, and has shown good clinical effects in the treatment of bronchial asthma. In this study, Xuanfei Pingchuan Decoction acted by improving lung function, attenuating lung histopathologic changes, and reducing airway resistance in AA mice. The formula also reduced the proportion of EOS in alveolar lavage fluid and lowered serum levels of IgE and IgG1, thereby reducing allergic reactions and inflammation. At the level of molecular mechanism, Xuanfei Pingchuan Decoction works by regulating the Th17/Treg balance. Xuanfei Pingchuan Decoction decreased the levels of pro-inflammatory factors IL-6 and IL-17A while increasing the level of anti-inflammatory factor IL-10. In addition, Xuanfei Pingchuan Decoction down-regulated the expression of RORγt and up-regulated the expression of Foxp3, thereby inhibiting the differentiation of Th17 cells, promoting the generation of Treg cells, and restoring the immune balance. This multi-target and multi-pathway mechanism of action provides a scientific basis for the treatment of asthma with Xuanlong Pingchuan Decoction and demonstrates the potential of Chinese medicine in regulating immune response and treating chronic inflammatory diseases.
Immune function is one of the most important causes of AA.30,31 IgE is mostly produced by the respiratory mucosa and has a high affinity for basophils and mast cells.32,33 Under normal physiological conditions, serum IgE levels are very low. However, in patients with allergic diseases or allergies, serum IgE levels are significantly greater and can be involved in inflammatory reactions.34,35 IgG1 is a diagnostic antigen that plays a regulatory role in the development of asthma. 36 Montelukast sodium (MKST) combined with budesonide (BUD) can ameliorate the pulmonary function of children with cough variant asthma (CVA), reduce IgE levels, and improve the curative effect and prognosis. 37 The results of this study showed that Xuanfei Pingchuan Decoction and budesonide treatment significantly decreased the levels of IgE, s-IgE, and IgG1.
In AA patients, inflammatory lesions in the airway are often associated with EOS infiltration.38,39 The results of this study showed that Xuanfei Pingchuan Decoction and budesonide treatment significantly reduced the proportion of EOS. Th17/Treg imbalance is also an important mechanism in the pathogenesis of asthma, as Th17 and Treg cells are functionally antagonistic and work together to maintain immune homeostasis. 40 IL-17A is the main effector secreted by Th17 cells and can promote the inflammatory response by promoting the recruitment of various inflammatory cells to inflammatory areas and producing various cytokines, such as IL-6.41,42 IL-6 is a key factor necessary for Th17 cell differentiation and interacts with TGF-β to promote Th17 cell differentiation and inhibit Treg cell proliferation, resulting in a Th17/Treg imbalance.43,44 It was found that Tuo-Min-Ding-Chuan Decoction increases Treg cells percentage and decreases Th17 cells percentage in the OVA-induced eosinophilic asthma mice model. 45 The results of this study showed that the levels of Th17 cells and the proinflammatory cytokines IL-6 and IL-17A were significantly greater in mice with asthma than in normal mice, while the levels of Treg cells and the anti-inflammatory cytokine IL-10 were significantly lower. The effect of Xuanfei Pingchuan Decoction on asthma may be related to the regulation of the Th17/Treg cell ratio and the expression of related cytokines in AA mice.
In conclusion, this study revealed that Xuanfei Pingchuan Decoction could regulate the levels of Th17/Treg cells and related cytokines in mice with AA, thus exerting an anti-AA effect. Its mechanism of action may be related to the regulation of Th17/Treg immune imbalance. These findings provide an experimental basis for the study of Xuanfei Pingchuan Decoction in the treatment of AA and further confirm the important role of Th17/Treg immune imbalance in the pathogenesis of AA. However, there are some limitations to this study. Although we observed that Xuanfei Pingchuan Decoction could regulate the Th17/Treg immune balance, the exploration of its specific mechanism of action and molecular pathway is not deep enough. In addition, this study was only validated in an animal model, and its effectiveness and safety in clinical application need to be further confirmed. Future studies will be devoted to in-depth exploration of the mechanism of action of Xuanfei Pingchuan Decoction in the treatment of AA, and to assess the safety of its long-term application. Moreover, clinical studies will be conducted to verify the effectiveness and applicability of Xuanfei Pingchuan Decoction in the treatment of AA.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X251316772 - Supplemental material for Xuanfei Pingchuan Decoction Attenuates Ovalbumin-Induced Allergic Asthma Mice Through Regulating Th17/Treg Immune Imbalance
Supplemental material, sj-doc-1-npx-10.1177_1934578X251316772 for Xuanfei Pingchuan Decoction Attenuates Ovalbumin-Induced Allergic Asthma Mice Through Regulating Th17/Treg Immune Imbalance by Dou Ding, Qi Zhang, Juan Gu, Mingxing Cai, Yuan Gan, Yanni Ye, Xun Yang, Xiaojun Dong, and Xiuwen Xia in Natural Product Communications
Footnotes
Acknowledgments
The technical assistance and support of Chengdu Lilai Biotechnology Co., Ltd (Sichuan, China) were appreciated.
Author Contributions
Dou Ding, Qi Zhang, and Juan Gu conceived and designed the experiments. Mingxing Cai and Yuan Gan performed the experiments. Yanni Ye and Xun Yang analyzed the data. Xiaojun Dong and Xiuwen Xia contributed reagents and materials. Dou Ding wrote the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zunyi Science and Technology Bureau (No. [2022]143) and the Guizhou Provincial Science and Technology Department Project (No. ZK[2023]587).
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of Zunyi Medical And Pharmaceutical College (ZMPC-2301-001).
Consent for Publication
Written informed consent for publication was obtained from all participants.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Zunyi Medical And Pharmaceutical College, China, and approved by the Administration Committee of Experimental Animals, Guizhou Province, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
