Abstract
Objective
To identify the blood-entering components of HanJing Decoction (HJT) after administration based on UPLC-QE-MS/MS, and the key components, therapeutic targets and mechanisms of HJT therapeutic coronary heart disease (CHD) were analyzed using network pharmacology and molecular docking.
Method
The UPLC-QE-MS/MS was used to analyze the blood-entering components of HJT before and after administration. The targets of blood-entering components were predicted by SwissTargetPrediction. Targets related to CHD were collected using multiple databases. The GO and KEGG enrichment analyses were used to predict the mechanisms of HJT therapeutic CHD, and PPI and “Components-Targets-Pathways” network were used to identify and elucidate the core targets. The key blood-entering components aimed at the core target are screened by molecular docking and QSAR analysis.
Results
A total of 14 blood-entering components were detected in serum samples of rat after administration, and the 32 potential targets of HJT therapeutic CHD were screened out. The result of PPI network showed that the core targets of HJT for the treatment of CHD include MMP1, GSK3B, EGFR and PTGS2, and the 5 key components with high degree were screened out. The GO and KEGG enrichment analyses indicate that HJT therapy for CHD is associated with the IL-17 and cGMP-PKG signaling pathways. The result of molecular docking indicate that the binding energy of coroglaucigenin to PTGS2 is the largest and it may be the key pharmacological component of HJT, and the QSAR analysis showed that Boldine and Coroglaucigenin had excellent activity in inhibiting PTGS2.
Conclusions
In this study, the blood-entering components of HJT were preliminarily identified, Combined network pharmacology and molecular docking analyses revealed that the PTGS2 may be a core target, and the IL-17 and cGMP-PKG signaling pathways may be the key pathways. Moreover, the coroglaucigenin and boldine may be the key pharmacological components of HJT.
Keywords
Introduction
Coronary heart disease (CHD for short), also known as Coronary atherosclerotic heart disease, has become the number one leading cause of mortality among chronic non-infectious diseases in the world, 1 results from cardiac insufficient blood supply and myocardial injury driven by narrow coronary arteries after atherosclerosis. 2 In recent years, the prevalence and mortality of CHD are increasing year by year, and the incidence of the population tends to be younger, which threatens human health and creates a heavy burden on the society. 3 At present, the western medicine is the major clinical treatment for CHD, but clinical research has found that the therapeutic effect of simple western medicine is not ideal, such as the symptoms of angina occur repeatedly, the total efficacy is not high, and the high incidence of adverse drug reactions.4,5 Therefore, it is urgently necessary to find safe and effective drugs for the treatment of CHD.
It is well known that traditional Chinese medicine (TCM) formulae often used to prevent and treat chronic and complex diseases owing to its multi-component, multi-target, multi-pathway, and low side effect characteristics, which are attributed to their combination of different herbs according to specific compatibility mechanism. 6 With the increasing development of TCM, many studies have confirmed that TCM therapy for CHD can significantly improve the efficacy and reduce the incidence of adverse reactions.7,8 In the view of TCM, CHD belongs to the category of “chest arthralgia”, and its main etiology are blood stasis and obstruction of the heart channel. The CHD is also related to spleen and kidney, and the major symptoms were congenital deficiency and severe external evil intrusion. 9 Thus, clinical treatment of CHD should focus on tonifying heart Yin and eliminating blood stasis.
Hanjing Decoction (HJT for short) is a TCM formula originates from Li Shimao, a national master of Chinese medicine. 10 This decoction comprises nine herbal medicines, ie, Ephedrae Herba (Mahuang), Cinnamomi cassia (Guizhi), Zingiberis Rhizoma Recens (Shenjiang), Jujubae Fructus (Dazao), Honey-Roasted Glycyrrhizae Radix Et Rhizoma (Zhigancao), Aconiti Lateralis Radix Praeparata (fuzi), Asari Radix Et Rhizoma (Xixin), Scorpio (Quanxie), and Scolopendra (Wugong). HJT is derived from a combination of Guizhi Qu Shaoyao Decoction, Mahuang fuzi Xixin Decoction, and Zhijing powder. The Mahuang, Xixin, Guizhi, Shenjiang and fuzi were used to activate blood circulation and disperse cold, the Dazao and Zhigancao to vigorate Qi and replenish the blood, the Quanxie and Wugong to activate blood circulation, remove blood stasis, and dredge meridians. Therefore, HJT can replenishes and activates blood, warming yang for dispelling cold, and resolves blood stasis systematically. 11 Recently, HJT has been employed to prevent and treat CHD in our hospital, and has achieved good efficacy. However, its key components and action mechanisms were still unclear.
According to the statement of serum pharmacochemistry, the entering of drug components into the blood and maintaining a certain exposure concentration is essential for drug to have a therapeutic effect, in which these components may be crucial ingredients. 12 The multiple-components, multi-targets and multi-pathways network was beneficial to elucidate the mechanisms of TCM. Thus, many studies demonstrated that network pharmacology is a useful tool to discover the potential mechanisms of TCM. 13 Hence, this study identified the blood-entering components of HJT using UPLC–QE–MS/MS method, and network pharmacology analysis was conducted for these components to find the core targets, key components, and potential mechanisms. Our study provides the theoretical basis for the clinical application of HJT therapy for CHD.
Materials and Methods
Reagent and Instrumentation
The palmatine hydrochloride (Lot: 18082103) and coptisine hydrochloride (Lot: 19042602) were supplied by Qiming Bio Co. Ltd (Shanghai, China). The acetonitrile, methanol, and formic acid (Mass spectrometry) were obtained from Fisher Scientific (Fair Lawn, USA). A Benchtop High-Speed Freezing Centrifuge (TGL-16MS) was obtained from Shanghai Lu Xiangyi Centrifuge Instrument Co. (Shanghai, China), a Shanghai Khannuo Instrument Co. (Shanghai, China) provided the TYXH-I Vortex Oscillator, a Shenzhen Fuyang Technology Group Co. (Shanghai, China) provided the F-060SD Ultrasonic Cleaning Machine, a Waters (USA) provided the ACQUITY UPLCI-Class Plus and an ACQUITY UPLC PDA detector and an ACQUITY UPLC HSST3 (100 mm × 2.1 mm, 1.8 µm) column.
Herbs Preparation
Ephedrae Herba (12 g, Mahuang, Lot: 201202), Cinnamomi cassia (18 g, Guizhi, Lot: 230201), Zingiberis Rhizoma Recens (18 g, Shenjiang, Lot: 23032401), Jujubae Fructus (18 g, Dazao, Lot: 22101201), Honey-Roasted Glycyrrhizae Radix Et Rhizoma(12 g, Zhigancao, Lot: 22070901), Aconiti Lateralis Radix Praeparata (18 g, fuzi, Lot: 22060601), Asari Radix Et Rhizoma(6 g, Xixin, Lot: 21012601), Scorpio (10 g, Quanxie, Lot: 230202), and Scolopendra (10 g, Wugong, Lot: 211001) were supplied by National Medical Hall of Hebei University of Traditional Chinese Medicine. The fuzi was decocted for an hour, then the all the other herbs are added together and decocted for an hour again, the supernatant of HJT was collected and concentrated to concentration of 2 g/mL equivalent to the raw drug concentration (Refer to commonly used clinical dose), which means that each milliliter solution equals to 2 g raw drug of HJT. 14 The HJT solution was filtered by 0.22 μm microporous filter membrane after centrifugation at 4500*g for 10 min ie to get HJT drug solution.
Animals Treatment, Dosing and serum Sample Collection
The animal study was approved by the Experimental Animal Ethical Committee of Hebei University of Chinese Medicine (approval no. DWLL202308001). A total of male 12 Sprague-Dawley rats (SPF grade, weighted 210-240 g) were provided by the Experimental Animal Facilities of Hebei Medical University. The rats were housed in environment with constant temperature (23 ± 2) °C, humidity (60 ± 5) % and a 12 h dark/light cycle, and diet and water were not restricted. Then, Referring to previous studies, 15 the CHD rat model was prepared and all rats were randomly divided into two groups, ie, Control group and HJT-treated group. Before experiment, all rats were fasted with free access to water for 12 h, the rats of HJT-treated group were orally administered 10 mL/kg HJT drug solution and the rats of Control group were treated with PBS instead of HJT drug solution. The rats were dosed for three days continuously, and the plasma was collected from the rats at 0.5, 1 and 2 h using anticoagulant tube after the last dose. 16 The plasma was centrifuged at 4500*g for 10 min 4 °C to obtain the drug-contained serum, which combined and stored at −80 °C.
Blood Sample Pretreatment
The pretreatment was performed for the plasma sample by protein precipitation. 17 Briefly, 150 μL serum sample was transferred into 1.5 mL EP tube and 450 μL methanol-acetonitrile mixture (v/v, 2:1) was added and vortex-mixed for 1 min. Then, the serum sample was centrifuged at 17500*g for 10 min at 4 °C, and the 100 μL supernatants were collected and the 5 μL supernatants were injected into the UPLC–QE–MS/MS system for analysis.
LC-MS Conditions
The chromatographic conditions were as follows: The chromatographic column was a ACQUITY UPLC I-Class plus (100 mm × 2.1 mm, 1.8 μm); the column temperature was 45°C; the mobile phase included 0.1% formic acid aqueous solution (A)–acetonitrile (B); the flow rate was 0.35 ml/min; and the injection volume was 5 μl.
The mass spectrometry conditions used were as follows: ESI ion source, the source temperature was 105 °C, Sample mass spectrometry signal acquisition was performed in positive and negative ion scanning modes. The data acquisition mode was DDA. The scanning mode used was Full MS/dd-MS2. The mass spectrometer are set follows: capillary voltage 4000 V, sheath gas flow 12 L/min, temperature 400 °C, dry gas (nitrogen) flow 10 L/min and temperature 350 °C, nebulizer pressure 45 psi.
Identification the Blood-Entering Components of HJT
The original LC‒MS data were processed by Progenesis QI V2.3 software (Newcastle, UK) for baseline filtering, peak identification, integration, retention time correction, peak alignment, and normalization. The main parameters were 5 ppm precursor tolerance and 10 ppm product tolerance. Compound identification was based on the mass‒charge ratio (M/Z), secondary fragments, and isotopic distribution using the LuMet-TCM and Herb_DB databases. The samples from the administered group and the blank group were compared and analyzed to determine the difference in mass spectral peaks between the 2 groups and combined with the retention time, charged ion information and ion fragmentation for in vivo identification of the blood-entering components of HJT.
Network Pharmacology Analysis
Blood-Entering Components of HJT Collection and Target Prediction
The chemical structures of the blood-entering components of HJT were downloaded from the PubChem database. As previously reported, 18 all blood-entering components predicted by the SwissADME database for drug-like properties. Then, the SwissTargetPrediction database (http://www.swisstargetprediction.ch/) was employed to screen the targets of blood-entering components, which were summarized and duplicates were removed to obtain targets of HJT.
Targets of CHD-Related Collection and Potential Target Screening
The keyword “Coronary Heart Disease” was used to collect targets of CHD-related by searching various databases, including the DrugBank 19 (https://go.drugbank.com/), OMIM 20 (http://www.omim.org), DisGeNET 21 (https://www.disgenet.org/home/), and GeneCard database 22 (https://www.genecards. org/), and all targets CHD-related were summarized and delete duplicates. Lastly, the Venn analysis was used to screen out the potential targets of HJT therapy for CHD, and the results were visualized using the Venndiagram package.
2.7.3 Protein-Protein Interaction (PPI) Network Construction
The potential targets of HJT therapy for CHD were imported into the STRING database (https://stringdb.org/) to analyze and obtain the PPI information, the confidence level was 0.4 and species was “Homo”. 23 The PPI information was imported into Cytoscape 3.9.1 software to construct PPI network, and the PPI network was topologically analyzed to identify the core targets of HJT therapy for CHD using cytoHubba plug-in. The results are displayed sorted by degree value.
2.7.4 Gene Ontology (GO) and KEGG Enrichment Analysis of Potential Targets
The GO and KEGG enrichment analyses can help to elucidate the potential biological function and mechanisms of drug therapy for disease. The potential targets were inputted into Metascape database 24 (http://metascape.org/) for GO (Includes Biological processes, Cellular components, and Molecular functions) and KEGG enrichment analysis, the reference values were set to P-value < 0.05 and Homo sapiens as the species. The top 10 GO term and top 20 KEGG pathways were visualized with the Bioinformatics online tool (https://www.bioinformatics.com.cn).
2.7.4 the “Component-Target-Pathway” Network Construction
Organizing the blood-entering components, potential targets and pathways interrelationships and importing them into the Cytoscape 3.9.1 software to create “Component-Target-Pathway” network network. It is more visualizable to elucidate the key components, core targets, and major pathways of HJT for CHD through the“Component-Target-Pathway” network.
Molecular Docking Preliminarily Verified the Mechanism of HJT Therapy for CHD
In order to further verify the key components and core targets of HJT therapy for CHD, molecular docking was performed to initially validate the binding between activity the components and the targets using the core targets as the receptor and the key components as the ligands. The protein structures, including EGFR, GSK3B, MMP1 and PTGS2, were downloaded from the PDB database 25 (https://www.rcsb.org/) screened by Homo sapiens, refinement resolution (Å) ≤ 2.5, and protein with small molecule ligands as conditions. Then, the protein were pretreated using PyMOL tool, such as remove water, add hydrogen and equilibrium electric fields. Binding energy is the general standard in evaluating molecular docking, lower binding energy signifies stronger appetency. Normally, when the binding energy is lower than 5 Kcal/mol is considered to be tight binding. 26 The AutoDock Vina software was used to molecular docking and Pheatmap package, PyMOL and Ligplot were used to visualize the results.
Quantitative Structure Activity Relationships (QSAR) Analysis
QSAR is an important methods for predicting the activity of unknown compounds based on the properties of known compounds. 27 The known inhibitors of PTGS2 were collected as training set for the development of QSAR models, and the HyperChem 7.52 was employed to modeled and optimized the chemical structures by MM + force field and steepest descent algorithm. Only considering the models with the best R2 (goodness of fit) and P value, a total of five QSAR models were created. Then, QSAR model was identified and derived for activity prediction by MDL QSAR v.2.2 software. Lastly, The potential activity of the key components of HJT therapy for CHD to inhibit PTGS2 was calculated.
Results
3.1 Component Identification of HJT
The CHD model rats were prepared according to previously reported studies and were evaluated based on selectrocardiogram and HE staining of myocardial tissue. The results revealed pathological Q waves in the electrocardiogram, and the HE staining displayed irregular morphology of cardiomyocytes, characterized by enlarged intercellular spaces and signs of degeneration and fibrosis (Figure S1). These findings confirm that the CHD rat model was successfully established, and that the rats were subsequently treated with HJT.
The drug components entering the bloodstream is a prerequisite for their effectiveness. Referring to previous studies,28‐35 this study identified a total of 14 blood-entering components of HJT through secondary mass spectra of each component (Figure 1 and Figure S2), and these compounds include boldine, hypaconitine, licorice saponin G2, neoline and talatisamine, etc (Table 1). The results demonstrated that these 14 blood-entering components may be potential pharmacological active components of HJT therapy for CHD. Moreover, a total of 74 components in aqueous extract of HJT were also identified by UPLC-QE-MS/MS analysis (Figure S3), including 24 flavonoids, 14 amino acids, 9 phenylpropanoids and so on. (Table S1), and the components quantity in aqueous extract of HJT were classified.

Chemical structure of 14 blood-entering components of rat serum after HJT administration.
The Identification of Blood-Entering of HJT by UPLC-QE-MS/MS Analysis.

3.2 Potential Target Screening and PPI Network Analysis
After removing the duplicates, a total of 145 targets of HJT and 1405 CHD related targets were obtained from various databases, and the 32 potential targets of HJT therapy for CHD were screened out by Venn analsis (Figure 2A). To further identify the core targets, a PPI network was established using STRING database, Cytoscape software, and cytoHubba plug-in. As shown in Figure 2B, The results indicated that EGFR, GSK3B, MMP1, PTGS2 and others targets in network centers may be the core targets of HJT therapy for CHD.
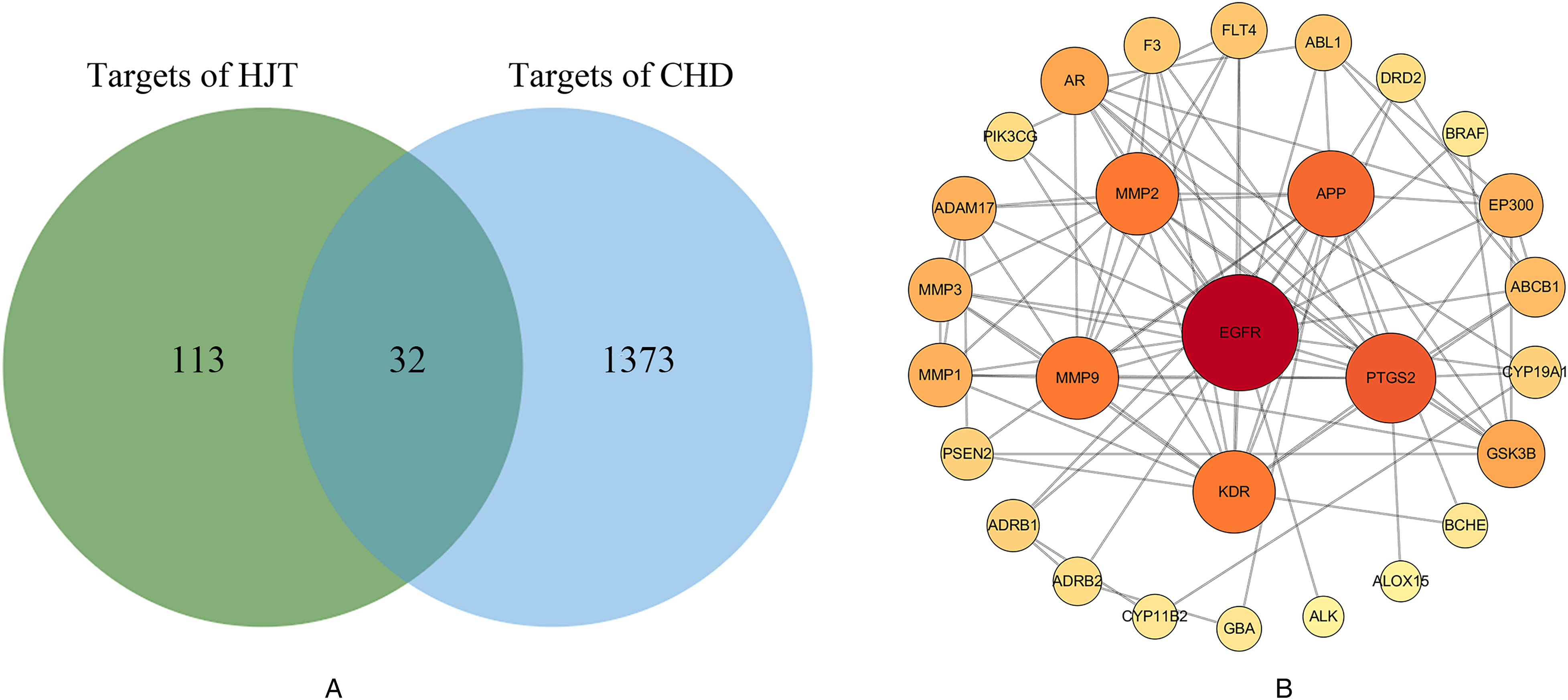
Potential target screening and protein- protein interaction analysis, A: Venn analysis between targets of HJT and CHD; B: the PPI network of 32 potential targets of HJT therapy for CHD.
3.3 GO and KEGG Enrichment Analysis of the Potential Target
The 32 potential targets of HJT therapy for CHDc were subjected to GO and KEGG enrichment analysis through the Metascape database, with P < 0.01 as the screening criterion. A total of 161 were biological process (BP) terms, 26 cellular component (CC) terms, and 38 molecular function (MF) terms were enriched (Figure 3A). The results of GO enrichment analysis indicated that the BP of HJT therapy for CHD associated with platelet activation, negative regulation of epithelial cell proliferation, and excitatory prominence of post-potentiation potential, CC was related to axons, mitochondria, and the perinuclear region of the cytoplasm, MF was mainly associated with endopeptidase activity, protein tyrosine kinase activity, and peptidase activity.

Results of enrichment analysis of potential targets, A: GO enrichment analysis; B: KEGG enrichment analysis.
To further clarify the potential mechanisms of HJT for the treatment of CHD, the Metascape database was also used for KEGG enrichment analysis. According to their P values and count, the top 20 signaling pathways layout in a bubble plot (Figure 3B). Even though some cancer-associated pathways were enriched to the top in KEGG bubble plot, the IL-17 and cGMP-PKG signaling pathways caught our attention as them play important roles in regulating CHD according to previous studies reported.
3.4 “Component-Target-Pathway” Network Construction
To systematically and comprehensively explore the mechanism that underlies HJT activity against CHD, “Component-Target-Pathway” network was constructed using Cytoscape 3.9.1 software (Figure 4). The blue triangle denotes the potential targets, and the orange V-shape represents the pathways. The results demonstrate that the coroglaucigenin, licoricesaponin A3, menadione, puerarin, and boldine were key blood-entering components of HJT, and the MMP1, GSK3B, EGFR, and PTGS2 were presumed to be as the core targets. This network showed that HJT exhibits beneficial effects on CHD via multiple components, multiple targets, and multiple pathways.

The “Component-Target-Pathway” network of HJT therapy for CHD.
3.5 Molecular Docking Validation
Molecular docking was used to validate the binding ability between the key components and the core target, and the results of heatmap showed that all key blood-entering components bind better to the core targets (Figure 5A), and the max binding energy is −11.1 Kcal/mol and min is −5.7 Kcal/mol. As shown in Figure 5B-C, the interaction patterns of the top2 docking results with highest binding energy were displayed, ie, coroglaucigenin binding to PTGS2 and boldine bingding to GSK3B. The coroglaucigenin binding to PTGS2 via residues such as Val-135 and Val-110, and the boldine bingding to GSK3B mainly through the Leu-352 and Tyr-385 residues. Notably, the binding energies between PTGS2 and the key components were all substantially greater than other core targets, the key components of HJT may be targeting PTGS2 inhibition for the treatment of CHD. Therefore, PTGS2 was selected as target for QSAR analysis.
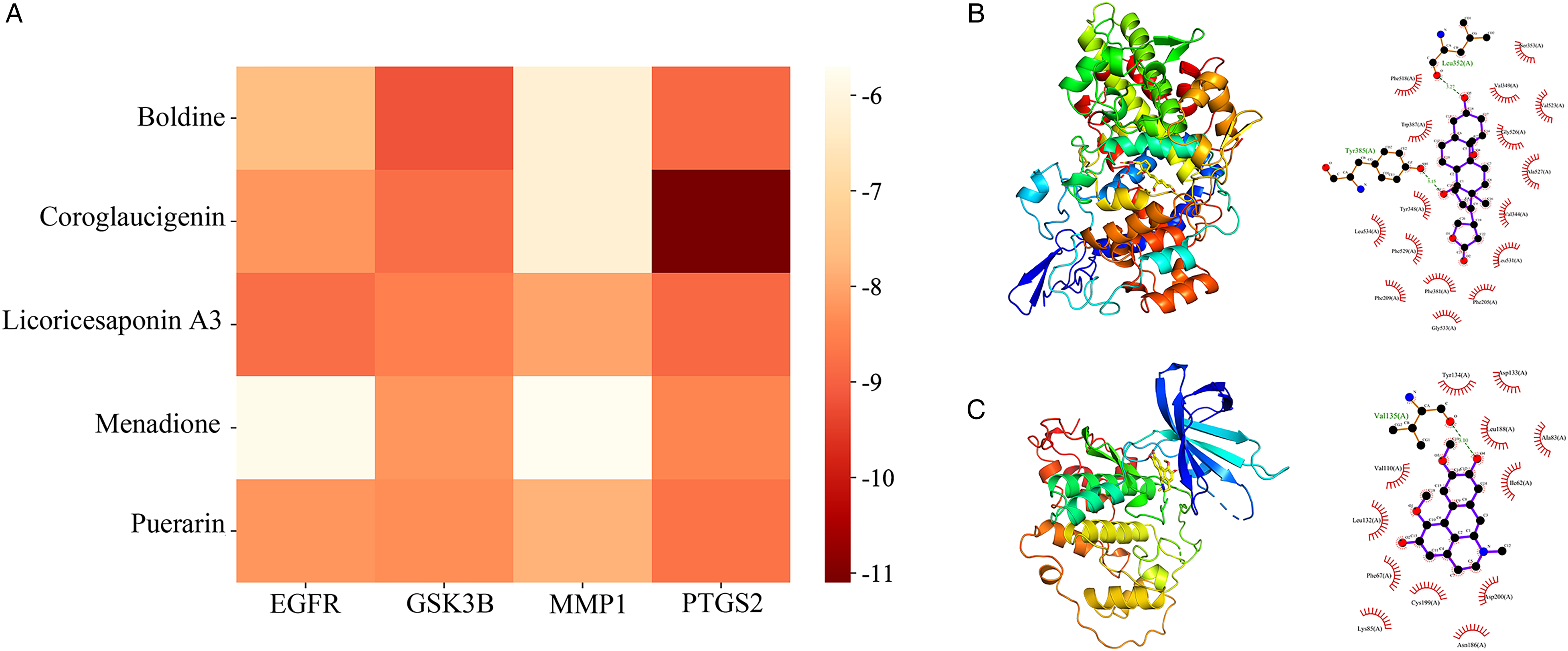
Results of molecular docking of the key components with the core targets, A: Heat map of the binding energy of the components with the targets; B: 3D conformation of molecular docking of Coroglaucigenin binding PTGS2; C: 3D conformation of molecular docking of Boldine binding GSK3B.
3.6 QSAR Analysis and Activity Prediction of key Components
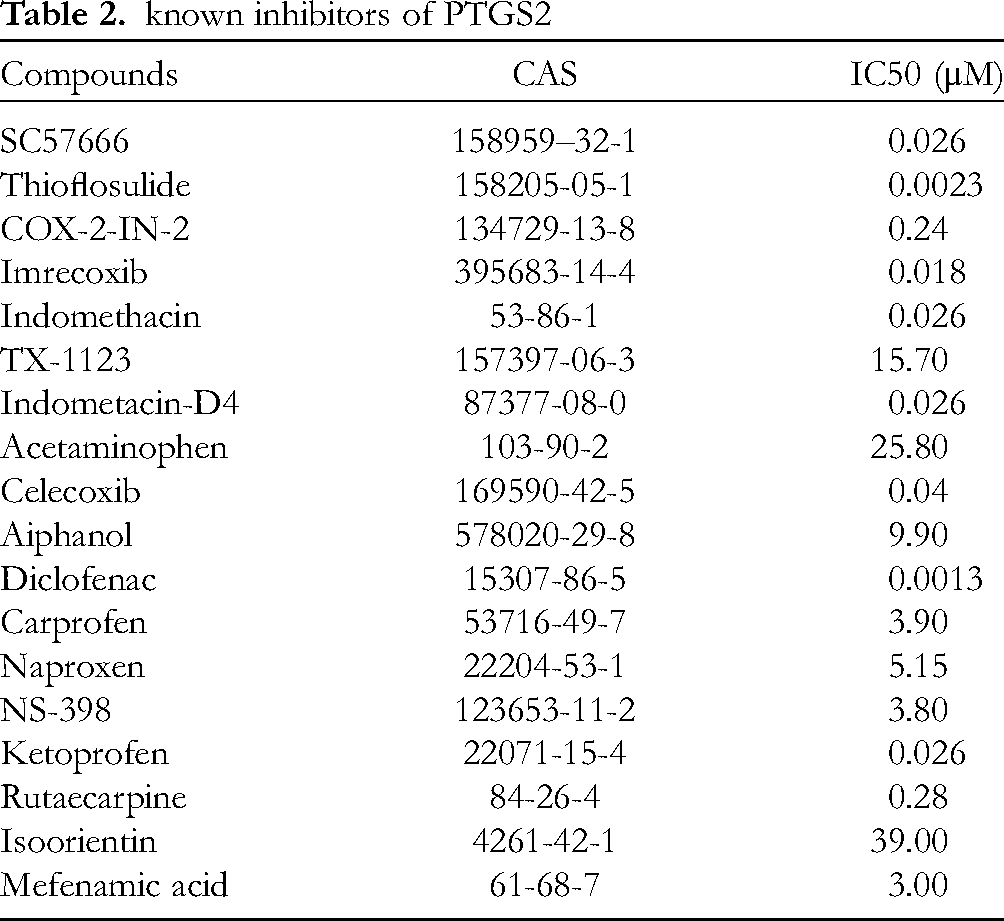
A total of 18 known PTGS2 inhibitors information was collected (Table 2), and five QSAR models were constructed (Table 3 and Figure S4). Then, the activity of the five key components was predicted, and the results showed that Boldine had the best activity (IC50 as low as 0.03 μM). In addition, Coroglaucigenin activity was also excellent, the lowest IC50 was 0.048 μM (Table 4). These results are consistent with the molecular docking description, and which further confirms the possibility of the key component of HJT for the treatment of CHD by inhibition of PTGS2. Many studies have reported the multifaceted pharmacological activities of boldine, which include anti-atherosclerotic, antioxidant, and anti-inflammatory properties. It also exhibits cardioprotective and nephroprotective effects.36‐39 Coroglaucigenin has been reported to be effective against colorectal cancer. 40 Licoricesaponin A3 has shown significant anti-inflammatory activity in both COVID-19 and colitis.41,42 Menadione (vitamin K3) is widely recognized for its antioxidant properties and its ability to scavenge oxygen radicals, contributing to the prevention of coronary heart disease (CHD) by alleviating oxidative stress. 43 Puerarin has been demonstrated to exert cardioprotective effects through AMPK, Wnt/β-catenin, and ferroptosis pathways, and has shown considerable therapeutic potential for coronary heart disease, myocardial ischemia-reperfusion injury, and myocardial hypertrophy.44‐47
known inhibitors of PTGS2
Parameters of the five QSAR models

The IC50 of key Components to Inhibit PTGS2.

Coronary heart disease (CHD) is a common chronic cardiovascular disease driven by a combination of multiple factors, such as hypertension, myocardial ischemia, and reperfusion injury. 48 Traditional Chinese medicine (TCM) has the advantages of multi-components, multi-targets and multi-pathways, and is commonly used in the treatment of various chronic diseases. The TCM can improve complex diseases systematically, which originates from the unique philosophical concepts of TCM. 49 Hanjing Decoction (HJT) was proposed by Shimao Li, a national Chinese medical master, and it has various effects such as activating blood circulation, warming yang for dispelling cold, removing blood stasis, relieving pain, and nourishing blood and vital energy. Previous studies have shown that HJT has a beneficial ameliorative effect on hypertension, oxidative stress injury in vascular smooth muscle, and vascular inflammation. 50 In our clinical observations, we found that HJT showed good therapeutic potential for CHD, but its pharmacological components and mechanisms are still unclear and need to be further elucidated.
Through UPLC-QE-MS/MS method, a total of 14 blood-entering components from rat serum and 74 chemical components from the aqueous extract of HJT were identified. Network pharmacology analysis identified four core targets for HJT in the treatment of CHD, namely MMP1, GSK3B, EGFR, and PTGS2. and also identified 5 key components, including coroglaucigenin, liquoricesaponin 3, menadione, puerarin, and boldine. The potential targets of HJT therapy for CHD are mainly enriched in the IL-17 signaling pathway and the cGMP-PKG signaling pathway. Molecular docking demonstrated the essential role of PTGS2 in the treatment of CHD with HJT, and the activity of key components inhibiting PTGS2 was further verified by QSAR analysis.
PTGS2, aka COX2, is a key rate-limiting enzyme in the biosynthesis of prostaglandins, have been reported to be involved in the inflammatory progression, and PTGS2 contribute to the inflammation involved in atherosclerosis. 51 Normally, PTGS2 is hardly expressed, but high PTGS2 expression can be induced after pro-inflammatory factor stimulation. 52 Therefore, inhibition PTGS2 is considered a promising therapeutic strategy for inflammation. Previous studies have shown that PTGS2 is a risk factor for CHD patients, and inhibiting PTGS2 activity or expression may lead to a better prognosis for CHD patients. 53 Ma et al study found that Tanshinone IIA could treat coronary artery disease by inhibiting COX2 expression, 54 and there were also studies showing that the FPS-ZM1 (a PTGS2 inhibitor) can improved myocardial ischemia-reperfusion injury. 55 These results again demonstrated that it is an effective strategy for the treatment of CHD through inhibiting PTGS2.
In this sudy, the results of molecular docking and QSAR analysis showed that all key components bind well to PTGS2 and may be potential PTGS2 inhibitors, especially boldine and coroglaucigenin. Study reported that the boldine can ameliorated the atherosclerotic lesion formation by reduced total, LDL cholesterol, and oxidation oxidative stress 56 ; additionally. a recent study of chronic liver injury revealed that PTGS2 expression levels were significantly reduced in mice after boldine administration. 57 Thus, in combination with our findings boldine may be key components of HJT in the treatment of CHD. At present, there are fewer studies on the coroglaucigenin, and no reports have been made linking the coroglaucigenin to PTGS2 or to CHD, and our findings first suggest that coroglaucigenin could treat CHD by inhibiting PTGS2.
This study still has some shortcomings need to be improved, as follows: 1) A total of 74 chemical components were identified in the aqueous extract of HJT, while only 14 were identified in the rat serum, and other components in the serum remain to be further characterized to make the results more complete. 2) Although this study used molecular docking and QSAR analysis to favorably analyze the binding between the key components and the core targets, especially with PTGS2, the conclusions still need to be supported by more experimental evidence. 3) We have observed the beneficial effects of HJT on CHD in clinical applications, and this phenomenon has been re-confirmed in rats, but the specific mechanism of HJT therapy for CHD needs to be further investigated.
Conclusion
In conclusion, this study identified five key components and four core targets of HJT in the treatment of CHD by combined UPLC-MS, network pharmacology, molecular docking and QSAR analysis. The key components against CHD may through inhibit PTGS2, and the boldine and coroglaucigenin had the most significant inhibition of PTGS2. The HJT may treat CHD by inhibiting PTGS2 and regulating the IL-17 signaling pathway.
Supplemental Material
sj-tif-1-npx-10.1177_1934578X241275005 - Supplemental material for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology
Supplemental material, sj-tif-1-npx-10.1177_1934578X241275005 for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology by Wang Qi, Zhong Jianyuan, Zhang Wenxia, Ren Yinghan and Yang Yang in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241275005 - Supplemental material for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology
Supplemental material, sj-docx-2-npx-10.1177_1934578X241275005 for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology by Wang Qi, Zhong Jianyuan, Zhang Wenxia, Ren Yinghan and Yang Yang in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X241275005 - Supplemental material for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology
Supplemental material, sj-docx-3-npx-10.1177_1934578X241275005 for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology by Wang Qi, Zhong Jianyuan, Zhang Wenxia, Ren Yinghan and Yang Yang in Natural Product Communications
Supplemental Material
sj-tif-4-npx-10.1177_1934578X241275005 - Supplemental material for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology
Supplemental material, sj-tif-4-npx-10.1177_1934578X241275005 for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology by Wang Qi, Zhong Jianyuan, Zhang Wenxia, Ren Yinghan and Yang Yang in Natural Product Communications
Supplemental Material
sj-docx-5-npx-10.1177_1934578X241275005 - Supplemental material for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology
Supplemental material, sj-docx-5-npx-10.1177_1934578X241275005 for Exploring the Potential Pharmacological Basis and Mechanism of HJT Activity in the Treatment of CHD Based on UPLC-QE-MS and Network Pharmacology by Wang Qi, Zhong Jianyuan, Zhang Wenxia, Ren Yinghan and Yang Yang in Natural Product Communications
Footnotes
Abbreviations
Author Contributions
WQ: data collection, perform experiment, and writing draft manuscript. ZJY: data processing, analysis results, and draft manuscript preparation. ZWX and RYH: analysis and interpretation of results, and draft manuscript preparation. YY: methodology, investigation, critical revision and editing of manuscript, and funding acquisition. WQ and ZJY made equal contributions to the manuscript and they are co-first authors. All authors approved the final version to be published.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Experimental Animal Committee of Hebei University of Traditional Chinese Medicine.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Research Projects of Hebei Administration of Traditional Chinese Medicine (Grant No. 2023110) and 2022 Introducing Overseas Educated Personnel Funded Project (Grant No. C20220110).
Statement of Informed Consent
Not applicable.
Statement of Human and Animal Rights
The animal experimental procedures involved in this study were carried out in accordance with the guidelines for animal experiments of the Experimental Animal Centre of Hebei University of Traditional Chinese Medicine and approved by the Experimental Animal Committee of Hebei University of Traditional Chinese Medicine.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
