Abstract
Introduction
Over recent years, coronary heart disease (CHD) has remained a health-threatening, catastrophic disease that imposes a huge economic burden on patients and their families all over the world. According to a recent report published in The Lancet, cardiovascular disease become the primary cause of death in China between 1990 and 2017.1,2 Recently, Traditional Tibetan medicine (TTM) was shown to have distinctive curative efficacy on cardiovascular diseases during clinical trials because of its multitargeted functions. 3
Wuwei Yuganzi San (WYS) has come from MaDiYiZhuXuanJi, a famous book in TTM, and has been widely used in the treatment of CHD for a long time.4,5 These prescriptions were used extensively in the treatment of angina pectoris and other cardiovascular diseases.4,5 WYS includes Phyllanthus emblica L. (YuGanZi, YGZ), Caragana jubata P. (ZangJinJiEr, ZJJE), Corydalis impatiens F. (SaiBeiZiJin, SBZJ), Rheum palmatum L. (DaHuang, DH), and Zingiberis Rhizoma (GanJiang, GJ). What is more, our research has shown that WYS had significant effects in protecting cardiomyocytes from myocardial ischemia-reperfusion injury.5,6 However, the underlying pharmacological mechanism of WYS in the therapy of CHD is unclear.
Recently, network pharmacology, a significant part of systematic biology, has provided a novel way to elucidate the mode of action of TTM. 7 It also highlights the function of multiingredient–multitarget–multipathway treatments. 8 Consequently, a network pharmacology strategy has been undertaken to explore the mechanisms of WYS in the treatment of CHD. The purpose of this study was to provide a convincing scientific basis for WYS to treat CHD. We screened the active ingredients and the underlying targets, predicted the associated targets and signaling pathways, and constructed an active ingredient-target-disease network to investigate the therapeutic mechanism of this prescription in CHD. The flowchart of this study is shown in Figure 1.

Flowchart of this study.
Methods
Active Ingredients of WYS Collection and Screening
The Traditional Chinese Medicine Systems Pharmacology (TCMSP: https://tcmspw.com/tcmsp.php) and the literature was used to collect the compounds of WYS.9–22 The selection of active ingredients of WYS followed the pharmacokinetic rules, which included absorption, distribution, metabolism, and excretion (ADME). 9 The oral bioavailability threshold was set as ≥30% and the drug-likeness threshold was set as ≥0.18.23,24
Acquiring the Potential Target Proteins of the Active Compounds of WYS
All the data related to active components of WYS were fed into the TCMSP and PubChem database platform (http://pubchem.ncbi.nih.gov) to acquire MOL2 structure and 2-dimensional (2D) structure of the ingredients, respectively. 25 The MOL2 structures of active ingredients were inputted into the PharmMapper (http://www.lilab-ecust.cn/pharmmapper/).26,27 A threshold (norm fit ≥0.88) was set to obtain targets for components in the PharmMapper. Data related to the 2D structure of active ingredients were fed in the Swiss Target Prediction (http://www.swisstargetprediction.ch/index.php). Another threshold (norm fit ≥0.88) was set up to obtain targets for compounds in the Swiss Target Prediction.
CHD-Related Target Screening
CHD-related targets were collected from the OMIM (http://omim.org/) database and GeneCards (http://www.gengcards.org/) database and NCBI Gene Expression Omnibus (GEO) DataSets (https://www.ncbi.nlm.nih.gov/gds/). 28 All gene symbols were retrieved and confirmed by the UniProt database (https://www.uniprot.org/). A Venn diagram was used to show the common targets of WYS and CHD, which was acquired from “ggvenn” packages and “ggplot2” packages of RStudio software (Version 1.3.1093). Heatmap and Volcano plots of expressed differential genes were obtained with the “pheatmap” packages and “ggplot2” packages.
Protein–Protein Interaction (PPI) Network Construction
Data related to WYS–CHD targets were fed into the String platform (https://string-db.org/). The confidence scores were set as ≥0.9, and the species was confined to “Homo sapiens.” The outcomes from the String platform were inputted into Cytoscape 3.7.2 to be utilized for further topological characterization to identify the hub genes of WYS treatment of CHD. 29
Enrichment Analysis
For further investigation of the mechanism of WYS in the treatment of CHD, GO enrichment analysis and KEGG enrichment analysis were performed with the “clusterProfiler,” “BiocManager,” “stringi,” “ggplot2,” and “pathview” packages in RStudio software. GO enrichment analysis contained biological process, molecular function, and cellular component. 30
Molecular Docking
Molecular docking was carried out to prove the crucial ingredients’ binding affinity to the core targets and to investigate their precise combination modes. Protein target receptors for macromolecules were obtained from the RCSB PDB online platform (https://www.rcsb.org/). AutoDock Vina (Version 1.5.6) was used to improve the average precision of binding model prediction. The outcomes were visualized by PyMOL (Version 1.7.x). 31
Statistical Analysis
Cytoscape version 3.7.2 was used to analyze the topological data. Go and KEGG enrichment analyses were carried out by RStudio software (Version 1.3.1093). Molecular docking was performed by AutoDock Vina Version 1.5.6 and PyMOL Version 1.7.x software. 32
Results
Active Ingredients in WYS
Three main components of WYS were collected from the TCMSP database and 92 ingredients in YGZ were found, 92 ingredients in DH, and 148 ingredients in GJ. Thirty-one ingredients in SBZJ and 33 ingredients in ZJJE were acquired from the published literature, individually.10–22 Among them, 34 active ingredients fulfilled the ADME parameter (Table 1), which were identified using PubChem database. Based on the active ingredients, we collected and identified 202 target genes in total.
Active Ingredients of WYS After ADME Screening.

Abbreviations: WYS, Wuwei Yuganzi San; ADME, absorption, distribution, metabolism, and excretion.
CHD-Related Targets
From the GeneCards, OMIM and NCBI-GEO databases, 445, 548, and 3556 relevant targets for CHD were collected, correspondingly. After filtering, 4424 CHD-related targets were obtained. Differentially expressed genes of GSE71226 were presented by making heat maps and volcano plots (Figure 2). Fifty-nine WYS–CHD common targets were obtained from the intersection between WYS-related targets and CHD-related targets (Figure 2).

Common targets of WYS–CHD-related targets. (A) Heatmap of the outcomes was obtained from GSE71226. Upregulation and downregulation of the genes are expressed as red and blue. (B) Volcano plots of the results were acquired from GSE71226. Red, blue, and grey dots are used to indicate upregulation, downregulation, and no significance, respectively. (C) Venn diagram of WYS–CHD common targets.
Construction of the PPI Network
WYS–CHD common targets (59) were imported into the String platform, with “Homo sapiens” chosen. The highest confidence was set up as 0.90; the PPI network was constructed (Figure 3).

The PPI network of WYS and CHD.
Construction of Medicine-Compound-Target-Disease Network
Cytoscape 3.7.2 was used to acquire 95 nodes and 298 edges from the intersection network diagram (Figure 4). Red represents WYS, purple represents ingredients, green represents CHD, and blue represents genes. The picture showed that the top three effective compounds were sennoside D_qt (Degree = 25), quercetin (Degree = 21), and procyanidin B-5,3'-O-gallate (Degree = 17) (Table 2). The top five ranked target genes were ADAM17 (Degree = 17), AKR1C2 (Degree = 16), ALB (Degree = 15), AKT1 (Degree = 15), and ADH1C (Degree = 14) (Table 3).

Effective medicine-compound-target-disease network. In the picture, red represents Wuwei Yuganzi San, purple represents ingredients, green represents coronary heart disease, and blue represents genes.
Key Compounds of WYS in the Treatment of CHD.

Abbreviations: WYS, Wuwei Yuganzi San; CHD, coronary heart disease.
Hub Genes of WYS in the Treatment of CHD.

Abbreviations: WYS, Wuwei Yuganzi San; CHD, coronary heart disease; ADAM17, a disintegrin and metalloprotease 17; AKR1C2, aldo-keto reductase family 1 member C2; ALB, albumin; AKT1, protein kinase B; ADH1C, alcohol dehydrogenase 1C; AR, androgen receptor; AR1, androgen receptor 1; ACADM, acyl-coa dehydrogenase medium; CA12, carbonic anhydrase 12; ARF1, adp-ribosylation factor 1.
Enrichment Analysis
Enrichment analysis was performed by GO and KEGG. A series of 84 items associated with the therapeutic effect of WYS on CHD was obtained through GO enrichment analysis, which could be classified into 3 categories, namely 59 items in the biological process, 15 items in the molecular function, and 10 items in the cellular component. The bubble diagram presents the top 10 items in the above 3 categories. Figure 5(A) shows the biological results; the putative ingredients in WYS responded mainly to negative regulation of the apoptotic and oxidation-reduction processes, and cell proliferation. Figure 5(B) shows the results of the molecular function; the active ingredients of WYS were closely correlated to protein binding, identical protein binding, and protein kinase binding. From the results of the cellular components shown in Figure 5(C), the active ingredients in WYS were associated with cytoplasm, cytosol, and the plasma membrane. The chordal diagram was constructed based on the connection between target genes and enrichment pathways, as shown in Figure 5(D). Fifty-two signaling pathways concerning the potential mechanisms by which WYS treatment affects CHD were obtained through KEGG enrichment analysis, as shown in Figure 6(A) and (B). The chord diagram shows the relationship between the signaling pathway and therapeutic targets of WYS in the treatment of CHD, as shown in Figure 6(C).
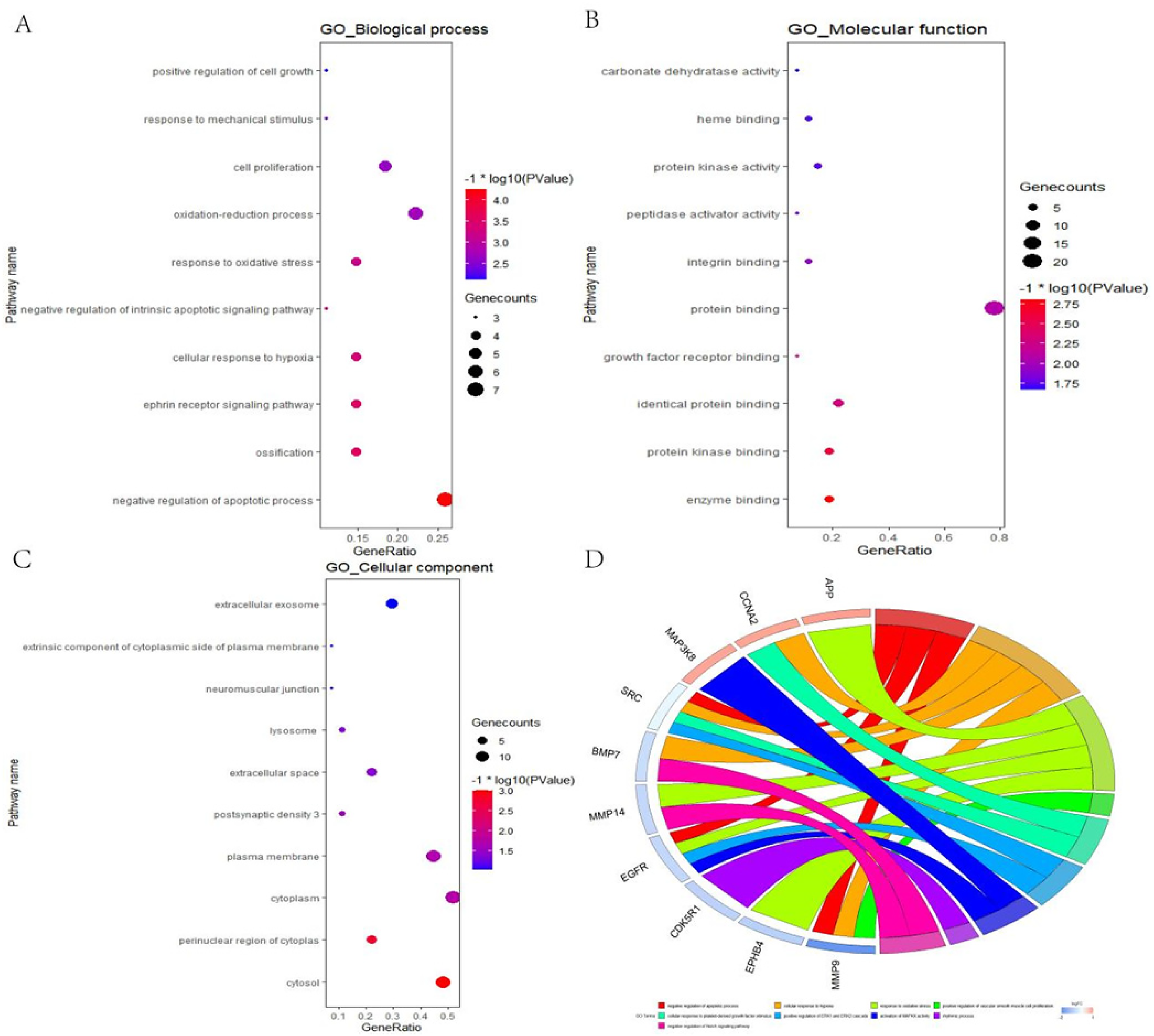
GO enrichment analysis. (A) Enriched biological processes from GO enrichment analysis. (B) Enriched molecular functions from GO enrichment analysis. (C) Enriched cellular components from GO enrichment analysis. (D) Chordal diagram of the GO term of the therapeutic targets of WYS in the treatment of CHD.

KEGG enrichment analysis. (A) and (B) Histograms and bubble plots of signaling pathways closely associated with treatment effects in WYS. (C) Chordal diagram of the pathway description of the therapeutic targets of WYS in the treatment of CHD.
Molecular Docking
The intersection between putative ingredients and target genes was investigated, and we have found that the putative ingredients were sennoside D_qt, quercetin, and procyanidin B-5,3'-O-gallate. The crucial target genes were ADAM17, AKR1C2, ALB, AKT1, and ADH1C. Analysis using AutoDock Vina 1.5.7 was carried out. The values of binding affinity were >5 kcal/mol, which indicated that the putative ingredients and target genes had stable combinations. The results of binding affinity are shown in detail in Figure 7 and Table 4.

The molecular docking of putative ingredients and crucial target genes. (A) Docking patterns of crucial targets (ADAM17, AKR1C2, ALB, AKT1, and ADH1C) and putative ingredients (sennoside D_qt, quercetin and procyanidin B-5,3'-O-gallate) of WYS. (B) Binding affinities (kcal/mol) of crucial targets and putative ingredients of WYS.
Outcomes of Molecular Docking Between Putative Ingredients and hub Genes.

Abbreviations: rmsd l.b, root-mean-square deviation lower bound; rmsd u.b, root-mean-square deviation upper bound; ADAM17, a disintegrin and metalloprotease 17; AKR1C2, aldo-keto reductase family 1 member C2; ALB, albumin; AKT1, protein kinase B; ADH1C, alcohol dehydrogenase 1C.
Discussion
The definitions of “syndromes” and “diseases” have been understood in TTM. The recognition of the disease has been based on its comprehensive state. Therefore, the underlying occurrence and development of the disease can be seen from a macroscopic point of view. 32 However, the underlying mechanisms and pathways of TTM have remained indistinct, because CHD treatment has required a comprehensive understanding of its etiology, pathogenesis, and drug selection. Recently, network pharmacology has emerged as an optimized approach to explore the “drug-ingredient-target” of TTM and traditional Chinese medicine.
WYS consists of 5 herbs, P emblica (YGZ), C jubata (ZJJE), C impatiens (SBZJ), R palmatum (DH), and Z Rhizoma (GJ). WYS had been used as a typical and effective prescription for CHD for a long time in Tibetan medicine. 5 In in vivo experiments in rats, WYS had been testified to reduce the levels of lactate dehydrogenase (LDH) and creatine kinase (CK) in serum, increase the activity of superoxide dismutase (SOD) and glutathione peroxidase (GPX), and reduce the activity of malondialdehyde (MDA) in myocardial tissues, which could play a protective role against myocardial ischemia-reperfusion injury. 6 Another study has reported that WYS protected injured cardiomyocytes by up-regulating the expression level of B cell lymphoma-2/B cell lymphoma-2-x (Bcl-2/Bax). 5 Based on previous research results, the object of this study was to investigate the mechanism of the therapeutic effect of WYS on CHD by using a network pharmacological approach and molecular docking. The results indicated that the putative ingredients of WYS were associated with core proteins of several biological processes and signaling pathways in the treatment of CHD. However, all these factors need to be further illuminated in animal experiments and clinical trials in our following schemes.
Firstly, 4549 CHD-related target genes were acquired from the GeneCards, OMIM and NCBI-GEO databases. At the same time, the TCMSP database and literature have obtained 34 candidate putative ingredients and 202 WYS-related target genes from the 5 herbs in WYS: P emblica, C jubata, C impatiens, R palmatum, and Z Rhizoma. Several articles have reported that YGZ and its extracts could protect RAW264.7 cells from H2O2-induced toxicity, reduce the size of atherosclerotic plaques, and inhibit endothelin-1 (ET-1) expression.33–35 Moreover, it could exert antiinflammatory effects by reducing the expression of nitric oxide (NO) and proinflammatory cytokines. 36 In addition, it could protect β-cells, scavenge-free radicals, alleviate inflammation, and reduce advanced glycation end products. 37 DH could protect mitochondria from oxidative stress by regulating lipid metabolism and increasing antioxidant activity. In addition, DH could also regulate Fas/Fas ligand (Fas/FasL)-mediated apoptosis and suppress β-cell apoptosis. 38 A study, using a network pharmacology and metabolomics approach, has reported that the combination of Aconiti Lateralis Radix Praeparata combined with Z Rhizoma could treat chronic heart failure through mitochondrial-mediated energy metabolism.39,40 SBZJ could inhibit the expression of tumor necrosis factor-α (TNF-α) and interleukin-6 by down-regulating the activation of nuclear factor-kappa B (NF-κB), extracellular signal-regulated kinase 1/2 (ERK1/2) phosphorylation, and the mitogen-activated protein kinase signaling pathway. 41 ZJJE could inhibit TNF-α, scavenge-free radicals, and inhibit lipid peroxidation to achieve cardioprotection.42–44 Consequently, WYS exerted cardiovascular protective effects through antiinflammatory and antioxidant effects.
From the results of network pharmacology, there were 59 WYS–CHD common targets from the intersection between WYS-related targets and CHD-related targets. Putative ingredients of WYS were ranked, which were engaged in the treatment of CHD by the degree in the ingredient-CHD target network. The top 3 putative ingredients were sennoside D_qt (Degree = 25), quercetin (Degree = 21), and procyanidin B-5,3'-O-gallate (Degree = 17). These ingredients have shown inhibition of inflammation. Quercetin could significantly alleviate endothelial dysfunction by inhibiting inflammation-associated signaling pathways. 45 Sennoside D_qt could reduce inflammation and improve insulin resistance. To some extent, sennoside D_qt had a cardiovascular protective effect. 46 Procyanidin protected the brain from ischemia-reperfusion injury by inhibiting the TLR4-NLRP3 inflammasome signaling pathway. 47 Gallnut protected coronary artery endothelial cells by inhibiting the NF-κB signaling pathway. So we speculated that procyanidin B-5,3'-O-gallate had advantages in the treatment of myocardial ischemia-reperfusion injury. 48 Thus, these results demonstrated that WYS might exert cardioprotective effects via these putative ingredients and their specific targets. The outcomes showed that in the GSE71226, compared with healthy people, the expression of the top 5 target genes were ADAM17, AKR1C2, ALB, AKT1, and ADH1C. ADAM17, tumor necrosis factor-alpha converting enzyme, played the main role in cardiovascular disease. Increased shedding of ADAMs could induce various cardiovascular diseases, which have been closely related to inflammation, tissue remodeling, and dysfunction. ADAMs might be promising therapeutic targets for hypertension and atherosclerosis. 49
Conclusion
In this study, we have shown that the underlying mechanisms of treatment of CHD are associated with processes such as negative regulation of the apoptotic and oxidation-reduction processes, and cell proliferation. Molecular docking outcomes demonstrated that the ingredients of WYS stably bind with crucial proteins, namely, ADAM17, AKR1C2, ALB, AKT1, and ADH1C. However, this study lacks experimental validation, so additional studies are needed. In summary, this work showed the systems biology of WYS. Subsequent studies are necessary to either develop prospective applications of WYS or to validate our findings.
Footnotes
Acknowledgments
We sincerely thank all members who have participated in and supported this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Department of Qinghai Province (grant number 2020-ZJ-922).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
