Abstract
Objectives
The aim is to discover α-glucosidase/acetylcholinesterase inhibitors as lead compounds from a traditional herbal prescription of Qi-Li-Qiang-Xin capsule (QLQX).
Methods
A novel strategy combining time-based fractionation, LC-QTOF-MS, enzymatic activity assay, molecular docking and component-target association analysis was performed to discover α-glucosidase/acetylcholinesterase inhibitors from QLQX. Time-based fractionation combined with enzymatic activity assay was used to find the distribution period of active compounds in the herbal prescription. LC-QTOF-MS was used to analyze the structure of active compounds. Molecular docking was applied to explore the interaction between active compounds and targets.
Results
According to time-based fractionation, the active components of QLQX for acetylcholinesterase were primarily concentrated in the highly polar region, whereas the active components for α-glucosidase were predominantly found in the moderately polar area. A total of 33 compounds were identified by comparing with chemical reference substances. Dihydrotanshinone Ⅰ (16.98 µM), hydroxysafflor yellow A (84.57 µM), salvianolic acid A (76.62 µM) and cryptotanshinone (112.68 µM) were identified as acetylcholinesterase inhibitors from QLQX. Similarly, rosmarinic acid (62.29 µM), isochlorogenic acid A (17.95 µM), 4,5-dicaffeoylquinic acid (117.93 µM), danshensu (207.88 µM), salvianolic acid A (1.31 µM), 3,4-dicaffeoylquinic acid (91.71 µM), formononetin (67.26 µM), ginsenoside Rd (3.43 µM), ginsenoside Rb1 (26.37 µM) and ginsenoside F1 (18.79 µM) were discovered as α-glucosidase inhibitors. Notably, salvianolic acid A inhibited both acetylcholinesterase and α-glucosidase. The results of molecular docking indicated that hydrogen bonds, hydrophobic interactions and Pi-Pi T-shaped interactions were crucial for inhibiting acetylcholinesterase. Meanwhile, hydrogen bonds, hydrophobic interactions and Pi-Pi stacked interactions were significant in suppressing α-glucosidase.
Conclusion
QLQX contains numerous acetylcholinesterase and α-glucosidase inhibitors, demonstrating its potential therapeutic benefits for Alzheimer's disease and diabetes.
Introduction
“Multiple components to multiple targets” was considered to be the action mode of herbs for a long time,1-5 but this opinion is now regarded as an inadequate understanding. Actually, how herbs act on multiple targets via multiple components is still unclear. A single herb commonly contains hundreds or thousands of ingredients, and they can usually be grouped into several structural analogues.6-8 From this perspective, despite the large number of ingredients, the complexity of a herb's composition is mitigated by the presence of structural analogues. In our previous work, we found that there is a natural structure-activity relationship of structural analogues in herbs, which is conducive to finding compounds with better activity. 9 In other words, the significance of a large number of structural analogues lies in increasing the probability of finding prominent molecules with high bioactivity. However, whether this phenomenon is widespread in herbs remains to be explored.
A herbal prescription contains more than two herbs and a variety of ingredients.10-12 One herbal prescription for multiple diseases is regarded as a feature in the medicinal system of traditional Chinese medicines.13-15 Qi-Li-Qiang-Xin capsule (QLQX) is a typical herbal prescription for the treatment of heart failure in China,16-19 and is composed of 11 herbs such as Astragali Radix, Ginseng Radix et Rhizoma and Aconiti lateralis Radix Praeparata.20,21 Most studies focus on its cardiac protection, and there are few studies on other pharmacological effects. In recent years, Astragali Radix, Alismatis Rhizoma and Polygonati odorati Rhizoma in QLQX have been reported to possess anti-diabetic effects.22-26 At the same time, this prescription has been proved to have the effect of alleviating Alzheimer's disease.27-31 Therefore, it is reasonable to speculate that QLQX has potential active components for Alzheimer's disease and diabetes.
Considering the abundant components, QLQX was chosen to discover prominent molecules. Acetylcholinesterase32,33 and α-glucosidase,34,35 which are key enzymes for Alzheimer's disease and diabetes respectively, were selected as targets. By integrating time-based fractionation, LC-QTOF-MS, enzymatic activity assay, molecular docking and component-target association analysis, the action mode of “multiple components to multiple targets” of QLQX was preliminarily revealed. A series of prominent molecules that selectively inhibited acetylcholinesterase or α-glucosidase were discovered as lead compounds. This work not only contributes to the exploration of traditional herbal prescriptions, but also accelerates the process of modern drug discovery.
Results and Discussion
Technical Route
The technical route for discovering inhibitors of α-glucosidase and acetylcholinesterase from QLQX is shown in Figure 1. The QLQX extract was divided into several fractions on a chromatographic column according to a fixed time. The α-glucosidase/acetylcholinesterase activity of each fraction was detected to depict the distribution of activity. Active fractions were then analyzed using LC-QTOF-MS to determine the structural information of the compounds. Detection of the activities of structurally similar compounds aids in identifying lead compounds with superior activity. Furthermore, molecular docking was used to explore the interactions between enzymes and compounds. Component-target association analysis was used to summarize the active compounds in the QLQX extract that act on α-glucosidase and acetylcholinesterase. Through this technical route, the activity of QLQX extract on different enzymes could be systematically studied in vitro.

A strategy consisting of time-based fractionation, LC-QTOF-MS, enzymatic activity assay, molecular docking and component-target association analysis for the research on herbal prescriptions.
Time-Based Fractionation of QLQX
Before the time-based fractionation, the activity of QLQX against acetylcholinesterase and α-glucosidase was tested. The results showed that QLQX had an inhibitory effect on acetylcholinesterase (Figure 2A) and α-glucosidase (Figure 2B) with IC50 values of 3.73 and 0.30 mg/mL, respectively. According to the chromatogram (Figure 3), QLQX extract was divided into 7 fractions, including 0–10 min, 10–20 min, 20–30 min, 30–40 min, 40–50 min, 50–60 min and 60–70 min. As shown in Figure 2C, the 7 fractions inhibited acetylcholinesterase with IC50 values of 1.00, 0.19, 0.21, 0.30, 0.48, 0.85 and 3.88 mg/mL, respectively. Among them, the fraction from 10–20 min (0.19 mg/mL) showed the highest activity. As shown in Figure 2D, the 7 fractions inhibited α-glucosidase with IC50 values of 1.20, 0.77, 0.20, 0.06, 0.05, 0.11 and 0.29 mg/mL, respectively. Among them, the fraction from 40–50 min (0.05 mg/mL) showed the highest activity. The above results indicated that the activity distributions of QLQX extract on acetylcholinesterase and α-glucosidase were different. From the perspective of polarity, the acetylcholinesterase inhibitors in QLQX were mainly concentrated in the highly polar region, while the α-glucosidase inhibitors were mainly present in the moderately polar region.

The inhibitory activities of QLQX extract (A) and time-based fractions (C) on acetylcholinesterase; and the inhibitory activities of QLQX extract (B) and time-based fractions (D) on α-glucosidase.

Distribution of compounds in the chromatogram of QLQX.
LC-QTOF-MS was employed to identify the chemical components in the QLQX extract, resulting in the identification of a total of 33 compounds through comparison with chemical reference substances. These compounds include 3,4-dicaffeoylquinic acid, danshensu, salvianolic acid A, salvianolic acid B, caffeic acid, formononetin, calycosin, rosmarinic acid, neochlorogenic acid, isochlorogenic acid A, hydroxysafflor yellow A, talatisamine, hesperidin, lithospermic acid, 4,5-dicaffeoylquinic acid, ginsenoside Rg2, ginsenoside F, ginsenoside Rg3, ginsenoside Rh1, ginsenoside Rb1, ginsenoside Rd, ginsenoside Re, ginsenoside Rg1, benzoylhypacoitine, benzoylaconitine, benzoylmesaconine, neoline, aconitine, tanshinone I, tanshinone IIA, dihydrotanshinone, cryptotanshinone and chlorogenic acid. The remaining compounds in QLQX were tentatively identified by comparing them with data from the literature.36,37 A summary of the compounds identified in each fraction is presented in Table 1.
The compounds in QLQX identified by LC-QTOF-MS.

These compounds were identified by comparing them with chemical reference substances.
Discovery of α-Glucosidase/Acetylcholinesterase Inhibitors
The inhibitory activities of 33 identified compounds in the QLQX extract against acetylcholinesterase and α-glucosidase were detected, including 6 salvianolic acids, 5 caffeoylquinic acids, 4 flavonoids, 4 tanshinones, 6 alkaloids and 8 ginsenosides. Their inhibition curves for acetylcholinesterase are shown in Figure 4. Among them, the IC50 values of dihydrotanshinone Ⅰ, salvianolic acid A, hydroxysafflor yellow A and cryptotanshinone for acetylcholinesterase were 16.98, 76.62, 84.57 and 112.68 μM, respectively (Table 2). Interestingly, one compound from each of the tanshinones, salvianolic acids and flavonoids exhibited significant activity. Among the tanshinones, dihydrotanshinone I exhibited the highest activity, with an activity at least one order of magnitude higher than the other compounds (Figure 4A). Among the salvianolic acids, salvianolic acid A showed the highest activity, while the activities of other compounds were weak (Figure 4B). Among the flavonoids, hydroxysafflor yellow A displayed the highest activity (Figure 4C). Notably, this compound was located in the fraction from 10–20 min with the highest activity. Additionally, the activities of caffeoylquinic acids (Figure 4D), alkaloids (Figure 4E), and ginsenosides (Figure 4F) were weak.

Inhibitory effects of tanshinones (A), salvianolic acids (B), flavonoids (C), caffeoylquinic acids (D), alkaloids (E) and ginsenosides (F) on acetylcholinesterase in QLQX.
Acetylcholinesterase and α-Glucosidase Inhibitory Activities of Compounds in QLQX.

n represents nM.
The similar result was observed on α-glucosidase. The IC50 values of salvianolic acid A, ginsenoside Rd, isochlorogenic acid A, ginsenoside F1, ginsenoside Rb1, rosmarinic acid, formononetin, 3,4-dicaffeoylquinic acid, 4,5-dicaffeoylquinic acid and danshensu for α-glucosidase were 1.31, 3.43, 17.95, 18.79, 26.37, 62.29, 67.26, 91.71, 117.93 and 207.88 μM, respectively (Table 2). Among salvianolic acids, salvianolic acid A had the highest activity (Figure 5A). Among ginsenosides, ginsenoside Rd exhibited the highest activity (Figure 5B). Among flavonoids, formononetin showed the highest activity (Figure 5C). Among caffeoylquinic acids, isochlorogenic acid A had the highest activity (Figure 5D). Notably, ginsenoside Rb1 and salvianolic acid A were in the fraction from 40–50 min with the highest activity. Alkaloids showed no activity (Figure 5E), while tanshinones had weak activity (Figure 5F). Interestingly, salvianolic acid A had an inhibitory effect on both α-glucosidase (1.31 μM) and acetylcholinesterase (76.62 μM).

Inhibitory effects of salvianolic acids (A), ginsenosides (B), flavonoids (C), caffeoylquinic acids (D), alkaloids (E) and tanshinones (F) on α-glucosidase in QLQX.
Interactions Between Bioactive Molecules and Target Proteins
The interactions between bioactive molecules and target proteins were obtained by molecular docking. A low interaction energy suggested a stable docking conformation, which represented a good affinity between the compounds and the proteins. For acetylcholinesterase, the interaction energies of salvianolic acid A, hydroxysafflor yellow A, cryptotanshinone and dihydrotanshinone Ⅰ were −55.52, −34.87, −34.12 and −30.57 kcal/mol, respectively. Salvianolic acid A showed the lowest interaction energy. The docking sites are shown in Figure 6A. The interaction energies and amino acid residues were summarized in Table 3. Among the four compounds, salvianolic acid A and hydroxysafflor yellow A could form hydrogen bonds with TYR 72, ASP 74, GLY 120, and GLU 202, which is consistent with the residues of acetylcholinesterase inhibitors reported previously. 38 It is noteworthy that salvianolic acid A could form Pi-Pi T-shaped interactions with TRP 86, TYR 124, and TRP 286, while hydroxysafflor yellow A did not exhibit such interactions. Unlike the above two compounds, cryptotanshinone and dihydrotanshinone I mainly interacted with acetylcholinesterase through hydrophobic bonds and Pi-Pi T-shaped interactions rather than hydrogen bonds. Specifically, cryptotanshinone and dihydrotanshinone I could both form hydrophobic bonds with HIS 447, and form Pi-Pi T-shaped interactions with TYR 337 and TYR 341.
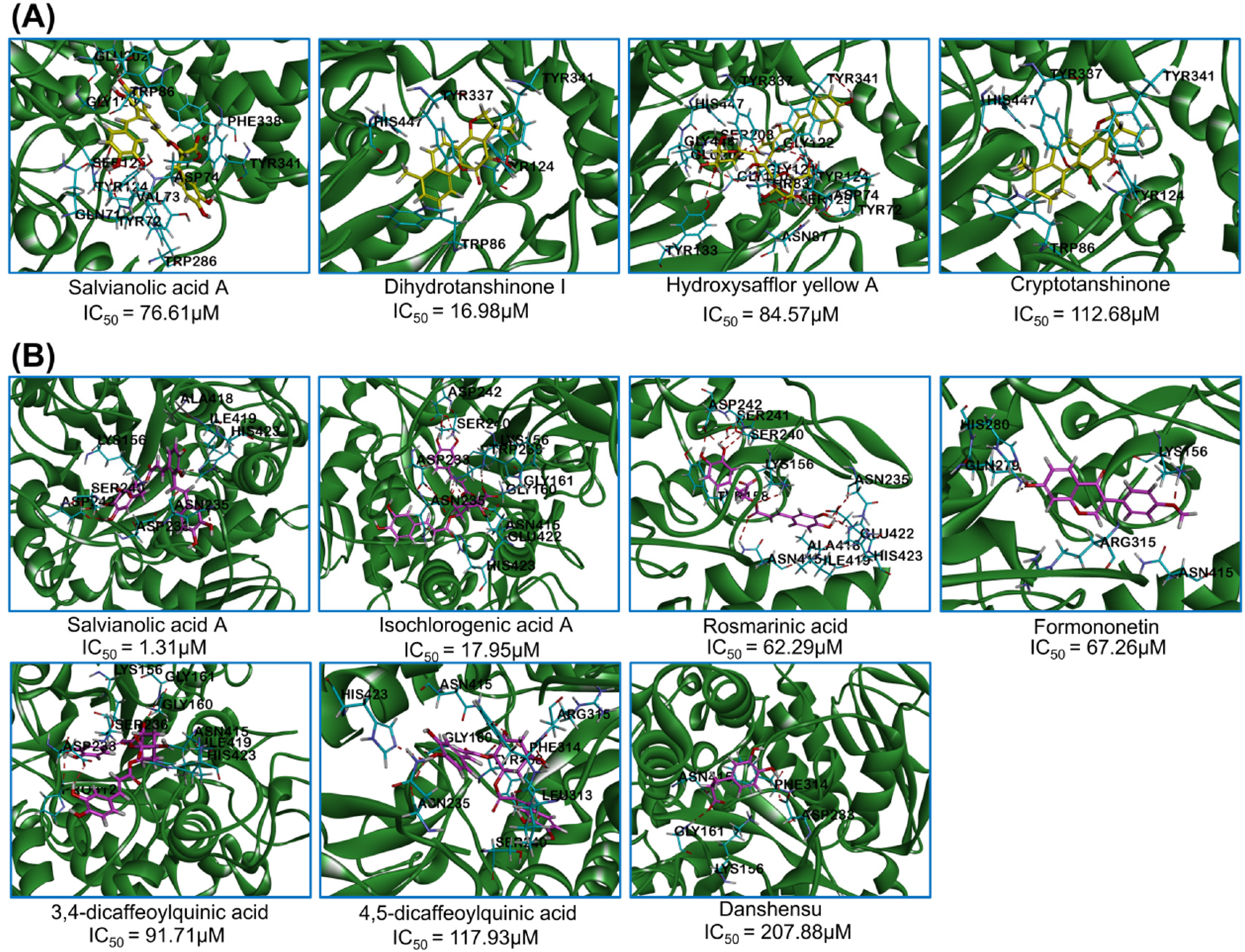
The molecular docking of active molecules with acetylcholinesterase (A) and α-glucosidase (B) in QLQX.
Interaction Energy and Amino Acid Residues in the Interaction Between Active Ingredients and Acetylcholinesterase.

The active compounds in QLQX were docked with α-glucosidase as shown in Table 4. The docking sites are shown in Figure 6B. The interaction energies of salvianolic acid A, rosmarinic acid, isochlorogenic acid A, 4,5-dicaffeoylquinic acid, danshensu, 3,4-dicaffeoylquinic acid and formononetin were −61.57, −54.85, −50.84, −40.14, −33.33, −25.13 and −22.65 kcal/mol, respectively. All the compounds could form hydrogen bonds with the amino acid residues of α-glucosidase. The binding sites of salvianolic acid A, rosmarinic acid and 4,5-dicaffeoylquinic acid were similar, and they could all form hydrogen bonds with ASN 235 and HIS 423. Isochlorogenic acid A, 3,4-dicaffeoylquinic acid, and danshensu exhibited similar binding sites due to their ability to form hydrogen bonds with GLY 161 and ASP 233. Formononetin could form hydrogen bonds with LYS 156, GLN 279, and HIS 280. It can be seen that hydrogen bonding is the main interaction between the above seven compounds and α-glucosidase. In addition, hydrophobic bonds and Pi-Pi stacked interactions have also been observed. For example, salvianolic acid A formed a hydrophobic bond with ALA 418, and 4,5-dicaffeoylquinic acid formed a Pi-Pi stacked interaction with PHE 314. The above amino acid residues have been reported to be close to the active site of α-glucosidase. 39 Notably, although ginsenoside Rd, ginsenoside Rb1 and ginsenoside F1 showed high inhibitory ability on α-glucosidase, the three ginsenosides were not successfully docked due to their large molecular weight.
Interaction Energy and Amino Acid Residues in the Interaction Between Active Ingredients and α-Glucosidase.

Component-Target Association Analysis
Based on these results, the action mode of “multiple components to multiple targets” was discovered in QLQX (Figure 7). Dihydrotanshinone Ⅰ, hydroxysafflor yellow A, salvianolic acid A and cryptotanshinone acted on the target of acetylcholinesterase. Rosmarinic acid, isochlorogenic acid A, 4,5-dicaffeoylquinic acid, danshensu, salvianolic acid A, 3,4-dicaffeoylquinic acid, formononetin, ginsenoside Rd, ginsenoside Rb1 and ginsenoside F1 acted on the target of α-glucosidase. Salvianolic acid A acted on both acetylcholinesterase and α-glucosidase. These results enrich the scientific connotation of “multiple components to multiple targets” of traditional Chinese medicines.

Association between chemical components and acetylcholinesterase targets (red) and α-glucosidase targets (blue) in QLQX.
Significance and Limitations of the Study
Many elderly people suffer from Alzheimer's disease and diabetes simultaneously, and discovery of inhibitors targeting acetylcholinesterase and α-glucosidase is of great significance. The significance of this study is mainly reflected in the following two aspects. Firstly, we explored the potential new application of the traditional herbal prescription of QLQX, which was originally used for chronic heart failure. In this study, the extract of QLQX was found to have inhibitory activity against both acetylcholinesterase and α-glucosidase, indicating that it might have a role in treating Alzheimer's disease and diabetes. Secondly, a series of selective inhibitors of acetylcholinesterase and α-glucosidase were discovered from the extract of QLQX. More importantly, salvianolic acid A was identified as an inhibitor that inhibited both acetylcholinesterase and α-glucosidase. These lead compounds are of great significance for the subsequent drug development.
The limitations of this study are mainly reflected in the following two aspects. Firstly, the detection of enzymatic activity was only conducted in vitro, and thus the lead compounds identified in this study are preliminary screening results. Therefore, the clinical value of this study needs further exploration. To investigate the potential of these compounds as drugs, a large number of pharmacological experiments need to be conducted. Secondly, this study does not fully explain the inhibitory effects of QLQX on acetylcholinesterase and α-glucosidase. The main reason is that the concentrations and interactions of compounds in QLQX remain unclear. Therefore, quantitative analysis of the components and exploration of compound-compound interactions in QLQX are necessary for further investigation.
Conclusion
A novel strategy that integrates time-based fractionation, LC-QTOF-MS, enzymatic activity assay, molecular docking and component-target association analysis was employed to explore the bioactivity of QLQX. With this strategy, a series of bioactive molecules, namely selective inhibitors on α-glucosidase or acetylcholinesterase, were discovered as lead compounds. Salvianolic acid A was found to be a dual-target inhibitor, which inhibits both α-glucosidase and acetylcholinesterase. The amino acid residues where they interacted with the enzyme were revealed by molecular docking. These findings suggest that QLQX has potential therapeutic value for Alzheimer's disease and diabetes. This study not only helps to clarify the action mode of QLQX, but also contributes to accelerating the process of drug discovery.
Experimental Section
Chemicals and Reagents
Acetylcholinesterase, α-glucosidase and p-nitrophenyl-α-D-glucopyranoside (PNPG) were purchased from Shanghai Yuanye Bio-Technology Co., Ltd Acetylthiocholine iodide (ATCI) and 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB) were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd QLQX and other chemicals were consistent with our previous work. 40
Sample Treatment and Analysis Conditions
QLQX was extracted and analyzed according to our previous work. 40 Differently, 1 mL of the extract was dried and redissolved in 0.1 ml of dimethyl sulfoxide (DMSO). The solution was diluted with phosphate-buffered saline (PBS) for the activity assay. The concentration of DMSO was below 2%. An Agilent HPLC system (Agilent Technologies, USA) was used for preparation. An Agilent ZORBAX SB-C18 column (9.4 × 250 mm, 5 μm) was used with a flow rate of 2 mL/min. Time-based fractionation was performed every 10 min for subsequent activity analysis. Each peak was collected for structural analysis on a 6550 QTOF mass spectrometer (Agilent Technologies, USA) with electrospray ionization (ESI) source. The detection mode was set as positive ion and negative ion respectively. The operation parameters were set as follows: the temperature of drying gas, 200 °C; the flow rate of drying gas, 11 L/min; the pressure of nebulizer gas, 35 psi; the temperature of sheath gas, 350 °C; the flow rate of sheath gas, 8 L/min; the capillary voltage, 4000 V. The m/z range was from 100 to 3000. The nozzle voltage was 1000 V. The fragmentor voltage was 75 V. Agilent Mass Hunter Qualitative Analysis software was used for data analysis.
Assay of Acetylcholinesterase Activity
The reaction principle is that acetylcholinesterase catalyzes the hydrolysis of ATCI to react with DTNB, generating a yellow substance with a maximum UV absorption wavelength of 405 nm. 41 The solutions for the reactions were prepared in PBS (0.1 M, pH = 8). The 96-well plate was placed in a microplate reader (Jinan Jiwei Medical Device Co., Ltd), and measurements were taken every 2 min for a total duration of 20 min. In the blank group, the sample solution was replaced with an equal volume of PBS. Each group was repeated 4 times.
Assay of α-Glucosidase Activity
The reaction principle involves the catalysis of the substrate (PNPG) by α-glucosidase to produce p-nitrophenol (PNP). PNP has a yellow color with a maximum UV absorption at 405 nm. 41 The solutions for the reactions were prepared in PBS (0.1 M, pH = 6.8). The 96-well plate was placed in a microplate reader, and measurements were taken every 3 min for a duration of 30 min. The sample solution in the blank group was replaced with an equal volume of PBS. Each group was repeated 4 times.
The inhibition rate (R) was calculated by the following formula:
Molecular Docking
The protein structures of acetylcholinesterase (PDB: 1C2B) from Electrophorus electricus and α-glucosidase (PDB: 3AJ7) from Saccharomyces cerevisiae were downloaded from Protein Data Bank. Molecular docking was conducted by Discovery Studio 2019. The structures of the above two proteins were prepared by removing water molecules and adding hydrogen atoms. CDOCKER was used to analyze the interactions between the compound and the protein. The pose cluster radius was set to 0.5. The information about interaction energy and amino acid residues was recorded.
Footnotes
Acknowledgements
This work was supported by the Liaoning Provincial Education Department project (JYTMS20231826 and JYTQN2023455), Shenyang Youth Science and Technology Innovation Talent Cultivation Project - U35 Top Youth Project (RC230846), National Natural Science Foundation of China (81803724) and the Youth Scientific and Technological Innovation Team Project at Liaoning University of Traditional Chinese Medicine.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Liaoning Provincial Education Department project, Shenyang Youth Science and Technology Innovation Talent Cultivation Project - U35 Top Youth Project, (grant number 81803724, JYTMS20231826, JYTQN2023455, RC230846).
Ethical Approval
Ethical Approval in not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
