Abstract
Objective
Postprandial hyperglycemia is considered an early sign of diabetes. Enzyme inhibitors, such as α-amylase and α-glucosidase inhibitors, are currently being studied as potential drugs for preventing postprandial hyperglycemia.
Methods
In this study, we investigated the effects of four purified 7-hydroxycoumarine derivatives from Ferula assafoetida: umbelliprenin, farnesiferol A, farnesiferol C, and samarcandin. We evaluated cell toxicity using the MTT method and also examined glucose uptake and inhibition of α-amylase and α-glucosidase enzymes in vitro. Additionally, we conducted a molecular docking study to investigate the mechanism of enzyme inhibition.
Results
The cell toxicity of the terpenoid coumarin derivatives (umbelliprenin, farnesiferol A, farnesiferol C, and samarcandin) on HepG2 cells was found to be approximately 28 to 37 µg/ml. The glucose uptake assay showed that these compounds (at a concentration of 25 µg/ml) were able to increase glucose consumption by HepG2 cells to a level comparable to that of the positive control (metformin at 50 µg/ml). Furthermore, umbelliprenin significantly inhibited the activity of α-amylase and α-glucosidase (by 35.07% and 4.98%, respectively). Molecular docking results indicated that umbelliprenin, with a farnesyl chain, had a more potent inhibitory effect.
Conclusion
Our findings suggest that umbelliprenin may be a valuable compound for controlling postprandial hyperglycemia and diabetes. However, further in vivo studies and clinical trials are necessary to validate these effects. While this research offers potential for the development of more effective compounds with coumarin structures, further studies are needed to confirm these findings.
Introduction
Diabetes is an important chronic metabolic disorder characterized by elevation in fasting and postprandial glucose levels resulting from due to insulin deficiency or insulin resistance. Postprandial hyperglycemia, which refers to high blood sugar levels after a meal, has been identified as an independent risk factor for cardiovascular disease. The underlying mechanism involves the occurrence of hyperglycemic spikes that induce oxidative stress in the body. Postprandial hyperglycemia is a plasma glucose level of 7.8 mmol/L (140 mg/dL) or higher 1 to 2 h after food intake.1,2
Absorption of glucose by the intestine depends on the function of the entire intestine. The pancreas and salivary gland in humans are responsible for synthesizing α-amylase, which plays a crucial role in breaking down dietary starch into disaccharides and trisaccharides through hydrolysis. Starch blockers are substances that inhibit amylase. α-amylase inhibitors (or starch blockers) lower the blood glucose level by delaying glucose absorption. 3 Furthermore, α-Glucosidase a glycoside hydrolase present in the brush border of the small intestine. α-Glucosidase plays a crucial role in glucose absorption and breaking down complex carbohydrates such as starch and glycogen into monomers. This enzyme cleaves α-1,4-glycosidic bonds between individual glucosyl residues of various glycoconjugates, including α- or β-linked glucose polymers. 4
F. asafoetida (Apiaceae family) grows wildly in Afghanistan and the central and southern mountains of Iran. Furthermore, it is commonly cultivated in the lands of central Asia. 5 Asafoetida (Anghouzeh in Persian) is an oleo-gum-resin obtained from the roots of the F. asa-foetida. The F. asa-foetida species is often considered the main source of asafoetida. 6
Asafoetida has been used as flavoring spice and folk herbal medicine for centuries. Asafoetida has a distinctive sulfur smell and bitter taste. Asafoetida was used in traditional medicine to treat various diseases such as epilepsy, gastrointestinal disorders, intestinal parasites, asthma, and influenza.7,8
Recent studies have also shown several pharmacological activities for asafoetida, such as antioxidant and anti-inflammatory, antimicrobial, anti-cancer, anti-diabetic, antispasmodic, hypotensive, and neuroprotective effects. 9
Asafoetida consists of three main fractions, including essential oil, gum and resin. The volatile fraction of asafoetida (10-17%) contains sulfur-containing compounds and monoterpenoids. The gum fraction of asafoetida (25%) contains monosaccharides and polysaccharides, which may contribute to its potential hypoglycemic effects. The resin fraction, which constitutes the majority of asafoetida (40-64%), contains various metabolites such as ferulic acid and its esters, coumarins, sesquiterpene coumarins, and other terpenoids.
Coumarins and sesquiterpene coumarins are most abundant in Ferula genus. Many different sesquiterpene coumarins have been reported from Asafoetida. Valuable sesquiterpene coumarins isolated from F. asa-foetida include umbelliprenin, furanesiferol A, furanesiferol B, furanesiferol C, galvanic acid, and gummosin. Overall, the diverse composition of asafoetida provides a range of bioactive compounds that can have targeted effects in several aspects.
Coumarin-derived dimers have C-C or C-O-C biaryl, terpene side chain linkages can inhibit α-glucosidase and cytochrome p450. Some of coumarin derivates have anti-inflammatory activity and antiviral activity. 10
Therefore, in this study, we evaluated the in vitro antidiabetic activity of coumarin compounds such as umbeliprenin, farnesiferol A, farnesiferol C, and samarcandin isolated from the dichloromethane extract of Asafoetida in comparison with in silico studies (Figure 1).

The chemical structures of coumarin compounds isolated from asafoetida.
Material and Methods
Chemicals
The purified compounds umbelliprenin, farnesiferol A, farnesiferol C, and samarkandin were extracted in a previous study 11 ; The HepG2 cell line was obtained from the Pasteur Institute of Iran, Tehran (Iran). RPMI 1640, penicillin-streptomycin solution, fetal bovine serum (FBS), 3-(4,5-dimethylthiazol-2yl)-2 and 5-diphenyltetrazolium bromide (MTT), and trypsin were purchased from Sigma. α-amylase, α-glucosidase, p-nitrophenyl-b-D-glucopyranoside, and glucose oxidase enzymes were purchased from Sigma.
Cytotoxicity Assay
HepG2 cells were cultured under incubation conditions such as 5% CO2 and 37 °C in RPMI-1640 containing 10 μS, penicillin (100 units/ml), and streptomycin (100 μg/ml). For in vitro cytotoxicity studies, 5000 HepG2 cells were seeded in 96-well plates. Different concentration of purified metabolites were added to the plates 24 h after cell seeding. Cell culture medium is considered blank. The plates were incubated for 48 h. Finally, MTT reagent (0.5 mg/ml) was added to each well. After incubation for 4 h an Epoch™ microplate spectrophotometer measured absorbance at 570 and 630 nm. IC50 concentration is the concentration of a compound that inhibits the proliferation of HepG2 cells in vitro by 50%.
Glucose Uptake Procedure on HepG2
The method was explained by Venter et al. 12 6000 HepG2 cells were seeded in 96-well plates, after 1 day without changing the culture medium, the cells were exposed to different concentrations of the purified metabolites. After 48 h, the supernatant was aspirated and the high glucose incubation medium containing 0.1% BSA and 8 mm glucose was added to 96-well plates. After three hours, 10 μl of the medium is removed and placed in a new plate with 200 μl of glucose oxidase enzyme to measure the amount of remaining glucose. After 15 min incubation at 37 °C, absorbance was measured at 492 nm. Metformin (0.1 μg/ml) was considered as the positive controls and untreated cells were considered as the negative control. The percentage of glucose uptake was calculated compared to untreated cells.
α-Amylase Inhibition Assay
The α-amylase inhibition assay was performed according to the method described by Odeyemi. 13 Different purified metabolites (25 μg/ml) were prepared in phosphate buffer and then added to 5 μl of enzyme into a 96-well plate. The plate was incubated for 10 min at 37 °C. Starch solution (20 μl) was added for starting reaction. The plate was incubated for 30 min at 37 °C. The reaction was stopped by adding 10 μl 1 M of HCl to each well followed by 75 μl of iodine reagent. Phosphate buffer (pH 6.9) was considered as blank (negative control) and acarbose (100 μM) was a positive control. The absorbance was measured at 580 nm.
α-Glucosidase Inhibition Assay
α-glucosidase inhibition assay was performed using the method described by Sagbo et al. 14 Briefly, 5 μl of the purified metabolites (25 μg/ml in phosphate buffer) was added to the solution of α-glucosidase into 96-well plate. In next step, 60 μl of 67 mM potassium phosphate buffer (pH 6.8) was added to each well. After incubation for 5 min, 10 μl of 10 mM ρ-nitrophenyl-α-D-glucoside solution was added. After incubation at 37 °C for 20 min, 25 μl of 100 mM sodium carbonate solution was added and the absorbance was measured at 405 nm. Deionized water was considered as a negative control and acarbose (100 μM) was used as a positive control.
Molecular Docking Studies
The crystal structures of α-amylase [PDB entry code: 1HNY] and α-glucosidase [PDB entry code:2ZE0] were obtained from the Protein Data Bank. The selection of protein codes was performed based on previous in silico study. 15 The co-crystallized ligand, all waters and non-interacting ions were removed from the receptor. Next, side chain atoms and all missing hydrogens were added. In the next step was to calculate the Gasteiger charges for the system. In the final step, the receptor was minimized and optimized using OPLS3. For ligand construction, these ligands were optimized using the OPLS3 (Optimized Potential for Liquid Simulations) level. Molecular docking studies were performed with XP mode in Glide (Schrödinger LLC, New York). Visualizing was performed using Chimera 1.8.1. 16
Statistical Analyses
All results are expressed as mean ± SD from three separate experiments performed parallelly. One-way ANOVA analysis of variance followed by the TUKEY post hoc test was used for statistical analysis between treatment groups. Significant differences between different groups are reported as *P < .05, **P < .01, and ***P < .001.
Results
Umbelliprenin, farnesiferol A, farnesiferol C, and samarcandin were tested for their antidiabetic properties in vitro. The outcomes were compared to a molecular docking study carried out using Schrodinger software.
Cytotoxicity
An in vitro MTT cytotoxicity test was used to assess the cytotoxic activity of asafoetida purified metabolites. Umbelliprenin, farnesiferol A, farnesiferol C, and samarcandin had IC50 values of 31.75, 28.14, 36.64, and 37.61 μg/ml, in that order. Compared to the others, farnesiferol A seems to be more cytotoxic (Figure 2). In a dose-dependent manner, farnesiferol A and farnesiferol C demonstrated their cytotoxic effects. Figure 2 demonstrated a significant cytotoxic effect of umbelliprenin, farnesiferol A, farnesiferol C, and samarcandin at 100 µg/ml (P < .001).

Cytotoxicity activity of umbelliprenin, farnesiferol A, farnesiferol C and samarcandin on the HepG2. Data expressed as mean ± SD (n = 3) (***P < .001).
Glucose Uptake
The rate at which hepatocytes absorbed glucose showed that asafoetida-purified metabolites could raise the amount of glucose that cells consumed. Treatment for postprandial hyperglycemia may involve increasing the rate at which glucose is absorbed. Figure 3 illustrates that the various examined sesquiterpene structures (25 µg/ml) were more effective in increasing the rate of glucose consumption than metformin (50 µg/ml). HepG2 cells’ ability to absorb glucose could be greatly increased by all of the isolated compounds (P < .001).
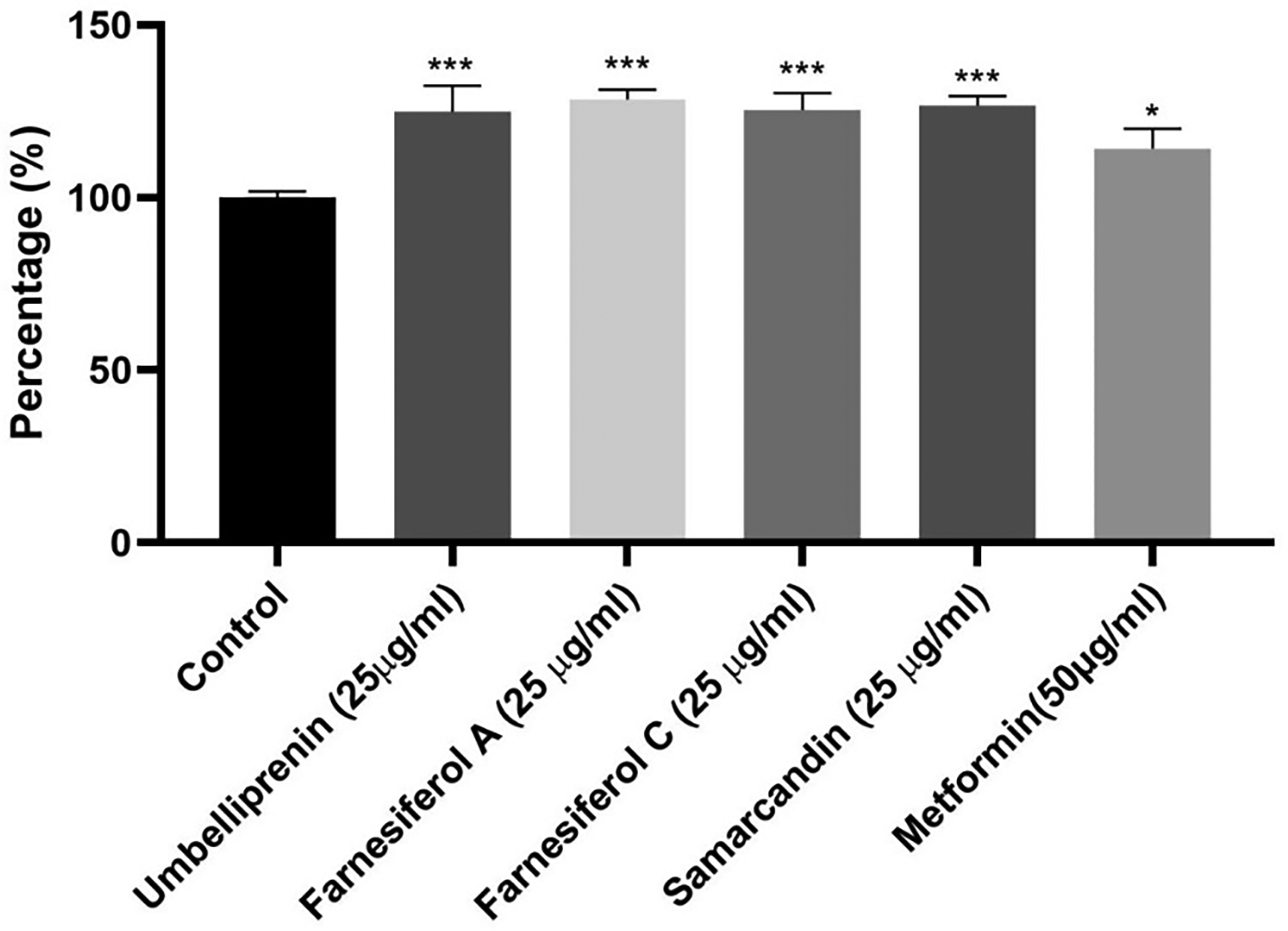
The percentage of glucose uptake on HepG2 after 48 h of exposure with the purified sesquiterpene from asafoetida. Metformin 50 µg/ml is positive control. (***P < .5) (***P < .01) (***P < .001).
α-Amylase Inhibition
The amount of α-amylase inhibition demonstrated the asafoetida purified metabolites’ potential for acting as inhibitors of starch hydrolase. The primary inhibitor is acarbose that could inhibit this enzyme by 28.77% at a concentration of 100 µM. The enzyme inhibition rate of umbelliprenin, farnesiferol A, farnesiferol C and samarcandin were 35.07, 18.82, 9.01 and 12.19%, respectively (Figure 4). Figure 4 illustrates that among the various examined sesquiterpene structures (25 µg/ml), farnesiferol A and umbelliprenin exhibited a significant inhibitory effect against α-amylase (P < .001).

α-amylase inhibition ability of coumarin compounds isolated from asafoetida. Acarbose 100 µM is a positive control (***P < .001).
α-Glucosidase Inhibition
The α-glucosidase inhibitors inhibit the intestinal carbohydrate decomposition. The α-glucosidase inhibition rate of umbelliprenin, farnesiferol A, farnesiferol C and samarcandin were 4.98, 4.45, 4.96 and 3.87%, respectively. Acarbose as the main inhibitor of α-glucosidase could inhibited this enzyme by 6.85% at a concentration of 100 µM (Figure 5). All of the isolated compounds could significantly inhibited the α-glucosidase (P < .01, P < .001).

α-glucosidase inhibition ability of coumarin compounds isolated from asafoetida. Acarbose 100 µM is a positive control (***P < .001).
Molecular Docking
The docking score of the asafoetida purified metabolites against α-amylase and α-glucosidase is shown in Table 1.
Docking Score for Coumarin Compounds Isolated from Asafoetida.

α-Amylase Docking Study
Although umbelliprenin showed the best inhibitory effect (Docking Score −4.408 kCal/mol) against α-amylase, acarbose demonstrated the most potent inhibitory activity (Docking Score −9.276 kCal/mol). According to the results of the molecular docking study, the central core of coumarin in 7-hydroxycoumarin derivatives can establish a pi-pi stacking connection with HIS 201. It can be considered a central core for α-amylase inhibitors. 2D and 3D visualization of binding interactions between assafoetida purified metabolites α-amylase was shown in Figure 6. Farnesyl chain in umbelliprenin could be effective in hydrogen donor activity and increase the α-amylase inhibitory activity.

(A) 2D and 3D visualization interactions of Umbelliprenin with α-amylase, (B) 2D visualization interactions of Samarcandin, Farnesiferol A and Faresiferol C with α-amylase (C) 2D and 3D visualization interaction of acarbose with α-amylase.
α-Glucosidase Docking Study
According to the molecular docking results, among the tested 7-hydroxycoumarin derivatives that were purified from asafoetida, umbelliprenin showed the best inhibitory effect against α-glucosidase (Docking score: −5.062 kCal/mol). 7-hydroxycoumarin in tested compounds can inhibit α-glucosidase through pi-pi stacking with PHE 144 and PHE 282. 2D and 3D visualization of binding interactions between assafoetida purified metabolites α-gucosidase was shown in Figure 7. Hydroxyl groups in acarbose structure has caused many hydrogen interactions such as ASP326, GLU256, ASN258, HIS203, ASN61 and ARG411.
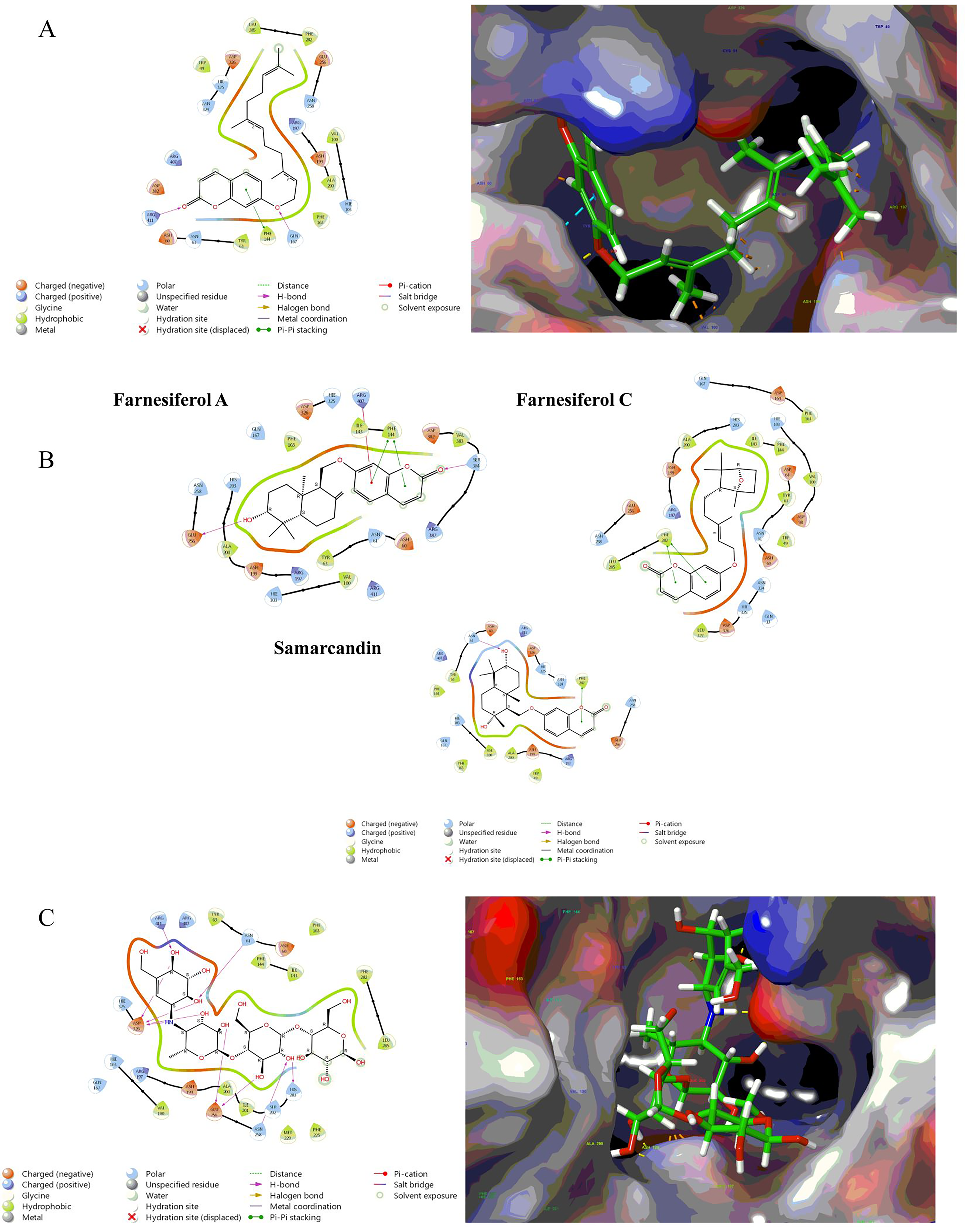
(A) 2D and 3D visualization interactions of Umbelliprenin with α-glucosidase, (B) 2D visualization interactions of Samarcandin, Farnesiferol A and Faresiferol C with α-glucosidase (C) 2D and 3D visualization interaction of acarbose with α-glucosidase.
Discussion
According to the literature, coumarin compounds showed significant cytotoxicity against cancerous cell lines. Yu and coworkers demonstrated that umbelliferone (a 7-hydroxycoumarin derivate) induced apoptosis in the HepG2. 17 Furthermore, umbelliprenin and Auraptene inhibited cell proliferation in M4Beu. Also, the toxic effects of umbelliprenin have been more than Auraptene. 18 Also, umbelliprenin has induced cytotoxic effects on lung cancer cells and adenocarcinoma through apoptosis induction. 19 Iranshahi and coworkers represent the strong cytotoxic effects of umbelliprenin and farnesiferol A against Raji cells activated by Epstein-Barr virus (IC50: 9.1 nM and 13.1 nM, respectively). According to the results of this study, prenylation in sesquiterpene coumarins structure could increase the antitumor activity. 20
In addition, the cytotoxicity of umbelliprenin, farnesiferol B and farnesiferol C on MCF-7 was about 50 μM. 21 Samarcandin has been shown to have toxic effects on AGS and WEHI-164 cancer lines. 22
Coumarin compounds have significant toxicity effects on different cancer lines and can induce apoptosis in cancer cells. Prenylation of coumarin structure and terpene coumarin formation also increase the cytotoxic activity.
Increasing the percentage of glucose consumption may be an effective treatment for postprandial hyperglycemia. Based on previous studies, oleo-resin from F. gummosa has the potential to prevent from diabetes complications by inhibiting the progression of hyperglycemia in a rat model of streptozotocin-induced diabetes. 23 Furthermore, the sesquiterpene coumarin and its semi-synthetic derivatives exhibited promising inhibitory effects against α-amylase, making them potential candidates for the future development of anti-diabetic agents. 24 Additionally, according to the literature, 9 different species of Ferula genus have been identified in articles for their anti-diabetic properties and totally the genus Ferula shows significant potential in reducing blood sugar. 25
Farnesiferol A has been demonstrated to enhance glucose uptake by hepatocytes more effectively than metformin. Previous studies have shown that conjugated coumarin structures with an imide ring in the fourth position can improve glucose utilization similarly to metformin. 26
In an in vitro enzyme inhibition assay, umbelliprenin exhibited significant inhibition of α-amylase and α-glucosidase enzymes (P < .001). The farnesyl chain, with three double bonds, acts as a hydrogen donor chain and shows efficacy in inhibiting α-amylase and α-glucosidase enzymes. The 7-hydroxycoumarin core can form a hydrogen bond with GLH 233 in α-amylase and ASN61 in α-glucosidase enzymes.
Coumarin and terpenoid coumarin compounds have demonstrated beneficial effects in managing diabetes and postprandial hyperglycemia. These compounds effectively inhibit α-amylase and α-glucosidase enzymes due to their coumarin ring structure. In 2022, Patil and coworkers conducted a molecular modeling study on over 80 coumarin derivatives as inhibitors of α-glucosidase and α-amylase enzymes. Through Pharmacophore Modeling, they illustrated that the central ring of coumarin can act as an inhibitor of α-glucosidase and α-amylase enzymes. 27
Anti-oxidant activity of terpenoid coumarins was studied in previous studies. Although, umbelliprenin did not indicate any significant antioxidant activity, it showed a potent inhibitory effect against soybean lipoxygenase. 28 Farnesiferol A and farnesiferol C increased intracellular ROS in dose dependent manner and induced apoptosis in cancer cell line.29,30 Furthermore, Abdel-Kader and coworkers demonstrated that samarcandin may be able to prevent testicular ischemia/reperfusion injury by mainly regulating the expression of Nrf2 and NF-B, which seems to mediate its promising antioxidant, anti-inflammatory, and antiapoptotic effects. 31
Limitation and Future Perspective
There are two major limitations in this study that could be addressed in future research. Firstly, the study focused solely on in vitro and in silico experiments and did not incorporate in vivo effects. The second limitation pertains to the need for more precise in silico studies, such as molecular dynamics and structure-activity- relationship (SAR). In conclusion, we recommend that future research endeavors include: 1) Investigating the in vivo impacts of terpenoid coumarin compounds as anti-diabetic agents. 2) Exploring the effects of terpenoid coumarin compounds on additional mechanisms involved in diabetes regulation, such as inhibiting aldose reductase and dipeptidyl peptidase. 3) Conducting SAR studies on terpenoid coumarin compounds to develop compounds that are more potent.
Conclusion
Based on the findings of this study, 7-hydroxycoumarin derivatives from asafoetida show potential effectiveness in treating postprandial hyperglycemia by inhibiting α-amylase and α-glucosidase enzymes, as well as enhancing glucose uptake by hepatocytes at non-cytotoxic concentrations. Among these compounds, umbelliprenin stands out as the most potent inhibitor of α-amylase and α-glucosidase enzymes. Therefore, coumarin terpenoid compounds, particularly umbelliprenin, hold promise as valuable agents for managing diabetes. While these results can inform the development of more potent compounds with coumarin structures, further in vivo studies and clinical trials are necessary to validate these effects
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable to this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Research Affairs of Mashhad University of Medical Sciences, the Specialized Research Fund (No. 992030) for the Pharmacy doctorate Program.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable
