Abstract
Introduction
Fruit wastes have excellent therapeutic potential, and their utilization is considered as an extremely attractive and cost-effective technology.
1
Commercially, it has been estimated that the most commonly consumed fruits are citrus fruits such as Mandarin, Kinnow, Lemon, Sweat orange, Citron and Grapefruit. Globally, 100 million tons of citrus fruit are cultivated per season, of which only 26% is utilized to meet the juice requirements.
2
The food industry pays little attention to the environmental pollution caused by its waste. Fruit wastes include pomace, peel (flavedo), rind, seeds and pith (Albedo). These fruit residues are natural sources of phenolic and flavonoid contents and have a rich carbohydrate polymer profile comparable to their pulps.
3
Tropical fruit
C. reticulata albedo is concentrated with antioxidants, carbohydrate polymers, polyphenols, enzymes, vitamins, minerals, essential oils, carotenoids and dietary fibres, especially pectin. Albedo is the white fibrous tissue attached to the peels of fruits, which protect the pulp inside. 5 A literature survey has disclosed that the consumption of pectin-rich products helps to control cholesterol and blood sugar levels. 6 Citrus albedo is rich in flavonoids such as naringenin and nesperidin, which enhance the body's natural defense system by boosting the activity of white blood cells. 7 Moreover, carbohydrate polymers extracted from citrus albedo include pectic polysaccharides, arabinans, cellulose, xyloglucans, heteroxylanes, heteromannans, soluble sugars, xylose, starch and neutral sugars. 5 These bioactive polysaccharides regulate the body's physiology by exhibiting antibacterial, anti-stress, antidiabetic, antiviral, immunomodulatory, antioxidant, renoprotective and anticancer properties. Citrus waste-derived nutraceuticals have demonstrated various biological characteristics such as anti-carcinogenicity, anti-allergenicity, anti-mutagenicity and anti-aging capacities. 5 Phyto-derivative antioxidant enzymes provide a cost-effective and sustainable way for wastewater treatment in various industrial sectors, 8 and the presence of these enzymes in the albedo extract of C. reticulata is also evidenced by our present study.
In developed countries, C. reticulata albedo undergoes various stages of treatments such as bioremediation, biochemical extraction, anaerobic fermentation and biotransformation for the production of many useful products like animal feed, bio-peroxidase catalysts, biodegradable plastics and soil fertilizers. 5 The primary goal of these processes is to maintain environmental sustainability and support the economy. Additionally, natural bioactive polysaccharides are considered biomedically valuable in the pharmaceutical industry due to their non-toxicity, biocompatibility, biodegradability and unique therapeutic activities. 9 Although there are numerous reports on the medicinal applications of raw fruit material, this is the first investigation on the antioxidant profile of the albedo extract of C. reticulata, considering two different aspects. The first aspect involves the proximate analysis of crude C. reticulata albedo samples and the preparation of an albedo extract using methanol. The methanolic extract of C. reticulata albedo was biochemically characterized via Liquid Chromatography with tandem mass spectrometry (LC-MS/MS) and FTIR analysis to validate the presence of phytochemical and polysaccharide components. The second aspect focuses on evaluating the antioxidant defense potential and carbohydrate profile of the prepared extract. In-vitro cytotoxicity evaluation of the albedo extract was performed through MTT assay using HepG2 cells.
Results and Discussion
Proximate Composition
Quantification through proximate analysis provides the macronutrient composition of C. reticulata albedo, including ash, moisture, crude fibre, crude protein and lipid contents. The results (Table 1) indicate that the albedo has an excellent moisture-holding capacity of 52.67 (g/100−g FW) which is considerably higher than the moisture content (15.00%) of citrus sinensis as evaluated by a previous study. 10 Recent studies have disclosed that moisture helps plants survive in wilting conditions and maintains the refreshing character of fruit. 10 In addition to its high moisture content, albedo showed a comparatively higher crude fibre content of 30.80 (g/100−g FW) than crude protein. The fiber composition of the C. reticulata albedo provides several health benefits such as reducing plaque formation along artery walls by lowering blood cholesterol levels, which ultimately prevents depression, aging, pulmonary disease, and colorectal cancer. 10 Citrus fibres are also used to extend the shelf-life of meat products, which is attributed to its antioxidant properties. 11 With relatively lower protein (9.62 Kcal/100−g FW) and lipid (5.58 g/100−g FW) content, C. reticulata albedo is a termed as a moderately caloric food. 12 The protein in orange albedo primarily consist of amino acids such as leucine, aspartic acid, glycine, glutamic acid, serine, alanine, valine, and lysine. 5 It has also been reported that citrus albedo sections generally have lower protein, ash, and lipid contents compared to other parts of the fruit. 10 In comparison to proximate analysis for biochemical profiling by Emelike and Akusu, 13 the high ash content (3.30 Kcal/ 100−g FW) confirmed that the albedo section of C. reticulata is rich in minerals. These findings were consistent with those reported in the literature by Osfor et al 14
Proximate Compositions (g/100−g Fresh Weight) and Carbohydrate Profiling (mg Saccharides/g Dry Weight) of the Albedo Extract of C. reticulata.

Results are expressed as mean ± SD.
Carbohydrate Quantification
Carbohydrate analysis revealed a rich polysaccharide profile in the albedo extract of C. reticulata, as reported in (Table 1). These observations were comparable to findings in Dimitrova et al, 15 where analysis was carried out on the leaves of Morus species. A comparatively lower amount of total soluble sugars (TSS), 13.5 mg/g DW, was found through analysis. These values were lower but comparable to the soluble sugars of albedo fractions of lemon. 16 TSS be isolated from albedo extract can be used in drugs formulation to enhance the bioactivity of other ingredients present in drugs. 17 It has been reported that these saccharides, especially sucrose, can be used in antiseptic bandages to prevent the wound putrefaction. 18 The albedo extract of C. reticulate contained 6.5 mg/g DW of maltose, providing an immediate source of glucose, and higher glucosamine content (5.05 mg/g DW) was also observed. Oral intake of glucosamine sulphate as a nutritional supplement has been shown to significantly reduce the prevalence of osteoarthritis by effectively binding to DNA in synovial cells and chondrocytes. 19
Moreover, the albedo extract contained a tremendous amount of neutral sugar, specifically D-xylose (35.5 mg/g DW), consistent with the levels found in grapefruit. 20 Recent investigations on COVID-19 have found that phyto-extracted D-xylose exhibits antiviral, antibacterial, anti-glycemic and anti-inflammatory properties against lung infections. 21 Starch, obtained as a white powder, yielded 26% on a dry basis, demonstrating the nutritional value of C. reticulata albedo. The cellulosic content (12.2 mg/g DW) of the albedo was higher than that of other polysaccharides. The average pectin content (18.5 ± 0.05 mg/g DW) confirmed the potential use of albedo in anti-metastatic therapy, as previously reported by Nicholas et al. 22 This result is consistent with earlier studies, indicating that the albedo section of citrus fruit contains up to 60% of pectin polysaccharides. 5 While no previously published data specifically on the carbohydrate analysis of C. reticulata albedo exists, these findings are comparable to findings in literature. 23
Quantification of Bioactive Components and Scavenging Potential
Bioactive component analysis revealed a rich profile of flavonoids and phenolics in the methanolic extract of C. reticulata albedo. These secondary metabolites defend the fruit against microorganisms through various signaling pathways and contribute to regulating their development through auxin transport. 24 The antioxidant activity of these bioactive components (phenolics and flavonoids) and their radical scavenging potential were evaluated (Table 2). The TPC analysis of the albedo revealed that methanolic extract at 100 mg/mL had the highest TPC of 2.30 mg GAE/ g DW. As the extract concentration decreased from 75 to 25 mg/mL, the TPC values also reduced from 2.2 to 2.05 mg GAE/ g DW, respectively. A similar trend was reported in another study. 25 However, these findings are not in line with a previous study, 26 which reported relatively lower TPC with 1882 µg/g FW and 7667 µg/g FW for flavedo extract of citrus fruit (lemon and tangor), respectively. 26 The high TPC in the current study suggested that citrus albedo was a good source of phenolic compounds. Major phenolic compounds identified by LC-MS/MS analysis included ferulic acid, vanillic acid, p-coumaric acid, resveratrol and caffeic acid.
Quantification of Bioactive Components and Antioxidant Enzyme Analysis of the C. reticulata Albedo Extract.
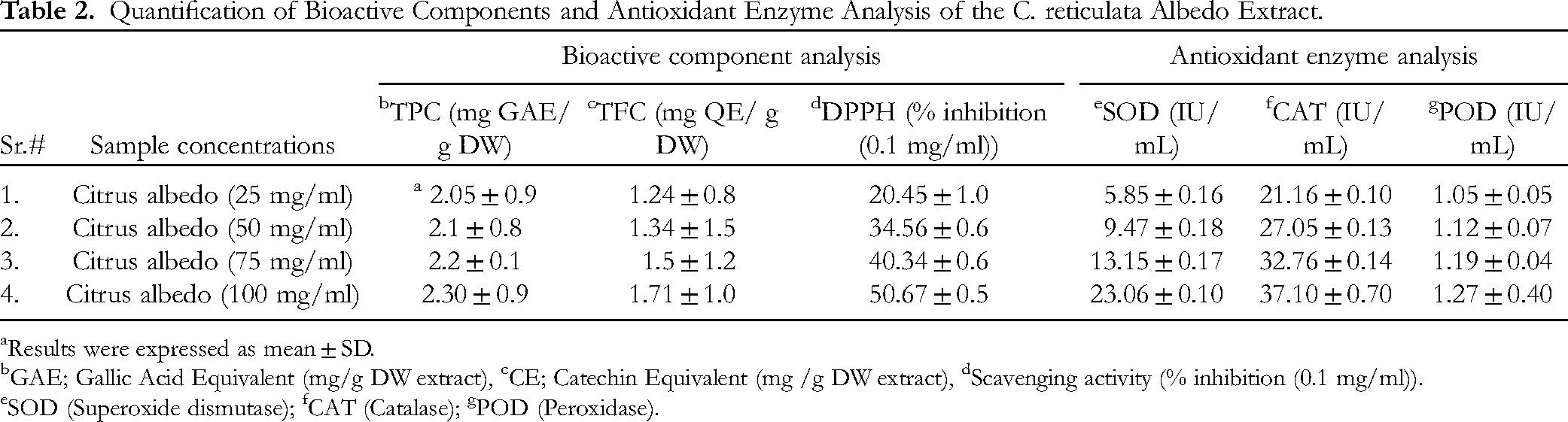
Results were expressed as mean ± SD.
GAE; Gallic Acid Equivalent (mg/g DW extract), cCE; Catechin Equivalent (mg /g DW extract), dScavenging activity (% inhibition (0.1 mg/ml)).
SOD (Superoxide dismutase); fCAT (Catalase); gPOD (Peroxidase).
Similarly, the TFC was also increased to 1.24, 1.34, 1.5 and 1.71 mg QE/g DW in the descending sequence of stock solutions of albedo extract, ie 25, 50, 75 and 100 mg/mL, respectively. The TFC in the present study was lower than those reported by Chang et al for the albedo section of Tambo. 25 The variation in TFC compared to previous studies could be associated to environmental conditions during cultivation or the solvents used for extraction. 27 Major flavonoids identified by LC-MS/MS analysis included quercetin, apigenin hexoside, hesperidin, naringenin, and pentahydroxy trimethoxy flavone. The quantification of C. reticulata albedo phytoconstituents against DPPH revealed excellent excellent radical scavenging activity of 50.67% per 100 mg/mL of extract. The least scavenging potential (20.45%) was observed at 25 mg/mL of methanolic extract of albedo. Other studies suggested that plants with varying degrees of antioxidant activity might be utilized to prevent oxidative stress and related disorders, 28 aligning with findings in the literature. 29
Antioxidant Enzyme Analysis of the C. reticulata Albedo Extract
Naturally occurring antioxidant enzymes in plants serve as reservoirs of bioactive components and aid in combating severe environmental challenges through various mechanisms of quenching reactive oxygen species. A significant difference in activity at varied concentrations of enzyme extract was observed (Table 2). The maximum SOD activity of 23.06 IU/mL was shown at 100 mg/mL of enzyme extract. In contrast, activity was reduced to 13.15, 9.47 and 5.85 IU/mL as the extract concentration decreased to 75, 50 and 25 mg/mL, respectively. These values were lower than that of those evaluated SOD activity in citrus albedo infected with Penicillium digitatum. 30 Catalase activity also gradually increased in a similar pattern, with values of 21.16, 27.05, 32.76 and 37.10 IU/mL at enzyme extract concentrations of 25, 50, 75 and 100 mg/Ml, respectively. Our results of catalase activity were in line with the previous study. 31 Evidence of rich accumulation of catalase in the albedo of C. reticulata suggests its role in defending the fruit from aging and senescence by scavenging hydrogen peroxide generated during fatty acids (β-oxidation) and photo-respiratory oxidation. 32 Peroxidase enzyme analysis of the albedo sample exhibited the highest POX potential of 1.27 IU/mL at an enzyme concentration of 100 mg/mL, which was significantly higher than the1.05 IU/mL observed at an enzyme concentration of 25 mg/mL. These findings were significantly higher than the POD activity values of flavedo (0.05 IU/mL) reported in previous study. 33 Recent studies have revealed that plant peroxidases, also known as heme groups-containing glycoproteins, plays a versatile role in reducing oxidative stress via hydrogen peroxide degradation, toxic substances elimination and defense against insects by triggering hormone signalling pathways. 33 To our knowledge, no reports have been found in the literature on the antioxidant enzyme analysis of C. reticulata albedo extract. However, it has been reported that the synthesis of these synthetic modulators protects fruits against oxidative damage during citrus fruit development. The ability of plants to cope with the multivariate natural environment depends on the scavenging activities of antioxidant enzymes and is a key factor in ensuring high yield. 34
Characterization of the Methanolic Extract of C. reticulata Albedo
Functional Group Analysis via FTIR Spectrometry
The interferometer of the FTIR spectrometer captured all the absorbed wavelengths of the albedo sample and the detector then analyzed the resulting signal to provide a spectrum with characteristic functional group regions (3500-1600 cm−1) and fingerprint regions (1500-800 cm−1). The spectral pattern (Figure 1) gives novel information on the molecular structure of organic molecules in C. reticulata pith. Phyto-constituents were detected in the functional group region with absorptions at 3400.45, 3235.24, 2900.48, 2750.23, 2345.67, 2230.90, 1850.10 and 1633.54 cm−1 while intense sharp peaks at 1498.06, 1311.23, 1190.99, 1123.45, 1051.18, 980.82, 960.78, 940.0, 910.78, and 799.0 cm−1 in the fingerprint region confirmed the presence of polysaccharides in the albedo sample. The broad absorption band at 3500–3000 cm−1 was due to the O-H stretching vibration of phenolic compounds like catechin and hydroxyl flavones. 35 The intense peaks at 2900–1900cm−1 were associated with C-H stretching and bending vibrations of saturated and aromatic compounds, respectively. 36 A sharp absorption peak at 1633.54 cm−1 was attributed to galactose or mannose ring stretching due to bending vibrations of the carbonyl functional group (-CHO). 37 The strong absorptions at 1051.18 & 910.78 cm−1, 980.82 & 1123.45 cm−1 and 960.78 & 940.0 cm−1 were the main characteristic bands of glucose, sucrose and fructose, respectively. 38 Moreover, the methanolic extract of C. reticulata albedo also showed sharp peaks, at 1311.45 & 1190.99 cm−1, which were associated with C-O & C-O-C stretching of polysaccharides. The extremely minor band at 799 cm−1 was due to skeletal vibrations of carbohydrates. The bending vibrations of NH at 1498.06 cm−1 give evidence of the presence of protein content in the albedo extract.

FTIR spectral pattern of methanolic extract of C. reticulata albedo.
LC-MS/MS Analysis of the C. reticulata Albedo Extract
Chromatographic extraction of major phytoconstituents from the albedo sample of C. reticulata yielded spectra with 78 prominent peaks along with their respective retention time in both positive and negative modes. These peaks were identified and compared with the NIST reference library. Each peak in the chromatograms of Figure 2 corresponded to a specific m/z compound or group of compounds present in the albedo extract. Table 3 demonstrated characteristic information and biomedical importance of bioactive compounds separated through LC-MS/MS analysis. Major identified bioactive compounds included tartaric acid, indole-3-carboxylic acid, p-coumaric acid, caffeic acid, ferulic acid and caftaric acid, which are potent antioxidant, exhibit bactericidal, anti-inflammatory, anti-aging, anti-diabetic, anti-cancer, neuroprotective, hepatoprotective properties and are effective in cutaneous wound healing by NRF2 expression.

LC-MS/MS chromatograms of methanolic extract of C. reticulata albedo, based on m/z (mass to charge ratio) value plotted against relative abundance. Peaks are the fingerprints of bioactive components in both positive and negative modes.
Major Phytoconstituents of Albedo Extract of C. reticulata Identified by LCMS-MS Analysis.
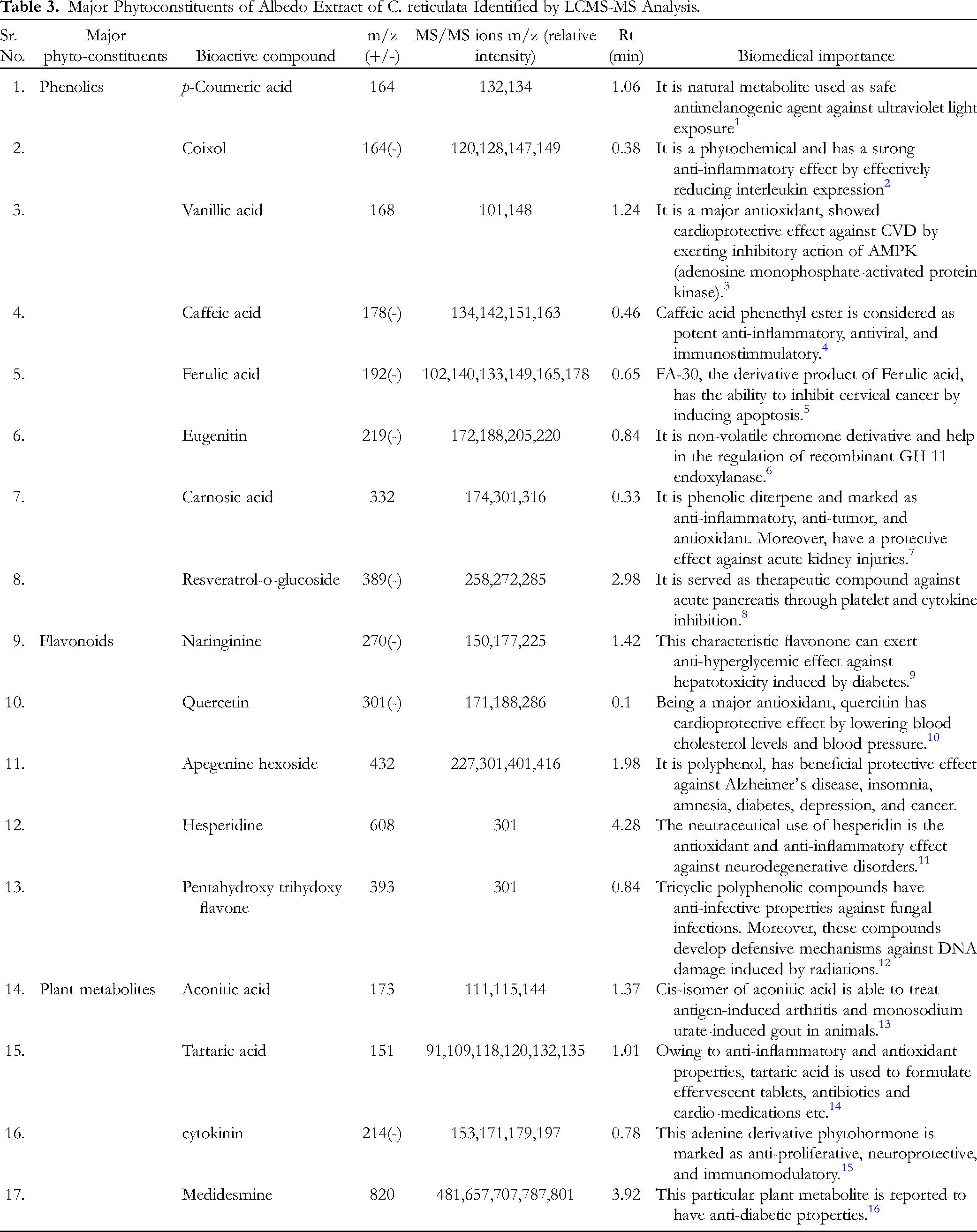
Furthermore, these compounds promote melanin synthesis by triggering tyrosine gene expression and can reduce hyperpigmentation in the skin. In addition, phenolics and flavonoids act as defensive compounds against biotic and abiotic stresses for plants. 39 Plant metabolites, with their antimicrobial properties, serve as chemical barriers against pathogens and are used as flavoring, coloring and texturizing agents in various food industries. 28
Cytotoxic Assessment of the C. reticulata Albedo Extract
The cell viability of four different concentrations of the albedo extract was evaluated using MTT assay with the HepG2 cell line (Table 4). The results revealed that treatment of selected cells with albedo extract and cisplatin induced apoptosis in a dose-dependent manner. The maximum apoptotic cells (IC50= 49.42 µg/mL) were observed at the higher concentration of 100 mg/mL of the albedo extract of C. reticulate used in this study. In contrast, a higher number of cells survived at concentrations of 50 mg/mL and 75 mg/mL, with survival rates (IC50) of 65.34 µg/mL and 57.79 µg/mL, respectively. The minimum cytotoxic potential (higher cell viability) was shown to be IC50 = 79.64 µg/mL at the concentration of 25 mg/mL of the albedo extract. Similar findings of increasing concentration of tested compounds leading to decreased cell viability were also reported by Bhandari et al 40 Previous cytotoxic evaluation of the methanolic extract of Lagerstroemia Speciosa, against the HepG2 cell line has demonstrated that bioactive components of plants cause apoptotic changes such as cytoplasmic and chromatin condensation, nuclear damage and cell shrinkage. 41 A study has depicted that 59% of phyto-based bioactive molecules have been approved for cancer therapy for years. 42 Therefore, the present in-vitro cytotoxic evaluation suggested using C. reticulata albedo in cancer treatment.
in Vitro Cytotoxic Evaluation of the Albedo Extract of C. reticulata Against HepG2 Cell Line.

IC50 (µg/ml of citrus albedo extract which cause cell death by 50% vs control).
The values represent relative number of viable cells, determined by ELIZA plate reader and are the mean ± S.D of triplicate samples (n = 3).
Blank (without any extract), c Cis-Platin (5 µl/mL).
LC-MS/MS and FTIR analysis of Citrus reticulata methanolic extract detected a rich composition of phytomolecules. The major identified bioactive compounds included tartaric acid, indole-3-carboxylic acid, p-coumaric acid, caffeic acid, ferulic acid, and caftaric acid. These compounds are potent antioxidant, bactericidal, anti-inflammatory, anti-aging, anti-diabetic, anti-cancer, neuroprotective and hepatoprotective. According to the findings of the research, C. reticulata albedo could be used to develop a variety of commercial products in industries such as food, pharmaceuticals and cosmetics as skin protectors
Although the study confirms the hepatoprotective properties of Citrus reticulata methanolic, however, further isolation and purification of the detected bioactive components are needed. This will enable their use as a replacement for synthetic additives in the food industry as well as in pharmaceuticals.
Conclusions
Flavonoids, phenolics and carbohydrate polymers are major value-added products extracted from C. reticulata albedo. Characterization via LC-MS/MS and FTIR analysis further confirmed these results, suggesting that citrus albedo extract could be utilized in phytotherapy, hormone replacement therapy, osteoporosis and coronary disorders. Analysis by enzyme assays revealed that the occurrence of three major antioxidant enzymes: catalase, superoxide dismutase and peroxidase. Therefore, C. reticulata albedo might be a source of exogenous enzyme for the organism's body. C. reticulata albedo can be used for biofuel production due to the presence of soluble sugars. The majority of the analyzed polysaccharides (cellulose, pectin, starch, and lignin) exhibited the potential to regulate body's anatomy due to their biological and pharmacological activities such as antimicrobial, anti-stress, anti-diabetic, anti-virus, immunomodulatory, antioxidant, renoprotective and anticancer. . Therefore, the processing of citrus waste should be mandated over food companies. Off-white albedo fibre was separated from C. reticulata peels, and an extract was prepared using the maceration method. Green synthesis was also carried out and analytical techniques including LC-MS/MS, SEM, XRD, UV-visible and FTIR were employed for the characterization of green synthesis and the presence of different phytoconstituents in the citrus peels. Antibacterial potential against a panel of gram-positive and gram-negative bacteria was also confirmed using the agar well diffusion method. To evaluate their clinical application in tropical healing, open wounds were induced on the dorsal surface of experimental animal (rabbits). Wound reduction and histopathology studies of both the green synthesis and crude extract were conducted. Based on biochemical potential and animal model study, future research will focus on molecular studies to highlight the biomedical potential of hidden bioactive constituents in citrus peel.
Materials and Methods
Chemicals and Reagents
All the chemicals and organic solvents used in this study were of analytical grade. Analyses were carried out in double distilled water. DPPH (2,2-diphenyl-1-picryl-hydrazyl-hydrate), pyrogallol and buffers for enzyme analysis were procured from Sigma Aldrich.
Sample Collection and Preparation
The fresh fruits of C. reticulata were purchased from the local fruit market in mid-January based on visual assessment of healthy and freshness, authenticated by Taxonomist Ms. Zahira Rafique Lecturer, Department of Basic Sciences (Section Botany), University of Veterinary and Animal Sciences (UVAS) (Jhang Campus), Pakistan. The albedo from C. reticulata was carefully peeled off using forceps. Approximately 1.5 Kg albedo was obtained from 50 kg of fresh peels (Figure 3a). The sample was shade-dried, weighed and stored in clear cellophane bags. Fifteen grams of dried albedo powder was mixed in 150 mL of methanol, and the whole mixture was magnetically stirred at 60 °C for 15 min. The extracted solution was filtered through Whatman No. 1 filter paper. The sample was then subjected to low-temperature dehydration process using the freeze-drying method. The lyophilized sample (Figure 3b) of albedo extract was then stored for further analysis.

(a) Citrus albedo and (b) Lyophilized CRP extract.
Proximate Composition of the C. reticulata Albedo
The proximate composition of albedo components including ash, moisture, fiber content, fat and crude protein contents was determined according to the AOAC method. 43 Lipid extraction from the albedo sample was performed using the standard soxhlet extraction method, as described by Emelike and Akusu. 13 Briefly, 2 g of dried sample was loaded into the porous thimble and placed in the Soxhlet apparatus using petroleum ether as an organic solvent. Moreover, lipid content was determined after eight hours of complete extraction. Crude protein from albedo was estimated using the micro-Kjeldahl method. 44
Carbohydrate Analysis of the C. reticulata Albedo
The prepared albedo extract of C. reticulata was quantified using carbohydrate analysis to ensure its biomedical importance. Total soluble sugars were estimated by calorimetric assay using anthrone reagent under acidic conditions and absorbance was taken spectrophotometrically at 620 nm as reported earlier by Calugar et al.
45
The DNS method was used to evaluate maltose concentration in citrus albedo extract using maltose as standard.
46
Neutral sugar (xylose) content was quantified, while, cellulose, pectin and glucosamine contents were evaluated following established procedure.
47
Starch was extracted from the methanolic extract of albedo and starch yield was estimated by using the following equation: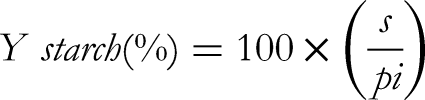
Total Phenolic and Flavonoid Content Estimation in the C. reticulata Albedo Extract
To quantify the total phenolic and flavonoids of citrus albedo, stock solutions of varied concentrations (25 mg/mL, 50 mg/ml, 75 mg/mL, and 100 mg/mL) of the extract were prepared. The total phenolic content (TPC) of the sample was evaluated by the Folin-Ciocalteus (F.C) method with minor modifications, as described by Ahmad et al. 29 To quantify TPC, 300 µL of each prepared stock solution was transferred into a sterilized test tube containing a mixture of F.C reagent (500 µL) and 800 mM Na2CO3 solution (1 mL). All the test tubes were vortexed for homogenization, and the samples were left at room temperature for 2 h. Then, 500 µL of each sample was transferred into a cuvette, and absorbance was taken at 765 nm against the blank containing all the reagents except the extract. The concentration of TPC was documented as mg Gallic acid equivalent (GAE) per g DW of extract. Total flavonoid contents (TFC) in albedo extract were quantified following the method described by Ahmad et al. 29 Briefly, 1 mL of each stock solution was transferred into a sterilized test tube, following the addition of 400 µL of 5% Na2NO3 and the mixture was diluted by adding 6 mL distilled water. After 5 min of incubation, 700 µL of 10% Al2Cl3 and 3 mL of 1 M NaOH solution were added to the mixture. Then, 500 µL of the resulting mixture was transferred into a cuvette, and the absorbance was taken at 510 nm against blank, containing all the reagents except the extract. Quercetin (10 mg/ml) was used as a standard solution. The concentration of TFC was documented as mg catechine equivalent (CE) per g DW of extract.
Determination of Free Radical Scavenging Potential Using DPPH Assay
The free radical scavenging potential of albedo extract was evaluated using a spectrophotometric DPPH method as described by Ahmad et al.
29
Stock solutions of C. reticulata albedo extract in four different concentrations (25-100 mg/mL) were prepared in an organic solvent. Then, 300 µl of ethanolic solution of DPPH was mixed with 40 µl of each stock solution of albedo extract. The mixture was allowed to stay in the dark for 30 min at room temperature for reaction, and then absorbance of the solution was taken at 517 nm to calculate the antioxidant activity as inhibition (%) against DPPH free radical using the following equation:
Antioxidant Enzyme Analysis of the C. reticulata Albedo Extract
The antioxidant defense system of bioactive components associated with the albedo section of C. reticulata was accessed through enzyme analysis. Enzyme extracts were prepared by mixing albedo extract in respective buffers and centrifuged at 15000 rpm for 20 min as described by Hameed et al 48 For antioxidant estimation, the resulting supernatant was used to prepare enzyme extract of varied concentrations (25 mg/mL, 50 mg/mL, 75 mg/mL and 100 mg/mL) and results were compared. Superoxide dismutase (SOD) activity was assessed in-vitro using pyrogallol as an autoxidant. Initially, 0.2 mL of prepared enzyme extract was mixed in 2.74 ml of tris-HCl buffer along with the addition of 60 µL pyrogallol and incubated at room temperature for 10 min. This preparation (500 µL) was transferred to cuvette and the SOD activity was determined at a wavelength of 420 nm. 49 The results of enzyme activity were expressed as SOD in IU/mg of protein. Catalase (CAT) activity in the albedo of C. reticulata was estimated by following the procedure described by Zhang et al 50 Briefly, 200 µL H2O2 was added in 600 µL prepared enzyme extract. A UV-Vis spectrophotometer was used to measure the change in absorbance at 240 nm at intervals of 15 s for 5 min, and catalase activity was expressed as IU/mg of protein. Peroxidase (POD) activity was assayed using method 48 with minor modifications. Evaluation of the in-vitro antioxidant activity of POD in albedo extract of C. reticulata was initiated by adding 0.3 mL enzyme extract in 2.4 ml phosphate buffer along with the addition of 0.6 mL guaiacol and mixing it well. Then 20 µL of 5% H2O2 was added to the prepared homogeneous mixture and incubated at room temperature for 30 min. POD estimation was based on an absorbance change of 0.1 units per minute at 470 nm and was expressed as IU/mg of protein.
Characterization of the C. reticulata Albedo
FTIR-Analysis
A Perkin Elmer infrared spectrometer was used to record the FTIR spectral of the albedo extract and quantify the functionality of phytoconstituents and polysaccharides. The sample was prepared by mixing 3 mg of lyophilized albedo extract in a small quantity of potassium bromide (KBr). The mixture was then loaded onto the KBr disc of the spectrometer. When the prepared sample was exposed to the infrared region of the electromagnetic region, a spectrum of fine absorption bands was obtained in the wave number range of 1000–3500 cm−1 with a resolution accuracy of 1cm−1.
Analysis of Phenolic Compound Composition by LCMS-MS
Identification of phenolic compounds in the albedo extract of C. reticulata was carried out through liquid chromatography equipped with mass spectrometry method. 51 Initially, the sample was prepared via solid phase extraction (SPE) using a multichannel SPE cartridge with Strata C18 columns and vacuum pump. The column was washed with methanol/water (50/50) to activate its sorbent surface. Then, 5 mL of methanolic albedo extract was loaded onto the SPE columns, followed by washing with 1 mL methanol/water (30/70). A full vacuum drying was applied for 5–10 min to remove residual solvent. To recover phenols, the cartridges were eluted 1 mL acetonitrile, 2 mL methanol, and 2 mL of 5% formic acid in methanol. All three fractions were combined before being subjected to LC/MS analysis. Due to the varying nature of phenolic compounds in albedo extract, each phytochemical was eluted from the LC column at a specific retention time (min) in both positive and negative ion modes. An LC-MS run provided retention time (RT) information in a chromatogram, mass-over-charge ratio (m/z) in the MS spectrum and relative ion abundance for each particular ion. MS signals were detected throughout the range of chromatographic separation and formatted in a three-dimensional map, which interpreted the data from a single LCMS-MS run.
Cell Viability (MTT) Assay
MTT assay was used for in-vitro cytotoxicity assessment of the albedo extract of C. reticulata following a method. 52 HepG2 cells were maintained on Dulbecco's Modified Eagle Medium (DMEM) supplemented with 100 mg/mL streptomycin and 10% heat-inactivated fetal bovine serum at 37 °C in a humidified atmosphere of 5% CO2 in air. After 24 h of incubation, the growth of the of HepG2 cell culture was observed using a reversal microscope. Stock solutions of four distinct concentrations (25-100 mg/mL) were prepared using albedo extract. HepG2 cells at a concentration of 5 × 104 cells/well were seeded into 96-well plates with a flat bottom following the incubation of 16 h. HepG2 cells were then treated with prepared stock solutions of albedo extract, using cisplatin (5 µL/well) as a positive control, and growth media only as a negative control (blank). After 48 h of treatment, the cell culture medium was replaced with fresh medium containing MTT reagent. After 4 h of incubation, HepG2 Cell viability was recorded by the reduction of the tetrazolium compound MTT (3-[4, 5-dimethylthizol-2-yl]-2, 5-diphnytetrazolium bromide) into an insoluble purple color formazan by a metabolically active mitochondrial enzyme. An ELISA (Enzyme-Linked Immunosorbent Assay) plate reader was used for absorbance measurement (extension of cell survival) at a wavelength of 570 nm. 53
Statistical Analysis
The data from antioxidant and biochemical analyses data were evaluated using Microsoft Excel 2013 software. All the experiments were conducted in triplicate and statistical analysis was carried out using analysis of variance (ANOVA), with STATISTICA 5.5 (Stat soft INC, Tulsa, USA) software. Statistically significance difference was mentioned by a probability value of P ≤0.05. All data were presented as mean values ± standard deviation (SD) for triplicated determinations. 54
Footnotes
Acknowledgements
The authors are thankful to the Deanship of Graduate Studies and Scientific Research at University of Bisha for supporting this work through the Fast-Track Research Support Program. The authors would like to thanks to the lab members of Institute of Molecular Biology and Biotechnology.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This article does not contain any studies with human or animal participants.
Statement of Human and Animal Rights
This article does not contain any human or animal study.
