Abstract
Among the phlorotannins of seaweed polyphenols, eckols which have a dibenzodioxin linkage are known to have various physiological functions. The purpose of this study was to investigate the intestinal epithelial absorption of eckols using Caco-2 cell monolayers of the small intestinal membrane model. Each compound permeated from the apical (AP) side to the basolateral (BL) side in the monolayers was identified and quantitated by liquid chromatography-mass spectrometry with electrospray ionization. In the transport assays using five types of eckols (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8'-bieckol), only the monomeric eckol showed limited transepithelial absorption with relatively small apparent permeability values (0.30 ± 0.04 × 10−8 cm/s). Analyzing the Hanks’ balanced salt solution in the receiver on the BL side showed that phloroglucinol was detected in all experimental sections using eckols, and it's concentration increased with time over the course of the incubation. The other molecules corresponding to the unconjugated and conjugated metabolites of eckols were not detected in the AP and BL sides through the assays. These results suggest that eckols, including monomeric eckol, may be decomposed into phloroglucinol in the intestinal epithelium and the resulting phloroglucinol permeates to the BL side.
Introduction
Phlorotannins, polyphenols peculiar to brown seaweeds, are a group of compounds having a polymerized structure of phloroglucinol (1,3,5-trihydroxybenzene). 1 According to the difference in the structural linkages between phloroglucinol units, phlorotannins can be classified into the following seven subclasses: fucols, phlorethols, fucophlorethols, fuhalols, isofuhalols, eckols, 1 and carmalols. 2 Among these subclasses, eckols in particular are attracting attention as novel bioactive substances derived from seaweeds because they have various physiological functions, 3 e.g., suppression of melanogenesis,4,5 antioxidant,4,6,7 anti-glycation,8–11 anti-diabetic, 12 and anti-inflammatory activities. 13 Although the beneficial effects of phlorotannins are well recognized, the pharmacokinetic properties of phlorotannins are poorly elucidated. Recently, two reports were published on the analysis of metabolism involving phlorotannins, and expression of their physiological functions in an oral administration test using a 60% ethanol extract from the brown seaweed Ascophyllum nodosum.14,15 The A. nodosum is one of the representative seaweed species rich in phlorotannins, 16 and compounds belonging to the subclasses of phlorethols and fucols have been mainly identified. 17 In the above reports, findings included the discovery that several phlorotannin-metabolites attributed to both unconjugated and conjugated metabolites in urine and plasma samples and indicated that ingested phlorotannins may be metabolized and absorbed predominantly in the large intestine.14,15 However, findings on the bioavailability of phlorotannins are limited to these two studies, and so far there are no reports analyzing the permeability and metabolism of isolated compounds using the evaluation system of the small intestine model, unlike the research field of terrestrial polyphenols.
Caco-2 cells are unique cells derived from colon cancer that have come to be known to express small intestinal resorption cell-like functions.18,19 By culturing on a porous membrane, Caco-2 cells exhibit a well-differentiated brush border on their apical (AP) surfaces, and express many typical transporters and enzymes found in the small intestine.18,19 Because of their biological properties, monolayer cultures of Caco-2 cells are widely used as a small intestinal membrane model to estimate the intestinal permeability of drugs and bioactive substances. In order to develop phlorotannins, especially bioactive eckols, as supplements and active ingredients of functional foods, it is extremely important to predict their absorption in the intestinal tract of humans. Therefore, the purpose of this study was to quantitatively investigate the intestinal epithelial absorption of five types of bioactive eckol isolated from the brown seaweed Eisenia bicyclis, using the Caco-2 cell monolayers of the small intestine model.
Results and Discussion
Eckol, which has a dibenzo-dioxin structure, is one of the basic constituent units of phlorotannins, 1 and many of its oligomeric compounds have been isolated and identified from Lessoniaceae.1,20,21 The brown seaweed E. bicyclis is a typical species belonging to the Lessoniaceae, and has been widely used for research of phlorotannins. 3 The chemical structures of the eckols (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8'-bieckol) isolated from E. bicyclis by chromatographies are shown in Figure 1. At the beginning of this study, the effects of the eckols and phloroglucinol on the viability of Caco-2 cells were investigated by using a 3-(4,5-dimethylthial-2-yl)-2,5-diphenyltetrazalium bromide (MTT) assay. As a result of adding each compound to the culture medium at a concentration from 1 to 500 μg/mL, the viability of Caco-2 cells was maintained at approximately 95%, or more (Figure 2). The results indicated that the eckols and phloroglucinol had no cytotoxicity to Caco-2 cells at the tested concentration. Thus, concentrations at 500 μg/mL were chosen for the eckols in the transport assays using Caco-2 cell monolayers.

Chemical structural formulas of the eckols used in this study.
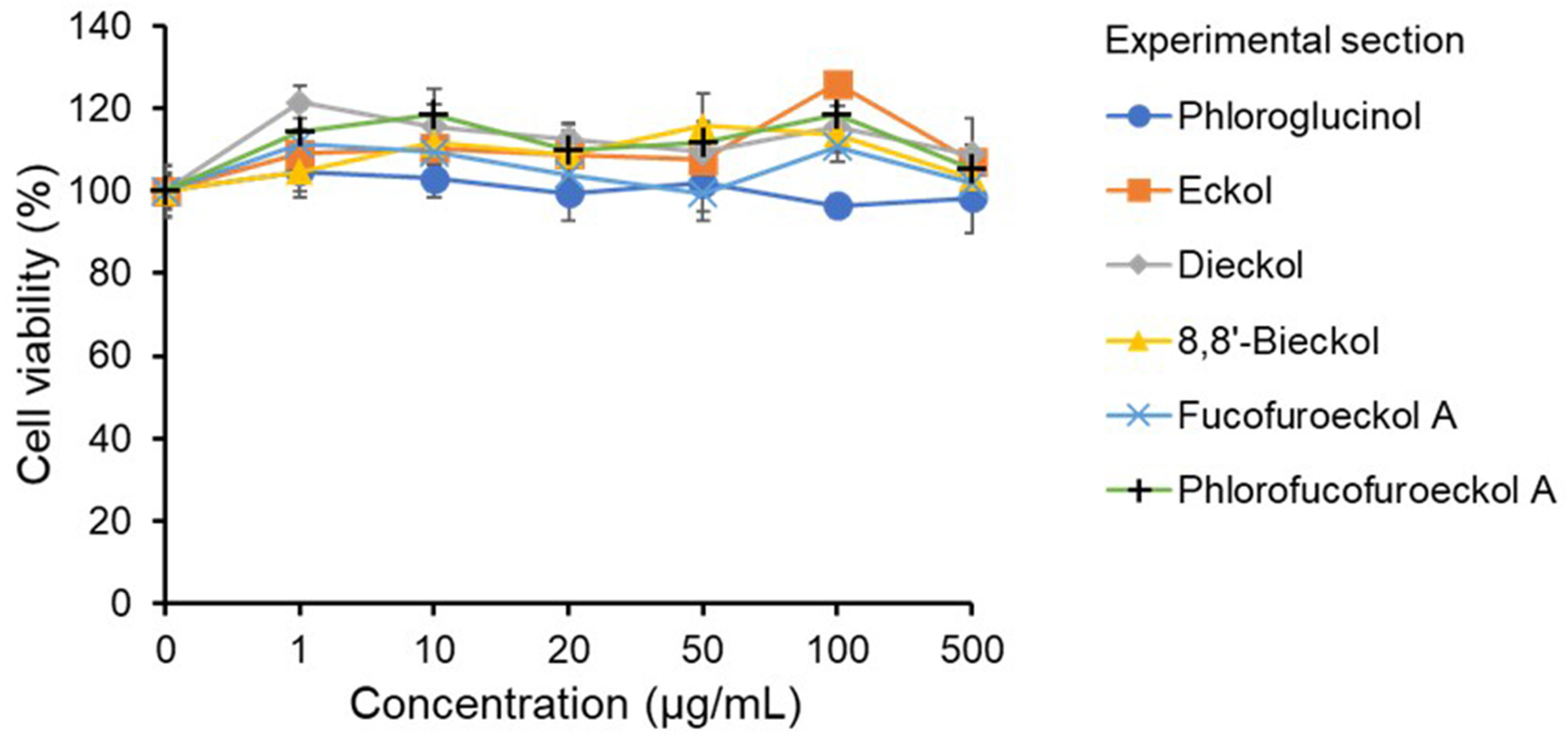
The viability of each Caco-2 cell treated with eckols and phloroglucinol.
In the transport assay, the concentration of the tested compound permeated to the basolateral (BL) side of the Caco-2 cell monolayers was measured using the selected ion monitoring (SIM) measurement of liquid chromatography mass spectrometry (LC/MS). The SIM measurement is suitable for identification and sensitive detection of a target compound in a sample, because it measures only the ions having a specific m/z. In order to obtain the optimum sensitivity for the measurement, the intensity of the fragmentor voltage in the mass spectrometer (MS) was investigated for each compound. As a result of the SIM measurement in the range from 70 to 150V, the peak intensity of [M-H]− was maximum at 150V for eckols and 100V for phloroglucinol. The data of the detection (LOD, S/N = 3) and quantification (LOQ, S/N = 10) limits of the eckols and phloroglucinol in the SIM measurement with the optimized fragmentor voltage were calculated as shown in Table 1.
LOD and LOQ for Eckols and Phloroglucinol Analysis Using a SIM Measurement of LC/MS.

The chromatographic conditions were described in the methods section.
Limit of detection (S/N = 3).
Limit of quantification (S/N = 10).
In order to the investigate intestinal absorption of the eckols (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8'-bieckol), each compound at a concentration of 500 μg/mL was added to the AP side of the Caco-2 cell monolayers with a transepithelial electrical resistance (TEER) value of more than 300 Ω·cm2. At each sampling time, Hanks’ balanced salt solution (HBSS) in the chamber of the BL side was collected and the concentration of compound contained therein was determined by LC/MS analysis. Table 2 shows the concentration of each compound detected in HBSS on the BL side of Caco-2 cell monolayers. Among the eckols tested, it was clearly confirmed that only monomeric eckol (phloroglucinol trimer) permeates the BL side of the Caco-2 cell monolayers. Eckol was detected in the HBSS collected at 30 min after the start of the transport assay, and its concentration decreased at the subsequent sampling times (Table 2). The apparent permeability (Papp) value for eckol calculated in this transport assay was 0.30 ± 0.04 × 10−8 cm/s. For oligomeric eckols of phloroglucinol tetramer and above, the amount of each compound in the HBSS was below the LOD at all sampling times, except for dieckol, which was below LOQ at all sampling times (Table 2). The Papp value obtained in a test using Caco-2 cell monolayers is considered to correlate with small intestinal absorption and bioavailability of the tested compound. 22 The permeability of the compound is determined to be poorly absorbed (from 0% to 20%) when the calculated Papp value is 1.0 × 10−6 cm/s or less, moderately absorbed (from 20% to 70%) when the Papp value is between 1.0 × 10−6 cm/s and 1.0 × 10−5 cm/s, and well absorbed (from 70% to 100%) when the Papp value is 1.0 × 10−5 cm/s or more. 22 The obtained Papp value which was much less than 1.0 × 10−6 cm/s suggests that the permeability of eckol through the lipid bilayer was not favorable and its oral bioavailability was low. Further analysis was performed by LC/MS on the HBSS in the receivers of the BL side collected in each transport assay. In the assays of eckols (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8'-bieckol), the phloroglucinol was detected in the HBSS of all experimental sections as early as 30 min after incubation, and its concentration increased with each sampling time (Figure 3). At the stage of 90 min in the transport assay, each of the HBSS samples on the AP side was also collected and analyzed by LC/MS. Analysis of the resulting chromatograms revealed that none of the tested eckols were detected in the HBSS and only phloroglucinol was found (data not shown). As shown in Table 2, the concentration of permeated eckol on the BL side decreased at sampling times of 60 and 90 min. On the other hand, the phloroglucinol concentration on the BL side in the experimental section of eckol increased significantly at the same sampling times (Figure 3). Summarizing these results, it is suggested that eckols are decomposed into phloroglucinol in the intestinal epithelium and the resulting phloroglucinol permeates to the BL side. The other molecules corresponding to the unconjugated and conjugated metabolites (glucuronides and/or sulphates) of eckols were not detected in the chromatograms through all of the assays.

The concentration of phloroglucinol in the HBSS taken from receivers on the BL side of Caco-2 cell monolayers in the transport assays of eckols.
The Concentration of the Compound in HBSS Taken from the Receiver on the BL side of the Caco-2 Cell Monolayers in the Transport Assay of Each.

Each value is the mean ± SD of the quantitative values obtained in three independent tests.
N.D: not detected (less than LOD).
To the best of our knowledge, this is the first report on the evaluation of the intestinal permeability of eckols using Caco-2 cell monolayers. The present results suggest the following two findings; (1) only monomeric eckol (phloroglucinol trimer) can penetrate through the intestinal epithelium directly, but it's transepithelial absorption is considerably limited, and (2) each eckol (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8'-bieckol) is decomposed into phloroglucinol on the AP side and then permeates to the BL side of the intestinal epithelial cell layer. The absorption rate of polyphenols in the small intestine depends on the structure of the compound, but is generally considered to be poor.23,24 It has been clarified that the biotranslocation rate of condensed tannins such as anthocyanidins and proanthocyanidins is extremely low, at about 0.1%. 23 In catechins known as tea polyphenols, it has been reported that the Papp values of epicatechin gallate (ECG) and epigallocatechin gallate (EGCG) were 9.6 ± 1.5 × 10−8 cm/s and 8.3 ± 2.4 × 10−8 cm/s in an assay using Caco-2 cell monolayers, with limited intestinal permeability of each. 25 Judging from the Papp values obtained in this study, it is conceivable that the level of absorption of eckol in the intestinal epithelium is much lower than those of ECG and EGCG. On the other hand, it was suggested that the eckols may be decomposed into phloroglucinol during the permeation process and absorbed into the small intestine. The low-molecular weight metabolite, phloroglucinol resulting from eckols, may exert various physiological functions in vivo when ingesting phlorotannins from Lessoniaceae. Song et al 26 reported that the intestinal epithelium absorption of catechin and EGCG could be improved by the use of a niosomal carrier. Vesicles such as niosomes and liposomes are used to improve the bioavailability of target drugs because they allow transport across lipid bilayers. Therefore, the development of vesicles encapsulating eckols may lead to improved absorption in the intestinal epithelium, similar to what was found in the study of catechins. Further studies, including a detailed analysis on the degradation mechanism of eckols in intestinal epithelial cells, will also be needed to accumulate findings on the bioavailability of phlorotannins.
Experimental
Materials
The brown seaweed E. bicyclis (Kjellman) Setchell was collected from the coast of the Itoshima Peninsula (33°38'N, 130°11'E) in Fukuoka prefecture, Japan. The collected seaweed was air-dried in the shade and pulverized via pulverizing mill (ABS-W, Osaka Chemical). The seaweed powder was stored at −30 °C until used for preparation of phlorotannins.
The phloroglucinol used was a special grade reagent (Fujifilm Wako). All other reagents used in this study were of analytical grade.
Preparation of Phlorotannins
The phlorotannins were extracted from the seaweed powder using chloroform, MeOH, and water as solvents, as described in a previous report. 27 After removing MeOH from the hydrophilic fraction of extract using a rotary evaporator (N-1000, Eyela), the fraction was extracted and partitioned with an equal volume of ethyl acetate. The ethyl acetate fraction was evaporated in vacuo using the rotary evaporator and the obtained residue was used as crude phlorotannins.
Each of the eckols (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8'-bieckol) was isolated from the crude phlorotannins using silica gel column chromatography (1.5 cm i.d. × 150 cm) on a Wakogel C-300HG (Fujifilm Wako) 6 and a preparative high-performance liquid chromatography (HPLC) system (Prominence, Shimadzu) with an Inertsil ODS-3 column (10 mm i.d. × 250 mm, GL Sciences) under the same conditions as in a previous report. 7 The isolated eckols were confirmed to have a purity of 98% or more by a three-dimensional HPLC (SPD-M10AV, Shimadzu) with an Inertsil ODS-3 column (4.6 mm i.d. × 250 mm, GL Sciences),6,7 and were used in each cell experiment.
Subculture of Caco-2 Cells
Caco-2 cells were obtained from Riken BioResource Research Center in Japan. The cells were cultured in Eagle's minimal essential medium (E-MEM, Fujifilm Wako) supplemented with 10% inactivated fetal-bovine serum (BioWest), 1% L-glutamine and 1% penicillin-streptomycin-amphotericin B suspension (Fujifilm Wako). Caco-2 cells were maintained at 37 °C in a humidified atmosphere of 5% CO2, and subcultured at 70 to 80% confluency.
Assay of Cell Viability
Similar to a preceding report,
9
MTT (Dojindo) was used as an indicator of cell viability. Briefly, Caco-2 cells were cultured in 96-well microplates (BioLite, Thermo Scientific) at a density of 2.0 × 104 cells per well (0.32 cm2). After 24 h of cultivation at 37 °C in a CO2 incubator (CPE-601, Hirasawa) with 5% CO2, the cell lines were washed with the E-MEM and then treated with each sample solution (10 μL) for 24 h in the incubator. Each sample (eckols and phloroglucinol) was dissolved in Dulbecco's phosphate buffered saline without calcium and magnesium (DPBS, Nacalai Tesque) at concentrations ranging from 1 µg/mL to 500 µg/mL, and they were used as a sample solution. For blank and control wells, DPBS was used instead of a sample solution. The cell lines were then rewashed with the medium, treated with 10 μL of MTT solution, and cultured at 37 °C for 4 h in the CO2 incubator. The MTT solution was prepared by dissolving MTT (25 mg) in the DPBS (5 mL). After removing the medium containing the MTT solution, 200 μL of the DPBS was added to each well and maintained at 37 °C for 1 min. Finally, in order to dissolve the formed formazan salt, dimethyl sulfoxide (DMSO, 200 μL, Fujifilm Wako) was added to each well from which DPBS had been removed. The absorbance of each well was measured at 535 nm using a microplate reader (Infinite 50, Tecan). The cell viability (%) was calculated using the following formula:
Culture of Caco-2 Cell Monolayers
Caco-2 cells were seeded at a density of 2.0 × 105 cells per well on a type-I collagen (Nippi) coated 12-well transwell inserts (ThinCert 665641, Greiner), and incubated at 37 °C under 90% humidity and 5% CO2 for 21 to 23 days. The medium was replaced every 2 to 3 days for both the AP and BL sides of the transwell inserts. The integrity of each cell monolayer was confirmed by measuring TEER values between AP and BL sides with Millicell ESR-2 (Merck) and a permeability test using a Lucifer Yellow CH dipotassium salt (Fujifilm Wako) known as a paracellular flux marker. Only cell monolayers with a TEER value above 300 Ω·cm2 and a permeability of the Lucifer Yellow less than 2% were used for the transport assays of eckols.
Transport Assay of Phlorotannins
The transport assay using monolayers of Caco-2 cell was performed with reference to the method of Kosińska and Andlauer. 28 Briefly, the medium was removed from the AP and BL sides of the transwell inserts, and the both sides were washed three times with HBSS (Fujifilm Wako). The monolayers were then incubated for 30 min with pre-warmed HBSS at 37 °C. The HBSS was added at 500 µL on the AP side and 1.5 mL on the BL side. Each of the eckols was pre-dissolved in DMSO, and then prepared to 500 μg/mL with HBSS to allow the final concentration of DMSO to be less than 0.1%. Each sample solution was sterilized by filtration through PTFE filter (Millex LG, Merck) with a pore size of 0.20 µm. After removing HBSS on the AP side, 500 μL of sample solution was added to the AP side. Incubation was performed at 37 °C for 120 min, while shaking at 50 rpm on a rotary shaker (NX-20, Nissin). At 30, 60, and 90 min, 100 µL of the solution from BL side was collected, and replaced with an equal volume of HBSS. The collected solutions were stored at −80 °C until LC/MS analysis.
Papp values for each compound were calculated according to the following formula:
LC/MS Analysis
LC/MS analysis was performed on a system consisting of 6120 quadrupole MS, 1260 Infinity LC, and OpenLAB chromatography data system (Agilent). Chromatography for the analysis of eckols and phloroglucinol was used under the same conditions as described in the previous reports.6,7 The HBSS collected in the transport assay was filtered by PVDF filter (Cosmonice filter W, Nacalai tesque) with a pore size of 0.45 µm and used for LC/MS analysis. Each compound (eckols and phloroglucinol) was monitored by ESI-MS measurement in negative mode. Other experimental conditions for the MS were as follows: dry gas, 12.0 L/min; nebulizer, 35 psi; dry temperature, 250˚C; vaporizer, 200˚C; scan from m/z 100 to 1500. The concentration of the test compound in the collected HBSS was determined using the SIM measurement of MS. In the SIM measurement, the m/z was set to 125 (corresponding to the molecular mass of the deprotonated form of phloroglucinol), 371 (corresponding to the molecular mass of the deprotonated form of eckol), 477 (corresponding to the molecular mass of the deprotonated form of fucofuroeckol A), 601 (corresponding to the molecular mass of the deprotonated form of phlorofucofuroeckol A), and 741 (corresponding to the molecular mass of the deprotonated form of dieckol and 8,8'-bieckol).
Supporting Information
This research was based on results obtained from a project subsidized by the New Energy and Industrial Technology Development Organization (NEDO) in Japan.
Footnotes
Acknowledgments
We acknowledge that the English was revised by Mr Andrew Rother (English Step, Tsu, Japan).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the New Energy and Industrial Technology Development Organization (NEDO) in Japan
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
