Abstract
Highlights
LC-MS analysis of F. insignis extract identified gallic acid, chlorogenic acid, and quercetin. F. insignis extracts showed significant antioxidant activity against several free radicals, such as DPPH, nitric oxide, hydroxyl, and ferric ions. Agar well diffusion and broth dilution assays demonstrated promising dose-dependent antibacterial activity of the F. insignis extract against Staphylococcus aureus. Quercetin, gallic acid, and chlorogenic acid exhibited strong molecular interaction with catalytic residues of S. aureus penicillin binding protein, indicating antibacterial efficacy.
Introduction
Human civilization has utilized the plant for their healing properties in a variety of illnesses since the time immemorial. 1 In due date, approximately 60% world's population, along with 80% of population in developing countries, relies on herbal medicine for primary health care due to its better compatibility and safety. 2 Consequently, phytoextract as a foundational resource for the discovery and development of novel drugs, has gained significant traction in the recent pharmaceutical industry due to its cost-effectiveness and renewability. 3 These plant-derived extracts are rich in bioactive principles, which possess an inherent ability to interact with a broad array of biological targets. This polypharmacological characteristics position phytoconstituents as advantageous alternatives to synthetic drugs in drug discovery and development pipelines. 4 The potential to discover novel leads from diverse phytobioactive compounds, coupled with their effectiveness and safety as medicines within the healthcare system, establishes phytoextracts as a significant source of essential medicines. 3 Moreover, the increasing trend of prescribing herbal medicines, often based on “cruder” preparations of phytoextracts that contain numerous bioactive compounds, has captured attention in the global pharmaceutical sector. This market has surpassed a trillion dollars and is projected to grow at an annual rate exceeding 8%, reflecting a rising consumer preference for natural products. 5 Thus, finding plant species with pharmacologically precious properties has become a focal area of research, promising significant contributions to the discovery of novel drugs.
Cellular respiration transforms approximately 2% to 3% of oxygen into reactive oxygen species (ROS) and other free radicals that cause damage to various biomolecules, such as DNA, proteins, small cellular molecules and membrane lipids. 6 This oxidative damage significantly contributes to the pathogenesis of serious diseases, including cancer, diabetes, aging, atherosclerosis, ulcers, gastrointestinal disease, neurodegenerative disorder, coronary heart disease, hypertensions, and hepatotoxicity.7–10 Therefore, an adequate intake of antioxidants has become crucial in preventing oxidative stress and promoting overall health. Consequently, the search for safe and natural antioxidants, such as ascorbic acid, tocopherols, flavonoids and phenolic compounds, from medicinal plants has gained increasing attention. 11
In parallel, the global emergence and spread of antibiotic resistance, along with the evolution of new bacterial strains, pose significant concerns for human health. 12 The World Health Organization (WHO) identifies antimicrobial resistance (AMR) as one of the most critical threats to health and development, accounting for approximately 1.27 million deaths in 2019 and contributing to a total of 4.95 million deaths. 13 The misuse and abuse of antimicrobials have significantly increased the prevalence of antibiotic resistant bacteria, especially among pathogens such as Escherichia coli, Klebsiella pneumoniae, Staphylococcus aureus, Pseudomonas aeruginosa, Acinetobacter baumanni, and Streptococcus pneumoniae. Alarmingly, the pace of discovery and development of novel antibiotics has lagged behind the rapid emergence of resistance. Thus, there is an urgent need to discover effective and safer alternatives to combat AMR, with focus turning to medicinal plants rich in bioactive compounds like alkaloids, flavonoids, and phenolic compounds, which have demonstrated broad-spectrum antibacterial activities in scientific studies. 14
Falconeria insignis Royle (Syn. Sapium insigne Royle) is a plant belonging to the Euphorbiaceae family, renowned for its traditional medicinal properties across various ethnic healers. It is locally known as Khirro in Nepal, 15 and is distributed in countries such as Nepal, India, China, and Sri Lanka. Traditionally in Nepal, the juice of F. insignis bark is applied topically to livestock wounds to eliminate bacteria and enhance the healing process. 16 Additionally, a few drops of milky latex mixed with water and administered orally for indigestion. 17 Ethnic tribes such as Raji, Gurung, Magar, and Majhi apply the plant`s juice around the navel region to treat cholera, diarrhea, and green stool.16,18 What's more, the leaves of F. insignis leaf have also been used in China for the treatment of snake bites. 19
Despite the prevalence of such beneficial claims, there exists a significant lack of rigorous scientific evidence supporting the bioactivities of these herbal remedies. Previous studies have identified numerous chemical compounds from this species that include phenols, flavonoids, diterpenes and phorbol esters.17,20–22 More precisely, Liu et al reported the presence of diterpenes known as sapinsignoids A−G, which exhibited significant cytotoxicity against the A-549 tumor cell line (IC50: 0.2−1.8 μM) and HL-60 cell line (IC50: 2.7−6.5 μM). 21 Additionally, research on related species, such as Sapium sebiferum, demonstrated promising antioxidant activity when assessed through various assays, including DPPH, ABTS, superoxide, and nitrite radical scavenging, as well as reducing power and β-carotene bleaching. 23 Rao et al highlighted the phenolic antioxidants, such as gallic acid, quercetin, ellagic acid, isoquercitrin, astragalin, and kaempferol, underlying the beneficial effect of Sapium sebiferum leaf extract against the allergic contact dermatitis. 24 Moreover, the compounds including diterpenes, triterpenes, and sapintoxins from Sapium haematospermum and Sapium indicum were found to exhibit significant antibacterial activity against Mycobacterium tuberculosis, with MIC values 3–25 μg/ml.25,26
These findings have sparked our curiosity in investigating the F. insignis. As of October 2024, the absence of comprehensive studies raises critical questions regarding their safety, effectiveness, and mechanisms of action. In light of that, our research aims to clarify consumer claims about its efficacy in treating bacteria-infected wounds. The study seeks to establish F. insignis as a safe and potent crude antibacterial drug by identifying its bioactive phytoconstituents and investigating its antioxidant and antibacterial properties for the first time, as illustrated in Figure 1. Furthermore, recent advance in computational approaches, particularly in silico molecular docking and ADME-toxicity studies, have emerged as vital laboratory tools for the rapid and accurate prediction of the biological activity and safety profile of plant secondary metabolites. Such methods improve our understanding of pharmacodynamics by uncovering the underlying mechanism of action of phytoconstituents, including identifying binding sites, interacting residues, and binding affinities.27,28 Numerous researchers in the field of drug discovery and development have successfully employed computational software, such as AutoDock Vina and Schrödinger Glide, to investigate pharmacological properties that corroborate in vitro, 29 cell line, 30 and in vivo 10 bioassay results. In line with these scientific trends, our study also incorporates in silico virtual docking studies of F. insignis’ native phytoconstituents to validate the observed in vitro antibacterial activities and enhance its further topical application in bacterial infections.

A Schematic Representation of Methods and Protocols Implemented in the Investigation of Falconeria Insignis from Nepal.
Materials and Methods
Chemicals and Bacterial Strains
In current study, ascorbic acid (Merck, India), hexane and methanol (Thermo Fisher Scientific, India), ethyl acetate and sulfanilamide (CDH, India), Mueller Hinton agar, meropenem discs, and quercetin dihydrate (HiMedia, India), as well as 2,2-diphenyl-1-picrylhydrazyl (DPPH) (Sigma-Aldrich, USA), were used. Staphylococcus aureus (ATCC 11238) and Escherichia coli (ATCC 11386) were gifted by Manipal Teaching Hospital, Pokhara, Nepal.
Plant Material
The leaves and stems of F. insignis were collected from Pokhara-30, Nepal. The scientific name of the plant specimen (31/02/2080/Herbarium specimen No. 14) was confirmed by Botanist Dhan Raj Kandel at the National Herbarium and Plant Laboratories, Godawari-3, Lalitpur, Nepal, referring to the deposited plant specimen.
Extraction Procedures
Leaves and stems were shade dried at room temperature (24 °C) and relative humidity (50-55%) for one week to remove the moisture content. Dry leaves were ground to powder using an electrical grinder (Baltra, India), while the dried stem sizes were reduced using a plant cutter. Successive maceration was performed at room temperature (24 °C) for 72 h using the sample-to-solvent ratio of 1:7 w/v, as per the previous method.
31
The leaves (125 g) and stem (100 g) were separately subjected to successive maceration using menstruum (1:7 w/v) in increasing polarity order: a) hexane, b) ethyl acetate, and c) methanol at room temperature with frequent agitation for 24 h. The liquid extract was filtered using Whatman No.1 filter paper and evaporated under reduced pressure at < 45 °C using a rotary evaporator (Biobase RE-2000B, Germany). The resulting liquid crude extracts of hexane leaf (HL), hexane stem (HS), ethyl acetate leaf (EL), ethyl acetate stem (ES), methanolic leaf (ML), and methanolic stem (MS) were used for qualitative phytochemical analysis. The concentrated extract obtained after rotary evaporation was further subjected to complete drying in a vacuum desiccator (1300-2-B, Cleatech) for quantitative analysis. After complete drying, the extracts were weighted to estimate the extraction yield and then stored at 4 °C.
Qualitative Phytochemical Analysis
The presence of phytochemicals, such as alkaloids, phenols, flavonoids, tannins, carbohydrates, saponins, terpenoids, quinines, steroids, and proteins in the extracts of F. insignis were tested according to the well-established protocols.11,27
Quantitative Phytochemical Analysis
Total Phenolic Content (TPC). The TPC of F. insignis extract was estimated spectrophotometrically by implementing the Folin-Ciocalteu reagent method.32,33 In brief, 100 μl of extract (1000 µg/ml), 6 ml of distilled water, and 0.5 ml of Folin-Ciocalteu phenol reagent (2 N) were homogenized in a test tube for 10 s using a vortex mixer. After 5 min, 1.5 ml of sodium carbonate (7.5%) and 1.9 ml of distilled water were added, homogenized, and incubated for 2 h in the dark. For the calibration curve, gallic acid (0.25 mg/ml–1.25 mg/ml) was considered a reference phenolic compound and treated similarly to the extract. At 750 nm, a single-beam UV-VIS spectrophotometer (Agilent Cary 60, Malaysia) was used to read the absorbance of the solution. In the blank solution, the extract (100 μl) was replaced with methanol (100 μl) and treated identically. The mean absorbance of triplicates per sample was used, and the TPC was expressed as milligram of gallic acid equivalents (mg of GAE)/g of extract.
Total Flavonoid Content (TFC). The TFC of F. insignis extracts was estimated using the AlCl3 method with slight modifications.11,33 Briefly, 2 ml of AlCl3 (2% in methanol) and 2 ml of extract (100 µg/ml) were mixed and incubated at room temperature for 10 min. For the calibration curve, quercetin (1.25-100 µg/ml) was considered a reference flavonoid and treated similarly to the extract. At 415 nm, a single beam UV-VIS spectrophotometer was used to read the absorbance of the solution against a blank containing the extract with methanol. The mean absorbance of triplicates per sample was used to express TFC as milligrams of quercetin equivalent (mg of QE)/g of extract.
TLC Profiling
The TLC profiling of F. insignis extracts was performed according to our previous method with slight modifications.27,33 Particle-free extract (1 mg/ml) was used for TLC profiling. The extract band was applied to silica gel 60 F254 plates using a microcapillary tube. The extract-loaded plates were developed in a saturated glass beaker containing solvents (chloroform, methanol and water) in proportions of 7: 3: 0.5. The developed plates were faced to a hot air blow, and the solvent was air-dried. Afterward, they were visualized in day light, UV-259 nm, and UV-365 nm followed by derivatization with 10% v/v FeCl3 spray/dry, 10% v/v H2SO4 spray/heat, and dipping in DPPH solution.
LC-MS Analysis
The methanolic leaf extract of F. insignis was analyzed using the liquid chromatography-mass spectrometric (LC- MS) technique, as per our previous method.27,28 The LC was carried out on an HPLC (Shimadzu) coupled with a C18 column (Water XBridge 50 × 4.6 mm, 3.5 µ), maintained at 35 °C. The linear gradient of mobile phases between 0.1% formic acid in water (A) and 100% acetonitrile (B) was programed to flow at a rate of 1.2 ml/min as follow: (Time- %A/% B): 0 min-85/15, 6 min-25/75, and 11 to 15 min-85/15. A 5 µl of particle free extract at a concentration of 1 mg/ml was injected for the analysis. Mass spectrometric detection was performed on a single quadrupole mass analyzer. The electron spray ionization source in both positive (ESI+) and negative (ESI-) ion modes was used with a scanning range of m/z 0–1000. MS source parameters were adjusted as follows: ESI capillary voltage: 3.03 KV; cone voltage: 13 V; source temperature: 118 °C; desolvation temperature: 246 °C; and gas flow rates: desolvation (500 L/hr) and cone (50 L/hr).
Antioxidant Activity
DPPH Scavenging Assay. The DPPH• scavenging activity of F. insignis was estimated spectrophotometrically based on the ability of DPPH• to absorb light at 517 nm as described by Chen et al 11 In brief, 2 ml of extract at different concentrations was treated with 2 ml of DPPH• methanolic solution (100 µM). The content was mixed for 5 s using a vortex mixer. After half an hour of dark incubation at room temperature, the absorbance of DPPH• (oxidized form) in the solution was measured at 517 nm against the DPPH• control (which contained 2 ml of methanol instead of extract) and blank (methanol). Ascorbic acid (0.6125 µg/ml–10 µg/ml) was used as a positive control (standard antioxidant). Each experiment was carried out in triplicate, and its mean absorbance was used to calculate the percentage of DPPH• scavenging. A decrease in absorbance (ie, the purple decolorizes to pale yellow) indicates strong antioxidant properties.
% DPPH• radical scavenging = [(A0-A1)/A0] × 100, where A0 represents the absorbance of the DPPH control and A1 represents the absorbance of the sample or positive control. The antioxidant potency of each extract sample and ascorbic acid was expressed as an IC50 value (mean ± standard deviation). The linear graph showing the percentage of DPPH scavenging versus extract and ascorbic acid concentration was utilized to calculate the IC50.
Nitric Oxide Scavenging Assay. The F. insignis extracts were assessed for their potency to scavenge nitric oxide by implementing the Griess reaction. 34 In an aqueous solution at physiological pH 7.4, sodium nitroprusside releases nitric oxide (NO) which reacts with oxygen to generate nitrite ions. These ions are assessed by utilizing Griess reagent. Antioxidants compete with oxygen and decline nitrite ion liberation. Phosphate-buffered saline (PBS) of pH 7.4 was prepared according to the Indian Pharmacopeia, 1996. In brief, 1 ml of test extract of different concentrations (125 µg/ml-1000 µg/ml) was treated with 1 ml of 10 mM sodium nitroprusside in PBS. The content was mixed for 5 s. After 150 min of incubation at room temperature, 2 ml of Griess reagent (1% sulfanilamide, 3% phosphoric acid, and 0.1% N-(1-napthyl) ethylenediamine dichloride) was added. As a result of diazotization, the pink chromophore appeared and was recorded at 546 nm against the control (containing 1 ml of methanol instead of 1 ml of extract) and blank (methanol). Curcumin (5 µg/ml–100 µg/ml) was used as a reference nitric oxide scavenger. The mean absorbance of triplicates per sample was utilized to calculate the percentage of NO scavenging as follows:
% NO scavenging = [(Absorbance of control – Absorbance of sample) / Absorbance of control] × 100
Ferric Ions (Fe3+) Reducing Antioxidant Power (FRAP). The ability of F. insignis extracts to reduce the Fe3+ complex to the Fe2+ complex were assessed through the electron-donating capacity, following the potassium ferric cyanide method. 35 In brief, 1 ml of test extract at various concentrations (12.5 µg/ml–200 µg/ml in phosphate buffer) was treated with 2.5 ml of phosphate buffer (0.2 M, pH 6.6) and 2.5 ml of 1% potassium ferric cyanide. After incubation at 50 °C for 20 min, the mixture was acidified with 2.5 ml of 10% trichloroacetic acid and then centrifuged at 3000 rpm for 5 min. The 2.5 ml of the mixture's top contents were separated, and mixed with 2.5 ml of distilled water and 0.5 ml of 0.1% FeCl3. The absorbance of the developed Perl's Prussian blue (Fe2+ complex) was measured at 700 nm against the control (1 ml of phosphate buffer instead of 1 ml of sample) and blank (distilled water). Ascorbic acid (12.5 µg/ml–200 µg/ml) was used as a standard antioxidant. The mean absorbance of triplicates per sample was expressed and compared with standard antioxidants.
Hydroxyl Radical (OH·) Scavenging Assay. The hydroxyl radical scavenging activity of F. insignis extracts was estimated spectrophotometrically by implementing the salicylic acid reagent. 36 In brief, 1 ml of test extract was treated with 1 ml of FeSO4 (3 mM) and 1 ml of H2O2 (3 mM). The content was homogenized for 5 s using a vortex mixer. Subsequently, after 10 min, 1 ml of salicylic acid (6 mM) was added and then mixed. Following a half-hour incubation in a 37 °C water bath, the reaction mixture was centrifuged at 4000 rpm for 5 min. The absorbance of the developed purple chromophore in the supernatant was measured at 500 nm against the control (0 µg/ml, containing 1 ml of methanol instead of 1 ml of extract) and blank (1 ml of water instead of 1 ml of salicylic acid solution). Ascorbic acid (31.25 µg/ml–500 µg/ml) was used as a positive control (standard OH· scavenger). The mean absorbance of triplicates per sample was utilized to calculate the percentage of OH· scavenging. A decrease in absorbance (ie, the purple color decolorizes to colorless) indicates a strong hydroxyl radical scavenging power.
Hydroxyl radical scavenging (%) = [1− (A1−A2) / A0] × 100%, Where A0 represents the absorbance of the control (0 µg/ml, containing methanol instead of extract), A1 represents the absorbance of the test sample, and A2 is the blank (water instead of salicylic acid solution).
Antibacterial Activity
The antibacterial activity of F. insignis extracts was evaluated using well diffusion and broth microdilution methods against the selected standard bacterial strains from the American Type Culture Collection: Staphylococcus aureus (ATCC 11238) and Escherichia coli (ATCC 11386), as per the our previous methods.27,33
Well Diffusion Assay. The Muller-Hinton agar was autoclaved and poured aseptically into sterile Petri plates. After solidification of the culture media, standard bacterial inoculums (0.5 McFarland) were swabbed over the entire surface of the MHA plates. Using a sterile tip (6 mm), five wells were created in each plate. 20 μl of molten MHA was dropped to seal the base of the wells. Different extracts of F. insignis (100 μl) at concentrations (25, 50, and 100 mg/ml in 5% DMSO) were added to the respective wells, and then the plates were placed in an incubator for 48 h at 37 °C. A negative control (5% DMSO) and a positive control (amikacin 30 μg disc) were used. After 48 h, the clear zone of inhibition (ZOI) around the wells was measured in millimeters (mm).
Broth Microdilution Assay. The active antibacterial extracts of F. insignis were further analyzed for their potency (MIC) against the sensitive bacterial strain implementing the broth microdilution method. 27 In brief, 100 µl of different extract concentrations (7.8125 µg/ml to 1000 µg/ml) and meropenem (0.78125 µg/ml to 100 µg/ml) were treated with 100 µl of S. aureus suspension (0.5 McFarland standard) in a 96-well titer plate and then incubated for 24 h at 37 °C. Additionally, resazurin dye was added to each well to monitor the growth of S. aureus (color changes from blue to pink). The minimum concentration of treatment showing no growth on visual observation (blue color) was considered as the minimum inhibitory concentration (MIC), compared to negative control containing Muller-Hinton broth instead of extract or antibiotics.
In Silico Studies
Optimization of Ligands and Proteins. In this molecular docking study, the phytoconstituents identified by LC-MS analysis were chosen as ligands. The 3D structure of phytoconstituents and standard meropenem was retrieved from the PubChem database in the SDF (Structure Data Format) and then converted to PDB (Protein Data Bank) format using BIOVIA Discovery Studio Visualizer. The ligands were optimized by removing extra moieties and joining crucial polar hydrogen and Kollman charge, and finally converted to the pdbqt files using AutoDock 1.5.6. software. Likewise, penicillin binding protein (PBP 2a) was selected as the target protein. The 3D crystal structure of PBP 2a (PDB ID: 6H5O) 37 was downloaded from the RSCB Protein Data Bank server and subjected to purification by removing (water and heteroatoms) and adding (polar hydrogens and Kollman charge). The energy of the protein was minimized and finally converted to pdbqt format using AutoDock 1.5.6. software.
Molecular Docking. AutoDock Vina 1.5.7. was used for docking the ligands against PBP 2a in a 3D space defined by the grid box of 20 × 20 × 20 sizes with coordinates: x = 36.71; y = -0.95; and z = 24.47, and spacing 0.375 Å. This space encompasses active site amino acid residues as shown in Figure 2. After docking, BIOVIA Discovery Studio Visualizer 2020 was utilized to analyze the interactions between the docked protein and ligands. Re-docking and superimposition techniques were applied to validate the docking protocol. 27

3D Structure of the Target Protein (PBP 2a) and its Active Site Amino Acids Represented by a Grid Box.
ADMET Analysis. The pharmacokinetic parameters, including absorption, distribution, metabolism and excretion data of F. insignis phytoconstituents were determined using the computation tool SwissADME (http://www.swissadme.ch/).27 Similarly, for the toxicity analysis, Computation software's pKCSM (https://biosig.lab.uq.edu.au/pkcsm/) and ProTox-3.0 (https://comptox.charite.de/protox3/ (Predicting of Rodent Oral Toxicity) were employed to forecast the potential AMES toxicity, hepatotoxicity, nephrotoxicity, carcinogenicity, cytotoxicity and mutagenicity of F. insignis phytoconstituents. 28
Statistical Analysis
Statistical analysis was performed using Microsoft Excel 2016 software. Each experiment was performed in triplicate and their data were presented as the mean ± standard deviation. The TPC, TFC, and antioxidant activity (IC50) were determined by using linear regression analysis.
Results
Extraction Yield
In successive maceration with increasing order of solvent polarity, such as n-hexane, ethyl acetate, and methanol, the extracts of F. insignis revealed an extraction yield ranging from 1.2% to 16.4%.
Phytochemical Analysis
The qualitative phytochemical analysis of the F. insignis extract indicated the presence of phenols, tannins, flavonoids, saponins, steroids, terpenoids, alkaloids, quinones and carbohydrates, while proteins were not detected, as presented in Table 1. Likewise, in case of the quantitative analysis, the methanolic leaf extract (ML) showed the highest phenolic and flavonoid content of 798.18 ± 5.81 mg GAE/g and 169.37 ± 6.26 mg QE/g extract, respectively, as shown in Table 2.
Qualitative Phytochemical Analysis of F. insignis Extracts.

(+) indicates presence; (–) indicates absence; (0) indicates no test.
Extraction Yield, Total Phenolic and Flavonoid Contents of F. insignis Extracts.

Extraction yield was expressed as % yield = (weight of dry extract/initial weight of dry sample) × 100. TPC: Total phenolic content. TFC: Total flavonoid content. GAE: Gallic acid equivalent. QE: Quercetin equivalent. TPC and TFC values are the mean ± standard deviation of triplicate experiments.
TLC Profiling
The TLC profile of different extracts from F. insignis are shown in Figure 3. They demonstrate the presence of several compounds band under UV light at the different retention factor. Similarly, several characteristic bands, such as green, blue and brown bands after 10% FeCl3 spray; yellow and reddish-brown band after H2SO4 spray/ heat and yellow bands after DPPH dipped were observed on TLC plate.

Thin Layer Chromatography (TLC) profiles of Methanolic Leaf (ML), Methanolic Stem (MS), Ethyl Acetate Leaf (EL), Ethyl Acetate Stem (ES), Hexane Leaf (HL), and Hexane Stem (HS) Extract of F. Insignis Using Silica Gel 60 F254 as Stationary Phase and Chloroform: Methanol: Water = 7:3:0.5 v/v as Mobile Solvent; Observed Under 1.) Daylight 2.) UV-259 nm; 3.) UV-365 nm; 4.) Using Spray Reagent 10% FeCl3 and dry; 5.) Using Spray Reagent 10% H2SO4 and Heated; 6.) Dipped in 500 µM DPPH Solution.
LC-MS Analysis
The LC-MS analysis of methanolic leaf extract from F. insignis identified three phytoconstituents, including gallic acid, chlorogenic acid, and quercetin, as shown in Figures 4, 5, 6 and 7.

Liquid Chromatography-Mass Spectra Acquired in Negative Mode Electron Spay Ionization (ESI-) for the Methanolic Leaf Extract of F. Insignis.

Mass Spectra of Peak 1(Retention Time: 1.25 min) Indicating the Deprotonated Molecular Ions at m/z 169.22 [M-H]−, Adduct Ion at m/z 205.29 [M + Cl]−, and Dimer Ion at m/z 339.35 [2M-H]− in Negative Mode ESI. Additionally, the Fragment Ion at m/z 97.14 was Formed After the Successive Loss of CO2 (44 Da) and CO (28 Da) from the Precursor Ion m/z 169.22.11,38 By Comparing these Spectral Data with Previously Isolated Compounds from ‘F. insignis’, Peak 1 was Identified as Gallic Acid. 20

Mass Spectra of Peak 2 (Retention Time of 1.96 min) Indicating the Deprotonated Molecular Ions at m/z 353.39 [M-H]−, Adduct Ion at m/z 389.37 [M + Cl]−, and Dimer Ion at m/z 707.63 [2M-H]−, in Negative Mode ESI. Additionally, the Fragment Ion at m/z 191.18 Indicates Quinic Acid Formed After the Loss of Caffeoyl (C9H6O3; 162 Da) from the Precursor Ion m/z 353.39.11,38 By Comparing these Spectral Data with Previously Isolated Compounds from ‘F. Insignis’, Peak 2 was Identified as Chlorogenic Acid. 17

Mass Spectra of Peak 3 (Retention Time of 5.7 min) Indicating the Deprotonated Molecular Ions at m/z 301.33 [M-H]−; Adduct Ions at m/z 337.32 [M + Cl]−, 639.47 [2M-H + Cl]−, and 942.65 [3M-H + Cl]−; a Dimer Ion at m/z 603.46 [2M-H]−; and a Trimer Ion at m/z 905.65 [3M-H]−, in Negative Mode ESI. By Comparing these Spectral Data with Previously Isolated Compounds from ‘F. Insignis’, Peak 3 was Identified as Quercetin. 17
Antioxidant Activity
Table 3 detailed the findings of the antioxidant activities evaluated through four different protocols. Among the extracts of F. insignis analyzed in DPPH assay, ML extract showed the highest radical scavenging activity, with a fifty-percentage inhibitory concentration (IC50) of 5.27 ± 0.5 µg/ml, which is comparable to the standard antioxidant ascorbic acid (IC50: 4.05 ± 0.3 µg/ml). MS extract came as the next most potent, with an IC50 of 10.84 ± 1.3 µg/ml, followed by EL extract (IC50: 28.85 ± 1.5 µg/ml), ES extract (IC50: 49.21 ± 2.5 µg/ml), HL extract (IC50:183.27 ± 8.1 µg/ml), and HS extract (IC50: 191.03 ± 5.7 µg/ml). In the case of the nitric oxide assay, ML extract exhibited good scavenging activity (IC50: 434.5 ± 6.45 µg/ml) compared to the standard curcumin (IC50: 139.8 ± 3.78 µg/ml). On the contrary, HL and HS extracts does not show any potential to scavenge the nitric oxide radicals. With regard to the FRAP assay, aside HL and HS extracts, all other extracts reduce the ferric ion. Particularly, ML extract revealed an absorbance value of 0.916 ± 0.04, compared to standard ascorbic acid (1.173 ± 0.03), indicating the prominent ferric ion reducing potential of ML extract of F. insignis. All extracts, except HL and HS extracts, revealed dose-dependent hydroxy radical scavenging activity comparable to standard ascorbic acid. Notably, ML extract exhibited promising hydroxyl radical scavenging with an IC50 of 92.8 ± 5.6, found to be equipotent with standard ascorbic acid (IC50: 125.5 ± 3.4).
Antioxidant Activity of F. insignis Extracts by DPPH, Nitric Oxide Radical, Hydroxyl Radical Scavenging and FRAP Assay.

DPPH: 2,2-Diphenyl-1-picrylhydrazyl. IC50: concentration required to scavenge 50% of free radicals in the sample solution. ML: Methanolic leaf; MS: Methanolic stem; EL: Ethyl acetate leaf; ES: Ethyl acetate stem; HL: Hexane leaf; HS: Hexane stem extract. AA: Ascorbic acid and CU: Curcumin used as standard antioxidant.
Antibacterial Activity
The in vitro antibacterial assay showed a dose-dependent antibacterial activity of F. insignis extract against S. aureus, as shown in Table 4 and Figures 8 and 9. Firstly, the disc diffusion assay indicated a zone of inhibition in the range of 11 to 24 mm compared to the standard antibiotic meropenem (24 ± 1.6). Specifically, at a higher concentration of 100 mg/ml, the ES extract was found to inhibit the bacteria most prominently, with the highest ZOI (24 ± 0.8 mm), followed by ML extract (22 ± 1.4 mm), MS extract (20 ± 1.2 mm) and EL extract (18 ± 1.4 mm), while for HL and HS extract, bacteria was found to be resistant. Secondly, the broth dilution assay demonstrated the antibacterial potency of active extracts, such as ML, MS, EL, and ES in terms of MIC value of 15.625, 31.25, 62.5, and 15.625 µg/ml, respectively, compared to standard meropenem (MIC: 1.56 µg/ml).
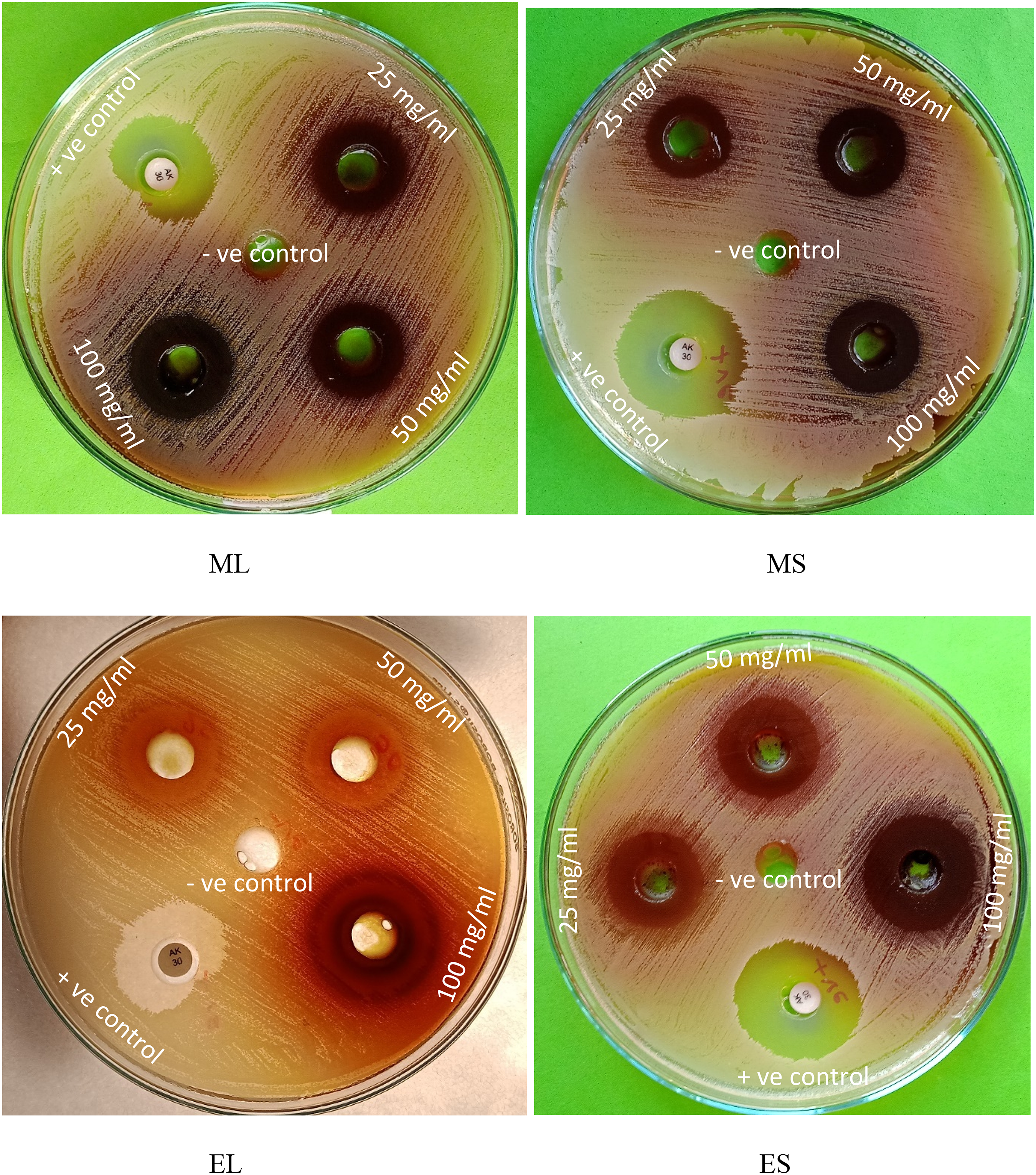
Antibacterial Activity (ZOI) of F. insignis Extracts Against S. Aureus. ML: Methanolic Leaf Extract. MS: Methanolic Stem Extract. EL: Ethyl Acetate Leaf Extract. ES: Ethyl Acetate Stem Extract. Three Different Concentrations (25, 50, 100 mg/ml) of F. Insignis Along with Negative Control (5% DMSO) and Positive Control (Amikacin: 30 μg) were Tested for the Antibacterial Activity.

Photograph Showing the Minimum Inhibitory Concentration of (MIC) of F. Insignis Extract (7.8125 to 1000 µg/ml) and Standard Meropenem (0.78125 to 100 µg/ml) Against S. Aureus. The Blue Color in the Extract or Meropenem Treated Well Indicates Growth Inhibition, While a Pink Color in the Negative Control and Lower Concentration of Extract or Meropenem Indicates Bacterial Growth.
Antibacterial Activity of F. insignis Extracts Using Agar Well Diffusion Method.

Standard antibiotics. AK: Amikacin disc- 30 µg. MP disc: Meropenem disc- 10 µg. ZOI: Zone of inhibition in mm expressed as mean ± SD.
In Silico Studies
Molecular Docking. Docking study revealed that the quercetin had a prominent binding energy of −7.1 kcal/mol, followed by chlorogenic acid (−6.9 kcal/mol) and gallic acid (−6.1 kcal/mol), compared to standard meropenem with a docking score of −7.4 kcal/mol. In BIOVIA Discovery Studio Visualizer, phytoconstituents were observed to bind with almost all catalytic residues through conventional hydrogen bond, carbon hydrogen bond, pi-sigma interaction, pi-pi T shaped interaction, pi-donor hydrogen bond, pi-alkyl interaction, and pi-pi stacked molecular interactions, as explored in Figure 10 and Table 5.
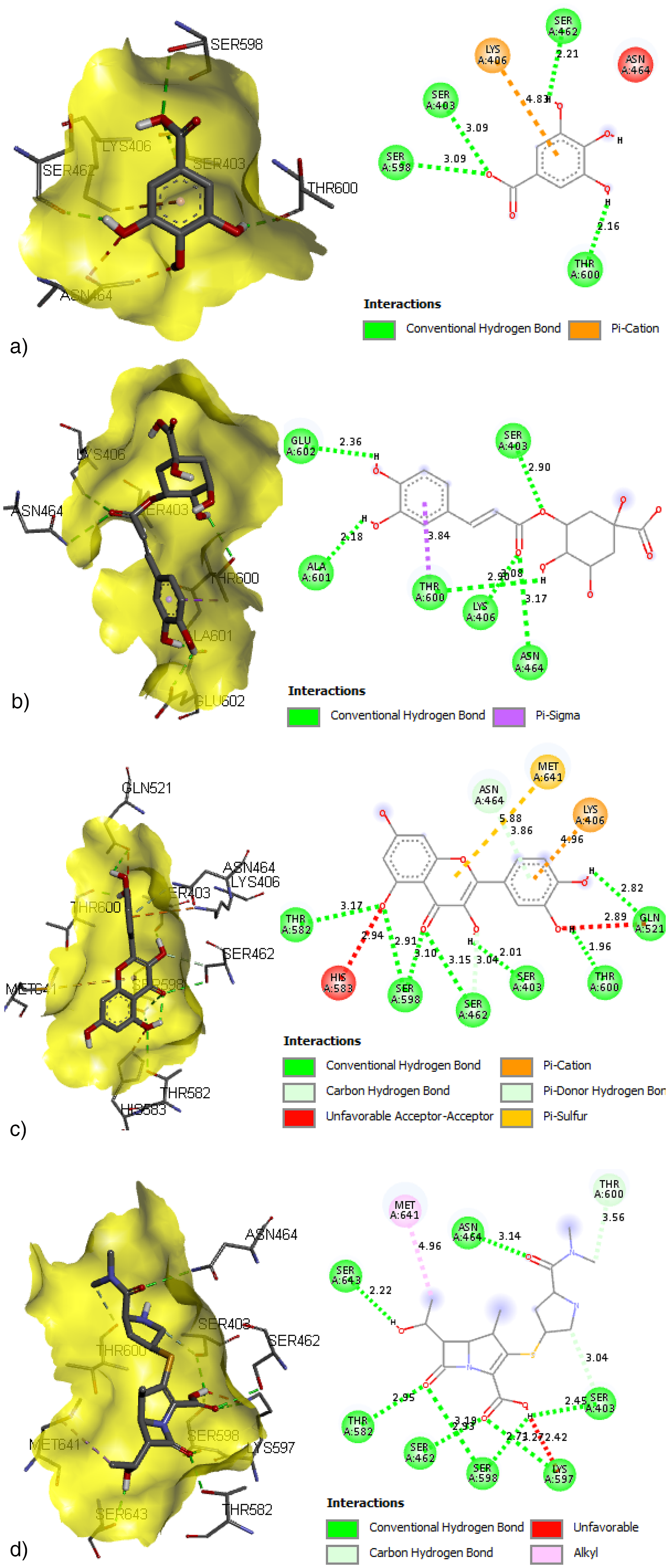
3D and 2D Molecular Interactions of Gallic Acid (A), Chlorogenic Acid (B), Quercetin (C) and Standard Antibiotics Meropenem (D) Within the Catalytic Pocket of PBP 2a.
Binding Energy (kcal/mol) and Molecular Interaction of F. Insignis Phytochemicals with Penicillin Binding Protein of S. aureus.

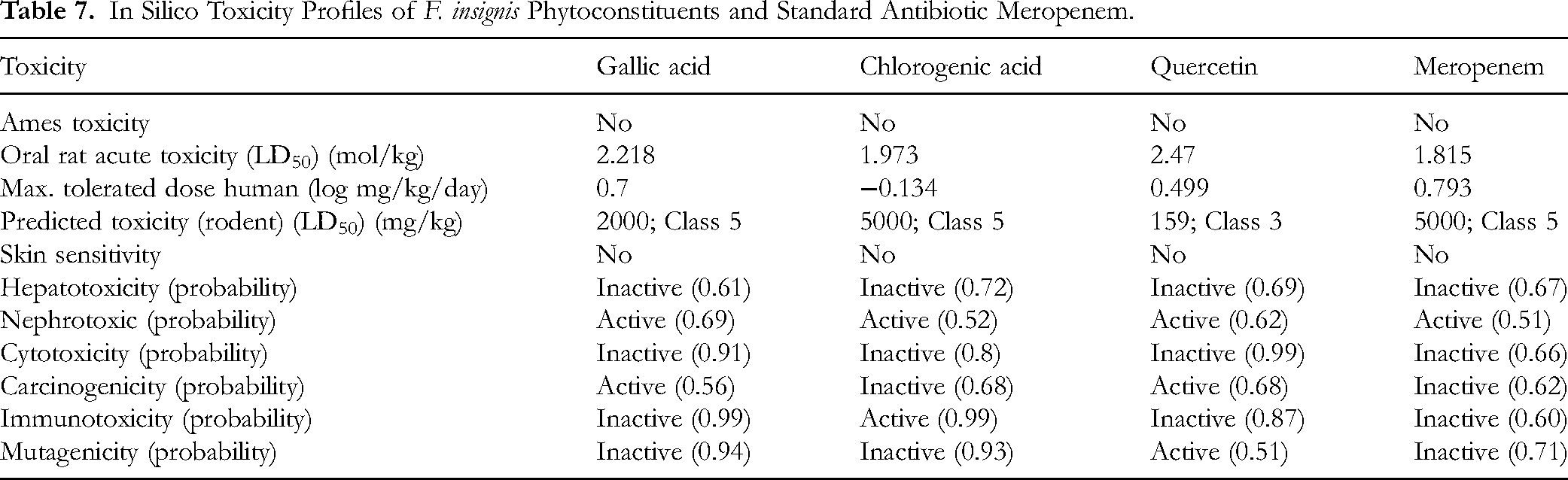
ADMET Analysis. Table 6 meticulously outlines the ADME parameters for F. insignis phytoconstituents and standard antibiotic meropenem. Notably, gallic acid, quercetin and meropenem, with the exception of chlorogenic acid, exhibited TPSA values below 140 Ų. Likewise, except chlorogenic acid, all test compounds (gallic acid, quercetin and meropenem) had passed the Lipinski's Rule of Five. In the realm of in silico toxicity forecasting, all phytoconstituents exhibited a robust safety profile concerning Ames toxicity, hepatotoxicity, and cytotoxicity. Conversely, they also displayed a low potential to induce nephrotoxicity, with a maximum probability of 0.69 for gallic acid, 0.52 for chlorogenic acid, and 0.62 for quercetin, compared to meropenem (0.51), as detailed in Table 7.
ADME Analysis of F. insignis Phytoconstituents and Standard Antibiotic Meropenem.

In Silico Toxicity Profiles of F. insignis Phytoconstituents and Standard Antibiotic Meropenem.
Discussions
Plants serve as the prime drug bank due to their enrichment in diverse phytoconstituents, including phenols, flavonoids, alkaloids, terpenoids, and carbohydrates, each with distinct biological properties. 39 The choice of solvent system for extraction is critical to maximize recovery of these compounds. 40 Polar compounds dissolve well in polar solvents, including methanol, ethanol, and water, while nonpolar compound like terpenes are better extracted using nonpolar solvents such as ethyl acetate and hexane. 41 Therefore, to encompass a full spectrum of phytoconstituents and diverse biological activity, this study employed successive extractions in increasing order of solvent polarity, such as hexane, ethyl acetate, and methanol. 40 Particularly, methanol as the polar solvent system showed the highest percentage of yield for the F. insignis leaf (16.4%) and stem (5.4%), indicating its richness in polar phytoconstituents, while for ethyl acetate and hexane, it decreased, suggesting the rare occurrence of low-polarity compounds in F. insignis.42,43 In contrast, Devkota and his coworker 17 had previously estimated an extraction yield of 7.1% and 10.7% from the successive maceration of F. insigne leaf from Nepal, using methanol and 40% methanol as a solvent, respectively. Moreover, a similar study in China claimed a 5% extraction yield following the percolation of leaves in 95% ethanolic solution. 21 As evidenced by quantitative phytochemical analysis, the methanolic extract showed the highest TPC and TFC contents for leaf and stem, which must be due to the high polarity of methanol and its strong penetrating, diffusing, and solubilizing capacity for polar phytochemicals such as phenolics, flavonoids, and alkaloids. 8 Unlike ethyl acetate's low polarity and hexane's nonpolar nature 44 produces a moderate to lowest content of phenolic and flavonoids due to its diminished capacity to partition the polar phenolic compounds and flavonoids, such as polyphenolic glycosides. 40 Clearly, these mild to moderate ranges of TPC and TFC in hexane and ethyl acetate could be contributed by less polar phenolic compounds, such as flavanones, methylated flavanone, phenolic terpenes and flavonoid aglycone, because it needs less polar solvent to be extracted.40,45
Thin layer chromatography (TLC) is one of the simple and widely employed techniques for the rapid evaluation of phytoconstituents in plant extract and herbal medicines. 46 It uses several derivatizing reagents for the characterization of a specific class of phytochemicals. For instance, FeCl3, H2SO4, and DPPH solution were sprayed for the identification of phenolics, flavonoids, and antioxidants, respectively. 28 In this investigation, the green, blue, and brown bands in the TLC of methanolic leaf extract (ML) and methanolic stem extract (MS), after spraying with 10% FeCl3 as a derivatizing agent, suggest the presence of phenolic compounds, such as catechol, galloyl, and phenol groups. Likewise, the yellow and reddish-brown bands on the TLC of ML and MS extracts, after H2SO4 spray/ heat, indicate the presence of flavonoids and terpenoids, respectively. 46 Moreover, the appearance of yellow bands on the TLC of ML and MS extracts after dipping in DPPH solution confirm the presence of antioxidants in F. insignis extracts. 33
Liquid chromatography-mass spectrometry (LC-MS) is a crucial analytical tool for both quantitative and qualitative analysis, renowned for its high sensitivity and specificity. 30 It is widely used in pharmaceuticals, forensic toxicology, clinical research, and food and environmental testing. 47 This research focus on qualitative analysis of phytochemicals in F. insignis extract using LC-MS equipped with featured electron spray ionization (ESI), a soft ionization source leading to a little fragmentation. 47 Identification of phytoconstituents were based on their mass-to-charge ratios (m/z). ESI typically produces a protonated molecule [M + H]+ in the positive mode (ESI+), and a deprotonated molecule [M-H]− in the negative mode (ESI-). Furthermore, depending on the composition of mobile phase, it also generates adduct ions such as cationized molecules: [M + Na]+, [M + NH4]+, [M + K]+, [2M + H]+, and [2M + Na]+ in the ESI + mode47,48; and anionized molecules: [M + Cl]−, [2M-H]−, [M + HCOO]−, and [M + CH3COO]− in the ESI- mode.47,49 For current analysis, the negative ion mode (ESI-) was selected because of its lower background noise and higher ionization efficiency. 50 Three phenolic phytoconstituents, such as gallic acid, chlorogenic acid, and quercetin were identified by analyzing their spectral data, including deprotonated ion, adduct ion, dimer ion, and trimer ion, as well as by comparing them with the existing literature.
Gallic acid, chlorogenic acid, and quercetin are phenolic secondary metabolites with significant health benefits. These compounds have raised considerable therapeutic interest due to their wide-ranging medicinal properties, such as antioxidant, anti-aging, immunomodulatory, cardioprotective, anti-inflammatory, and anticarcinogenic effects.8,51 Gallic acid is abundantly found in tea leaves; it exhibits strong antioxidant and anti-inflammatory activities, primarily through the scavenging of free radicals and inhibition of inflammatory cytokines and enzymes, thereby protecting cell from oxidative stress. 52 Chlorogenic acid, a rich component of coffee beans, mainly modulates anti-inflammatory and antioxidant mechanisms, as well as metabolic homeostasis by targeting key components like inhibiting the NF-kB signaling cascade and activating the Nrf2 pathway. It also promotes metabolic balance through AMPK pathway stimulation, aiding glucose and lipid homeostasis. 53 Quercetin, a flavonoid renowned for its antioxidant properties, scavenges free radicals, reduces oxidative stress, and inhibits inflammatory cytokines. Quercetin also demonstrates anticancer effects by inhibiting cancer cell proliferation and inducing apoptosis. 54 What's more, it supports cardiovascular health by lowering blood pressure, reducing cholesterol, and enhancing endothelial function. 55
Given the accumulation of these bioactive compounds in F. insignis, it is essential to evaluate their biological activities, particularly their antioxidant and antibacterial properties prior to establish F. insignis as a bioactive crude drug However, due to the complexity and variability of the cellular oxidation process, it is inadequate to execute a single protocol for confirming antioxidant potential comprehensively.11,56 As a result, we performed a set of in vitro screening protocols, including DPPH, NO, OH
The DPPH method is one of the reliable and widely adopted protocols for screening the free radical scavenging potential of natural products. 57 In the current investigation, ML extract was found to have equipotent DPPH free radical scavenging activity as standard antioxidant, ascorbic acid. A previous study on a similar genus, Sapium glandulosum, has reported the DPPH scavenging activity of its ethanolic leaf extract, with an IC50 of 58.5576.35 mg/ml. 58 Likewise, the ethyl acetate extract of Sapium sebiferum leaf exhibited a significant lower IC50 of 4.91 ± 0.08 μg/ml compared to standard ascorbic acid (IC50: 4.797 ± 0.01 µg/ml), 23 which is consistent with our result for the ML extract (IC50: 5.27 ± 0.5 µg/ml). The notable antioxidant properties of the ML extract are attributed to its high TPC and TFC, stemming from phenolic and flavonoid compounds such as gallic acid, quercetin, and chlorogenic acid.51,59 Conversely, HL and HS extracts lacking polyphenolics had demonstrated poor scavenging activity. Previously, Joshi et al reported the promising DPPH scavenging activity of gallic acid, chlorogenic acid, and quercetin, with IC50 values of 43.3 µM, 176.2 µM and 37.7 µM, respectively. 60 These phytochemicals have strong antioxidant activity that protects human tissue against oxidative stress and damage caused by free radicals and ROS, 8 suggesting the prominent potential of the ML extract as a novel antioxidant agent for further drug development.
Nitric oxide is a gaseous endogenous free radical which plays a vital role in physiological and pathological processes. Its excess release leads to several diseases, such as chronic inflammation, asthma, rheumatoid arthritis, carcinoma, hypertension, septic shock, and ulcerative colitis.35,61 In a previous study, Ethyl acetate extract of Sapium sebiferum leaf showed an IC50 of 240.097 ± 29.18 μg/ml compared to standard ascorbic acid (IC50: 13.01 ± 2.53 μg/ml). 23 In contrast, it was found to dominate the scavenging potential of ML extract of F. insignis (IC50: 434.5 ± 6.5 μg/ml) by twice. Phenolics and flavonoids had capabilities to scavenge these radicals. 34 For instance, phytoconstituents, such as gallic acid, chlorogenic acid, and quercetin act as an antioxidant system to compete with oxygen and enable it to react with nitric oxide, consequently reducing harmful nitrite production. 35 As a nitric oxide scavenger, F. insignis helps to treat or prevent diseases related to excessive nitric oxide radicals. However, further cell line and animal experiment-based validation deemed necessary for advancing their clinical use.
Hydroxyl radical is one of the highly oxidizing free radicals, 36 which causes oxidative damage to biomolecules such as lipids, peptides, and nucleic acids, and consequently leads to organ damage, mitochondria dysfunction, cancer, and body aging.36,62 All extracts revealed dose-dependent hydroxy radical scavenging activity comparable to standard ascorbic acid. Notably, the ML extract exhibited promising hydroxyl radical scavenging with an IC50 of 92.8 ± 5.6 μg/ml, found to be more potent than that of standard ascorbic acid (IC50: 125.5 ± 3.4 μg/ml), indicating its significant potential to be developed as an alternative hydroxyl radical scavenger to prevent or treat the above-mentioned pathogenesis.
The FRAP assay is based on the capability of antioxidant marker of plant extract to reduce Fe3+ complex to the Fe2+ complex through electron donating ability, Where the Fe2+ complex can be monitored by measuring absorbance of the developed Perl's Prussian blue color at 700 nm. A higher absorbance of the reaction mixture at 700 nm indicated the potential ferric reducing power of the extract.35,63 ML extract displayed the highest absorbance value 0.916 ± 0.04 at 200 μg/ml, indicating their strong ferric ion reducing potential, which may be attributed to the reductant nature of its intrinsic polyphenolic compounds. 35
The global proliferation and dissemination of antibiotic resistance, alongside the emergence of novel bacterial strains, pose a significant threat to public health.12,64 Consequently, the effective management of infectious diseases necessitates rigorous research and the development of innovative therapeutics derived from potent phytochemical sources. 64 Natural products have gifted a diverse array of antibacterial lead compounds that serve as effective agents in combatting infections. Notable examples include allicin from Allium sativum (garlic), Berberine from Berberis vulgaris (barberry), piperine from Piper nigrum (black pepper), catechin from Camellia sinensis (green tea), thymol from Thymus vulgaris (thyme), curcumin from Curcuma longa (turmeric), limonene and α-pinene from Zingiber officinale (ginger), and eugenol from Syzygium aromaticum (cloves).65,66 These phytochemicals have been commercialized within healthcare settings as plant-derived antimicrobials. Their intrinsic antibacterial properties not only demonstrate significant efficacy, but it also facilitate the rejuvenation of the clinical application of older antibiotics by enhancing their antimicrobial potency. 66 This synergistic effect potentially mitigates the emergence of antibiotic resistance and further contributes to control the infections more efficiently.
Our investigation revealed a dose-dependent antibacterial activity against S. aureus, with the ES extract showing the most pronounced effect. Similarly, a prior study reported that the ethyl acetate fraction of Sapium sebiferum leaves, rich in phenolic compounds, had an MIC of 256 μg/mL against S. aureus. 26 This MIC of value of S. sebiferum was significantly higher than that of F. insignis extracts: ML (15.625 µg/mL), MS (31.25 µg/mL), EL (62.5 µg/mL), and ES (15.625 µg/mL), underscoring the superior efficacy of F. insignis. The significant antibacterial activity of these extracts against S. aureus, which is characterized by a high zone of inhibition and low MIC values, may be attributed to the synergistic effects of the cognate phenolic compounds such as quercetin, cholinergic acid, and gallic acid.67–73 Moreover, cytotoxic diterpenes such as sapinsignoids A−G and phorbol ester reported previously from the leaf and stems of F. insignis,17,21,22 may also account for its antibacterial properties. 74 Several previous studies have demonstrated that phenolic acids and flavonoids bind via hydrogen bonds to disrupt the cell membrane, 75 inhibit its synthesis, suppress enzyme synthesis, and reduce ATP formation, ultimately leading to bacterial death.33,67,71,76–78 Specifically, quercetin causes cellular electrolytes leakage by significantly disrupting the cell walls and plasma membranes of S. aureus. 76 Likewise, Sang et al 79 observed that gallic acid effectively inhibited the formation of new biofilms and the maturation of existing biofilms, which are responsible for the first line defense and exaggerative resistance in S. aureus. Furthermore, chlorogenic acid demonstrated significant inhibitory activity against S. aureus by reducing intracellular ATP levels, lowering PH, altering membrane polarity, destroying the membrane, and causing the release of intracellular constituents. 80 On the contrary, the resistance profile of bacteria to the extracts like HL and HS, could be due to the lack or a lower amount of antibacterial phytoconstituents, including phenolics and flavonoids in these extracts. 81 Furthermore, none of the F. insignis extracts showed inhibitory activity against E. coli. These resistance observed in E. coli to extracts of F. insignis and their phytoconstituents may be due to the restricted penetration offered by the presence of impermeable lipopolysaccharide in the outer cell membrane of E. coli. 82 Furthermore, the upregulation of hydrolytic enzymes and efflux pump, and mutation of porin, along with the downregulation of PBP and influx pump were also considered to be the prime reason behind the resistance of gram negative bacterial strain.33,75
Molecular docking is a computational technique used in drug discovery and molecular biology to aid a deep understanding of the binding mechanisms between drugs and their target proteins, facilitating the design of novel therapeutic compounds. 83 With regards to the promising in vitro antistaphylococcal activity of F. insignis extracts, we conducted in silico molecular docking studies to uncover the mechanism of action underlying the antibacterial activity. 28
Penicillin-binding protein (PBP 2a) is a membrane protein of S. aureus that catalyzes the critical transpeptidation cross-linking polymerization reaction in bacterial peptidoglycans cell wall synthesis and is considered a potential target for novel antibacterial compounds.84,85 Antibiotic meropenem, which inhibit these enzymes, have been used as antibacterial drugs in clinical settings globally for a long period of time. In predocking, an RMSD value of 0.8177 Å (< 2) was obtained when the docked pose was superimposed with the native co-crystal ligand, which validated the accuracy and precision of the docking process. 86 Furthermore, the co-crystalized piperacillin was mapped using BIOVIA Discovery Studio to identify the catalytic triad and active residues within the 3D crystal structure of host proteins; it discovered numerous active residues, including, SER 403, LYS 406, TYR 446, SER 462, ASN 464, TYR 519, GLN 521, SER 598, GLY 599, THR 600, and MET 641 for site-specific docking studies. 87
The molecular docking results unveiled compelling insights regarding binding energy, interacting residues, bond types, and bond lengths, showcasing a relationship in which stronger biological activity is associated with lower (more negative) binding energies. 27 The interactions involving catalytic residues, along with the prevalence of conventional hydrogen bonds and reduced bond lengths, underscore the significant antimicrobial potential. Notably, gallic acid, chlorogenic acid, and quercetin demonstrated docking scores comparable to the standard antibiotic meropenem, indicating their formidable antibacterial capabilities. The dominance of conventional hydrogen bonds in these interactions further underlines the stability of phytoconstituent-enzyme complexes, 30 reinforcing our in vitro findings and providing robust scientific evidence for the therapeutic efficacy of F. insignis as a potential antibacterial crude drug. Given these promising results, we recommend further in vivo and cell line studies not only to validate current findings, but also to pave their ways for potential clinical applications.
Accurate prediction of absorption, distribution, metabolism, and excretion (ADME) parameters, as well as toxicity, is critical to ensure the safety and efficacy of bioactive phytoconstituents in drug discovery and development processes. 28 Implementing innovative in silico approaches such as SwissADME, pKCSM and ProTox-3.0 offers a valuable and reliable methods for forecasting ADME-toxicity properties, presenting significant advantages such as cost-effectiveness, rapid analysis, eco-friendly predictions, and the ability to bypass animal testing. 27 Its key concept, such as Lipinski's Rule of Five predict the pharmacokinetic attributes of the test compounds with more precision. Compounds that adhere the Lipinski's Rule of Five, including molecular weight (≤500 g/mol), lipophilicity (Log P ≤ 5), number of hydrogen bond acceptors (≤10), hydrogen bond donors (≤5), and TPSA (≤140 Ų), had excellent solubility, membrane permeability, and efficacy, positioning them as strong candidates for drug development.88,89 In the current investigation, gallic acid and quercetin had satisfied all five rules of Lipinski; therefore, they confirmed excellent absorption and superior bioavailability. 90
Lipophilicity and hydrophilicity are critical parameters that influence not only the solubility, but it also affects the absorption, distribution, metabolism, and excretion (ADME) profiles, potency, and toxicity of drugs. 28 Notably, the consensus Log Po/w is represents an average across five predictive methods and serves as a pivotal metric, where a Log P value between 1 and 4 signifies the optimal characteristics of ADME for oral administration. 91 Our findings indicate that quercetin falls within this desirable range, while gallic acid, chlorogenic acid, and meropenem (with a Log P of 0.21, −0.39 and −0.37, respectively) signify suboptimal ADME for oral routes, reinforcing their limited usability as oral medications. 28 It is critical to note that highly lipophilic drugs (Log P > 5) often face challenges such as poor aqueous solubility, inadequate oral absorption, and increased risk of toxicity. 91 The more negative Log Kp values for meropenem suggest its poor skin permeation compared to the phytoconstituents of F. insignis. 88
The cytochrome P450 (CYP) isoenzyme is responsible for the metabolism of approximately 80% of drugs and plays a crucial role in influencing drug bioavailability and toxicity risk. 88 Especially, its subtype, CYP3A4, metabolizes around 50% of therapeutic agents. 92 Gallic acid and quercetin displayed inhibitory characteristics against CYP isoenzymes, suggesting a higher likelihood of extensive metabolism through these pathways. 88 Moreover, the P-glycoprotein (P-gp) transporter in enterocytes serves to limit oral bioavailability by actively returning drugs back into the intestinal lumen. 28 Only gallic acid was identified as a P-gp substrate; thus, it affects its absorption efficiency.
In terms of drug development properties, quercetin meets the lead-likeness criteria, while gallic acid and chlorogenic acid exhibit one violation concerning molecular weight. Notably, the gallic acid and quercetin showed a bioavailability score of 0.55 and 0.56, indicating a probability of 55% and 56% of achieving effective oral bioavailability, while for chlorogenic acid, it dropped to 11%. The synthetic accessibility scores for all phytoconstituents were under 10, which suggested they can be synthesized with relative ease in laboratory settings. 88
The toxic dose of the compounds was displayed as the medium lethal dose (LD50), a dose in mg/kg body weight at which 50% of the test subjects die upon administration. The thresholds for toxicity were classified into diverse classes according to the LD50 figures. Especially, Class I is categorized as fatal if swallowed (LD50 ≤ 5); Class II is fatal if taken (5 < LD50 ≤ 50); Class III is labeled as toxic if swallowed (50 < LD50 ≤ 300); Class IV is harmful if administered (300 < LD50 ≤ 2000); Class V indicates that it may be harmful if swallowed (2000 < LD50 ≤ 5000); and Class VI is considered as non-toxic (LD50 > 5000). This standard scale was implemented to assess the potential risks linked with exposure to the compounds of F. insignis. 27 All of the compounds were safe for the liver, while they had minimal potential to cause harm to the rodent kidney. Therefore, despite these insights, it is imperative to emphasize that robust molecular, cell line, genetic, and in vivo studies are essential for comprehensive validation of these in silico safety claims.
Conclusion
This study provides an insightful exploration into the phytochemical composition and biological activity of F. insignis extract, identifying key phenolic compounds such as gallic acid, chlorogenic acid, and quercetin through LC-MS analysis. The in vitro bioassays revealed significant antioxidant and antibacterial properties, particularly against S. aureus, thereby supporting the ethnomedicinal use of the extract for treating bacteria related infections. Additionally, in silico molecular docking and ADME-toxicity studies underscored the potential molecular interactions of these phytoconstituents with catalytic residues of the penicillin-binding protein of S. aureus, further reinforcing the therapeutic potential of F. insignis. However, due to the lack of sophisticated laboratory setup, this study is limited in its scope, primarily focusing on in vitro and in silico analyses without comprehensive identification of bioactive compounds responsible for the biological activities. The absence of cell line and in vivo evaluations restrict the robustness of our claims regarding the efficacy and safety of F. insignis. To advance this research, future studies should aim to isolate and thoroughly characterize individual bioactive compounds, conduct detailed toxicity assessments, and perform in vivo experiments to better understand the pharmacological profiles and therapeutic applications of F. insignis in treating bacterial infections. These directions will not only validate the current findings, but it also paves the way for the development of novel antibacterial agents from this promising plant species.
Footnotes
Acknowledgments
The authors would like to express sincere thanks to Dhan Raj Kandel, Assistant Research Officer of National Herbarium and Plant Laboratories, Godawari, Lalitpur, Nepal for the authentication of plant specimen.
ORCID iDs
Author Contributions
Ram Kishor Yadav participated in the conceptualization, investigations, methodology, data curation, original draft writing, review and editing; Sushil Panta participated in methodology, conceptualization, supervision and validation; Sindhu KC, Prabhat Kumar Jha, Siddha Raj Upadhyaya, Sandesh Poudel and Bipindra Pandey, participated in methodology, formal analysis, original draft writing, review and editing; Kushal Subedi, Susmita Baral, and Rekha Bhandari assisted the investigation.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw data required to reproduce these findings are available in the body and illustrations of this manuscript.

