Abstract
Objectives/Background
Several plants with various chemical constituents have been widely explored for managing diabetes mellitus. One of the most common strategies is the inhibitors of the α-amylase and α-glucosidase, key regulatory enzymes in diabetes. This study aims to investigate the anti-diabetic activity of methanolic extract of Coccinia barteri leaves using in vitro α-amylase enzyme inhibition assay and in silico molecular docking study.
Methods
The dried pulverized leaf of Coccinia barteri was extracted by maceration using methanol. Qualitative and quantitative phytochemical analyses of the powdered leaf extract were carried out using standard procedures. The extract was fractionated using n-hexane, ethyl acetate, n-butanol and aqueous methanol. In vitro anti-diabetic study of the different fractions and sub-fractions was investigated by α-Amylase Inhibition Assay. Sub-fractions of the ethyl acetate fraction of C.barteri leaf with the highest in vitro anti-diabetic activity were subjected to GC-MS analysis. The compounds detected by GC-MS were selected as ligands for α-amylase and α-glucosidase in the molecular docking study.
Results
The phytochemical analysis revealed the presence of saponins, alkaloids, tannins, glycosides, steroids, flavonoids, and cyanide. Compared to the control drug, the ethyl acetate fraction of Coccinia barteri leaf gave a 2-fold lower IC50. GCMS chromatogram of the most active sub-fraction revealed the presence of 18 major compounds. Piperine showed good binding affinity (−6.9 kcal/mol) to α-amylase and displayed hydrogen bonding with ARG 344 and HIS 210, along with pi alkyl bonds with TRP 83, HIS 80, LEU 166, and LEU 232. For α-glucosidase, piperine and 2,4-di-tertbutylphenol surpassed the standard with binding energies of −7.1 kcal/mol and - 6.9 kcal/mol, respectively. Drug likeness and toxicity assessments confirmed adherence to Lipinski's rule, with both compounds showing non-mutagenic and non-tumorigenic properties.
Conclusion
The ethyl acetate fraction of the Coccinia barteri leaf exhibits potential anti-diabetic activity, which may be attributed to the inhibition of α-amylase and α-glucosidase, by its constituents, piperine and 2,4-di-tertbutylphenol.
Introduction
Diabetes mellitus is one of the most common chronic diseases globally. It is a complex metabolic disease characterized by hyperglycaemia resulting from the deficiency or malfunctioning of insulin. 1 Out of a hundred diabetic patients, about five suffer from Type 1, ninety from Type 2 and the remaining five patients from other subtypes of diabetes. 2 The morbidity and mortality rate of diabetes is high as approximately 420 million people suffer from the disease annually, and 1.5 million people die of diabetes-related issues. 3 Both the incidence and the prevalence of diabetes have continued to increase at alarming rates over the past few decades, especially in developing countries. 4 The associated complications of diabetes include glaucoma, cataracts, diabetic foot, diabetic retinopathy, peripheral retinopathy, stroke and heart attack. 5
One of the clinical signs of Type 2 diabetes mellitus is postprandial hyperglycemia. To reduce the absorption of glucose, the hydrolytic action of carbohydrate digestive enzymes should be inhibited or at least moderated. The major enzyme involved in carbohydrate digestion is α- amylase. During digestion, α- amylase released from the salivary glands and the pancreas digest carbohydrates to many oligosaccharides, which are further digested by α-glucosidase to glucose to be absorbed in the human intestine. Consequently, there is a surge in blood glucose levels, resulting in hyperglycaemia in diabetic patients. Hence, the inhibition of this key enzyme retards carbohydrate digestion, decreasing the absorption and distribution of glucose into the bloodstream, consequently reducing postprandial hyperglycaemia. 6 Based on this, inhibition of α-amylase is recognized as an essential tool in managing Type 2 diabetes mellitus.
The conventional drugs used in the treatment of Type 2 diabetes include insulin, metformin, glipizide, glimepiride and pioglitazone. The failure in therapeutic outcomes and adverse effects associated with these commercially available drugs have warranted the search for better alternatives. The well-known α-amylase inhibitors such as Acarbose, Miglitol and Voglibose exhibit adverse effects, including hepatotoxicity and gastrointestinal effects such as diarrhoea, flatulence and bowel bloating. 7 There is renewed interest in α-amylase inhibitors derived from plants as alternatives in treating diabetes mellitus because of their efficacy, minimal side effects and low cost.8,9
The development of computational screening methods has drastically reformed drug discovery. Computational approaches facilitate a drug screening process with significantly reduced time and cost. Molecular docking is widely used to predict the binding affinity of a drug to an enzyme or receptor when their structures are known. 10 It has been extensively used to identify drug-like compounds interacting with a specific enzyme or receptor to inhibit its function. 11 Structurally, α-amylase is composed of three domains, as shown in Figure 1. The central N-terminal domain A is the largest and comprises an eight-stranded parallel beta (β/α)8)-barrel. The active site residues (Asp197, Glu233, and Asp300) are at one domain's end. A bound chlorine ion is also found in the domain. Domain B is the smallest and forms a calcium-binding site against the wall of domain A. Domain C consists of an anti-parallel beta-structure loosely associated with Domain A.12,13
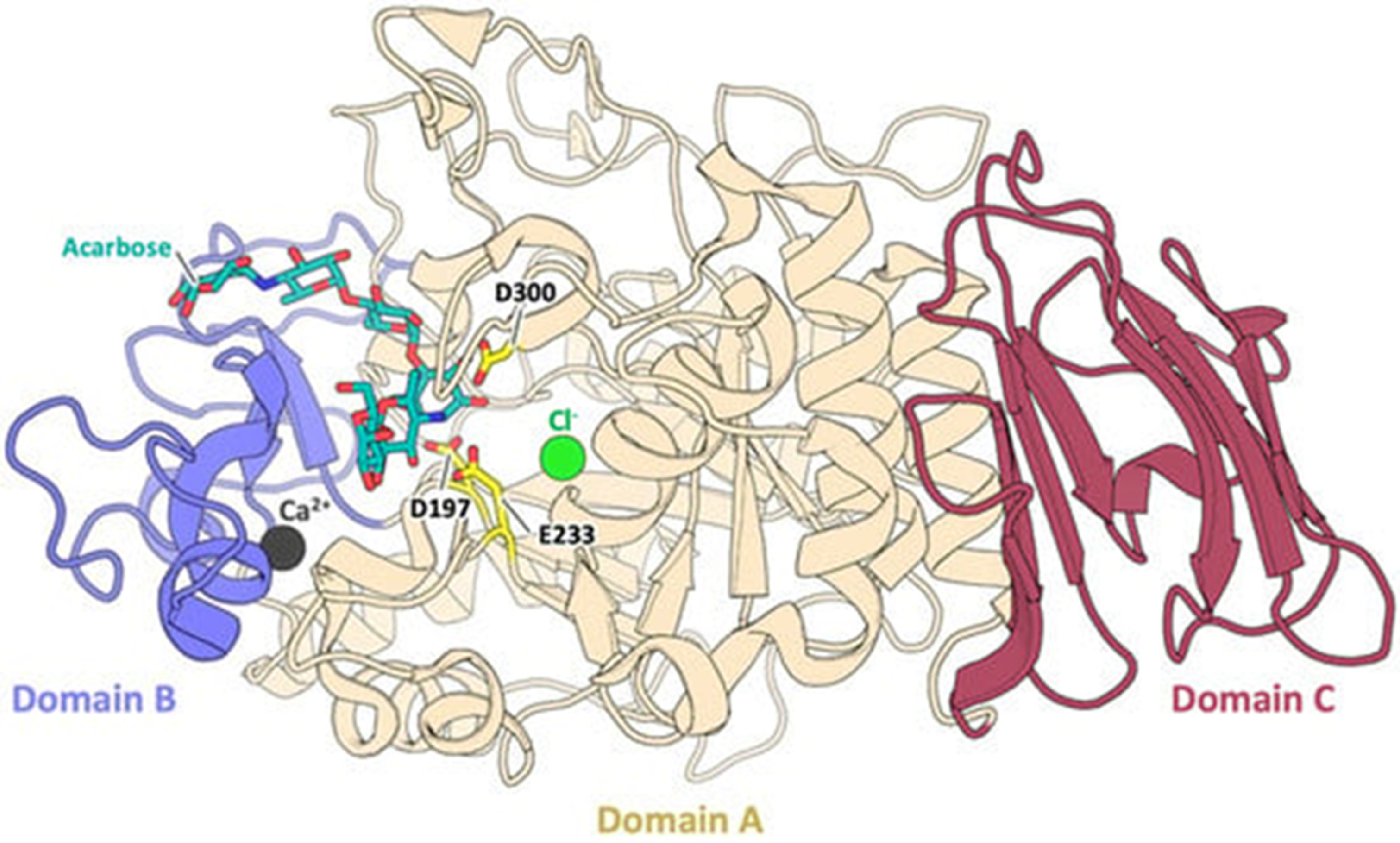
Domains of α-amylases. The three domains (A, B, and C) are shown in different colours. 13 .
Coccinia barteri is a plant from the family Cucurbitaceae that is widely distributed across Asia, Africa, and Australia. The plant exhibits a climbing or trailing growth habit, adorned with distinctive lobed leaves and small, elongated fruits. 14 Some species of Coccinia, such as C.grandis and C.indica, have been reported to have anti-diabetic activity.15,16 Coccinia barteri, with its unique phytochemical profile, presents an intriguing reservoir for bioactive compounds that could aid in ameliorating oxidative stress, which has been implicated in the development and progression of diabetes and other diseases. 17 The antimalarial, antimicrobial and antioxidant activities of Coccinia barteri leaves have been reported in the literature.18,19 We previously reported the in vivo anti-diabetic activity of ethyl acetate fraction of Coccinia barteri leaf. 20 In the present study, we investigated the in vitro anti-diabetic activity of the ethyl acetate fraction of Coccinia barteri using the alpha-amylase inhibition assay. We further analyzed the plant extract using GC-MS to identify the bioactive compounds. The detected compounds were subjected to in silico molecular docking studies to expedite the screening process and provide insights into the structural basis of enzyme inhibition.
Materials and Methods
Materials
Chemicals and Reagents
Glibenclamide (Aventis Pharma Ltd, Goa, India), α - amylase (Sigma Chemical Company, St. Louis, USA), alloxan monohydrate (Sigma Chemical Company, St. Louis, USA). All other chemicals and reagents used in this study were of analytical grade.
Database and Software Used in in Silico Molecular Docking
Database used were Protein Data Bank (https://rcsb.org/, Natural Product Database ((http://african-compounds.org/) and PubChem (https://pubchem.ncbi.nlm.nih.gov/). The software used were AutodockVina 1.1.2 on the Ubuntu operating system, Pymol, AutoDock 1.5.6, Ubuntu 18.04, Biovia Discovery studio, MS Office, Swiss ADME and Data Warrior
Plant Material
The fresh leaves of Coccinia barteri were collected from Nsukka (6o51ʹ24ʺN7o23ʹ45ʺE), Enugu State, in southeastern Nigeria. The plant was identified and authenticated by a taxonomist of the Pharmacognosy and Environmental Medicine Department, University of Nigeria Nsukka, Nigeria. The plant specimen with voucher number UNN/11712 was deposited at the University of Nigeria, Nsukka Herbarium. The collected plant leaves were air-dried at room temperature for two weeks, and they were ground to a fine powder and stored in a moisture-free, airtight container.
Preparation of Plant Extract
The extraction was carried out using the cold maceration technique. 21 300 g of the powdered dry leaves of C.barteri was macerated with 1500 mL of methanol for 72 h with intermittent agitation every 6 h to ensure complete extraction. The methanolic extract was filtered through the Whatman No. 1 filter paper. The filtrate was concentrated with a rotary evaporator and evaporated to dryness over a water bath to obtain the dried extract.
Preliminary Qualitative and Quantitative Phytochemical Analysis
Preliminary qualitative and quantitative phytochemical analyses of the leaves of Coccinia barteri were done according to standard procedures.22,23
Qualitative Phytochemical Analysis
Test for Saponins
Briefly, 5 mL of the extract was mixed with 20 ml of deionized water. The mixture was observed for persistent foaming on vigorous shaking.
Test for Alkaloids
A 0.1 mg extract was added to 6 ml of dilute HCl. The mixture was boiled for a few minutes, allowed to cool, and then filtered. The filtrate was divided into three portions. Two drops of Dragendorff's reagent were added to the first portion. The production of a crimson precipitate revealed the presence of alkaloids. Two drops of Meyer's reagent were applied to the second portion. The formation of creamy white precipitates indicates the presence of alkaloids. Two drops of Wagner's reagent were applied to the third portion. A reddish-brown precipitate indicated the presence of alkaloids.
Test for Tannins
A 10 ml volume of deionized water was mixed with 1 ml extract. Three drops of ferric chloride were added to the mixture. A greenish-brown precipitate indicated the presence of tannins.
Test for Flavonoids
1 ml of the extract was mixed with an equal volume of diluted ammonia solution. A bright yellow colouration indicated the presence of flavonoids.
Test for Terpenoids
1 g of the extract was mixed with 9 ml of ethanol and shaked for a few minutes. The alcoholic mixture was filtered and the filtrate was concentrated to 2.5 ml by boiling over a water bath. Hot distilled water (5 ml) was added to the mixture and allowed to stand for an hour before the waxy material was filtered out. The filtrate was extracted using a separating funnel and 2.5 ml of chloroform. After drying in a water bath, the chloroform extract was dissolved in 3 ml of strong sulfuric acid and heated in the water bath for 10 min. A grey colour indicated the presence of terpenoids.
Test for Steroids
5 ml of chloroform was used to dissolve 0.5 g of extract. An equal volume of concentrated sulphuric acid was added to the mixture. A steroidal ring was seen as a reddish-brown colour at the interphase.
Test for Glycosides
0.1 g of the extract was combined with 5 ml of diluted sulfuric acid in a test tube. The mixture was then heated for 3 min in 10 ml of ethyl acetate, filtered, and cooled. The filtrate (4 ml) was cooled and neutralized with a 20% potassium hydroxide solution after being heated for 15 min in a water bath. A mixture of equal parts of Fehling's solutions A and B was added and heated for 5 min. The appearance of a dense brick-red precipitate indicated the presence of glycosides.
Test for Cyanide
In a test tube containing 0.1 g of the extract, 15 ml of distilled water was added. An alkaline picrate paper was hung and fastened with a rubber bung over the mixture. The setup was left to stand at room temperature for 18 h. The colour change from yellow to orange identified cyanide's presence.
Quantitative Phytochemical Screening of Coccinia barteri Leaf Extract
Test for Saponins
The extract (1 g) was macerated in 10 ml of petroleum ether and decanted into a beaker. An additional 10 ml of petroleum ether was added to the beaker, filtered and evaporated to dryness. The residue was dissolved in 6 mL of ethanol. 2 ml of the solution was mixed with 2 ml of chromogen solution, and the mixture was allowed for 30 min. The absorbance was measured at 550 nm, and the quantity of saponins was calculated from the standard curve of saponin. 24
Test for Alkaloids
1 g of the plant extract was macerated using 1:1 v/v of 20% H2SO4 and ethanol. 5 ml of 60% H2SO4 was mixed with 1 ml filtrate. After 5 min, 5 ml of 0.5% formaldehyde in 60% H2SO4 was added to the mixture and left to stand for 3 h. The absorbance at 565 was obtained, and the amount of alkaloid was calculated from the standard curve of atropine. 24
Test for Tannins
After macerating the extract in 50 ml of methanol, 0.3 ml of 0.1N ferric chloride in 0.1N HCl and 0.3 ml of 0.0008 M potassium ferric cyanide were added to 5 ml of the filtrate. The absorbance was measured at 720 nm amount of tannins determined from a standard curve of tannic acid.
Test for Flavonoids
20 ml of ethyl acetate was added to 1 g of the extract and macerated for 5 min. The mixture was filtered. 5 ml of the filtrate was mixed with 5 ml of dilute ammonia, and the mixture was agitated for 5 min. The supernatant was obtained, and the absorbance was measured at 490 nm from the standard curve of quercetin. 24
Test for Terpenoids
1 g of the extract was macerated in 50 ml of ethanol. The filtrate (2.5 ml) was combined with 2.5 ml of concentrated H4SO4 and 2.5 ml of a 5% aqueous phosphomolybdic acid solution. The mixture was allowed to stand for 30 min, after which ethanol was added to increase the volume to 12.5 ml. The absorbance was measured at 700 nm, and the concentration of terpenoids was calculated using a standard curve equation of linalool. 24
Test for Steroids
20 ml of ethanol was added to 1 g of the extract, macerated and then filtered. 2 ml of the chromogen solution was added to the filtrate, and the mixture was allowed to stand for 30 min before the absorbance was measured at 550 nm.
Test for Glycosides
1 g of the extract was macerated with 50 ml of distilled water and then filtered. 4 ml of alkaline picrate solution was added to 1 ml filtrate. The mixture was boiled for 5 min, cooled and the absorbance measured at 490 nm. The concentration of glycosides was obtained using a standard calibration equation of glucose. 24
Test for Cyanide
1 g of the extract was macerated with 50 ml of distilled water and then filtered. 4 ml of alkaline picrate solution was added to 1 ml filtrate. The mixture was boiled for 5 min and then cooled, and the absorbance was measured at 490 nm.
Fractionation of the Methanol Extract of the Leaves of Coccinia barteri
Fractionation of the crude extract was carried out using four solvents: n-hexane, ethyl acetate, n-butanol, and aqueous methanol. Briefly, the crude extract was dissolved in aqueous methanol solution. Then, an appropriate volume of n-hexane was added to the solution and poured into a separating funnel. The mixture was allowed to stand for 20 min for proper separation, and the upper layer was collected. More n-hexane was used to wash the aqueous methanol portion repeatedly. The different n-hexane fractions were collected. 25 The process was repeated using ethyl acetate and n-butanol.
The most active fraction of Coccinia barteri leaf extract was sub-fractionated using the Vacuum Liquid Chromatography. The column was loaded with the analyte-stationary phase (silica gel 200-400 mesh size, 400 g, sintered glass 5L), after which the apparatus was connected to the vacuum pump, and the elution process began.
Firstly, the analyte was eluted with 500 ml each of dichloromethane and ethyl acetate in the ratio of 100:0, 90:10, 80:20, 70:30, 60:40, 50:50, 40:60, 30:70, 20:80, 10:90, 0:100 . Then, butanol and ethyl acetate was used next to wash the analyte in the ratio of 10:90, 20:80, 30:70, 40:60, 50:50, 60:40, 70:30, 80:20, 90:10, 100:0 ethyl acetate: butanol. Finally, 100% methanol was used. At the end of each elution, the column was allowed to run completely dry before adding the next solvent system. The sub-fractions were pooled together using the preparative thin-layer chromatography method.
Αlpha-Amylase Inhibition Assay
The α-amylase inhibition assay was performed using the 3,5-dinitrosalicylic acid (DNSA) method.
26
A total of 100 μl of sample and positive control of Acarbose were pipetted into a test tube, mixed with 25 μl of porcine pancreatic α-amylase solution (Sigma Chemical Company, St. Louis, USA) and buffered to 500 μL with phosphate buffer, which was then incubated for 10 min at 25 °C. 100 μl starch substrate was added to the mix and incubated at 25 °C for 10 min. The reaction was stopped by adding 400 μl of 3,5-dinitrosalicylic acid solution (DNS) and heating in a water bath for 15 min. The mixture was cooled at room temperature, and the absorbance was measured using the UV-Vis spectrophotometer instrument at a wavelength of 540 nm. A blank reaction was similarly prepared using the plant extract at each concentration without the enzyme solution. A control sample was also prepared without the plant extract or standard drug, and the same procedure was followed. The procedure was carried out in triplicates. The α-amylase inhibitory activity was expressed as percent inhibition and was calculated using the equation given below:
Gas Chromatography and Mass Spectroscopy (GC–MS) Analysis
The most active sub-fraction of the ethyl acetate fraction of C. barteri was subjected to GC-MS analysis using GC-MS QP-2010 (Shimadzu, Japan). A BPX – 35 capillary column (60 m x 0.25 mm and 0.25μm film thickness) was used, and pure helium gas (99.995%) was employed as the carrier gas with a flow rate of 1 ml/min at head pressure of 104.1 kPa. A 1μl split injection (230 °C, 37 min) was used. The initial temperature was 50 °C for 10 min, followed by 3 °C/min rise up to 150 °C. At last, the temperature was raised to 300 °C at a rate of 10 °C/min. High energy electrons (70 eV) were used in an electron ionization system for the spectroscopic detection.27,28
GC–MS data was interpreted using the National Institute of Standards and Technology (NIST) database. The spectra of the detected compounds of Coccinia barteri leaf extract were compared with the spectra of known compounds stored in the NIST library. 29 The retention time, peak area and quality of the compounds were recorded.
In Silico Molecular Docking Studies
The in silico molecular docking studies were carried out using some databases and software.
Literature Mining, Selection and Preparation of Targets
Some biological databases and journal publications were mined to discover possible targets/receptors for the phytochemicals. The 3D structures of the targets/proteins selected during the literature mining were obtained from Protein Data Bank (https://rscb.org). The PyMOL software was used to prepare the pdb files to choose the needed chains and delete multiple ligands and non-protein parts, such as water molecules.
This software was also used to view the 3D ligand-receptor binding and to prepare the individual targets for molecular docking. Another software, Autodock tools, was used to add polar hydrogens to the protein/targets. Then, they were exported as a Pdbqt file (the file format recognized by the molecular docking simulator for both ligands and receptors/targets). The Autodock tools software was used to create grid boxes of different sizes and centres around the active site of the protein. 30
Selection and Preparation of Ligands
The SDF-3D format of 18 Phytochemicals obtained from Coccinia barteri sub-fraction were obtained from PubChem (http://pubchem.ncbi.nlm.nih.gov/) and the Natural Products Database ((http://african-compounds.org/). The ligands co-crystallized with the receptor site on the proteins were used as the reference ligand/standard. Some known inhibitors of the different enzymes were also downloaded from Pubchem in the same format. Using Autodocktools, the ligands were prepared for molecular docking. All rotatable bonds and torsions were added, and the ligands were saved as Pdbqt files. Alternatively, the compounds/ligands downloaded in Sdf format were converted to Pdbqt format using Open Babel on Ubuntu in the Linux Operating System. The prepared ligands and the protein targets/receptors were used in molecular docking.
Validation of Docking Protocol
In order to validate the molecular docking protocol for the selected proteins, the PDB structure of these proteins in complex with their reference inhibitors were reproduced in silico. The deletion of the reference compounds from the protein was done. Polar hydrogen, grid box sizes and centres at a grid space of 0.35Ǻ were determined with AutoDock 1.5.6. The reference inhibitors were docked using the grid box properties. The reference compound used for both proteins was “ACARBOSE”.
Molecular Docking of the Phytochemicals on the Selected Enzymes
The Phytochemicals identified from Coccinia barteri sub-fraction using GC-MS were downloaded and batched for molecular docking against the α-amylase (at center x,y,z: 39.755, 37.555, 30.442) and size (x,y,z: 48, 54, 40) and α-glucosidase (at center x,y,z: −13.247, −37.553, 95.166) and size (x,y,z: 26, 54, 26) at with respective PDB ID of 7TAA and 5NN8, respectively, using virtual screening scripts. Molecular docking was carried out in four replicates on the Linux platform using AutodockVina® and associated tools after the validation of docking protocols. Binding free energy values (kcal/mol ± SD) were ranked using an Excel spreadsheet to identify the frontrunner phytochemicals.
Post-Docking Analysis
The mean binding affinities and standard deviations were calculated and recorded on the Microsoft Excel platform. The binding affinities of the 18 phytochemicals were compared with the reference compounds’ binding affinities. The phytochemicals with binding affinities higher or equal to that of the reference compounds for each protein were collected and checked for drug-likeness. Meanwhile, the phytochemicals with affinities lower than that of the reference compounds for each protein were screened out.
Drug-Likeness and Toxicity Assessment
The drug-likeness of the frontrunner phytochemicals was evaluated using Datawarrior software.31,32 Lipinski's rule of five was utilized to filter the frontrunners with drug-like properties. 33 Frontrunner phytochemicals with no Lipinski violation were subjected to in silico toxicity analysis using Datawarrior software. Datawarrior assesses compounds for the following toxicities- mutagenicity, tumorigenicity and reproductive effect. Frontrunners that showed any of this toxicity were deleted. Discovery Studio shows the 2D interaction of the frontrunner phytochemicals and the receptor site to identify which amino acids the compounds interact with. The interaction was also compared to the standard.
Statistical Analysis
Statistical analysis of the results was done using one-way ANOVA using GraphPad software, followed by the Tukey comparison test for significance. Significance was set at P < 0.05
Results and Discussion
Percentage Yield of Extract
The percentage yield of the methanol extract of C.barteri leaves was 10.31% (30.93 g). We chose maceration as the extraction method for the dried leaves of C. barteri because it is simple (does not involve special armamentarium), cheap and has been reported to yield better extract. 34 According to the findings by some researchers, better antimicrobial and hypoglycemic activity was obtained from samples extracted by cold maceration than the one obtained through the Soxhlet and microwave-assisted extraction methods.35,36
Qualitative and Quantitative Phytochemical Analysis
Phytochemical analysis assesses the presence and amount of secondary metabolic compounds present in plants. The quantitative and qualitative estimation of secondary metabolites in plants is vital for discovering and exploring new bioactive compounds in the pharmaceutical and agrochemical industries.
From our result (Table 1), the methanol extract of the leaves of Coccinia barteri contains high proportions of tannins and flavonoids, moderate amounts of alkaloids and glycosides and low proportions of saponins, cyanide, terpenoids and steroids. Generally, plants are reservoirs of natural compounds having diverse structures and pharmacological activities. The medicinal properties of C. barteri leaves, such as antioxidant, anti-inflammatory and antimicrobial properties, can be attributed to these secondary metabolites. These phytochemicals might be responsible for the anti-diabetic properties of this leaf via their individual or synergistic effects. 37
Phytoconstituents of the Methanol Extract of Coccinia barteri Leaf.

Key: + = Present.
Results of Αlpha-Amylase Inhibition Assay
The in vitro anti-diabetic activity of the Coccinia barteri leaves was determined using the α-Amylase Inhibition Method. In this method, the activity of alpha-amylase is measured through a REDOX reaction. Alpha-amylase hydrolyzes starch into reduced sugars, which react with DNS reagents to form a dark orange-coloured complex of 3-amino-5-nitrosalicylic acids. If the hydrolysis of starch is inhibited, there will be little or no reducing sugars to react with DNS, resulting in fewer 3-amino-5-nitrosalicylic acids. Therefore, inhibition of α-amylase results in a decrease in the concentration and absorbance of 3-amino-5-nitrosalicylic acid.
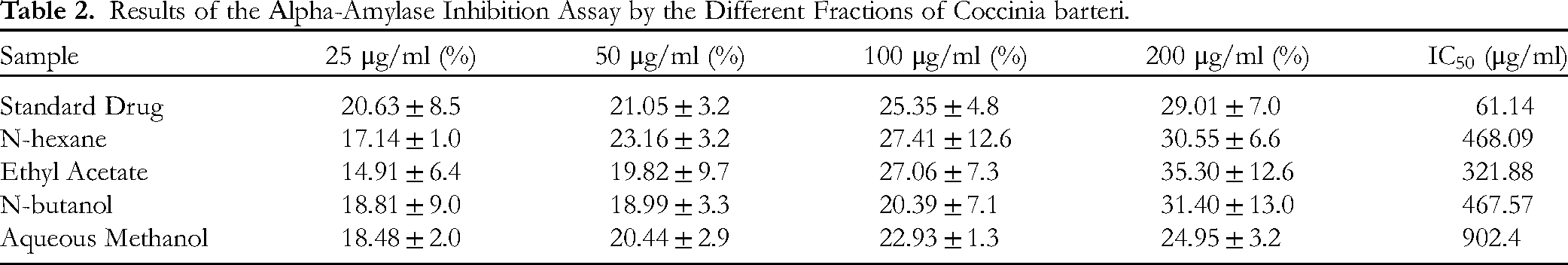
The percentage inhibition of α-amylase enzyme caused by the different concentrations of the various fractions of C. barteri leaf extract is presented in Figure 2. From the plot, the concentration of the extract that will cause 50% enzyme inhibition (IC50) was calculated, and the result is presented in Table 2. The ethyl acetate fraction exhibited the lowest IC50 of 321.88 μg/ml, and the IC50 values of n-butanol, n-hexane, and aqueous methanol fractions were 467.57 μg/mL, 468.09 μg/mL and 902.4 μg/mL, respectively. The positive control showed an IC50 of 61.14 μg/ml.

Enzyme inhibition as a function of concentration of different fractions of Coccinia barteri leaf extract.
Results of the Αlpha-Amylase Inhibition Assay by the Different Fractions of Coccinia barteri.
The ethyl acetate fraction with the lowest IC50 was considered the most active fraction. Twenty-two sub-fractions of the active fraction of C. barteri were obtained from the column chromatography and merged into seven sub-fractions by preparative Thin-layer chromatography (TLC) based on their retention factor. The sub-fractions were again subjected to α-amylase inhibition assay. The result is presented in Table 3.
Results for Percentage Inhibition of sub-Fractions of Ethyl Acetate Fraction of Coccinia barteri Leaf Against the α-Amylase Enzyme.

The sub-fractions of the ethyl acetate fraction of C. barteri gave IC50 of 4.88–24.93 mg/mL. The sub-fraction 2 gave the lowest IC50 value, while sub-fraction 4 gave the highest value. Based on this result, sub-fraction 2 was selected as the fraction with the most potent anti-diabetic activity and used for further analysis.
GC-MS Analysis
The GS-MS chromatogram of Coccinia barteri is presented in Figure 3. The GS-MS chromatogram revealed the presence of many bioactive compounds, including dodecane, piperine, cyclohexadecane, oxalic acid, 2,4-Di-tert-butylphenol, cetene, Bis(2-ethylhexyl)phthalate, Cis-vaccenic acid and 9-octadecenoic acid. These compounds have been reported to have different pharmacological activities, including hypoglycaemic, anti-obesity and antioxidant activities. The peak number, retention time, area, quality and reported activities of the eighteen major compounds detected in the ethylacetate fraction of Coccinia barteri are presented in Table 4. 2,4-Di-tert-butylphenol and piperine had the highest peak areas, indicating their abundance in the leaf of Coccinia barteri. Piperine is an alkaloid with a plethora of pharmacological activities, including antiproliferative, anti-diabetic, antioxidant, anti-obesity, cardioprotective, antimicrobial, hepatoprotective, anti-allergic, anti-inflammatory, neuroprotective and immunomodulatory activities. 38 Similarly, 2,4-Di-tert-butylphenol is a phenolic compound reported to have antifungal, antibacterial, anticancer and antioxidant effects.39,40

Chromatogram of GC MS of Coccinia barteri.
Bioactive Constituents of Ethyl Acetate Fraction of Coccinia barteri Leaf Detected by GC-MS.

In Silico Molecular Docking Results
Molecular docking is an in silico structure-based technique extensively used in drug discovery to identify new therapeutic compounds, predicting their molecular interaction with specific targets. 41 It has brought about a paradigm shift in drug discovery and development. Several authors have used in silico molecular docking to identify potential inhibitors of α-amylase and α-glucosidase.42,43 The molecular interaction of the major constituents of the ethyl acetate fraction of Coccinia barteri with two diabetes mellitus-related target proteins was investigated. Of the constituents, three of these, piperine, 2,4-di-tert-butylphenol and cyclohexadecane, exhibited good enzyme inhibition and served as the ligands, while the enzymes α-amylase and α-glycosidase were the target proteins. The 3D structure of α-amylase and α-glucosidase is shown in Figure 4.

3D structural representation of a) Αlpha-amylase (7TAA) and b) Αlpha-glycosidase (5NN8).
In preparing the targets, water molecules were removed as they can pose serious problems. The grid box was generated by docking the ligands at the centroid of the binding sites. Generation of the grid box is vital for fixing the active site of the target.
Cyclohexadecane and piperine exhibited substantial negative binding energies (−7.0 and −6.9 Kcal/mol, respectively) with α-amylase, designating them as frontrunner compounds (Table 5). Despite falling short of the standard Acarbose's binding energy (−11.3), their interactions merit attention. A binding energy value less than zero implies a spontaneous binding of the ligand to the target. 44
Frontrunner Compounds for α-Amylase (7TAA) and α-Glucosidase (5NN8) with Their Binding Energies.

The interaction of piperine with α-amylase is shown in Figure 5. Piperine displayed hydrogen bonding with ARG 344 and HIS 210, along with pi alkyl bonds with TRP 83, HIS 80, LEU 166, and LEU 232. Conversely, cyclohexadecane showed no interactions with any amino acid in α-amylase, eliminating it as a contender.
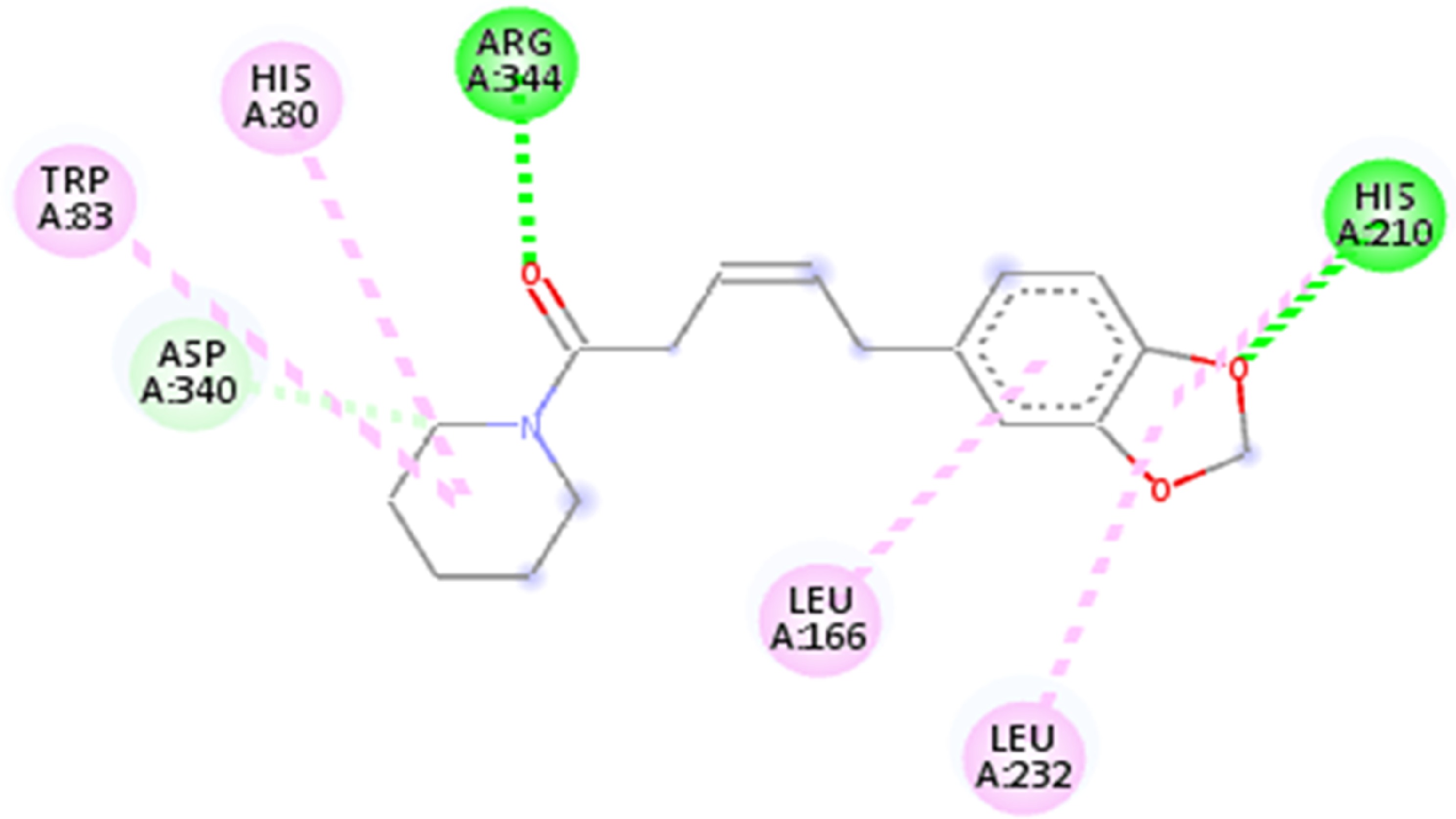
Interaction of piperine with α-amylase.
The metabolism of carbohydrates follows sequential steps involving both α-amylase and α-glucosidase. Therefore their actions are complementary. During digestion, α-amylase released from the salivary glands and the pancreas digest carbohydrates to many oligosaccharides, which are further digested by α-glucosidase to glucose to be absorbed in the human intestine. Therefore, we also decided to study the phytochemicals’ inhibitory effect on the α-glucosidase enzyme. From our results, piperine and 2,4-Di-tertbutylphenol surpassed the standard with binding energies of −7.1 and −6.9, respectively (Table 5). The interaction of these ligands with α-glucosidase is shown in Figure 6. Piperine showcased extensive hydrogen and alkyl bond interactions with HIS 674 and multiple amino acids, outperforming Acarbose. This result is in agreement with findings from previous studies. For instance, Magaña-Barajas et al reported that piperine binds at the α-glucosidase and α-amylase through hydrogen bonds and charge interactions with amino acid residues. 45 In another study, piperine was shown to inhibit α-amylase and α-glucosidase enzymes in vitro and in vivo, as well as to improve insulin sensitivity and glucose tolerance in diabetic rats. 46 Similarly, 2,4-di-tertbutylphenol, isolated from Moringa oleifera leaves, was reported to inhibit α-glucosidase enzyme and reduce postprandial hyperglycemia in diabetic mice. 47

Interaction of a) Piperine with α-glucosidase b) 2,4-di-tertbutylphenol with α-glycosidase.
The validation of the docking protocol was performed to ascertain the accuracy of the process. This was done by using the PyMOL software to check if the docked reference ligand overlapped on the same receptor site as the co-crystallized ligand. The docking protocols were validated, as shown by our result in Figure 7.
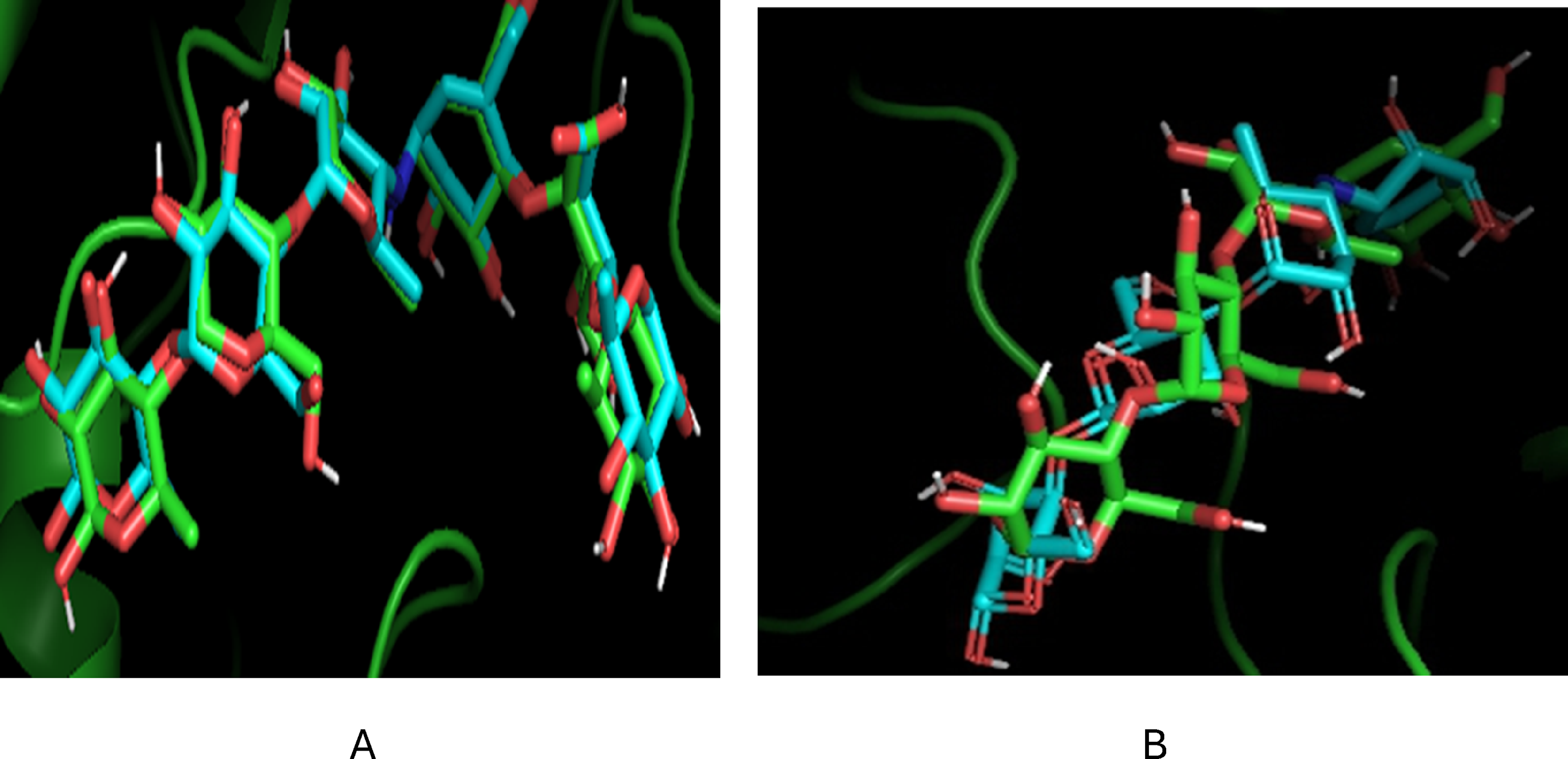
Validation of docking protocols a) α-amylase (7TAA) and b) α-glucosidase.
Biovia Discovery Studio revealed that both piperine and 2,4-di-tertbutylphenol adhered to Lipinski's rule, indicating drug-likeness with no violations. 48 Their molecular weights were less than 500, cLogP values less than 5, and hydrogen acceptors and donors than 10 and 5, respectively. Both compounds demonstrated non-mutagenic and non-tumorigenic characteristics (Table 6). Both ligands exhibit high reproductive effects. However, piperine exhibited a higher reproductive effect than 2,4-di-tertbutylphenol.
Drug-Likeness and Toxicity of the Phytocompounds.

The criteria for the six physicochemical properties investigated for the bioavailability radar of piperine were fulfilled. These properties are flexibility, lipophilicity, size, polarity, insolubility and insaturation. Therefore, piperine (represented by the red lines) is considered drug-like as it is wholly within the physicochemical range on each axis, as shown in Figure 8.

Bioavailability radar of piperine.
Our study provides new insights into the phytochemistry and pharmacology of Coccinia barteri and validates its ethnomedicinal use. This study confirms that piperine and 2,4-di-tertbutylphenol from Coccinia barteri are promising candidates for the development of novel anti-diabetic drugs. They are derived from natural sources, which may enhance their acceptability and availability for the patients. However, our study also has some limitations that need to be considered. First, our study was based on in silico screening methods, which may not fully reflect the in vivo situation and may have some errors and uncertainties. Second, our study focused on only two enzymes involved in carbohydrate metabolism, which may not capture the complexity and diversity of the pathophysiology of diabetes. Therefore, further investigation of the effects of these compounds on other targets and pathways related to diabetes is warranted. Third, our study used only one plant source to extract the phytoconstituents, which may limit the generalizability and reproducibility of our findings. Therefore, further exploration of the diversity and variability of the phytoconstituents from different plant sources and parts is needed.
Conclusion
The present study investigates the phytochemical constituents of Coccinia barteri leaf as well as the in vitro and in silico anti-diabetic activities. The study shows that Coccinia barteri leaves have many phytochemicals of therapeutic importance. The ethyl acetate fraction of the plant leaf exhibited significant inhibitory activity on α-amylase in vitro. In addition, this study identified piperine and 2,4-di-tertbutylphenol from Coccinia barteri as potent inhibitors of α-amylase and α-glucosidase, which are key targets for managing diabetes. These compounds also showed favourable drug-likeness and toxicity profiles, suggesting their potential as novel anti-diabetic agents in the management of diabetes mellitus. However, isolation of these compounds and further studies should be carried out.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies on human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.

