Abstract
Introduction
Ulcerative colitis (UC), a significant subtype of inflammatory bowel disease (IBD), is characterized by diffuse, superficial inflammation localized to the intestinal mucosa. Inflammatory lesions are situated within the mucosal and submucosal layers, rendering it a refractory intestinal disorder.1,2 Restricted exclusively to the colon, UC typically manifests with bloody diarrhea, accompanied by symptoms such as abdominal pain, urgency, mucous-laden stools, and tenesmus. Additionally, systemic manifestations may include fatigue, anorexia, fever, and pyoderma gangrenosum. Investigations indicate that apart from complications such as fulminant colitis, toxic megacolon, perforation, and hemorrhage, the progression of UC to colorectal carcinoma is a frequently encountered phenomenon.3,4 Given its intricate pathogenesis, efficacious therapeutic modalities remain elusive, necessitating a pharmacological approach encompassing various agents such as aminosalicylates, corticosteroids, immunosuppressants, biologics, and small molecules. Despite the widespread clinical use of these agents, prolonged usage causes considerable hepatorenal toxicity, with a propensity for disease relapse following discontinuation. 5 The empirical application of traditional Chinese herbal compounds reveals novel therapeutic advantages in the management of UC. This approach offers dual benefits: maintaining sustained remission and reducing relapse rates while mitigating gastrointestinal reactions and minimizing the emergence of complications. Moreover, this strategy demonstrates cost-effectiveness, promising a favorable outlook within the therapeutic landscape of UC.
Zuo Jin pill (ZJP), a traditional Chinese herbal compound, consists of 2 medicinal ingredients, Coptis chinensis, and Evodia rutaecarpa, in a ratio of 6:1. It stands as a quintessential representative formulation within traditional Chinese medicine for the management of gastrointestinal disorders.6,7 Esteemed for its safety, efficacy, and prevention of disease recurrence, ZJP has found extensive utility in clinical practice and boasts a clinical therapeutic history spanning over 600 years. 8 Multiple studies have substantiated the efficacy of ZJP in addressing UC9,10; however, the precise mechanistic basis of its therapeutic action in UC remains incompletely elucidated. In recent years, network pharmacology, employing a holistic research paradigm characterized by “multi-component-multi-target-complex disease,” has provided preliminary insights into the mode of action of traditional Chinese medicine in disease treatment. 11 This approach is bolstered by subsequent validation through animal experimentation or clinical investigation, employing factor detection or protein analysis, thereby providing novel methodological avenues for exploring traditional Chinese medicine in disease management.12,13
In this study, we aimed to explore the potential signaling pathway underlying the therapeutic effects of ZJP in UC through a comprehensive analysis. This analysis involved the integration of drug targets present within the constituent ingredients of ZJP and disease targets associated with UC. The potential signaling pathways of ZJP for the treatment of UC were identified by conducting an enrichment analysis of these targets. Furthermore, our findings substantiated the efficacy of ZJP in treating UC through animal experimentation, thereby validating the accuracy of the pathways identified via network pharmacology. Additionally, the study offers a detailed elucidation of the operative mechanism of these pathways, thus introducing novel insights for the treatment of UC and concurrently establishing a theoretical foundation for the clinical application of ZJP.
Results and Discussion
Results
Active Compounds in ZJP
A comprehensive array of 66 potentially active chemical constituents sourced from 2 Chinese herbal entities was meticulously compiled using the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP) database. Through a systematic exploration of the PubChem and SwissTargetPrediction databases, the prospective target genes of the 66 active constituents in ZJP were thoroughly scrutinized. Considering species as humans, targets of potential active ingredients were selected based on a probability threshold of ≥ 0.15. The target corresponding to Evodia remained elusive. Following a rigorous filtration process aimed at eliminating redundancy, a refined set of 189 discerning drug targets was successfully obtained.
Identification of Active Compounds in ZJP as Potential Targets for UC Treatment
By analyzing the repositories of OMIM, Therapeutic Target Database (TTD), DisGenet, DrugBank, and GeneCards, an exhaustive search was conducted to identify targets pertinent to UC. By carefully removing duplicate disease targets, we identified a total of 3175 distinct UC-related targets. The integration of the active constituents of ZJP and UC-associated targets was performed using the “Venn Diagram” software. (Figure 1a). Following this integration, a group of 103 pivotal targets, including EGFR, PI3KR1, ALOX5, and ESR2, was obtained. These targets are hypothesized to exert a substantial influence on the therapeutic landscape of UC through the utilization of ZJP. The set of 103 intersecting targets was entered into the STRING database to construct a multiprotein interaction network, thus providing a comprehensive insight into their intricate associations.
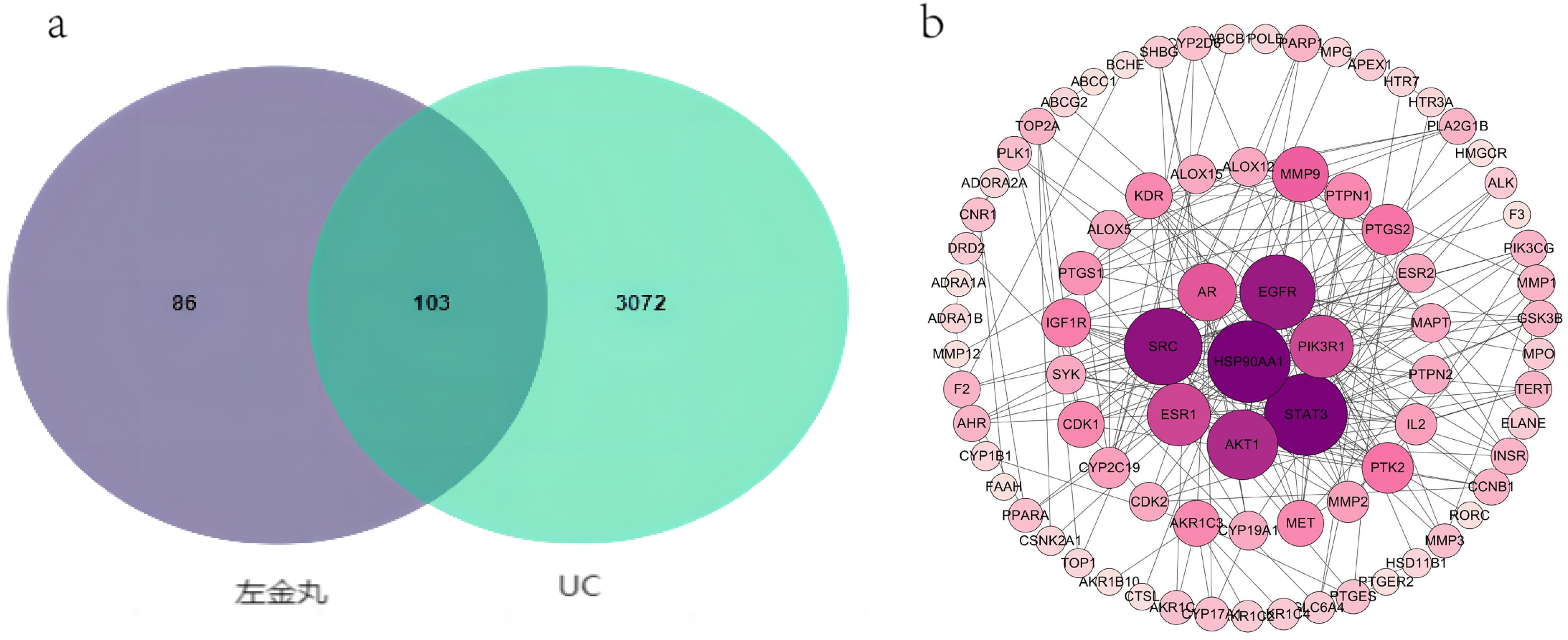
One hundred and three targets of ZJP in UC treatment: (a) Venn diagram of intersection targets of ZJP and UC, and (b) ZJP acts on the protein–protein interaction network and core target proteins of UC.
The protein–protein interaction network encompassed a set of 103 nodes interconnected by 239 edges, with each node representing a protein-coding gene. The size of a node corresponded to the degree value of the encoded protein, indicating its significance within the network. The connections established between nodes represented functional relationships between specific proteins, and the thickness of these connections reflected the strength of the correlation between the paired proteins. The mean degree value of each target protein averaged at 5.9. Proteins such as STAT3, HSP90AA1, SRC, AKT1, EGFR, ESR1, and PIK3R1 exceeded the mean degree value and were ranked significantly high, underscoring their pivotal roles within the network (Figure 1b).
Results of Gene Ontology (GO) Enrichment and Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Analyses
To elucidate the intricate biological implications of the 103 potential targets, we conducted a comprehensive exploration via GO and KEGG enrichment analyses. The GO analysis revealed that these 103 targets exerted influence over a broad range of 337 biological processes, 56 cell components, and 123 molecular functions. Applying a significance threshold of P < .05, we selected the top 20 attributes encompassing molecular functions, biological processes, and cellular components for compilation into a bar chart using the Weishengxin platform (Figure 2a). The therapeutic actions of ZJP in UC primarily involved coordinating responses to exogenous stimuli, eliciting drug responses, moderating the aging process, facilitating positive regulatory effects on vasoconstriction, and promoting protein autophosphorylation. The major influenced cellular components included the plasma membrane, components of presynaptic membranes, cytoplasm, extracellular exosomes, and binding organelles of cell intima (Figure 2b). On the molecular front, ZJP primarily exerted its effects through enzyme binding, protein kinase activity, protein kinase binding, ATP binding, isoprotein binding, and REDOX enzyme activity. Collectively, these findings underscore the multifaceted biological mechanisms through which ZJP exerts its therapeutic potential in the context of UC.

Enrichment analysis of GO and KEGG pathways in 103 UC targets using ZJP: (a) GO analysis of 103 potential targets associated with ZJP therapy for UC. The number of significantly enriched genes is plotted on the X-axis and the BP, CC, and MF terms (P < .05) are plotted on the Y-axis. (b) Point plots showing the top 20 KEGG enrichment pathways. The size of each dot corresponds to the number of genes for annotation, and the color of the dot corresponds to the corrected P-value.
Analysis Results of Disease-Pathway-Target-Active Ingredient-Drug Visualization Network
Using Cytoscape 3.7.1 software, we created a comprehensive visual representation that links disease, targets, active constituents, and pathway-drug associations. Our analysis revealed that ZJP stood out due to its multifaceted composition of active ingredients, a diverse array of targets, and intricate interactions with various pathways. Key active compounds in ZJP included quercetin, berberine, β-sitosterol, and evodipine. These compounds exhibited the ability to activate crucial targets such as STAT3, AKT1, EGFR, ESR1, PTGS2, and PIK3R1, thereby modulating a range of signaling pathways. This orchestrated action encompassed metabolic pathways, cancer pathways, the PI3K-AKT signaling pathway, chemical carcinogenic-reactive oxygen species pathways, proteoglycans in cancer pathways, neuroMars receptor–ligand interactions, and chemocarcinogenic receptor pathways. By exploiting this intricate network, ZJP employed a multipronged approach to effectively combat UC (Figure 3).

Disease-pathway-target-active ingredient-drug visualization network diagram.
Measurement of Body Mass, Colon Length, and Disease Activity Index in Each Group
To assess disease activity among mice in each group, the disease activity index (DAI) score was used. This composite score not only incorporated alterations in the weight of mice (Figure 4a), but also considered stool characteristics and the presence of occult blood. The inflammation score of colonic tissue among mice in the model group exhibited a marked elevation compared with that in the control group (P < .0001). Following therapeutic interventions, UC-afflicted mice within the medication group experienced a substantial reduction in their DAI scores (Figure 4b).

(a) Change in body weight of mice; (b) disease activity index (DAI) scores for different groups of mice (x ± s, n = 15). Compared with the control group, ***P < .001; compared with the model group, ###P < .001.
Upon completion of the experimental regimen, the length of the colon in distinct mouse groups was measured using a tape (Figure 5). The findings revealed that the colon length within the blank group averaged 10.17 ± 0.90 cm. In contrast, the colon length within the model group decreased significantly (P < .001), settling at ∼ 5.25 ± 0.96 cm. In the presence of drug intervention, a noticeable enhancement in abbreviated colon length among all experimental cohorts occurred (P < .001). The colon length within the positive drug group was 9.62 ± 0.10 cm, whereas the low-dose ZJP group recorded a measurement of roughly 7.07 ± 0.18 cm. The medium-dose ZJP group exhibited a colon length of 8.05 ± 0.19 cm, whereas the colon length of the high-dose ZJP group was ∼ 9.61 ± 0.11 cm.

Change in colon length of mice. Compared with the control group, ***P < .001; compared with the model group, ###P < .001.
Effect of ZJP on Colon Histopathology in UC Mice
The colonic tissue architecture adheres to a hierarchical and orderly arrangement. As depicted in the initial illustration in Figure 6, the colon exhibits a layered composition comprising 4 distinct components: the mucous membrane, submucosal layer, muscular layer, and serosal layer. In the control group, the colon maintained a state of health and normalcy, featuring an intact mucosal epithelial structure. Although the intestinal mucosa displayed slight wrinkles, its contours remained natural, displaying a typical arrangement of columnar surface colon cells (Co), numerous goblet cells (GC), and crypts (represented by dashed black lines) on the surface of the colonic tissue. Transitioning to the model group, as depicted in the second illustration, a substantial loss of crypts and goblet cells was observed, accompanied by hyperplasia of connective tissue. Inflammatory cells, primarily composed of lymphocytes, macrophages, and granulocytes, extended from the mucosal layer into the submucosal layer (indicated by blue arrows), accompanied by a degree of submucosal tissue edema (denoted by green arrows). Additionally, the colonic lumen contained remnants of necrotic cells (marked by red arrows), appearing in a pale purple hue. Following drug intervention, marked amelioration of colonic histopathological conditions was evident. This included the alleviation of dextran sulfate sodium (DSS)-induced histological damage, the regeneration of new colonic crypts, diminished infiltration of inflammatory cells largely confined to the mucosal layer, and relief of submucosal. It further comprised a significant reduction in the presence of necrotic cells within the colonic cavity. Among the treatment groups, the mesalazine and high-dose ZJP groups exhibited highly intact colonic structures and decreased inflammatory cell infiltration, indicative of a favorable therapeutic response.
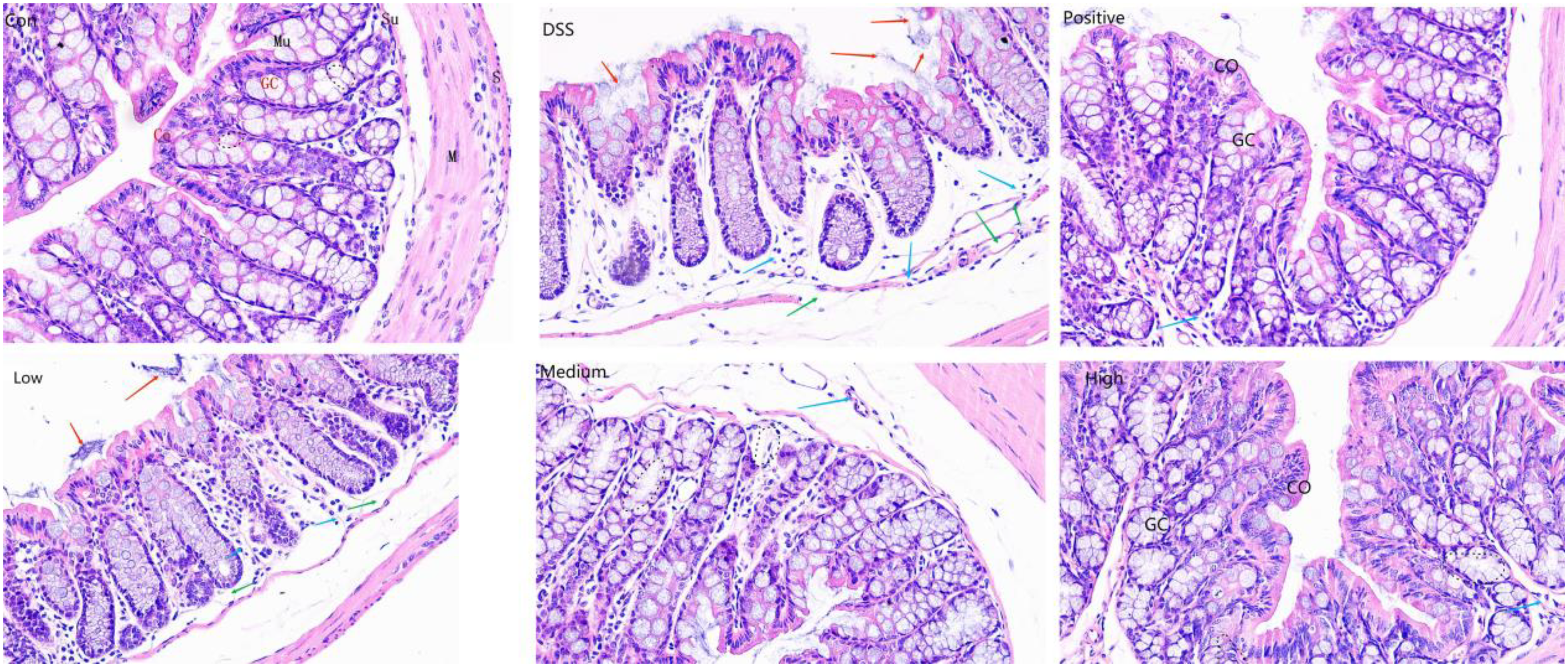
Pathological examination of colonic tissue (hematoxylin and eosin [HE] 200 ×).
Colonic cells (Co), cupular cells (GC), mucosa (Mu), submucosa (Su), muscularis (M), and plasma layer (S). Black dashed lines represent colonic crypts. Blue arrows indicate inflammatory cell infiltration, green arrows indicate submucosal edema, and red arrows indicate necrotic exfoliated cells.
Determination of Levels of Inflammatory Factors IL-6, IL-10, and TNF-α in Colon Tissue Using ELISA
To further delineate the extent of colon inflammation, we assessed the expression levels of several common inflammatory factors within colonic tissue, specifically IL-6, IL-10, and TNF-α (Figure 7). Comparison of IL-6 levels in colonic tissues across different mouse groups revealed a significant upregulation of the inflammatory factor in the model group compared with that in normal mice (P < .001). Following treatment with mesalazine and high, medium, and low doses of ZJP, the expression of IL-6 within the colonic tissues of mice was reduced. Particularly, the expression of IL-6 in the mesalazine-positive group and medium- and high-dose ZJP groups exhibited a significant decrease (P < .001). Additionally, compared with the model group, the low-dose ZJP group demonstrated a decrease in IL-6 expression (P < .01). Moreover, statistical analyses indicated no substantial difference in efficacy between mesalazine and high-dose ZJP (P > .05), suggesting a similar effectiveness of the 2 drugs.

Comparison of IL-6, IL-10, and TNF-α expression in colonic tissues of UC mice. Compared with the control group,***P < .001; compared with the model group, ###P < .001, ##P < .01, and #P < .05; compared with the positive control group, ▴▴▴P < .001.
Compared with that in the control group, a significant reduction in IL-10 expression was observed in the DSS-induced model group of mice (P < .001). Subsequent treatment resulted in varying degrees of increased IL-10 expression within the colonic tissues of each group of mice. Specifically, the medium- and high-dose ZJP groups demonstrated a significant increase in IL-10 expression within colonic tissues (P < .01). Similarly, compared with the model group, the low-dose ZJP group exhibited an increase in IL-10 expression (P < .05). However, no significant difference in IL-10 expression was observed between the positive drug and model groups (P > .05).
Following DSS induction, the expression level of TNF-α in the model group exhibited a notable increase (P < .001). Subsequent treatment with mesalazine and high-, medium-, and low-dose ZJP resulted in a significant reduction in TNF-α expression levels within the colonic tissues of all groups of mice (P < .001).
Determination of p-PI3K/PI3K, p-AKT/AKT, p-mTOR/mTOR, and Bcl-2/Bax Expression in Colon Tissues Using Western Blotting
To further elucidate the effective mechanism of ZJP in treating UC, we investigated the impact of varying doses of ZJP and mesalazine on the well-established PI3K/AKT/mTOR signaling pathway (Figure 8). This pathway holds significant relevance in the pathogenesis of UC. The ratios of P-PI3K/PI3K, p-AKT/AKT, and p-mTOR/mTOR in the DSS-induced model group demonstrated a substantial increase compared with that in the control group (P < .001). Conversely, the ratio of Bcl-2/Bax proteins, closely linked to apoptosis, experienced a notable decrease (P < .001). Postadministration of the drugs, the protein ratios within the colonic tissues underwent modifications as follows: The ratios of p-PI3K/PI3K, p-AKT/AKT, and p-mTOR/mTOR were downregulated across all groups, whereas the Bcl-2/Bax ratio was consistently upregulated to varying degrees. When comparing the outcomes of different drug groups, the positive control, mesalazine, and high-dose ZJP groups exhibited marked suppression of the P-PI3K/PI3K, p-AKT/AKT, and p-mTOR/mTOR ratios (P < .001). Concurrently, the Bcl-2/Bax ratio was significantly upregulated (P < .001). No significant difference in the regulation of the Bcl-2/Bax ratio was observed in the low-dose ZJP group compared with that in the DSS-induced model group. The difference between the 2 groups was not statistically significant (P > .05).

Comparative expression of related proteins in colonic tissues of ulcerative colitis (UC) mice. Compared with the control group, ****P < .0001 and ***P < .001; compared with the model group, ###P < .001, ##P < .01, and #P < .05.
Discussion
UC is a gastrointestinal disease characterized by diffuse edema of intestinal mucosa, superficial ulcers, and the presence of pus and bloody stool. As a representative prescription in traditional Chinese medicine, ZJP exhibits functions such as inhibiting gastric acid secretion and exerting anti-ulcer and antibacterial effects, effectively ameliorating severe gastrointestinal reactions in patients.8,14,15 Although ZJP holds therapeutic potential for UC, its mechanism remains incompletely elucidated. ZJP has been established to regulate intestinal flora and Treg cells, along with their subsets, and exert anti-oxidative stress effects in the treatment of UC.9,10,16 The primary constituents of ZJP include Coptis chinensis and Evodia officinalis. Pharmacological investigations have identified berberine, palmatine, evodiamine, and rutaecarpine as its principal active ingredients, among which evodiamine and rutaecarpine may play a major role in UC treatment.16,17 Berberine has been shown to downregulate Geboes scores in UC patients and ameliorate DSS-induced intestinal mucosal barrier dysfunction. This effect is achieved through microbiome-dependent mechanisms, the Wnt/β-catenin pathway, and the regulation of inflammatory infiltration involving macrophages, dendritic cells, and lymphocytes.18–22 Evodia exhibits antidiarrheal, antiemetic, and anti-ulcer effects, potentially mitigating DSS-induced colitis by modulating levels of Lactobacillus acidophilus and acetate and regulating the inflammatory environment of the intestine.23,24 Palmatine can attenuate colon damage by regulating tryptophan metabolism, intestinal microbes, and NLRP3 inflammasome.25–27 Rutin can induce NrF2-mediated antioxidant response, thereby alleviating DSS-induced colitis. 28 The infiltration of diffuse inflammatory cells in the intestinal laminae propria and elevated TNF-α and IL-6 levels, which promote macrophage and effector T cell infiltration, respectively, are primary inducers of UC. 29 IL-10, an essential immunosuppressant cytokine, particularly inhibits the production of monocytes, Th cells, and interferon (IFN)-γ, thus promoting intestinal epithelial barrier repair. ZJP may treat the disease by regulating the immune system. Some studies have suggested that ZJP could mitigate inflammation through mechanisms involving NLRP3 inflammasome and inflammatory factors. 30 In our study, treatment with mesalazine and ZJP significantly reduced the expression levels of TNF-α and IL-6 in the colon tissue of mice, and IL-10 levels were significantly recovered, with efficacy positively correlating with dose. Concurrently, pathological sections from each group demonstrated the definitive effect of ZJP on DSS-induced UC.
In our study, the potential active chemical components of ZJP in UC treatment were identified using network pharmacology. These identified compounds included berberine, phellobutanone, quercetin, phellobutanonic acid, rutin, epiberine, and evodipine. Furthermore, 103 potential targets of ZJP for UC treatment were obtained. Cancer pathways such as STAT3, AKT1, EGFR, PIK3R1, chemical carcinogen-reactive oxygen species, and other related signaling pathways exhibit greater than average significance, playing a key role in UC treatment and inhibiting UC progression to colorectal cancer. Additionally, GO enrichment analysis indicated that UC treatment with ZJP primarily affected processes involving drug reaction and protein autophosphorylation. It was closely associated with molecular functions such as protein kinase activity and binding. The intracellular protein kinase PI3K/AKT, as identified through KEGG analysis, may hold significant relevance in the treatment of UC with ZJP.
The PI3K/AKT/mTOR signaling pathway is involved in various biological processes such as cell growth, cell survival, apoptosis, and autophagy. It plays a crucial role in T cell metabolism, differentiation, and stability through lower-level effector factors.31,32 Among them, PI3 K activates PIP3 to provide an anchor point for phosphorylated AKT, subsequently activating downstream target mTOR to participate in the subordinate linkage reaction. This activation triggers glycolysis, cell growth promotion, cell proliferation, apoptosis, immune T cell synthesis, and tumor formation, thereby promoting the occurrence and development of UC. 33 Concurrently, inhibiting the high expression of the PI3K/AKT/mTOR signal can promote oxidative phosphorylation and inhibit the glycolytic process of proteins. This modulation serves to maintain immune defense ability, alleviate inflammatory damage to the intestinal mucosa, and promote the repair of intestinal UC to achieve the purpose of treatment. 34 Overactivation of the PI3K/AKT/mTOR signaling pathway leads to increased release of downstream genes regulating the proliferation of colon epithelial cells, such as cyclin and mTOR, and continuous renewal and repair of epithelial cells, thereby exacerbating UC. 35 UC has been shown to maintain intestinal barrier integrity through the PI3K/AKT/mTOR signaling pathway.36–38 However, research suggests that ZJP may alleviate UC by regulating the PI3K/AKT pathway and interfering with crosstalk between Treg cells and intestinal microbiota. 10
Bcl-2 and Bax, as anti-apoptotic and pro-apoptotic proteins, respectively, are frequently mentioned in the process of apoptosis and play a key role in its regulation.32,39 Following apoptosis stimulation, the Bax protein relocates to mitochondria, causing the release of cytochrome C and initiating the apoptotic cascade. 40 Bcl-2, by stabilizing mitochondrial membrane function, reduces the decreases in transmembrane potential and cascade activation of caspase, thereby inhibiting the process of apoptosis. 41 Hence, the Bcl-2/Bax ratio can serve as a molecular marker for monitoring apoptosis. As the ratio decreases and cell sensitivity to death signals increases, accelerated apoptosis of intestinal epithelial cells can disrupt the intestinal mucosal barrier. 42 In our study, the PI3K/AKT/mTOR signaling pathway detected by western blotting and the protein expression of downstream apoptotic protein Bcl-2/Bax were further verified to elucidate the mechanism of ZJP in UC treatment. The results showed that ZJP inhibited the expression of p-PI3 K, p-Akt, p-mTOR, and Bax proteins in the colon tissue of UC mice while increasing the expression of Bcl-2 protein. These findings verified the mechanism elucidated through network pharmacology regarding ZJP in UC treatment.
Based on multicenter and multitarget modern network pharmacology, 43 the relationship between 66 active drugs in traditional Chinese medicine and 189 key targets in ZJP along with 3175 UC-related targets was analyzed. Among these, 103 targets were implicated in the potential treatment of UC by ZJP through cancer, chemical carcinogenic-reactive oxygen species, and other pathways. It was closely associated with biological processes such as drug reaction and protein autophosphorylation as well as molecular functions such as protein kinase activity and binding. Results of KEGG analysis indicated that PI3K/AKT, as an intracellular protein kinase, may play a pivotal role in the treatment of UC with ZJP. To validate these findings, we intervened with ZJP in DSS-induced UC mice, confirming the ability of ZJP to ameliorate the pathological condition and inflammatory factor infiltration of UC mice through the PI3K/AKT/mTOR signaling pathway. Furthermore, ZJP was found to regulate downstream apoptosis and promote the recovery of anti-inflammatory factor levels, thereby establishing a theoretical basis for the clinical application of ZJP in UC treatment. In the present study, we integrated network pharmacology and animal experiments to elucidate the therapeutic mechanisms of ZJP in treating UC, specifically highlighting its interactions with the PI3 K/AKT/mTOR pathway and apoptosis. Nevertheless, our study exhibits certain limitations. It lacks a comprehensive analysis of the active pharmaceutical ingredients and their metabolic profiles. This gap underscores the need for additional experimentation to strengthen and refine the findings, suggesting a degree of limitation to the current results. Future investigations should entail a more thorough consideration of the concentrations of traditional Chinese medicine components and extend validation to a wider array of constituents and targets. This approach will enhance the reliability and practical applicability of the study.
Materials and Methods
Screening for Bioactive Ingredients and Targets of ZJP
We conducted a retrieval of the effective chemical constituents present in the composite ZJP formulation, specifically the ingredients “Coptis chinensis” (Huang Lian) and “Evodia rutaecarpa” (Wu Zhu Yu), using the TCMSP database (https://tcmspw.com/tcmspsearch.php). The screening process for pharmacokinetics involved applying defined thresholds: compounds with an oral bioavailability (OB) equal to or exceeding 20% and a drug-likeness (DL) value of 0.1 or higher were retained for further analysis. All active ingredients included in the PubChem and Swiss Target Prediction databases were searched, with the species filter set to “Homo sapiens.” Subsequently, the targets of each active ingredient were screened based on a probability threshold ≥ .15. This process yielded the predicted targets for 2 herbal ingredients, Huang Lian and Wu Zhu Yu, in the compound Zuo Jin Wan.
Screening of Drug Component Targets in ZJP Formulation
Each chemical constituent of ZJP was retrieved from PubChem (https://pubchem.ncbi.nlm.nih.gov) in a standardized linear format using their corresponding SMILES codes. These codes were subsequently subjected to drug molecule target prediction using the online database Swiss Target Prediction (http://www.swisstargetprediction.ch). The resultant output included the predicted target interactions for the 2 ingredients, Coptis chinensis, and Evodia rutaecarpa, present within the ZJP formulation.
Prediction of Molecular Targets for UC
Using the repositories of OMIM, TTD, DisGeNET, DrugBank, and GeneCards, a compilation of genes associated with UC was assembled. Subsequently, an intersection analysis was performed between the predicted targets of ZJP and the disease-related targets of UC. This process facilitated the identification of the pertinent targets through which ZJP exerts its therapeutic effect on UC.
Construction of Key Target-Molecule-Disease Target Network
The identified effective target genes linked to the therapeutic mechanisms of ZJP in UC were subjected to analysis within the STRING database. This analysis generated data representing the protein–protein interaction network, wherein higher scores indicated increased reliability of the data. Following this, outcomes with scores of 0.7 or higher were selected, preserved, and subsequently imported into the Cytoscape 3.7.1 software platform. Employing the NetworkAnalyzer module within this software, exhaustive assessments of the data on each node were undertaken, facilitating the extraction of valuable insights from the intricate network.
GO Enrichment and KEGG Pathway Analyses
The combined exploration of disease-associated drug molecules and shared prospective disease targets was executed using the bioinformatics resource DAVID 6.8 (https://david.ncifcrf.gov). Subsequent computations were conducted using R software version 4.0.2. Consequently, visual representations of the outcomes were illustrated through bar charts and bubble plots, portraying the outcomes of GO and KEGG enrichment analyses. Statistical significance was set at P < .05, indicating statistically significant differences.
Construction of a Visualized Network Linking Disease, Pathways, Targets, Effective Components, and Drugs
This methodology entailed systematically mapping the active constituents of ZJP, identifying potential targets from enriched pathways, and analyzing disease-correlated outcomes associated with these pathways. Subsequently, this amalgamation of elements was imported into the Cytoscape 3.7.1 platform to facilitate visual analysis. This stepwise process ultimately culminated in the creation of a comprehensive visual network, intricately linking disease states, pathways, targets, efficacious components, and therapeutic agents.
Validation of Animal Experimentation
Animals
Ninety male KM mice of SPF-grade quality, aged 6 weeks, with an average body weight of 22.43 ± 0.9 g, were procured from Jiangxi University of Traditional Chinese Medicine (Nanchang, Jiangxi) under the experimental animal production license number SCXK (Gan) 20180003. These mice were housed in the Barrier System Laboratory of the Experimental Animal Technology Center at Jiangxi University of Traditional Chinese Medicine. This facility meticulously maintained environmental conditions, ensuring a temperature range of 22 °C to 25 °C, relative humidity at 45 ± 5%, and a standardized light-dark cycle spanning 12 h (6:00 am-6:00 pm). The mice were granted ad libitum access to water and provided a standard pellet diet. After a 1-week acclimatization period, the experimental protocols commenced. Both the feed and water sources were consistently supplied by the Experimental Animal Technology Center at Jiangxi University of Traditional Chinese Medicine. The care of the mice adhered to the highest ethical standards and conformed to the guidelines stipulated by the Animal Care Committee of Jiangxi University of Traditional Chinese Medicine.
Preparation of Experimental Medication
ZJP (batch number: 20220614), sourced from Hubei Xiang Lian Pharmaceutical, underwent pulverization and subsequent sieving. The resultant powder was dissolved in an optimal volume of distilled water and then subjected to robust ultrasonic agitation for 5 min. This process yielded ZJP solutions of distinct concentrations, namely 75, 150, and 300 mg/mL, denoting low, medium, and high concentrations, respectively. The stipulated ZJP administration dose was established at 300 mg/kg. To facilitate the administration of mesalazine enteric-coated tablets (batch number: 221020) from Kuihua Pharmaceutical, the tablets were finely ground, sieved, and subjected to ultrasonication. This resulted in the formation of a mesalazine stock solution, characterized by a concentration of 15 mg/mL, which was subsequently administered at a dose of 300 mg/kg. The benchmark positive control group, DSS (36-50 kDa) sourced from MP Biomedicals (batch number: 160110), was judiciously employed. To prepare the 2.5% DSS solution, an appropriate quantity of DSS was dissolved in distilled water.
Grouping, Modeling, and Administration Procedures
The mice were randomly allocated into distinct groups using the SPSS random number method. These groups included the normal, model, and mesalazine groups (positive control group administered at 0.2 g/kg) and the low-, medium-, and high-dose ZJP groups. The dosages for the ZJP groups were determined from a table converting animal–human weight dosage factors, resulting in 1.5, 3, and 6 g/kg, respectively. Each group consisted of 15 mice, except for the control group. The remaining 5 groups, excluding the control group, were granted unrestricted access to a freshly prepared 2.5% DSS solution daily for 7 consecutive days to induce UC development in the mice. After the modeling process, an 8-hour interval was observed before the commencement of intervention administration. The control and model groups received intragastric administration of 0.2 mL/10 g of physiological saline. The mesalazine group was administered an equivalent dosage of the mesalazine stock solution. The low-, medium-, and high-dose ZJP groups were administered corresponding ZJP stock solutions at a volume of 0.2 mL/10 g of body weight. Administration solvents were prepared immediately prior to dosing each day. All experimental groups received daily administrations once a day consistently over 7 consecutive days.
Assessment of DAI Scoring
Throughout the modeling and intervention period, diligent daily monitoring was undertaken to record changes in mouse body weight, stool characteristics, and occurrences of rectal bleeding. Disease activity was evaluated using the DAI, calculated by averaging the scores obtained from assessments of weight loss, stool consistency, and rectal bleeding. 44 The comprehensive scoring standards are listed in Table 1.
Disease Activity Index (DAI) Scoring Items and Standards.

Paraffin Sections of Colonic Tissue and HE Staining
Following the final intervention, a 24-hour fasting period was initiated, during which water access remained unrestricted. After cervical dislocation, the colon was carefully excised from the cecum to the rectum. Adherent adipose tissue was removed from the dissected tissue and weighed, and its length was measured. A 1 cm segment from the proximal cecum was immersed in a 4% paraformaldehyde solution for 24 h to ensure fixation. Subsequently, the treated tissue was immersed overnight in running water before embedding. Upon successful sectioning, the samples underwent HE staining, followed by mounting with neutral gum. Finally, the samples were examined under an inverted microscope. 45
ELISA
Colonic tissue samples were procured from the −80 °C freezer. Following homogenization using a homogenizer for 5 min, the resultant samples underwent centrifugation, yielding a measurable supernatant. After reaching room temperature, the Elisa kit was used for sample loading, followed by sequential washing steps. Upon addition of the stop solution, the absorbance of each well was measured. 46
Western Blotting
Protein samples were collected from each experimental group and combined with SDS-PAGE protein loading buffer in a 4:1 ratio. An appropriate volume of this mixture was then loaded onto an SDS-PAGE gel for electrophoresis. Subsequently, the proteins were effectively transferred onto a polyvinylidene difluoride (PVDF) membrane, which was then blocked with a 5% skim milk solution for 1 h. Primary antibodies against p-PI3K (1:1500), PI3K (1:1000), p-AKT (1:3500), AKT (1:1000), p-mTOR (1:6000), mTOR (1:30 000), Bcl-2 (1:2500), and Bax (1:9000) were applied onto the membrane and allowed to incubate. Following this incubation period, the membrane underwent another round of incubation with secondary antibodies for 1.5 h. After washing the membrane with PBS, an enhanced chemiluminescence reagent was used to facilitate the visualization of the protein bands. 47
Statistical Analysis
Data analysis was conducted utilizing GraphPad Prism 8.3.0 software. Quantitative data are expressed as mean and were subjected to a one-way analysis of variance to assess potential intergroup variances. Subsequently, post hoc comparisons were conducted using the least significant difference approach for pairwise assessments among the groups. Results at P < .05 were considered statistically significant.
Footnotes
Acknowledgments
We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Committee of Animal Ethics and Welfare of Jiangxi University of Chinese Medicine (JZLLSC20210024).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangxi Regional Science Foundation of the National Natural Science Foundation of China [82260938] and the National Natural Science Foundation of China [82374454]. The funders were mainly responsible for the conceptual design, data analysis and interpretation, and manuscript revision of this study.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Ethics Committee of the Animal Center of the Jiangxi University of Chinese Medicine (JZLLSC20210024)* approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
