Abstract
Introduction
An abdominal aortic aneurysm (AAA) is characterized by the weakening and dilatation of the abdominal aorta, most commonly affecting the infrarenal part. 1 AAA is a common critical cardiovascular disease with high morbidity and mortality rates. It is the tenth leading cause of death in men over 65 years of age. The incidence of aortic aneurysm can reach 8% in men over 65 years of age. 2 The major complication of AAA is aortic rupture, which results in an overall mortality rate of approximately 90%. 1 The primary intervention for AAA is surgical repair; however, early intervention does not improve the survival rate of patients. Surgical treatment can be performed when a male aortic aneurysm is larger than 5.5 cm in diameter or female aortic aneurysm is larger than 5 cm in diameter. However, effective intervention methods for the treatment of small aortic aneurysms are lacking and if not treated promptly, they can continue to grow until rupture. 3 In addition to surgical treatment, effective pharmacological interventions, especially for the early treatment of AAA are lacking. 4 Therefore, it is crucial to explore potentially effective drugs for AAA treatment.
The receptor for advanced glycation end products (RAGE) is expressed in various cancer cells including pancreatic, 5 colon, breast, prostate, liver, and lung cancers. 6 Clinical studies have shown that soluble in human blood can reflect RAGE activity and is correlated with the progression of chronic diseases, providing a useful biological marker for RAGE-mediated pathogenesis. 7 RAGE activation increases inflammation and promotes the development of multiple cancers. 8 Related studies have shown that knocking out the myeloid RAGE prevents AAA formation in mouse models. 9 This indicates that RAGE plays an important role in the occurrence and development of AAA. We need to further explore the molecular mechanism by which RAGE acts after ligand binding and activation in AAA tumor wall tissue. It was also important to clarify whether blocking RAGE signaling can inhibit the occurrence and development of AAAs. This will provide new ideas for the non-surgical treatment of clinical AAAs.
Traditional Chinese medicine (TCM) is an important medical resource. Several recent in vivo and in vitro experimental studies have shown that many herbs and phytochemicals have potential benefits in preventing or reducing AAA progression with fewer side effects. 10 Quercetin is a flavonol, found in many TCMs, and has the molecular formula of Oc1cc(O)c2c(c1)oc(c(c2 = O)O)c1ccc(c(c1)O)O. It exhibits a wide range of biological activities and has various pharmacological effects such as antioxidant, 11 tumor inhibition, anti-inflammatory, 12 and lipid-lowering properties. 13 Studies have shown that quercetin exerts anti-cancer effects through multiple pathways, including antioxidant, anti-proliferation, cell cycle arrest, induction of apoptosis, and inhibition of tumor metastasis. 14 Previous studies have shown that quercetin treatment significantly inhibited RAGE protein expression in pancreatic cancer cells, 15 providing useful references for future research.
In summary, quercetin may alleviate the occurrence and development of AAA by inhibiting RAGE, but its target and mechanism for treating AAA are not completely clear. Therefore, quercetin can be used as a small-molecule drug to prevent aortic aneurysm formation and delay aortic aneurysm expansion. However, its specific mechanism of action is unknown. This study established a PPE perfusion animal model to simulate the pathogenesis of human AAA, used quercetin for treatment, elucidated the specific mechanism of action of quercetin, identified relevant drug targets, and provided a clinical basis for the treatment of AAA with quercetin.
Material and Methods
Animals and Ethics
Adult male C57/BL6 rats were obtained from the Second Affiliated Hospital of the Harbin Medical University (Harbin, China). These rats weighed approximately 200 ± 20 g and were acclimatized for 7 days under alternating light and dark conditions (12 h each), at a stable temperature of 20 ± 2°C and humidity of 50 ± 5%. Throughout the acclimatization period, the rats were randomly assigned to cages in groups of three to ensure continuous access to food and water. All animal care and experimental procedures were carried out in compliance with the regulations of the Animal Experimental Committee at the Second Affiliated Hospital of Qiqihar Medical University (QMU-ECC-2023-1115-2, China) and adhered to the guidelines provided by the National Institutes of Health for the proper care and utilization of laboratory animals.
Animals and Treatment
The C57/BL6 rats were randomly divided into three groups (n = 6 per group): the sham operation group (Sham group), abdominal aortic aneurysm model group (AAA group), and the quercetin treatment group (Que group). The abdominal aortic aneurysm model was established by elastase perfusion (XT119517, Cool Chemical science and technology, Beijing, China) and saline perfusion was given in the sham-operated group. The AAA model was established as follows: rats were anesthetized with isoflurane, and an abdominal incision was made to expose the abdominal aorta and inferior vena cava. The abdominal aorta was isolated from the renal artery branch to the common iliac artery and each branch was ligated. The proximal end of the left common iliac artery was suspended using a live button. A PE10 catheter was inserted from the live port into the left common iliac artery, guided to the transient closure of the abdominal aorta configured 8 U/mL elastase solution was injected and the filling pressure was maintained for 30 min. The rats were weighed and administered 0.1 mL (60 mg/kg) of quercetin daily via a gavage needle from 1 week preoperatively to 4 weeks postoperatively. 16 The 60 mg/kg dose administered to the rats was equivalent to a dose of 5 mg/kg for humans, which corresponds to a dose of 300 mg for an individual weighing 60 kg.
Measurement of Blood Pressure in Rats
At the end of the modeling, the determination of blood pressure in mice was performed. The sensor (MD3000) was applied to the tail of the mouse, and the blood flow signal was monitored while pressurizing and releasing pressure on the tail artery by inflation and deflation to derive the blood pressure value.
Tissue Sample Collection
After conventionally anesthesia with 1.5% isoflurane (Yipin Pharmaceutical Co., Ltd, Hebei, China), the chest and abdominal cavities of the rats were quickly opened to expose the heart. The left ventricle was punctured with a 10 mL syringe, the aorta was flushed with pre-cooled PBS buffer, and the blood was washed away from the blood vessel. The abdominal aorta below the renal artery was then dissected under a microscope. Portions of the aneurysm tumor tissue were embedded in OCT-frozen section-embedding medium, ideally on dry ice, with the tissue placed in the center of the medium. Another portion of the aneurysm tissue was stored in a −80 °C ultra-low temperature refrigerator for future testing. Venous blood samples were collected and allowed to stand at room temperature for 2 h before being centrifuged at 3000 rpm for 10 min at 4 °C. After centrifugation, the supernatant was carefully aspirated and stored at −80 °C for later analysis. All mice initially received inhalation anesthesia.
Serum Biochemical Assay
After blood collection, the supernatant was collected by centrifugation at 3000 rpm for 10 min at 4°C. Serum levels of creatinine (CREA), blood urea nitrogen (BUN), aspartate aminotransferase(AST), and alanine aminotransferase (ALT) were measured using a biochemical analyzer (UniCel DxC800 Synchron, Beckman, USA).
Histopathological Analysis
The infrarenal abdominal aorta segments (n = 6 per group) were fixed in 10% formalin solution and embedded in paraffin. The mouse abdominal aorta tissue was fixed with 4% paraformaldehyde, then dehydrated using graded alcohol, and further embedded in paraffin. Tissue sections (4∼5 μm thick) were prepared and stained according to the instructions of the eosin staining solution. Histopathological observations were performed by light microscopy (400× magnification, BX-FM; Olympus Corp., Tokyo, Japan) and evaluated by individuals who were blinded to the study group assignments.
Elastic Van Gieson
Elastic fiber staining Elastic fiber destruction can be observed. This was assessed by paraffin sectioning and dewaxing mouse basilar artery tubes, followed by rehydration. The Elastic Van Gieson (EVG) staining solution was prepared using ethanol, hematoxylin, ferric chloride, and iodine in a 5:2:2 ratio. The sections were stained with EVG staining solution for 30 min, then washed with pure water. Subsequently, saturated picric acid and acidic magenta were mixed in a 9:1 ratio with EVG staining solution for 1–3 min. These sections were then rinsed with pure water, dehydrated with anhydrous ethanol, made transparent with xylene, and sealed by neutral gum in turn. Images were acquired and analyzed using a microscope (Canon EOS 550D camera, Tokyo, Japan). Elastin degration: Tidak ada atau rusak ringan, 1; Fraktur sedang, 2; Fraktur berat, 3; Total kerusakan serat elastis, 4.
Immunohistochemistry Staining
Following deparaffinization, hydration, and antigen retrieval, samples of tissue from the abdominal aortic vessel were incubated at 4°C overnight with a primary antibody targeting alpha- smooth muscle actin (α-SMA) (1:100, Abcam). The membranes were then incubated with a biotinylated secondary antibody (1:1000, ZSGB-Bio). Visualization was carried out using DAB horseradish peroxidase color development kits (Beyotime), and the sections were counterstained with hematoxylin. Images were captured using a Leica DM4B microscope.
ELISA Analysis
The Malondialdehyde assay kit (MDA, A003-1-2, Nanjing Jiancheng Bioengineering Institute, Nanjing, China), Reduced Glutathione assay kit (GSH, A006-2-1, Nanjing Jiancheng Bioengineering Institute, Nanjing, China), Catalase assay kit (CAT, A007-2-1, Nanjing Jiancheng Bioengineering Institute, Nanjing, China), and Superoxide Dismutase assay kit (SOD, A001-3-2, Nanjing Jiancheng Bioengineering Institute, Nanjing, China) were used to measure the collected tissue from the abdominal aorta. Tissue samples were processed according to the instructions, and assayed using Model F4500 fuorescence spectrometer (Hitachi, Tokyo, Japan).
Molecular Docking
The structure of the protein of interest was obtained by searching the PDB database (https://www.rcsb.org). Irrelevant ligands and water molecules were removed from the protein structure using PyMOL. Subsequently, the protein was prepared in AutoDock Tools 1.5.6, including hydrogenation, charge calculation, and non-polar hydrogen merging. The prepared protein was saved in PDBQT format for docking. AutoDock Vina was executed via CMD prompt to conduct docking simulations between quercetin and the complete structure of the target proteins. The docking conformation was visualized using PyMOL.
Western Blot
Frozen abdominal aortic tissue was subjected to protein extraction. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels with a 10% concentration were prepared according to the manufacturer's instructions. Equal amounts of protein samples (28 μg) were loaded onto the SDS-PAGE gels and transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were then blocked for 2 h. with 5% skim milk. The primary antibodies and their dilutions were as follows: RAGE (1:1000, Santa Cruz Biotechnology, sc-80652), phospho-mTOR (1:100, Santa Cruz Biotechnology, sc-293132), mTOR (1:500, Santa Cruz Biotechnology, c-517464), Akt (1:1000, Cell Signaling Technology, 9272), phospho-Akt (1:500, Cell Signaling Technology, 4060), PI3K (1:500, Cell Signaling Technology, 4257f), phospho-PI3K (1:100, Cell Signaling Technology, 278545), and β-actin (1:1000, Cell Signaling Technology, 4967). After washing thrice with Tris-Buffered Saline and Tween 20 (TBST), the membranes were incubated with the appropriate secondary antibodies. Protein detection on each membrane was carried out using an enhanced chemiluminescence (ECL) method (Meilunbio, Dalian, China). Protein levels on the membranes were normalized to GAPDH. A TImager 600 fully automated chemiluminescence analysis system was used for detecting target protein chemiluminescence signals, and results were acquired and saved. ImageJ software was used to analyze the grayscale values of the protein bands.
Quantitative Real-Time Reverse Transcription Polymerase Chain Reaction
The mRNA expression levels of RAGE, mTOR, Akt, and PI3K were detected using Real-time reverse transcription polymerase chain reaction (RT-PCR). Total RNA was isolated with TRI-zol reagent (Invitrogen), followed by reverse transcription of 1 μg total RNA into cDNA using the AMV First-Strand cDNA Synthesis Kit (Invitrogen, USA). RT-PCR was employed for the quantification of gene transcripts, utilizing the ABI SYBR Green PCR Master Mix (Thermo Fisher) and the ABI StepOnePlus system, according to the manufacturer's instructions. The resulting data were analyzed using StepOnePlus software. To validate the fluorescent RT-PCR reaction, the constitutively expressed gene β-actin was used as an internal control. The primers were custom-synthesized by Shanghai Sangong Bioengineering Technology Service Co., and their sequences are listed in Table 1.
The Primer Sequences for the RT-PCR.

Statistical Analysis
Mean values ± SEM were used to represent the data. SPSS Version 25.0 (Chicago, IL, USA) was utilized for the tatistical analysis. Graphs were generated using GraphPad Prism version 10.0 (San Diego, CA, USA). The data were subjected to one-way ANOVA followed by Tukey's post-hoc test. Statistical significance was defined as P < .05 and high significance was defined as P < .01.
Results
Abdominal Aorta Size and Changes in Body Condition in Rats
All rats survived for 6 weeks after surgery and were euthanized after anesthesia. Visual tissue changes are shown in Figure 1A. In the AAA model group, the diameter of the perfused abdominal aorta was significantly expanded, with an uneven surface, rough and stiff walls, and the outer membrane adhering to surrounding tissues. The aorta was visible during dissection and adhesion to surrounding tissues made the separation more difficult However, after quercetin treatment, the external mold adhesion of the tube wall was reduced, and the wall returned to a smoother appearance. Quercetin treatment significantly reduced the incidence of AAA. During the operation, the abdominal aortas from all three groups of mice were photographed, and their dimensions were measured. Statistically significant differences were observed in the aorta diameters in each group. Compared to mice in the sham group, the outer diameter of the abdominal aorta in the AAA group was significantly increased (P < .01). In contrast, the outer diameter of the aorta in quercetin-treated mice was significantly reduced compared to the AAA group (P < .01) (Figure 1B).

A. In vivo photographs of the abdominal aorta in rats. B. Dimensions of the external diameter of the abdominal aorta in rats. C. Measurement of noninvasive blood pressure in rats. D-G: Serum for biochemical analysis of rats. Data are presented as means ± SEM (n = 6). * P < .05, **P < .01 versus the Sham group; # P < .05, ## P < .01 versus the AAA group.
The blood pressure of each mouse was measured before surgery. Rats in the AAA group exhibited significant increase in the blood pressure compared to those in the Sham group (P < .05). Following quercetin treatment, blood pressure in the mice significantly decreased (P < .05) (Figure 1C). Biochemical tests were conducted on the serum of mice in each group, and the results of various indicators are shown in Figure 1D-G. Renal function indicators, including creatinine (CREA) and blood urea nitrogen (BUN), were significantly elevated in the AAA group compared to the sham group (P < .01). Liver function indicators included AST and ALT levels. The AST and ALT levels were significantly higher in the AAA group compared to the sham group (P < .01). After quercetin treatment, levels of CREA, BUN, AST, and ALT significantly decreased (P < .01).
Structural Changes in the Abdominal Aortic Wall
Hematoxylin and Eosin (H&E) staining showed no obvious pathological changes in the sham group. In the AAA model group, the inner, middle, and outer membranes of the tumor wall were severely damaged. The three-layer structure was disorganized and unclear, with evident intimal hyperplasia and disorderly arranged smooth muscle cells in the media. Significant fibroblast proliferation was observed and a large number of infiltrating inflammatory cells, particularly in the middle layer. In contrast, the quercetin-treated group showed only slight inflammatory cell infiltration, a clear membrane structure, and no significant damage (Figure 2A).

Structural changes in the abdominal aortic wall. A. Pathological tissue section observation. B. Elastic Van Gieson results for the abdominal aorta. C.α-SMA immunohistochemical staining. D. Elastin degradation score. E. Semi-quantitative immunohistochemical analysis of α-SMA. Data are presented as means ± SEM (n = 6). * P < .05, **P < .01 versus the Sham group; # P < .05, ## P < .01 versus the AAA group.
EVG staining results: The sham group showed no elastin degradation. In the AAA group, the black elastic fibers in the abdominal aortic wall of mice were severely broken, degraded, and damaged, with a significant reduction in their number (P < .01). They also lost their normal wavy appearance and lamellar arrangement, exhibiting flak-like fractures. The quercetin treatment group showed only mild elastin degradation and no significant change in the wavy appearance (P < .01) (Figure 2B and D).
Representative images and semi-quantitative analysis of α-SMA immunohistochemical staining showed that α-SMA levels were significantly higher in the Sham group compared to the AAA group (P < .01). In the AAA model group, considerable smooth muscle cell apoptosis was observed in the abdominal aortic wall tissue. Compared with the degree of death, the positive area of smooth muscle cells in the aneurysm wall tissue almost disappeared. After quercetin treatment, the SMA of the abdominal aortic wall was significantly restored ((P < .01) (Figure 2C and E).
Quercetin Alleviates AAA-Induced Oxidative Stress Injury in Abdominal Aorta
CAT, MDA, GSH, and SOD were identified as the oxidative stress biomarkers (Figure 3A). Compared to the SHAM group, the levels of MDA in the AAA group increased significantly (P < .01) (Figure 3A). After Quercetin administration, the levels of MDA in the quercetin treatment group decreased significantly (P < .01) compared with those in the AAA group. The levels of CAT, GSH, and SOD in the AAA group were significantly lower (P < .01) than those in the SHAM group (Figure 3A). CAT, GSH, and SOD levels in the quercetin group were significantly lower than those in the AAA group (P < .01). Thus, quercetin treatment significantly alleviated AAA-induced oxidative stress caused by AAA.

Oxidative stress detection and molecular docking. A. Oxidative Stress Indicator Results. B. Molecular docking. Data are presented as means ± SEM (n = 6). * P < .05, **P < .01 versus the Sham group; # P < .05, ## P < .01 versus the AAA group.
Molecular Docking
Molecular docking was used to simulate the binding of the top ten hub targets to quercetin. Quercetin occupies the amino acid residue LYS-170 in the structural domain of the RAGE protein, with a minimum molecular binding energy of 7.6 kcal/mol. Quercetin could then bind to the docking pocket with effective docking activity between RAGE proteins (Figure 3B).
Abdominal Aorta-Related Protein Expression Results
The RAGE/PI3K/AKT/mTOR pathway was examined by western blotting to investigate the mechanism by which quercetin protects against abdominal aortic injury. The ratios of p-PI3K/PI3K, p-AKT/AKT, and p-mTOR/mTOR represented the phosphorylation levels of the three proteins, respectively. Western blot analysis showed that RAGE protein expression was significantly higher in the AAA group than in the CON group (P < .01). After treatment with quercetin, RAGE protein expression significantly reduced (P < .01) (Figure 4A and C). Compared with the CON group, the phosphorylation levels of PI3K, AKT, and mTOR in the AAA group were significantly increased, (P < .01) (Figure 4B and C). After quercetin treatment, the phosphorylation levels of these three proteins were significantly reduced (P < .01). Thus, it is evident that quercetin inhibited the occurrence and development of AAA through the RAGE/PI3K/AKT/mTOR pathway.

The phosphorylated protein levels of PI3K/AKT/mTOR as this figure presents the results of western blot. A. RAGE, p -PI3K and p-AKT protein expression result. B. Results of p-mTOR protein expression. C. Quantitative analysis of RAGE/PI3K/AKT/mTOR protein in the abdominal aorta. Data are presented as means ± SEM (n = 6). * P < .05, **P < .01 versus the Sham group; # P < .05, ## P < .01 versus the AAA group.
The mRNA Expression Assay for Pathway-Related Factors
The mRNA expression levels of abdominal aorta-related genes were examined in each group of rats. The gene expression levels of RAGE, mTOR, Akt, and PI3K in the AAA group were significantly higher than in the sham group (P < .01). After quercetin treatment, RAGE, mTOR, Akt, and PI3K gene expression levels were significantly reduced (P < .01) (Figure 5A, B, C and D).
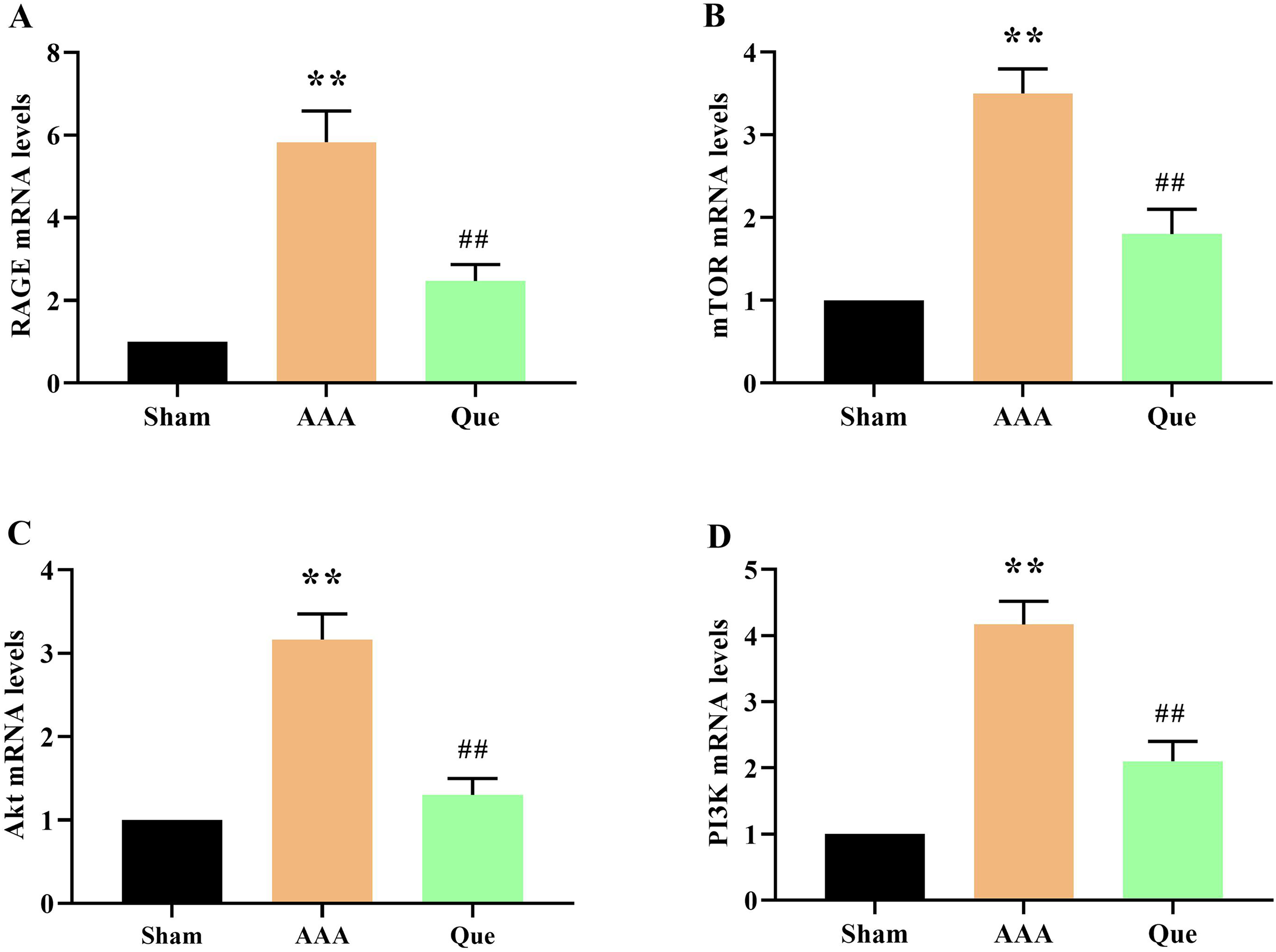
The mRNA expression assay for pathway-related factors. Data are presented as means ± SEM (n = 6). * P < .05, **P < .01 versus the Sham group; # P < .05, ## P < .01 versus the AAA group.
Discussion
AAA typically shows no symptoms unless it ruptures, which can be deadly if surgical repair is unsuccessful. In 2017, AAA was associated with 167,200 deaths and 3 million years of disability-adjusted life globally. 17 Although minimally invasive surgical devices, such as endovascular aneurysm repair (EVAR) are available, the durability of these treatments remains a problem and rupture can still occur. 18 Today, many of the therapeutic benefits of herbal medicines have been highlighted. Currently, spotlight on the numerous healing advantages of herbal medicines. Multiple pieces of evidence indicate that quercetin, the most prevalent flavonoid, exhibits anti-angiogenic properties in various human cancer cell lines. 19 Notably, quercetin is a primary flavonoid present in fruits and vegetables. Its daily consumption through diet has significant ethnopharmacological value, particularly in managing cardiovascular disease.16,20 Research suggests that quercetin's ability to protect blood vessels is linked to its anti-inflammatory properties and potential impact on oxidative stress. Previous studies have suggested that quercetin can help prevent aneurysmal degeneration by leveraging these characteristics.11,21
Currently, most researchers believe that the formation, expansion, and rupture of AAA are related to factors such as extracellular matrix metabolic imbalance, tissue hypoxia, atherosclerosis, inflammation and immune response, apoptosis, blood pressure regulation, and hemodynamic abnormalities. 22 The PI3K/AKT/m TOR signaling pathway is cross-linked and dialogues with the pathways involved in the above pathophysiological processes, potentially contributing to the occurrence and development of AAA. According to the results of this study, quercetin treatment of AAA rats significantly alleviated the deformation and diameter expansion of the abdominal aorta and prevented tissue adhesion. During the experiment, it was found that AAA can increase blood pressure in rats, consistent with previous research findings. 16 Biochemical tests indicated that AAA mice had impaired liver and kidney function. However, after quercetin treatment, the CREA, BUN, AST, and ALT levels decreased significantly (P < .01).
The thickening of the arterial wall in patients with AAA is often associated with rupture of elastic fibers of the arterial wall, degradation of collagen fibers, and increased fibrosis. 23 In this study, H&E staining revealed that the abdominal aorta wall of mice in the AAA group was significantly thickened, with an increase in infiltrative cells in both the media and adventitia of the artery. Quercetin treatment alleviated this phenomenon, indicating the anti-abdominal aortic effect of quercetin. These effects can be achieved by reducing the abdominal aortic wall thickening and cellular infiltration of both the media, and adventitia of the artery. EVG staining showed significant reduction in elastic fibers in the abdominal aortic wall of mice in the AAA group. While quercetin treatment reduced collagen loss and preserved the aortic elastin structure. α-SMA is an important signature protein for the transformation of VSMC into myofibroblasts. Decreased α-SMA expression leads to vascular remodeling. Therefore, this study conducted immunohistochemical staining of α-SMA in the blood vessel wall. It was found that the positive staining of α-SMA in the media of sham-operated mice was mainly positive, while α-SMA expression was significantly reduced in AAA mice (P < .01). However, quercetin treatment significantly increased α-SMA expression (P < .01). Oxidative stress levels are significantly elevated in AAA patients and experimental animal models. Along with increased ROS levels, inflammatory cell infiltration, and chemokine release, these pathological changes promote AAA occurrence and progression, whereas antioxidant stress treatments such as vitamin E and N-acetyl-L-cysteine (NAC) can reduce the incidence of AAA and inhibit its formation. In the present study, quercetin treatment significantly inhibited oxidative stress. MDA levels decreased significantly (P < .01), while levels of CAT, GSH, and SOD increased significantly following quercetin treatment (P < .01).
Several researchers have confirmed that RAGE is involved in the pathological processes of many clinical diseases and have conducted in-depth studies on the mechanisms related to its pathological effects, such as inflammatory bowel disease (IBD),24,25 cardiac infarction, 26 diabetic nephropathy, 27 atherosclerosis, 28 and cancer. 29 RAGE plays an important role in inflammatory vascular diseases. RAGE is a multi-ligand receptor located on the cell surface and is widely expressed on the surface of inflammatory cells. 30 Recent studies have shown that knocking out the RAGE gene in mice significantly slows the development of AAA. Compared with the AAA model group of mice, the maximum diameter of AAA was smaller and the growth rate was reduced. The aneurysm rate decreases and the aneurysm growth curve slows. 31 This is consistent with the hypothesis of the present study that RAGE plays an important role in the occurrence and development of AAA. After quercetin treatment, the RAGE protein expression in the abdominal aorta was significantly decreased (P < .01). RAGE activation occurs upon ligand binding, which then transmits extracellular signals to activate downstream signaling pathways. The KK-Ay mouse diabetes model has confirmed that RAGE activation can activate the PI3K/Akt/mTOR pathway, causing glucose and lipid metabolism disorders and renal function damage. 32 This is consistent with the results of the present study.
The PI3K/AKT/m TOR signaling pathway is inactive in normal tissue cells. Once cells are stimulated by internal and external stimuli, this pathway is activated and participates in the regulation of various cell functions by influencing many downstream factors. 33 The mTOR plays an important role in regulating protein cell survival, differentiation, and proliferation. 34 Li et al demonstrated that p-mTOR expression was significantly increased in an elastase infusion-induced AAA mouse model. This finding emphasizes that activation of p-mTOR signaling may be involved in the pathological process of AAA. 35
We introduced and confirmed the effect of quercetin in treating AAA, and research showed that this effect may be mediated by the RAGE/PI3K/AKT/mTOR pathway. However, this study has some limitations. It did not delve deeply into the relevant therapeutic mechanism but this will be addressed in the future research. Our research group plans to conduct in-depth studies on the antitumor effects of quercetin and its clinical applications. The expression of PI3K in AAA was significantly higher than that in normal individuals. It is not significantly related to the patient's sex, age, tumor diameter, smoking history, hypertension history, and blood lipid levels, but is closely related to AAA occurrence. 36 Recent studies have shown abnormal activation of the PI3K/AKT/mTOR pathway in AAA, 37 and inhibiting this pathway can suppress the occurrence and development of AAA. 38 This study showed that the PI3K/AKT/mTOR signaling pathway was activated in the aneurysm tissues of AAA rats. The protein and mRNA expression levels of PI3K/AKT/mTOR in the AAA group were higher than those in the control group (P < .01). After quercetin treatment, the expression of PI3K/AKT/mTOR pathway proteins was significantly reduced (P < .01), indicating that quercetin inhibits AAA occurrence and development by regulating this signaling pathway.
In this study, quercetin was shown to alleviate AAA through the PI3K/AKT/mTOR pathway; however, some limitations should be noted. The effect of quercetin on AAA was preliminarily confirmed by establishing a mouse model of AAA. However, this study did not explore the mechanism of action in depth. Future studies will focus on more detailed investigation of the mechanism and targets of quercentine's action.
Supplemental Material
sj-jpg-2-npx-10.1177_1934578X241286438 - Supplemental material for Quercetin Promotes Abdominal Aortic Aneurysm Regression Through the RAGE/PI3K/AKT/mTOR Axis
Supplemental material, sj-jpg-2-npx-10.1177_1934578X241286438 for Quercetin Promotes Abdominal Aortic Aneurysm Regression Through the RAGE/PI3K/AKT/mTOR Axis by Xin Yang, Ying Wang, Guangyu Xin, Jie Li, Lei Xu, Guohua Zhang and Haotian Yang in Natural Product Communications
Footnotes
Acknowledgments
We thank the clinicians in this group for their perseverance and dedication, and the editors for their comments on this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The Second Affiliated Hospital of Qiqihar Medical University approved the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by Qiqihar Science and Technology Program Joint Guidance Project (LSFGG-2023067).
Statement of Animal Rights
All procedures in this study were conducted in accordance with The Second Affiliated Hospital of Qiqihar Medical University Ethics Committee (QMU-ECC-2023-1115-2, China) approved protocols.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
