Abstract
The inhibition of intimal hyperplasia (IH) is an effective strategy to improve the long-term outcome of endovascular therapy and prevent restenosis. Farrerol, a naturally occurring dihydroflavone with a variety of bioactivities, exerts inhibitory effects against balloon injury-induced IH in rats. In the present study, bioinformatics analysis, in combination with in vitro experimental validation, was performed to elucidate the underlying inhibitory mechanisms. The protein–protein interaction (PPI) network was assessed to identify farrerol-related protein targets in the context of IH, based on which biological functions and pathway enrichment were analyzed. The proliferation and cell cycle distribution of vascular smooth muscle cells (VSMCs) were investigated using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide and 5-ethynyl-2-deoxyuridine incorporation assays and flow cytometric analysis, respectively. The level of pro-inflammatory cytokines in the cell culture medium was estimated using an enzyme-linked immunosorbent assay (ELISA). Protein expression in A7r5 cells was determined by western blotting. Forty-six IH-related targets of farrerol were identified, and the PI3K/Akt/mTOR pathway was highly enriched among the 43 predicted pathways (P < .05). In serum (10% fetal bovine serum)-induced A7r5 cells, farrerol inhibited proliferation through non-cytotoxic effects, induced cell cycle arrest in the G0/G1 phase, and suppressed the activation of the PI3K/Akt/mTOR pathway. In H2O2 (300 µM)-induced A7r5 cells, farrerol reduced the release of IL-1β and TNF-α and reversed the suppressive effect on the PI3K/Akt/mTOR pathway in response to H2O2 stimulation. In conclusion, farrerol inhibits the proliferation of VSMCs and protects VSMCs from oxidative injury via the bidirectional modulation of the PI3K/Akt/mTOR signaling pathway, which might contribute to the suppression of neointima formation.
Keywords
Introduction
Vascular interventions, such as angioplasty, endarterectomy, or surgical bypass grafting, are performed to treat various vascular diseases, including atherosclerosis, a major global cause of human morbidity and mortality.1-3 However, severe intimal hyperplasia (IH) and consequent vascular restenosis are major drawbacks of such vascular interventions.4,5 Thus, therapeutic strategies against IH could be beneficial for the treatment of vascular diseases, and it is important to discover agents to efficiently treat IH and prevent the further progression of restenosis. 6 Multiple events contribute to the pathogenesis of IH, including the excessive proliferation of vascular smooth muscle cells (VSMCs), oxidative stress, and inflammation. 7 Indeed, accumulating evidence shows that compounds with anti-inflammatory, antioxidant, and inhibitory effects against VSMC proliferation have beneficial effects in the treatment of IH.8-10
Farrerol, a dihydroflavone, is the main pharmacologically active ingredient of Rhododendri Daurici Folium. It exhibits extensive biological properties, exerting anti-inflammatory, 11 antibacterial, 12 antitumor, 13 and preventive effects against cardiovascular disease. 14 Our previous study has demonstrated that farrerol has a protective effect on aortic injury in rats with spontaneous hypertension, and can effectively inhibit balloon injury-induced IH of the common carotid artery. This compound also shows good potential for the prevention and treatment of vascular diseases, which partly results from the regulation of the phenotypic transformation of VSMCs by modulating the ERK1/2 signaling pathway 15 ; however, its mechanism of action has not been fully elucidated.
Bioinformatics analysis can be used to predict the potential mechanism of action and map the unexplored target spaces of compounds based on abundant network big-data resources and multi-analysis software. 16 Bioinformatics analysis combined with experimental validation has become an effective means to study the actionable targets and elucidate the mechanisms underlying the effects of natural compounds. 17 Therefore, in the present study, bioinformatics analysis combined with experimental validation was used to evaluate the therapeutic effects of farrerol, screen its key targets in the treatment of IH, and explore its underlying mechanisms in serum-induced proliferation and H2O2-induced inflammatory responses in cultured A7r5 cells. The present study provides experimental support to elucidate the mechanism of action of farrerol in the suppression of vascular IH.
Materials and Methods
Drugs and Reagents
Farrerol (purity by HPLC, 99%) was synthesized in the laboratory. 18 PI3K p85α, mTOR, and β-actin polyclonal antibodies were purchased from Bioworld Technology (Nanjing, China). Phospho-Akt1-S473, Phospho-PI3KP85α/γ/β-Y467/Y199/Y464, Phospho-mTOR-S2448, and Akt1 rabbit mAbs were purchased from ABclonal (Wuhan, China). The DNA Content Quantitation Assay (Cell Cycle) was purchased from Solarbio (Beijing, China). The Cell-Light EdU Apollo488 In Vitro Kit was purchased from Ribobio (Guangzhou, China).
The database websites used in the present study are as follows: http://tcmspw.com/tcmsp.php, https://www.genecards.org/, http://www.swisstargetprediction.ch/, https://www.uniprot.org/, https://string-db.org/, https://www.omicshare.com/, https://david.ncifcrf.gov/.
Evaluation of Farrerol Druggability
The Traditional Chinese Medicine System Pharmacology Database and Analysis Platform (TCMSP) can query the relationships among compounds, targets, and diseases, as well as the absorption, distribution, metabolism, and excretion (ADME) of compounds. 19 The chemical structure of farrerol and its ADME characteristics were obtained from the TCMSP database.
Prediction of IH-Related Targets and Screening of Potential Targets of Farrerol
IH-related targets were predicted by entering the keywords “vascular intimal hyperplasia” into the GeneCards database. 20 The structure of farrerol was drawn using ChemDraw 18.0, and saved in the SDF format. The chemical structure (SDF format) was then imported into the Swiss Target Prediction database for target matching and prediction. The Uniprot database was used for the normalization of the names of target proteins. Using the Venny online analysis tool, potential farrerol targets, and IH-related targets were intersected to obtain shared targets. 21
Construction of Protein–Protein Interaction and Compound–Target–Pathway Networks
The shared targets were imported into the STRING database to obtain the protein–protein interaction (PPI) relationships, and the results were analyzed and saved in TSV format. 22 The organism was set as “Homo sapiens,” and the minimum required interaction score was 0.4. The PPI and compound–target–pathway networks were constructed using Cytoscape v3.7.2 software.
GO and KEGG Enrichment Analyses
The potential IH-related targets of farrerol were imported into the DAVID database for GO biological function and KEGG pathway enrichment analyses. 23 A KEGG enrichment bubble diagram was drawn using the OmicShare online analysis platform.
Cell Culture and Treatment
A7r5 cells (rat thoracic aorta smooth muscle cell line, passage no. 5-12) from the Cell Bank of the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China) were cultured in DMEM (BOSTER, Wuhan, China) containing 10% FBS (EVERY GREEN, Hangzhou, China) and 1% penicillin-streptomycin under stable conditions (5% CO2 at 37 °C). Before any experimental procedure, cells were grown to 70% confluence and then incubated in DMEM (0.5% FBS) for 24 h to induce quiescence and synchronization.
A7r5 cells were seeded in 96-well plates (3 × 103 cells/well) or 6-well plates (3 × 105 cells/well). In the viability assay, cells were pretreated with farrerol (0.01, 0.1, 1, 10, 50, and 100 µM) for 24 h. In the analysis of cell proliferation, cells were pretreated with farrerol (0.1, 0.3, 1, 3, 10, and 30 µM) for 24 h and induced with 10% FBS for 24 h. In the H2O2-induced inflammatory model of A7r5 cells, the cells were pretreated with farrerol at concentrations of 0.3, 3, and 30 µM for 24 h and incubated with H2O2 (300 µM) for 6 h to induce oxidative damage.
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyl-2H-Tetrazolium Bromide (MTT) Assay
The MTT assay was used to analyze the effect of farrerol on the viability and proliferation of A7r5 cells. The entire testing process was performed in accordance with the manufacturer's instructions. In brief, after incubation, MTT (5 µg·mL−1, 20 μL) reagent was added to each well, and cells were incubated at 37 °C for 4 h, and washed with PBS. Then, to dissolve the formazan product, DMSO (200 µL) was added to each well. The absorbance of the resulting solution was assessed at 570 nm via a microplate reader (Thermo Scientific, USA), and the optical density was determined. 24
EdU Incorporation Assay
EdU reagent was added to the culture medium and incubated for 2 h. The cells were stained using the CLICK-iT assay. EdU-positive cell numbers were estimated and standardized using the total number of Hoechst 33342-stained cells. 25
Flow Cytometric Analysis
The cell cycle distribution of A7r5 cells was analyzed using flow cytometry. In brief, after treatment, cells were digested with trypsin, centrifuged, and washed twice with PBS. Then, the cells were fixed with 70% ice-cold ethanol (4 °C) overnight. Next, the fixed cells were centrifuged and stained with 0.4 mL of propidium iodide (PI) diluent (50 mg·L−1 PI and 100 mg·L−1 RNase A) for 30 min at 25 °C in the dark. The cells were then analyzed using a flow cytometer. 26 Finally, the ratio of the cells at different phases of the cell cycle in each group was calculated.
Western Blotting
After different treatments, the cells were collected and lysed at 4 °C for 1 h with pre-cooled lysis buffer and centrifuged at 12,000 × g for 10 min. The protein concentration in the supernatant was determined quantitatively using the BCA protein assay. The quantified proteins were heated at 100 °C for 10 min in an appropriate amount of loading buffer. Equal amounts of sample were then subjected to 8% or 10% SDS-PAGE, and the strips were transferred to a nitrocellulose membrane, sealed with 5% skim milk for 1 h, washed three times with TBST (10 min each time), and incubated with the rabbit monoclonal antibodies against p-Akt (1:1000, ABclonal: AP0637, China), Akt (1:1000, ABclonal: A17909, China), PI3K (1:1000, Bioworld: BS6291, China), m-TOR (1:1000, Bioworld: BS3611, China), and β-actin (1:1000, Bioworld: AP0060, China) overnight at 4 °C. After washing three times with TBST, the secondary antibodies (1:2000, BOSTER: BA1054, China) were added to the samples for 1 h, which were then washed three times with TBST, placed in a developing solution, and analyzed with Image-J software. 15
Enzyme-Linked Immunosorbent Assay
The secretion of pro-inflammatory cytokines was quantified using enzyme-linked immunosorbent assay (ELISA). The linear regression equation of the standard curve was calculated using the standard concentrations. The OD value of the sample was tested and substituted into the equation of the standard curve to calculate the sample concentration. 27
Statistical Analysis
GraphPad Prism v7.0 was used for statistical analysis. Experimental data are presented as the mean ± SEM. Results were analyzed by Student's t-tests for two-group comparisons. Statistical variation was considered significant at P < .05.
Results
The Medicinal Properties of Farrerol Satisfy the Drug-Like Principles
The oral bioavailability (46.25%), drug-like properties (0.26%), and Caco-2 values (5) of farrerol (Figure 1a), with a relative molecular mass of 300.33, were obtained from the TCMSP database. These data satisfied drug-like principles, indicating that farrerol is a natural compound with potential for drug development.
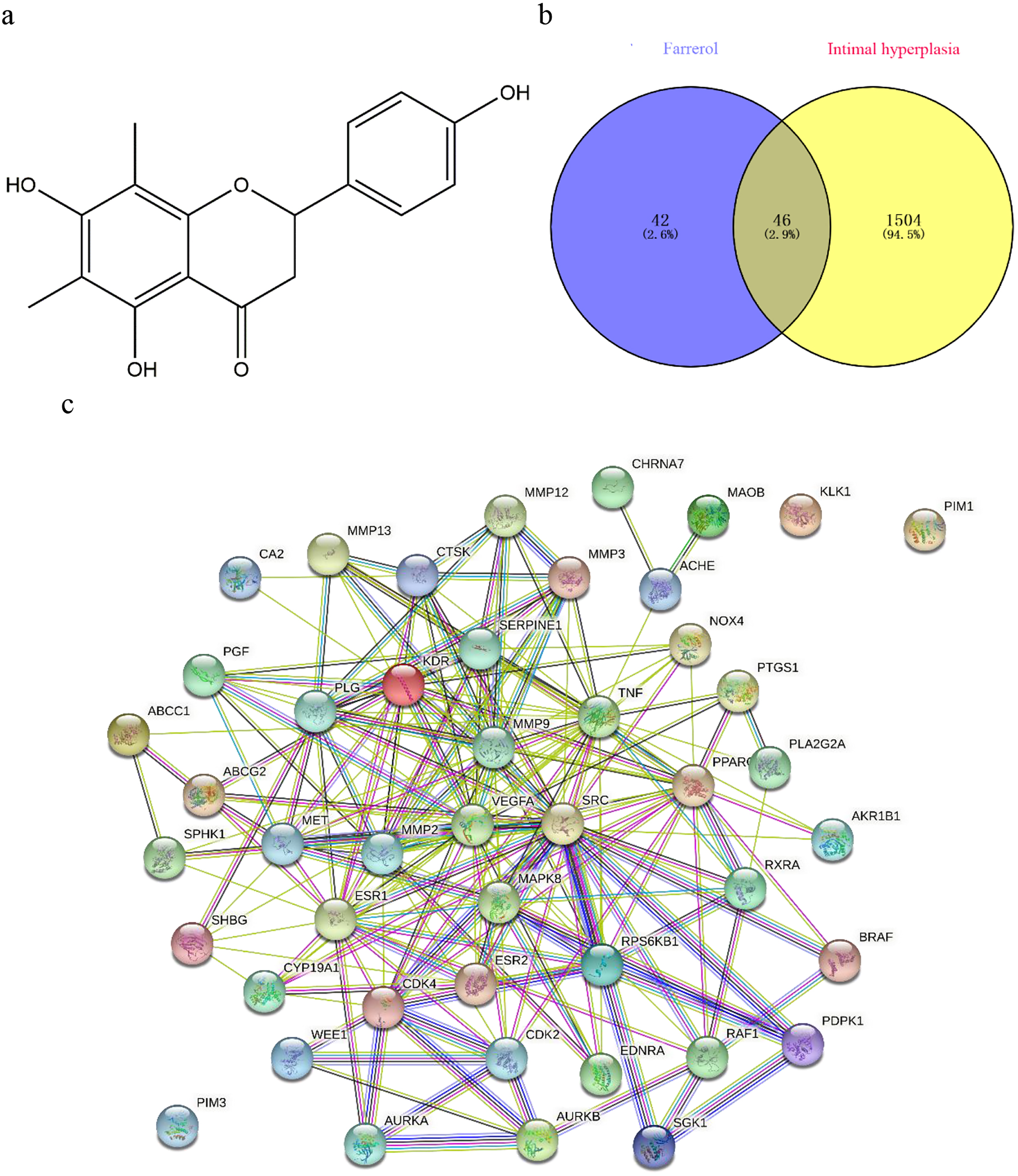
Screening of core targets and construction of protein–protein interaction (PPI) network for farrerol with respect to vascular intimal hyperplasia. (a) Chemical structure of farrerol. (b) Venn diagram of potential targets of farrerol and vascular intimal hyperplasia (IH)-related targets. The potential targets of farrerol with respect to IH were identified at the intersection of the potential targets of farrerol and IH-related targets using the Venny online tool. (c) Interactive PPI network of farrerol and IH-related targets.
Target Prediction, Screening, and Construction of PPI Network
Eighty-eight targets of farrerol and 1550 IH-related target genes were obtained. In total, 46 potential targets of farrerol with respect to IH (Figure 1b) were identified using the Venny online tool. These 46 targets were presented in the form of a PPI network (Figure 1c), which systematically summarizes the interactions among farrerol targets associated with IH treatment. In addition to three free protein target nodes (PIM1, PIM3, and KLK1), the network showed 43 viable protein target nodes connected by 207 edges with an average node degree of 9, and an average local clustering coefficient of 0.64. The p-value for PPI enrichment was < 1.0×10−16. These 43 interacting genes were used for further investigation.
GO Analysis, KEGG Analysis, and Construction of Compound–Target–Pathway Network
GO biological function analysis yielded GO items (P < .05), including 87 biological process (BP) items, 17 cellular component (CC) items, and 32 molecular function (MF) items. The first 10 items were selected for each category (Figure 2a). BPs were involved in ATP binding (P = 5.24 × 10−8), protein kinase activity (P = 3.54 × 10−7), protein serine/threonine kinase activity (P = 5.37 × 10−5), enzyme binding (P = 1.86 × 10−4), and steroid hormone receptor activity (P = 3.88 × 10−4), whereas MFs included signal transduction (P = 5.95 × 10−6), negative regulation of apoptosis (P = 2.22 × 10−3), protein phosphorylation (P = 1.49 × 10−6), positive regulation of angiogenesis (P = 9.59 × 10−6), and CCs involved the cell nucleus (P = 6.37 × 10−4), plasma membrane (P = 4.6 × 10−4), extracellular matrix (P = 4.04 × 10−4), and cell surface (P = 8.66 × 10−3). These findings suggest that the effects of farrerol on IH might result from a complex of multiple pathways with synergistic effects.

Go and KEGG pathway enrichment analyses of intimal hyperplasia-related candidate target genes and compound–target–pathway network. (a) GO enrichment analysis of candidate target genes in terms of biological processes (BPs), cellular components (CCs), and molecular functions (MFs). (b) KEGG enrichment analysis of candidate target genes (advanced bubble chart). (c) The farrerol–target–pathway network model. Yellow nodes represent the compound (farrerol) and pink nodes and blue nodes represent co-targets and pathways, respectively.
KEGG pathway enrichment analysis revealed 43 pathways (P < .05), including PI3K-Akt, VEGF, FoxO, Rap1, ErbB, Ras, the thyroid hormone, and the mTOR signaling pathway. The top 20 enriched KEGG signaling pathways are shown in Figure 2b. To further determine the underlying mechanism of farrerol against IH, an overall compound–target–pathway network model was constructed based on the top 20 enriched KEGG signaling pathways along with their corresponding targets using Cytoscape v3.7.2 software. This compound-target-pathway interaction network contained 46 nodes (1 compound, 25 targets, and 20 pathways) and 155 edges. The yellow oblong, pink rectangles, and blue diamonds correspond to farrerol, target genes, and signal pathways, respectively (Figure 2c). Betweenness Centrality and Closeness Centrality of the network were 0.08058 and 0.48913, respectively. In addition to the two signaling pathways involved in the development of cancer, the PI3K/Akt signaling pathway (degree = 12) was the most highly enriched pathway. The mTOR signaling pathway (degree = 4), a downstream pathway of PI3K/Akt, was also among the top 20 enriched KEGG signaling pathways. It was found that the PI3K/Akt/mTOR signaling pathway (degree = 16) was linked with 16 target genes. Accordingly, the network analysis suggested that the protective role of farrerol in the suppression of IH might be related to the PI3K/Akt/mTOR signaling pathway.
Inhibitory Effect of Farrerol on the Proliferation of VSMCs
In our preliminary experiments, the cellular toxicity of farrerol toward cultured A7r5 cells was evaluated within the concentration range of 0–100 µM using the MTT assay. Farrerol did not affect cell viability even at a concentration of 100 μM (Figure 3a), indicating that the inhibitory effect of farrerol on cell proliferation did not result from its cytotoxicity.

Farrerol inhibits 10% FBS-induced proliferation of vascular smooth muscle cells (VSMCs). A7r5 cells were pretreated with farrerol at specific concentrations for 24 h and then stimulated with 10% FBS for 24 h. (a) Effect of farrerol on the viability of A7r5 cells (n = 3). (b) Inhibitory effect of farrerol on 10% FBS-induced A7r5 cell proliferation (n = 6). ### P < .001 versus control group (0.5% FBS); * P < .05 versus model group (10% FBS); ** P < .01 versus model group (10% FBS); *** P < .001 versus model group (10% FBS). (c) DNA synthesis in VSMCs determined by an Edu incorporation assay. Blue fluorescence (Hoechst 33342) shows cell nuclei and red fluorescence (Edu) indicates cells with DNA synthesis. Scale bars = 150 μm. (d) Flow cytometric analysis of each group.
Then, the effects of farrerol on VSMC proliferation induced by 10% FBS were investigated using MTT assays. The results showed that treating A7r5 cells with 10% FBS markedly increased cell proliferation, whereas the 10% FBS-induced proliferation of A7r5 cells was inhibited by various concentrations of farrerol (Figure 3b).
Furthermore, DNA synthesis in A7r5 cells was detected using the EdU incorporation assay. Accordingly, farrerol (0.3, 3, and 30 µM) pretreatment reduced the number of EdU-positive cells during 10% FBS-induced cell proliferation in a concentration-dependent manner (Figure 3c).
Farrerol Induces Cell Cycle Arrest in the G0/G1 Phase
Subsequently, the effect of farrerol on the A7r5 cell cycle was investigated using flow cytometric analysis. The results showed that farrerol-induced cell cycle arrest at the G0/G1 phase in A7r5 cells, indicating that farrerol inhibited cell proliferation via the regulation of the cell cycle in A7r5 cells (Figure 3d and Table 1).
Cell Cycle Composition Ratio (n = 3).
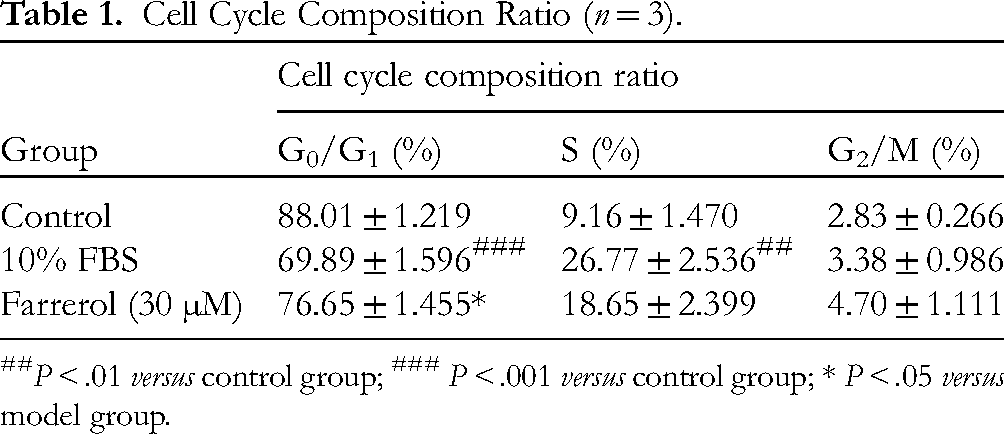
P < .01 versus control group; ### P < .001 versus control group; * P < .05 versus model group.
Farrerol Alleviates the Inflammatory Response in H2O2-Induced VSMCs
Inflammation plays a substantial role in the pathogenesis and development of IH, and farrerol possesses anti-inflammatory properties. 28 To investigate the protective effects of farrerol on the inflammatory response in H2O2-induced VSMCs, the release of pro-inflammatory cytokines induced by H2O2 (300 µM), such as IL-1β, IL-6, and TNF-α (Bioswamp, Wuhan, China), in the cell culture medium was estimated using an ELISA. A7r5 cells were treated with H2O2 (300 µM) for 6, 12, 18, and 24 h. The results indicated that H2O2 (300 µM) treatment for 6, 12, 18, and 24 h significantly enhanced the secretion of IL-1β and TNF-α, but not IL-6, compared to that in the control group (Figure 4a). Therefore, we examined the effect of farrerol on the secretion of IL-1β and TNF-α induced by H2O2 (300 µM) for 6 h. The results showed that farrerol pretreatment significantly reduced the H2O2-induced release of these two pro-inflammatory cytokines (Figure 4b), suggesting a potent anti-inflammatory effect of farrerol.

Farrerol alleviates the inflammatory response in H2O2-induced A7r5 cells. The release of IL-1β, IL-6, and TNF-α induced by H2O2 (300 μM) in the cell culture medium was estimated using ELISA. (a) Effect of H2O2 on the release of IL-1β, IL-6, and TNF-α by A7r5 cells (n = 3). (b) Inhibitory effect of farrerol on the release of IL-1β and TNF-α induced by H2O2 (n = 3). ## P < .01 versus control group; ### P < .001 versus control group; #### P < .0001 versus control group; ** P < .01 versus model group (300 μM H2O2) *** P < .001 versus model group (300 μM H2O2).
Farrerol Regulates the PI3K/Akt/mTOR Pathway in Both 10% FBS- and H2O2-Induced VSMCs
To confirm that the PI3K/Akt/mTOR pathway is involved in the effects of farrerol on 10% FBS-induced proliferation and H2O2-induced oxidative damage, total protein levels of PI3K, Akt, and mTOR, as well as those of phosphorylated PI3K, Akt, and mTOR proteins, in A7r5 cells were determined by western blotting. The results showed that the 10% FBS-induced phosphorylation of Akt (Ser473) and expression of PI3K and mTOR were increased, whereas the H2O2-induced phosphorylation of Akt (Ser473), PI3K, and mTOR and expression of mTOR was decreased, compared to levels in the corresponding control group. Farrerol (0.3, 3, and 30 µM) pretreatment significantly reversed the 10% FBS-induced protein expression of p-Akt, PI3K, and mTOR (Figure 5a and b) and H2O2-induced suppression of p-Akt, p-PI3K, and mTOR. Total protein expression of Akt in 10% FBS-induced A7r5 cells, along with Akt and PI3K in H2O2-induced A7r5 cells, remained unchanged (Figure 5c and d). These results indicate that farrerol inhibits the proliferation of VSMCs and protects VSMCs from oxidative injury via bidirectional modulation of the PI3K/Akt/mTOR signaling pathway.
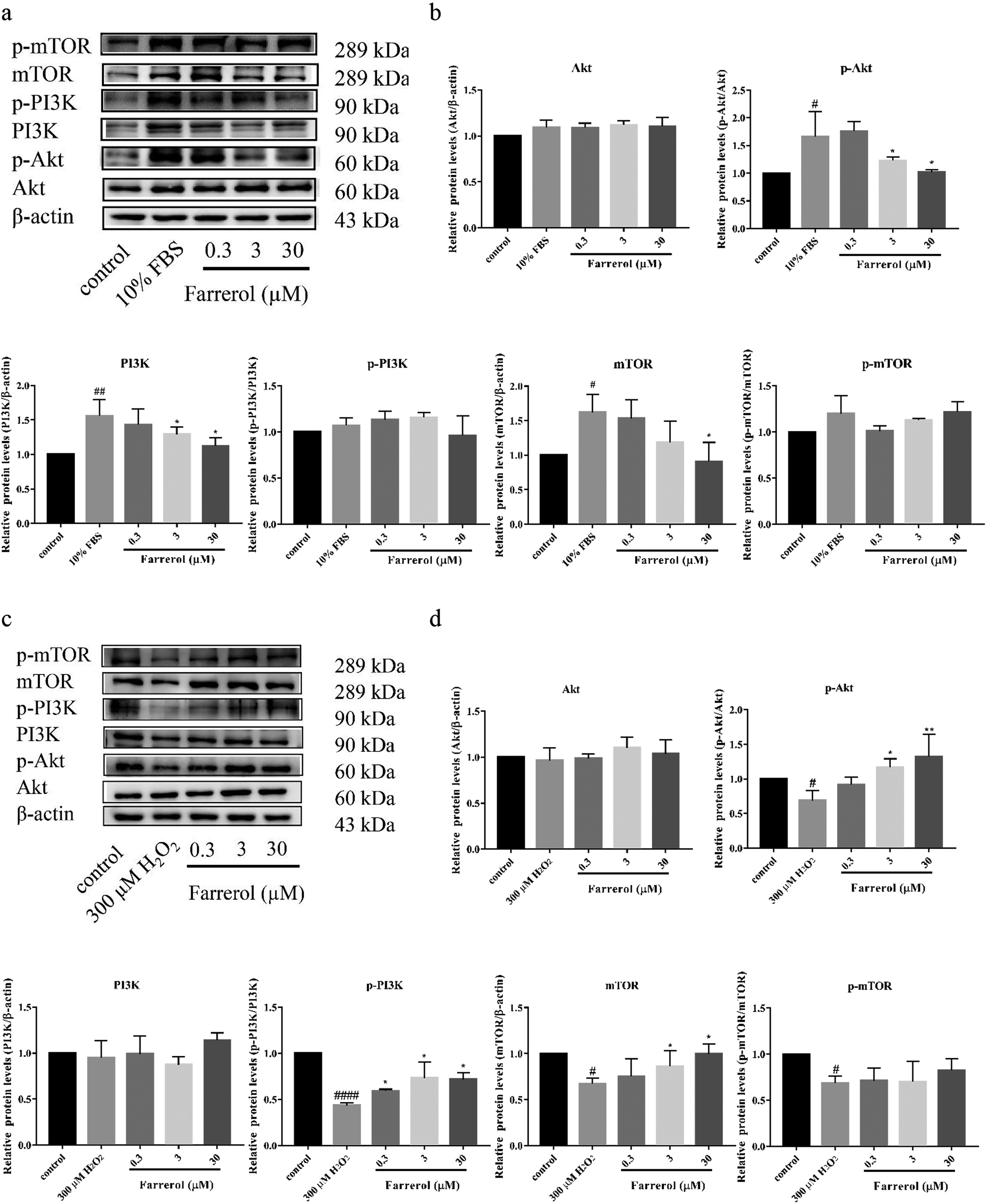
Farrerol regulates the PI3K/Akt/mTOR pathway in both 10% FBS- and H2O2-induced A7r5 cells. Cells were pretreated with farrerol (0.3, 3, and 30 μM) for 24 h and then treated with 10% FBS for 24 h, or incubated with H2O2 (300 μM) for 6 h. Total protein levels of PI3K, Akt, and mTOR, as well as the p-PI3K, p-Akt, and p-mTOR, in A7r5 cells were determined by western blotting. PI3K, Akt, and mTOR levels were normalized to those of β-actin, whereas p-PI3K, p-Akt, and p-mTOR levels were normalized to PI3K, Akt, and mTOR levels (mean ± SEM, n = 3 in each group). (a) Western blotting analysis of p-PI3K, p-Akt, p-mTOR, PI3K, Akt, and mTOR in vascular smooth muscle cells (VSMCs) treated with or without 10% FBS. (b) Quantitative analysis of the relative protein expression levels of p-PI3K, p-Akt, and p-mTOR, as well as Akt, PI3K, and mTOR in VSMCs treated with or without 10% FBS. (c) Western blotting analysis of p-PI3K, p-Akt, p-mTOR, PI3K, Akt, and mTOR in VSMCs treated with or without H2O2. (d) Quantitative analysis of the relative protein expression levels of p-PI3K, p-Akt, and p-mTOR, Akt, PI3K, and mTOR in VSMCs treated with or without H2O2. # P < .05 versus control group; ## P < .01 versus control group; #### P < .0001 versus control group; * P < .05 versus model group; ** P < .01 versus model group.
Discussion
Our present study revealed that farrerol inhibits VSMC proliferation and protects them from oxidative injury via bidirectional modulation of the key proteins in the PI3K/Akt/mTOR signaling pathway. This demonstrates that it is a common pathway of farrerol for the inhibition of proliferation and protection from oxidative damage in VSMCs, which might contribute to the suppression of neointima formation. VSMCs are the main constituents of the neointima in restenotic lesions. Arterial restenosis after percutaneous transluminal coronary angioplasty is mainly caused by the inflammatory response and excessive proliferation of VSMCs in the injured vascular wall. 29 In response to vascular injury, VSMCs undergo phenotypic transition, switching from a quiescent contractile mode to an active synthetic phenotype. VSMCs with a synthetic phenotype proliferate and secrete diverse pro-inflammatory factors. The accumulation and inflammatory response of VSMCs both contribute to IH and the subsequent vascular restenosis. 30 Hence, restraining the proliferation of VSMCs or interfering with their inflammatory response is an effective measure to prevent restenosis. Our previous study has demonstrated that farrerol significantly inhibits the formation of neointima induced by balloon injury. However, the mechanism underlying the effects of farrerol has not been completely demonstrated.
Given the evidence indicating that excessive proliferation and inflammatory response of VSMCs in the injured vascular wall contribute to the development of restenosis, 31 in the present study, we evaluated the effects of farrerol on cell proliferation and inflammation with VSMCs. The 10% FBS-induced proliferation of cultured A7r5 cells was established to mimic the pathological conditions of IH in vitro. We found that farrerol inhibits VSMC proliferation induced by 10% FBS, which was due to its cell cycle arrest at the G0/G1 phase, rather than its cytotoxicity. In cultured A7r5 cells, farrerol did not affect the cell viability even at a concentration of 100 μM. This characteristic suggests that this compound might have a protective effect against damage to VSMCs.
Among various pathological features of injury-induced VSMCs, the inflammatory response is important. Therefore, we evaluated the effects of farrerol on the inflammatory response of VSMCs in vitro. Vascular injury often leads to the accumulation of reactive oxygen species (ROS) and triggers an acute inflammatory response.32,33 The acute inflammatory response is mainly characterized by the elevated expression of pro-inflammatory cytokines in the vessel wall. 30 H2O2 is involved in many inflammatory cascades, and treating VSMCs with exogenous H2O2 could induce ROS generation. 34 Hence, H2O2-induced oxidative damage of A7r5 cells, characterized by the release of pro-inflammatory factors such as IL-1β and TNF-α, was used to mimic the inflammatory response of VSMCs at the site of vascular injury in vivo. We found that H2O2 treatment significantly increased the secretion and release of IL-1β and TNF-α, which were suppressed by farrerol pretreatment, suggesting that farrerol has a protective effect against oxidative injury in VSMCs, as expected.
The evident effect of farrerol on the abnormal proliferation and inflammatory response of VSMCs prompted us to investigate the underlying mechanism. The PI3K/Akt/mTOR signaling is a key pathway in neointimal hyperplasia, as well as cellular survival and proliferation.35-37 In addition, many studies have confirmed that this signaling pathway plays an active role in inhibiting the inflammatory response. 38 Hence, we explored the role of this signaling pathway in the inhibition of proliferation and the protective effect of farrerol against oxidative damage in A7r5 cells. Our observations revealed that farrerol exerts an inhibitory effect on 10% FBS-induced A7r5 cell proliferation by suppressing the PI3K/Akt/mTOR pathway, while exerting a protective effect against H2O2-induced oxidative damage by promoting the PI3K/Akt/mTOR pathway. These results suggest that farrerol inhibits VSMC proliferation and protects these cells from oxidative injury via bidirectional modulation of the PI3K/Akt/mTOR signaling pathway. However, the mechanism for how farrerol differentially regulates the key protein levels of the PI3K/Akt/mTOR signaling pathway in 10% FBS- and H2O2-induced A7r5 cells need to be further explored.
Conclusion
In summary, farrerol inhibits VSMC proliferation and protects these cells from oxidative injury via bidirectional modulation of the PI3K/Akt/mTOR signaling pathway, suggesting that it is a promising natural product for the treatment of vascular restenosis. For future studies, the molecular target of farrerol should be explored and identified to precisely delineate its mechanism of action.
Footnotes
Author Contributions
Enli Liu and Qingshan Li conceived and designed the experiments. Jiacheng Fang, Huanhuan Jiang, and Rui Ge performed the experiments and data analysis. Jiacheng Fang wrote the first draft of the manuscript, Enli Liu revised the manuscript, and all authors commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Natural Science Foundation of Shanxi Province (grant numbers 20210302123318), the Open Fund of Shanxi Key Laboratory of Innovative Drug for the Treatment of Serious Diseases Basing on the Chronic Inflammation, Shanxi University of Chinese Medicine (grant numbers 2021sxcxyw08).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subject.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
