Abstract
Patchouli alcohol (PA) has multiple pharmacological activities, but its protective effect against SH-SY5Y cell injury induced by Aβ25-35 has not been reported. It has been recorded that phosphatidylinositol 3-hydroxykinase (PI3K)/protein kinase B (Akt)/mammalian target of rapamycin (mTOR) pathway plays an important role in neuroprotection. The purpose of this study was to investigate the protective effect of PA against SH-SY5Y cell injury induced by Aβ25-35 and its underlying mechanism. The results showed that compared with that in the Aβ25-35-induced injury group, the survival rate of SH-SY5Y cells increased (P < .01) in the different PA-treated groups and the lactic dehydrogenase activity decreased significantly (P < .01) in the 10, 20, and 40 μg/mL PA groups; compared with those in the Aβ25-35-induced injury group, the malonyldialdehyde contents in SH-SY5Y cells decreased (P < .05 or P < .01), while the superoxide dismutase, glutathione peroxidase, and catalase activities increased significantly (P < .05 or P < .01) in the different PA-treated groups; compared with those in the Aβ25-35-induced injury group, the apoptosis rates, and the mRNA and protein levels of Caspase-3 and Bax in SH-SY5Y cells decreased (P < .05 or P < .01), while the mRNA and protein levels of Bcl-2, and phosphorylated Akt (p-Akt) and phosphorylated mTOR protein levels increased significantly (P < .05 or P < .01) in the different PA-treated groups. The above results indicate that PA can inhibit the oxidative stress and apoptosis of SH-SY5Y cells induced by Aβ25-35 by regulating the PI3K/Akt/mTOR pathway, to protect the SH-SY5Y cells from the injury induced by Aβ25-35.
Introduction
Alzheimer's disease (AD) is a degenerative disease of the central nervous system characterized by memory and cognitive dysfunction, and its main pathological characteristics are senile plaque deposition, neurofibrillar tangles, and a large loss of neurons. 1 AD is most often diagnosed in the elderly, and its incidence is <1% in the population under 60 years old, but increases rapidly with age, and is >30% in those over 85 years old. 2 The treatment cost of AD is up to hundreds of billions of dollars in the world every year, bringing a heavy burden to societies and families. The amyloid protein β (Aβ) deposition plays an important key role in the pathogenesis of AD. 3 Aβ is composed of 39 to 43 amino acids and generated from amyloid precursor protein (APP) through proteolytic cleavage; Aβ monomers can be polymerized to form water-insoluble Aβ oligomers that can cause neurotoxicity, leading to the occurrence and development of AD.4,5
At present, the main drugs for the clinical treatment of AD are antioxidants, estrogens, lipidemic-modulating agents, brain metabolism improving drugs, nonsteroidal anti-inflammatory drugs, calcium antagonists, and acetylcholinesterase inhibitors, but the treatment effect of these drugs is associated with side effects, high price, and single target,6,7 so it is of great significance to develop drugs with low toxicity, low price, multiple targets, and good efficacy for the treatment of AD. 8 As one of the main active components of Pogostemon cablin (Blanco) Benth. (family Labiatae), patchouli alcohol (PA) is a sesquiterpene with multiple pharmacological effects such as anti-inflammation, antibacterial, anticancer, and prevention of mastitis, colitis, and atherosclerosis.9-11 The protective effect of PA against various tissue injuries was reported. It was found that PA can improve the learning and memory of mice with scopolamine-induced memory impairment, which is related to its increasing the density of M1 receptors and the activity of choline acetyl transferase, and inhibiting the activity of acetylcholinesterase in the brain. 12 It was found that PA has a protective effect against the acute lung injury induced by lipopolysaccharide (LPS) in mice. 13 PA can be converted to β-patchoulene, which can protect the gastric injury caused by ethanol. 14 PA can protect against brain ischemia/reperfusion (I/R) injury in healthy and obese mice by inhibiting inflammation. 15 Furthermore, PA can also protect against the liver steatosis induced by a high-fat diet by mitigating the stress of endoplasmic reticulum and regulating the metabolism of very-low-density lipoprotein. 16 Up to now, the protective effect of PA against the SH-SY5Y cell injury induced by Aβ25-35 has not been reported. Therefore, PA was used to intervene with the SH-SY5Y cell injury model induced by Aβ25-35, to explore its protective effect against the injury and underlying mechanism, in order to provide an experimental basis for the further development of new drugs for the treatment of AD.
Results and Discussion
Aβ, a kind of amyloid peptide with a β-fold structure, is produced by the abnormal metabolism of β-APP, and can oligomerize into Aβ oligomers outside neurons. 17 Aβ oligomers can directly destroy the ion channels to enter the cell for aggregation and can also activate the intracellular signaling pathways by binding to a variety of receptors on the cell membrane surface, causing cell injuries such as lipid peroxidation, increase of reactive oxygen species (ROS), dysfunction of mitochondria, and inducing cell apoptosis. 18 Aβ25-35, the active fragment of a synthetic β amyloid protein, is similar to Aβ1-42 in biological activities, and is commonly used to induce both in vitro and in vivo AD models. 19 In recent years, great progress has been made in the application of active components of traditional Chinese medicines in the treatment of AD, such as saponins, flavonoids, and polysaccharides. 20 The thiazolyl blue tetrazolium bromide (MTT) assay results showed that compared with that in the normal control group, the cell survival rate of SH-SY5Y cells treated with different concentrations of PA was not significantly different (P > .05), as shown in Figure 1A. Compared with that in the normal control group, the cell survival rate of the SH-SY5Y cells in the 25 μmol/L Aβ25-35-induced injury group was significantly reduced (P < .01); compared with that in the Aβ25-35-induced injury group, the cell survival rate of the cells in each PA-treated group was significantly increased in a dose-dependent manner (P < .01), as shown in Figure 1B, suggesting that PA could alleviate the SH-SY5Y cell injury induced by Aβ25-35.

Effect of PA on the survival rate of Aβ25-35-induced SH-SY5Y cells. (A) SH-SY5Y cells were treated with different concentrations of PA (5, 10, 20, and 40 μg/mL) for 24 h and the cell survival rate was determined using the MTT assay and (B) SH-SY5Y cells were treated with Aβ25-35 (25 μmol/L) and PA (5, 10, 20, and 40 μg/mL) for 24 h. Then, the cell survival rate was measured using the MTT assay. Values are expressed as mean ± SD, n = 5. Compared with the normal control group: **P < .01; compared with Aβ25-35-induced injury group: ##P < .01.
The lactic dehydrogenase (LDH) activity in the supernatant of SH-SY5Y cells in the normal control group maintained at a lower level; compared with that in the normal group, the LDH activity in the supernatant of SH-SY5Y cells in the Aβ25-35-induced injury group increased significantly (P < .01); compared with that in the Aβ25-35-induced injury group, the LDH activity in the supernatant of SH-SY5Y cells was significantly reduced in the other PA-treated groups, except the 5 μg/mL PA-treated group (P < .01), further suggesting that PA could alleviate the SH-SY5Y cell injury induced by Aβ25-35 (Figure 2).

Effect of PA on the LDH activity of Aβ25-35-induced SH-SY5Y cells. SH-SY5Y cells were incubated with Aβ25-35 (25 μmol/L) and PA (5, 10, 20, and 40 μg/mL) for 24 h. Values are expressed as mean ± SD, n = 5. Compared with the normal control group: **P < .01; compared with Aβ25-35-induced injury group: ##P < .01.
Oxidative stress has attracted more and more attention connected with the pathogenesis of AD. 21 Dysfunction of the antioxidant system and an increase in the free radical level may lead to damage of brain nerve cells, and then induce the pathological changes of AD. 22 Aβ can mediate the production of ROS in nerve cells, and the excessive ROS will attack the membrane of nerve cells if they cannot be scavenged in time and damage the basic structure of nerve cells to cause the apoptosis or death of cells. 23 It has been confirmed that the three main pathological manifestations of AD, ie senile plaques, nerve fiber tangles, and neuron apoptosis, are closely related to oxidative stress. 24 Antioxidants can prevent or eliminate the generation of ROS and alleviate the degeneration of nerve cells, such as melatonin with the effect of scavenging free radicals, which can significantly inhibit the abnormal activation of glial cells and the deposition of Aβ, thus improving the learning and memory of AD model mice.25,26 The activities of antioxidant enzymes superoxide dismutase (SOD), glutathione peroxidase (GSH-Px) and catalase (CAT), and the content of malonyldialdehyde (MDA), an end product of lipid peroxidation, are often used as indicators to evaluate the body's antioxidant capacity. 27 Compared with those in the normal control group, SOD, GSH-Px, and CAT activities in SH-SY5Y cells were significantly decreased (P < .01), while the MDA content was significantly increased (P < .01) in the Aβ25-35-induced injury group; compared with those in the Aβ25-35-induced injury group, SOD, GSH-Px, and CAT activities in SH-SY5Y cells were significantly increased (P < .05 or P < .01), while the MDA content was significantly decreased (P < .05 or P < .01) in the different PA-treated groups (Figure 3), indicating that PA could lower the oxidative stress level of SH-SY5Y cells induced by Aβ25-35.

Effects of PA on SOD, GSH-Px and CAT activities and MDA content in Aβ25-35-induced SH-SY5Y cells. (A) SOD activity; (B) GSH-Px activity; (C) CAT activity; and (D) MDA content. SH-SY5Y cells were incubated with Aβ25-35 (25 μmol/L) and PA (5, 10, 20, and 40 μg/mL) for 24 h, respectively. Values are expressed as mean ± SD, n = 5. Compared with the normal control group: **P < .01; compared with Aβ25-35-induced injury group: #P < .05, ##P < .01.
Neuron apoptosis, one of the pathological characteristics of AD, is mainly manifested in the significant reduction of the number of neurons. 28 Aβ has a strong toxicity to neurons, which can directly lead to neuron apoptosis, and it can also induce the apoptosis of neurons by lowering the function of the K+ channel, destroying the calcium homeostasis in cells, promoting the formation of free radicals, and enhancing the mediation of active molecules. 29 Flow cytometry was used to observe the effect of PA on the apoptosis rate of SH-SY5Y cells induced by Aβ25-35. As shown in Figure 4, compared with that in the normal control group, the apoptosis rate of SH-SY5Y cells increased significantly in the Aβ25-35-induced injury group (P < .01); compared with that in the Aβ25-35-induced injury group, the apoptosis rate of SH-SY5Y cells decreased significantly in the different PA-treated groups (P < .05 or P < .01), suggesting that PA should have an antiapoptosis effect on SH-SY5Y cells induced by Aβ25-35.

Effect of PA on the apoptosis rate of Aβ25-35-induced SH-SY5Y cells. (A) Representative profiles of cell apoptosis detected by flow cytometry and (B) percentages of apoptotic cells. SH-SY5Y cells were treated with Aβ25-35 (25 μmol/L) and PA (5, 10, 20, and 40 μg/mL) for 24 h, respectively. Values are expressed as mean ± SD, n = 5. Compared with the normal control group: **P < .01; compared with Aβ25-35-induced injury group: #P < .05, ##P < .01.
The process of neuron apoptosis is regulated by heterodimer proteins Bax and Bcl-2; the former promotes and the latter resists apoptosis, so the proportion of Bax and Bcl-2 can be used to evaluate the process of apoptosis. 30 Bax can directly induce the release of cytochrome C, and further activate Caspase-3, the downstream key molecule of apoptosis, while the overexpression of Bcl-2 can prevent the release of cytochrome C from the mitochondria and inhibit the apoptosis. 31 It was found that ginsenoside Re can inhibit the apoptosis of SH-SY5Y cells induced by Aβ by increasing the ratio of Bcl-2/Bax, reducing the release of cytochrome C, and downregulating the expression of Caspase-3. 32 The effect of PA on the level of apoptosis-related genes and proteins in Aβ25-35-induced SH-SY5Y cells was investigated by real-time quantitative polymerase chain reaction (qPCR) and Western blot. As shown in Figure 5, compared with those in the normal control group, the mRNA and protein levels of Caspase-3 and Bax increased significantly (P < .01), while the Bcl-2 mRNA and protein levels decreased significantly (P < .01) in the Aβ25-35-induced injury group; compared with those in the Aβ25-35-induced injury group, the mRNA and protein levels of Caspase-3 and Bax in SH-SY5Y cells decreased significantly (P < .05 or P < .01), while the Bcl-2 mRNA and protein levels increased significantly in the different PA-treated groups (P < .05 or P < .01), further confirming that PA had an antiapoptosis effect on SH-SY5Y cells induced by Aβ25-35.
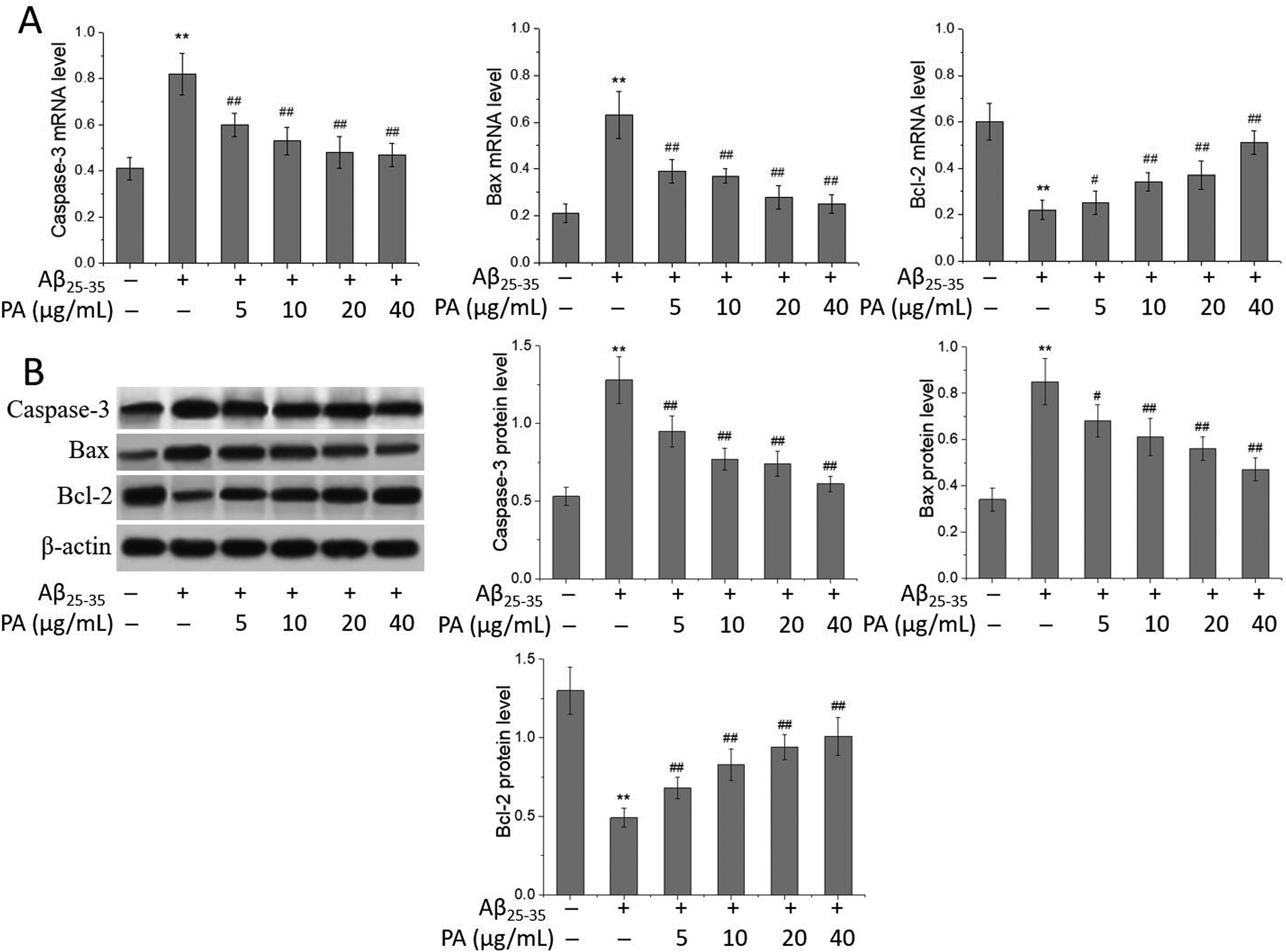
Effects of PA on the levels of apoptosis-related genes and proteins in Aβ25-35-induced SH-SY5Y cells. (A) Caspase-3, Bax and Bcl-2 mRNA levels were determined by real-time qPCR and (B) Caspase-3, Bax, and Bcl-2 protein levels were determined by Western blot. SH-SY5Y cells were treated with Aβ25-35 (25 μmol/L) and PA (5, 10, 20, and 40 μg/mL) for 24 h, respectively. Values are expressed as mean ± SD, n = 5. Compared with the normal control group: **P < .01; compared with Aβ25-35-induced injury group: #P < .05, ##P < .01.
Phosphatidylinositol 3-hydroxykinase (PI3K)/protein kinase B (Akt)/mammalian target of rapamycin (mTOR) pathway, an important signaling pathway for the survival of cells, is involved in the proliferation and apoptosis of nerve cells. 33 Akt, the expression product of proto-oncogene c-Akt, is located at the center of the PI3K/Akt/mTOR pathway and its activation is regulated by PI3K, and it is necessary for neuron survival, information transmission among synapses, synaptic structure formation, axon and dendrite formation, and nerves and angiogenesis. 34 The activated Akt can protect nerve cells from the apoptosis induced by oxidative toxins such as Aβ protein in the oxidative damage of cells, which is essential for the survival of cells. 35 The mTOR can be directly activated by Akt, and also plays an important role in the metabolism, growth, and apoptosis of the nerve cells. 36 It was found that PA can inhibit the autophagy in hippocampus, repair synapses, and restore the autophagy flux by activating the mTOR signaling pathway, with an antidepressant effect on rats with chronic unpredictable mild stress. 37 The results of this study also showed that compared with those in the normal control group, p-Akt and p-mTOR protein levels in the Aβ25-35-induced injury group were significantly reduced (P < .01), and compared with those in the Aβ25-35-induced injury group, p-Akt and p-mTOR protein levels in Aβ25-35-induced SH-SY5Y cells increased significantly in the different PA-treated groups (P < .05 or P < .01, Figure 6). The PI3K/Akt/mTOR pathway plays an important role in neuroprotection. It was found that hydrogen-saturated saline can play a neuroprotective role by regulating the activation of the PI3K/Akt/mTOR pathway in a rat model of early and medium-term Parkinson's disease induced by rotenone. 38 Mangiferin can enhance the neuroprotective effect of isoflurane against the neonatal hypoxic brain injury by reducing oxidative stress and PI3K/Akt/mTOR pathway activation. 39 The neuroprotective effect of panaxatriol saponins on PC12 cells and zebrafish is also related to PI3K/Akt/mTOR. 40 Based on these findings and the results of this study, it can be deduced that the neuroprotective effect of PA may be related to the regulation of the PI3K/Akt/mTOR pathway. The mitogen-activated protein kinases (MAPK) signal pathway also participates in PA-mediated pharmacological activities. Studies have shown that PA treatment can alleviate the dysfunction of the blood brain barrier and inhibit the level of TNF-α, IL-1β mRNA and proteins, and regulate the activation of the MAPK signaling pathway. 15 In addition, antitumor and antiviral effects of PA are also related to regulation of the MAPK signaling pathway.41,42 However, whether the MAPK signaling pathway can play a role in the protection of PA against the Aβ25-35-induced injury of SH-SY5Y cells needs further study.

Effect of PA on the PI3K/Akt/mTOR pathway in Aβ25-35-induced SH-SY5Y cells. (A) Western blot analysis of p-Akt protein levels in SH-SY5Y cells and (B) Western blot analysis of p-mTOR protein levels in SH-SY5Y cells. SH-SY5Y cells were treated with Aβ25-35 (25 μmol/L) and PA (5, 10, 20, and 40 μg/mL) for 24 h. Values are expressed as mean ± SD, n = 5. Compared with the normal control group: **P < .01; compared with Aβ25-35-induced injury group: #P < .05, ##P < .01.
Conclusions
PA can play a neuroprotective role by inhibiting the oxidative stress and apoptosis of SH-SY5Y cells induced by Aβ25-35. Based on the important role of the PI3K/Akt/mTOR pathway in neuroprotection and the results, it is speculated that the neuroprotective effect of PA may be related to its regulation of the PI3K/Akt/mTOR pathway.
Experimental
Materials
PA (purity ≥ 95%) was purchased from DASF Biotech Co, Ltd and dissolved in dimethyl sulfoxide to make a 1 mg/mL stock solution. Aβ25-35 (purity ≥ 97%, A4559) was purchased from Sigma Chemical Co. and dissolved in sterile double-distilled water at a concentration of 1 mmol/L stock solution, and the solution was left standing at 37 °C for 4 days and then stored at –20 °C before use. The detection kits of MTT (G020-1-2), LDH (A075-1-1), SOD (A001-4-1), GSH-Px (A005-1-2), CAT (A007-1-1), and MDA (A003-1-2) were provided by Jiancheng Biology Engineering Institute. TRIzol reagent (15596026) was purchased from Invitrogen and qPCR kit (LS2062) from Promega. The polyclonal antibodies of Caspase-3 (BA2142), Bax (BA0315-2), Bcl-2 (BM0200), and β-actin (BA2305) were purchased from Boster Biological Technology, and Akt (4691), p-Akt (4060), mTOR (2983), and p-mTOR (5536) from Cell Signaling Technology.
Cell Culture
The SH-SY5Y cell line was obtained from American Type Culture Collection (ATCC) and maintained in high glucose Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum and penicillin/streptomycin (100 μg/mL) at 37 °C with 5% CO2.
Measurement of Cell Viability by MTT Assay
SH-SY5Y cells were cultured to the logarithmic growth stage, and the cells counted after they were digested and centrifuged; the cell density was adjusted to 5 × 105 cells/mL with DMEM complete culture medium. The cells in 100 μL of the cell suspension were seeded in 96-well cell culture plates (5000 cells), in which a normal control group, and 5, 10, 20, and 40 μg/mL PA groups were set up. After culture for 24 h, the optical density (OD) values at 562 nm were measured by the MTT method for the calculation of cell survival rates and the effect of PA on the survival rate of normal SH-SY5Y cells was observed. The cell treatment method was the same as above in the observation of the effect of PA on the survival rate of SH-SY5Y cells induced by Aβ25-35 in the different PA-treated groups, in which a normal control group, Aβ25-35-induced injury model group, and 5, 10, 20, and 40 μg/mL PA groups were set up. After culture for 2 h, Aβ25-35 at a final concentration of 25 μmol/L was added to the wells in the Aβ25-35-induced injury model group, and 5, 10, 20, and 40 μg/mL PA groups, respectively, and the cells were cultured continuously for 24 h. Then, the effect of PA on the survival rate of SH-SY5Y cells induced by Aβ25-35 was observed.
Measurement of Oxidative Stress and Antioxidant Response
SH-SY5Y cells in their logarithmic growth phase were seeded at a density of 2 × 105 cells/well in 6-well culture plates. A normal control group, Aβ25-35-induced injury group, and 5, 10, 20, and 40 μg/mL PA groups were set up. After culture for 2 h, to the wells of the Aβ25-35-induced group, and 5, 10, 20, and 40 μg/mL PA groups, Aβ25-35 was added at a final concentration of 25 μmol/L, and the cells were cultured continuously for 24 h. Then, the culture liquid of SH-SY5Y cells was centrifuged at 4000 rpm for 10 min to obtain the supernatant, and the LDH activity in the supernatant was detected according to the methods proposed by the kit’s instructions. After adding 100 μL of lysis buffer to the wells of all groups, the cells in each group were fully lysed on ice for 30 min. The lysates were then centrifuged at 4000 rpm for 10 min and the supernatant taken. SOD, GSH-Px, and CAT activities and MDA content in the SH-SY5Y cells were measured according to the methods proposed in the kit’s instructions.
Detection of Apoptosis by Flow Cytometry
Flow cytometry was performed following previous protocols. 43 Briefly, SH-SY5Y cells were cultured, grouped and treated according to the above procedures. The cells were digested with trypsin without ethylene diamine tetraacetic acid, and centrifuged at 4000 rpm for 10 min to collect the cells. The cells (1 × 105) were resuspended in phosphate buffered saline (PBS) and annexin V-fluorescein isothiocyanate was added to the cell suspension in a dark place at room temperature, and the cells were cultured for 10 min; then propidium iodide was added to the cell suspension under the same conditions and the cells were cultured for 5 min; the apoptosis rate of the cells in each group was detected by flow cytometry.
RNA Isolation and Real-Time qPCR
Total RNA was extracted from SH-SY5Y cells using the Trizol method. The ImProm-II reverse transcription system (Promega) was applied to generate the first strand cDNA. The primers for qPCR were designed according to the gene sequences published by Primer Bank. Primers used for amplifying the Caspase-3 were 5'-CAGCTCATACCTGTGGCTGT' (forward) and 5'-TT CCCTGAGGTTTGCTGCAT-3 (reverse), Bax were 5'-CCCAGAGGCGGGGTTTCA-3' (forward) and 5'-GGAAAAAGACCTCTCGGGGG' (reverse), Bcl-2 were 5'-CATGTGTGTGGAGAGCGTCA-3' (forward) and 5'-CACTTGTGGCTCAGATAGGCA-3' (reverse), and β-actin were 5'-CTTCGCGGGCGACGAT-3' (forward) and 5'-CCACATAGGAATCCTTCTGACC-3' (reverse). The real-time qPCR was carried out by the routine method 44 and based on the amplification curve of qPCR, the relative expression of the target genes was calculated by the 2−ΔΔCt method.
Analysis of Protein Expression by Western Blot
Western blot analysis was performed following previous protocols. 45 Briefly, the cells from the different groups were added to 100 μL lysis buffer and fully lysed on ice for 30 min, then centrifuged at 4000 rpm for 10 min to obtain the supernatant. The total protein concentration was determined by the bicinchoninic acid method. The target proteins in 10 μL of the supernatant were separated by polyacrylamide gel electrophoresis, then transferred onto polyvinylidene fluoride membranes and blocked with the blocking buffer containing 5% skim milk powder for 1 h. After washing with PBS, primary antibodies (1:1000 dilution) were added to the membranes, which were incubated at room temperature for 2 h. Then, the membranes were washed with PBS and the second antibody (1:5000 dilution) was added to them; the membranes were kept at room temperature for 1 h, and visualized using an enhanced chemiluminescence (ECL) kit according to the manufacturer's instructions.
Statistical Analysis
Results are shown as the mean ± standard deviation (SD) of 5 separate experiments and the statistical significance analysis was performed using SPSS 16.0 (SPSS Inc) with 1-way analysis of variance and Tukey's post hoc test. P < .05 was considered to be a statistically significant difference.
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of People's Hospital of Suzhou New District, China and approved by the Administration Committee of Experimental Animals, Jiangsu Province, China.
Footnotes
Ethical Approval
This study was approved by the Administration Committee of Experimental Animals, Jiangsu Province, China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the project of Educational Department of Jilin Province (JJKH20210055KJ and JJKH20210059KJ) and project of Suzhou New District.
