Abstract
Introduction
Mume Flos, also known as white plum blossom, is the dried flower bud of the Rosaceae plant plum (Prunus Mume (Sieb.) Sieb. et Zucc.). It is a common Qi-regulating medicine used clinically in traditional Chinese medicine and has the effects of soothing the liver, harmonizing, resolving phlegm, and dispersing knots. 1 Mume Flos is described in the “Compendium of Materia Medica” as “clearing the mind,” in the “Supplement to the Compendium of Materia Medica” as having the ability to “soothe the mind and calm the soul,” and in other ancient texts as having the ability to treat madness when combined with pig's heart blood. This suggests that Mume Flos has the effect of regulating neurological diseases. According to several studies,2-6 Mume Flos contains a range of chemical components, including polyphenols, organic acids, and volatile compounds. The primary ingredients of Mume Flos are polyphenols, specifically hyperoside and chlorogenic acid.7,8 Modern pharmacological studies have shown that the extract of Mume Flos and the total flavonoids of Mume Flos have antidepressant effects and can significantly improve nerve damage.9-12 Wang et al 4 found that the compounds isolated from the ethyl acetate part of Mume Flos had a protective effect on corticosterone-induced SH-SY5Y cells and could significantly improve the survival rate of SH-SY5Y cells.
With an aging population and increasing work and life stress, the incidence of neurological disorders such as Alzheimer disease (AD), depression, anxiety disorders, and Parkinson disease is increasing year by year and is now one of the leading causes of disability and death in the population, 13 while the complex pathogenesis of these neurological disorders has not yet been elucidated, as well as the lack of effective treatments. 14 Therefore, research on the treatment and prevention of neurological disorders has significant clinical therapeutic and social value.
Caenorhabditis elegans (C. elegans), as a model organism, has various advantages over other animal models such as rats and mice, such as easy culture, low cost, short generation cycle, and clear cell lineage, it has been commonly used in more complex diseases such as neurodegenerative diseases,15,16 the most widely used C. elegans is N2 wild-type C. elegans, and the established AD transgenic C. elegans model GMC101 and CL4176 can induce Aβ1-42 gene expression at 25 °C, resulting in Aβ aggregation in body wall cells to simulate Aβ aggregation in the brain of AD patients.
It was found that the ethanol extract of Mume Flos has a certain protective effect on the SH-SY5Y nerve cell model injured by Aβ1-42. 17 In order to further study the anti-dementia activity of alcohol extract of Mume Flos, the n-butanol extraction site of methanol extract of Mume Flos (MFB) was selected as the research object, and C. elegans (N2 and GMC101) was selected as the model to study the neuroprotective effect of MFB. The MFB components were characterized and identified by High Resolution Mass Spectrometry, and the identified components were analyzed by a network pharmacology method to further clarify the substance basis of MFB and to lay the foundation for further studies on the neuroprotective effects of Mume Flos.
Results and Discussion
Results of Protective Effect of MFB on AD C elegans Model
Effects of MFB on paralysis
To evaluate the effect of MFB on Aβ protein, this study used transgenic C. elegans GMC101 to conduct a paralysis assay. The results showed that, compared with the blank group, the paralysis rate of C. elegans in each concentration group was reduced (Figure 1A), the effect of reducing the paralysis rate of C. elegans were very significant when the treatment concentration was 0.25 mg/mL (P < .001). The paralysis rate of C. elegans in the blank control group reached 50% at 110 h, while the paralysis rate of C. elegans in the 0.25 mg/mL administration group was at 150 h. However, the effect of MFB in delaying C. elegans paralysis decreased when the concentration of the administered drug continued to increase, and the paralysis rate reached 50% at 140 h and 90 h in the 0.5 mg/mL and 1 mg/mL administration groups, respectively, in which the 1 mg/mL administration group showed an inhibition instead when compared with the blank control group; therefore, the concentrations of 0.0625, 0.125, and 0.25 mg/mL were selected for the present study for subsequent experiments.

The effect of MFB on Caenorhabditis elegans. (A) Effects of different concentrations of MFB on paralysis of transgenic C. elegans GMC101; (B) Effects of different concentrations of MFB on the life span of transgenic C. elegans GMC101; (C) Effects of different concentrations of MFB on juglone-induced oxidative damage in C. elegans N2; (D) Effect of different concentrations of MFB on heat stress damage of C. elegans N2. *P < .05, **P < .01, ***P < .001, ****P < .0001.
Effect of MFB on life span
In order to investigate whether MFB affects the life span of C. elegans, different concentrations of MFB were used to treat C. elegans for life span determination in this study. As a result, different concentrations of MFB administered for 1 to 7 days did not adversely affect the survival of C. elegans, and the life span of C. elegans was significantly prolonged in the groups administered at all concentrations compared with the blank control group (P < .001). The results indicated that MFB had no toxic effect on C. elegans in this concentration range and had the effect of prolonging the life span of C. elegans (Figure 1B).
Effects of MFB on oxidative stress injury in C elegans
Oxidative stress injury is an important factor in the occurrence of AD. On the one hand, the excessive production of reactive oxygen species can lead to cell damage or apoptosis. On the other hand, the oxidative stress response is closely related to the accumulation of Aβ. Oxidants and oxidation products can increase the expression of amyloid precursor protein (APP), leading to the aggregation of Aβ, 18 which in turn leads to apoptosis. 19 In this study, juglone was used as an oxidant to investigate the antioxidant capacity of MFB, compared with the other groups, the administration group of each concentration prolonged the life span of C. elegans, indicating that the administration group of each concentration could reduce the oxidative damage caused by juglone, suggesting that MFB has a certain antioxidant capacity (Figure 1C).
Effect of MFB on nematode thermal damage
In the heat stress response, increased levels of the heat shock protein HSP70 can reduce the aggregation and toxicity of tau and Aβ, 20 and it has also been found that increasing the expression of related genes can improve C. elegans resistance to heat stress and prolong life span,21,22 so this study used a heat stress test to observe whether each administration group of C. elegans can improve heat stress ability. The results showed that the C. elegans life span was prolonged in all concentration administration groups compared to the blank control group (P < .01) (Figure 1D), indicating that MFB could alleviate the damage caused by heat stress.
MFB Component Identification Results
The MFB sample solution was analyzed by UHPLC-Q-Orbitrap-MS/MS according to the chromatographic and mass spectrometric conditions, a total of 45 compounds were identified and inferred by analyzing the secondary mass spectrometric fragments using Xcalibur software (Thermo Fisher Scientific) and Compound Discoverer 3.1 software (Thermo Fisher Scientific) and in combination with literature reports,23,24 including 23 flavonoid components, 11 phenylpropanoid components, 8 amino acid components, and 3 other components (Figure 2, Table 1).

Total ion chromatograms of UHPLC-Q-orbitrap-MS/MS. (A) pos ion pattern of MFB; (B) neg ion pattern of MFB.
UHPLC-Q-Orbitrap-MS/MS Tentative Identification Analysis of Chemical Composition of MFB.

Abbreviations: tR, retention time; FA, formic acid.
Results of Network Pharmacological Analysis
MFB active ingredient targets
The 45 compounds tentatively identified by UHPLC-Q-Orbitrap-MS/MS were initially screened by the database to obtain 39 compounds, and a total of 280 targets were obtained by screening the 39 compounds against the targets retrieved from different databases and removing duplicates.
Screening results of differentially expressed genes
Gene chips GSE122063 and GSE76826 were screened from the GEO database with P < .05, |log2FC|>1 as the screening condition, a total of 1391 DEGs were screened in GSE122063, of which 885 were downregulated genes and 506 were upregulated genes; and a total of 336 DEGs were screened in GSE76826, of which 192 were downregulated genes and 144 were upregulated genes. The differential gene volcano maps of the 2 GEO GeneChips and the heat maps of the top 20 differential genes are shown in Figure 3 and Figure 4.
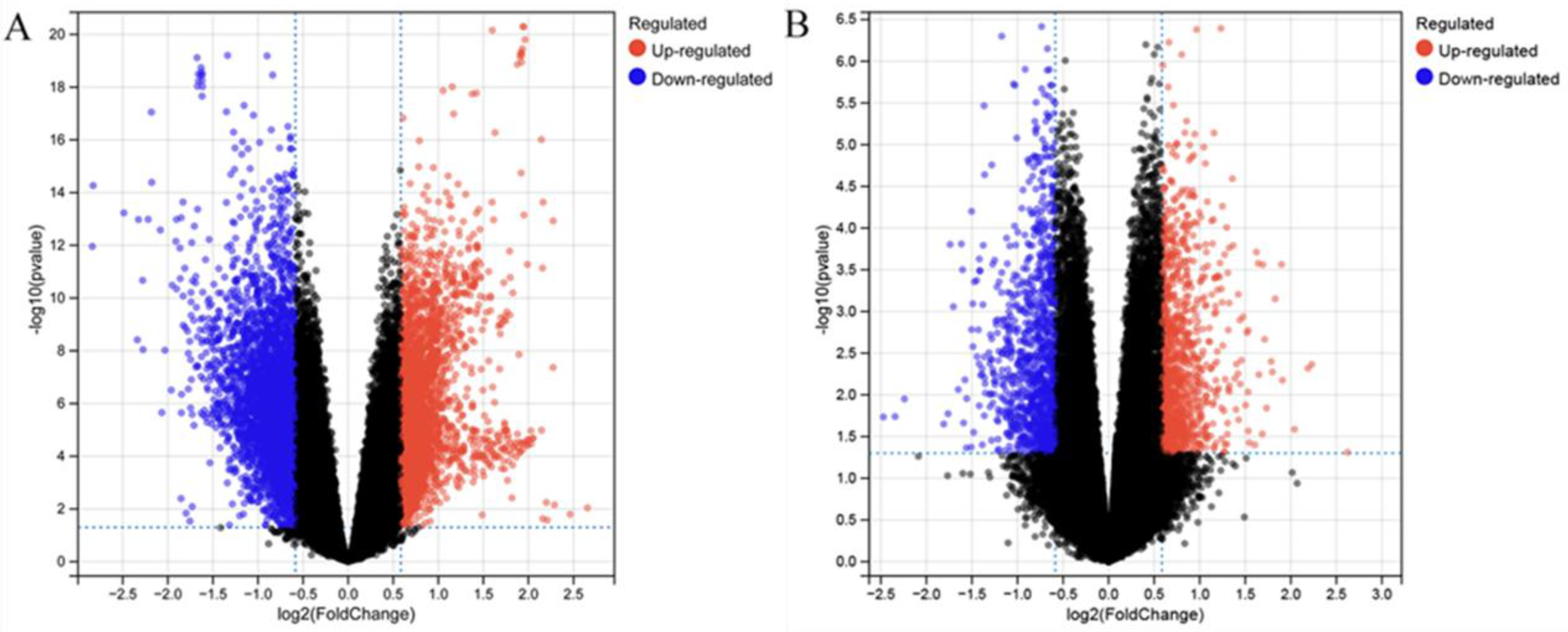
Volcano map of DEGs in GEO GeneChip, red and blue color represent upregulated and downregulated genes, respectively, black color indicates no significant difference; (A)GSE122063; (B) GSE76826.

Heatmap of top 20 upregulated, downregulated DEGs in GEO GeneChips; (A) GSE122063; (B) GSE76826.
Disease-related targets
Relevant targets were collected by searching GeneCard, Online Mendelian Inheritance in Man (OMIM), pharmgkb, and drugbank databases, and 2948 disease targets were obtained after screening, merging, and removing duplicates. After combining the targets predicted by the gene chip with those of the above disease databases and removing duplicates, a total of 4193 relevant targets were obtained.
MFB active ingredient-disease co-acting targets
The screened 280 potential active ingredient action targets and 4193 neurological disease-related targets were imported into online Venny 2.1.0, 130 potential active ingredient and disease co-action targets were obtained (Figure 5).
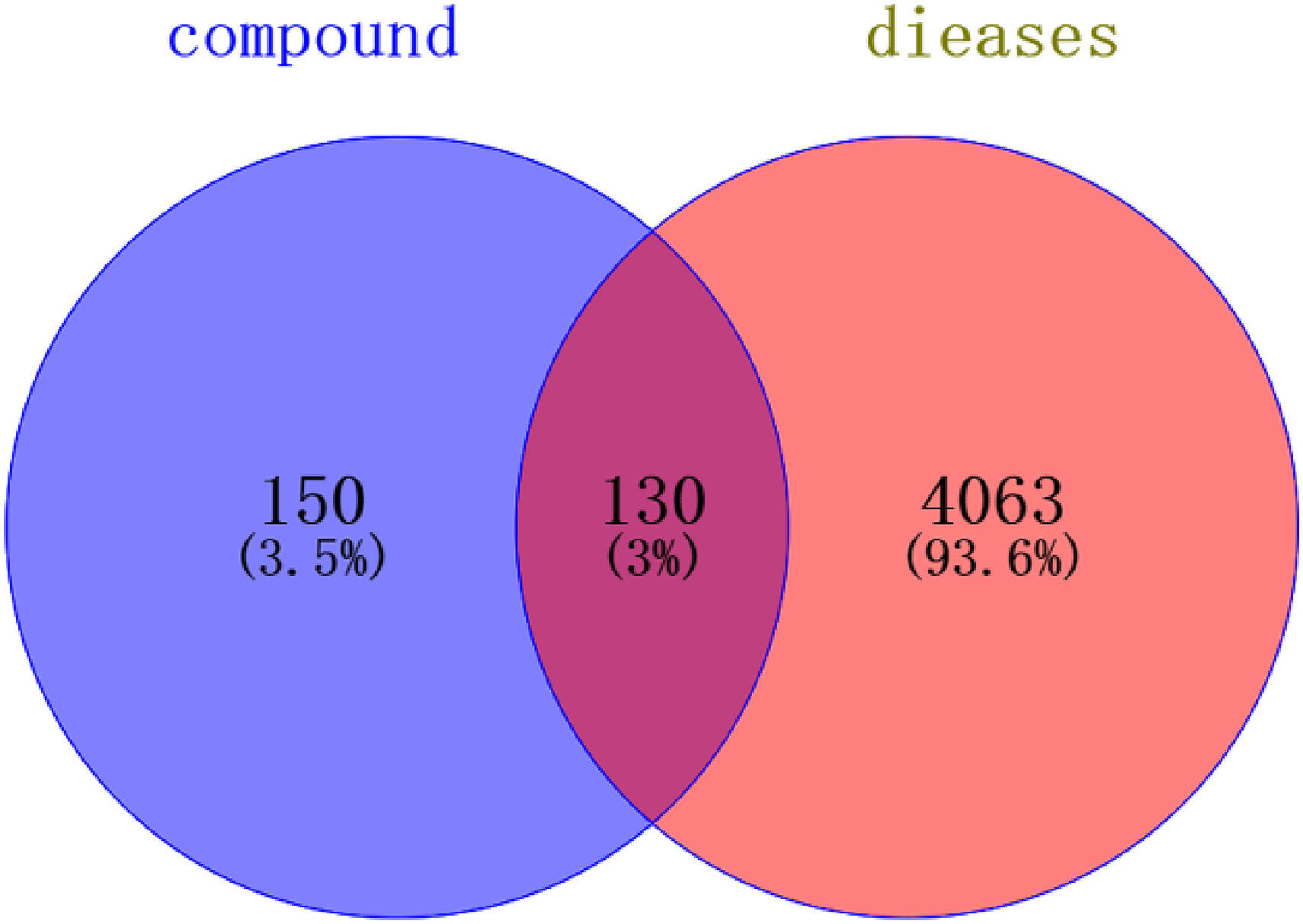
Intersection distribution of components and disease targets.
MFB—active ingredient—associated target—disease network diagram construction and analysis
The potential active ingredients of MFB and 130 co-acting targets of “active components of MFB-disease” were imported into Cytoscape 3.9.1, and the interaction network diagram of “MFB-ingredients-targets-disease” was drawn. The network consisted of 171 nodes and 684 edges, and the results of the topological analysis showed that 32 of these potential active ingredients had 10 or more targets associated with neuroprotective effects. The top 5 compounds in descending order according to degree values were adenosine, rutin, neochlorogenic acid, chlorogenic acid, and hyperoside, the degree values were 33, 28, 24, 23, and 19 in order, respectively (Figure 6, Table S1).

“MFB-ingredients-targets-disease” network.
PPI Network
The 130 co-acting targets obtained by mapping the potential active ingredient targets to the disease targets were imported into String database for PPI network analysis, and the PPI network information was imported into Cytoscape 3.9.1 for network topology analysis, and the PPI network diagram was made according to the degree (Figure 7A), where the nodes represent each protein, and the lines between nodes and nodes represent the interaction relationship between proteins. The darker the nodes, the greater the degree value. The core targets were screened according to degree, Betweenness Centrality (BC) and Closeness Centrality (CC), and those with degree, BC, CC above the median were taken as the core targets; 27 core targets were obtained after 2 screenings, and the core targets were ranked according to the degree value, the top 5 were GAPDH, TNF, IL6, INS, and TP53, in order (Figure 7B, Table 2).

PPI network. (A) PPI protein interaction network; (B) Core targets of MFB for neurological diseases.
Core Targets Identified in the PPI Network.

Gene Ontology and Kyoto Encyclopedia of Genes and Genomes Analysis
Gene ontology (GO) is one of the most widely used biological ontologies for functional annotation of gene products, including 3 separate ontologies for molecular functions, cellular components, and biological processes, which provide information on the various biochemical activities of gene products, the various processes in which they may be involved, and the places in the cell where genes may be active. 36 The Kyoto Encyclopedia of Genes and Genomes (KEGG) is a comprehensive database that integrates data on biological cellular processes, genetic information, human diseases, and metabolism. It is widely used for genomic data analysis and pathophysiological mechanism research. 37 Through GO and KEGG analysis of 130 co-acting targets, 1819 GO entries and 201 KEGG pathways were obtained (P < .01), 1533 biological processes, 112 cellular components, and 174 molecular functions were included in the GO entries, and the top 10 entries were selected for histogram plotting according to P values, respectively (Figure 8A). Biological processes are significantly enriched for “ cellular response to nitrogen compound, response to hormone, modulation of chemical synaptic transmission, regulation of trans-synaptic signaling”; cellular components are significantly enriched for “membrane raft, membrane microdomain, synaptic membrane”; and molecular functions are significantly enriched for “endopeptidase activity, oxidoreductase activity, postsynaptic neurotransmitter receptor activity, neurotransmitter receptor activity.” The top 20 KEGG pathways were screened according to their P values for visual analysis, and bubble plots were drawn (Figure 8B, Table S2), mainly involving cancer pathways, lipid and atherosclerosis pathways, neurodegeneration pathways, AD pathways, IL-17 signaling pathway, and MAPK signaling pathway.

(A) Gene ontology (GO) biological function analysis; (B) Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis.
Effect of Active Ingredients on C elegans Paralysis
Based on the results of network pharmacology, the neuroprotective effects of rutin, neochlorogenic acid, and chlorogenic acid were verified using CL4176 transgenic C. elegans, which began to express the Aβ gene in the body wall at 25 °C, and with the aggregation of Aβ proteins, it resulted in the C. elegans to exhibit a rigid paralytic behavior. Therefore, we analyzed the effects of rutin, neochlorogenic acid, and chlorogenic acid on the paralysis rate of CL4176 C. elegans, and the results showed that different concentrations of rutin, neochlorogenic acid, and chlorogenic acid delayed the paralysis rate of CL4176 C. elegans. Compared with the control group, the time for CL4176 C. elegans paralysis rate to reach 50% was prolonged by 16 h, 6 h, and 8 h after the treatment of 150μM rutin, neochlorogenic acid, and chlorogenic acid, respectively (Figure 9).

The effect of active ingredients on C. elegans paralysis. (A) Effect of different concentrations of rutin on paralysis in CL4176; (B) Effect of different concentrations of neochlorogenic acid on paralysis in CL4176; (C) Effect of different concentrations of chlorogenic acid on paralysis in CL4176.
Thioflavin T Fluorescence Staining of CL4176 C elegans
In order to determine whether the effect of rutin and other active ingredients on the paralytic effect of CL4176 C. elegans was related to the expression of Aβ gene and the aggregation of Aβ in the C. elegans, in this study, we used Thioflavine T staining to determine the aggregation of Aβ in the body of CL4176 C. elegans after the treatment of rutin, neochlorogenic acid, and chlorogenic acid, and the results are as shown in Figure 10. The control group had the largest number of C. elegans Aβ deposition plaques. There was a significant decrease in Aβ deposition in the C. elegans after the administration of drug intervention, which indicated that rutin, neochlorogenic acid, and chlorogenic acid could play a neuroprotective role by decreasing the level of Aβ expression and Aβ deposition in the C. elegans of CL4176.

Effect of active ingredient treatment on Aβ accumulation. (A) Thioflavin T staining of transgenic C. elegans in the control group; (B) Thioflavin T staining of transgenic C. elegans in the rutin administration group; (C) Thioflavin T staining of transgenic C. elegans in the neochlorogenic acid administration group; (D) Thioflavin T staining of transgenic C. elegans in the chlorogenic acid administration group.
Discussion
Caenorhabditis elegans is widely used as a model organism for studying neurodegenerative diseases, and among them, the transgenic C. elegans GMC101, which overexpresses Aβ protein under high temperature induction, is an important model used in AD as well as Aβ protein toxicity studies.38,39 In related studies using GMC101 as a model, aqueous extracts of Salvia miltiorrhiza 40 and flavonoid extracts of date palm 41 were found to inhibit Aβ aggregation and reduce Aβ toxicity, suggesting that natural medicines as well as natural medicine extracts have the potential to reduce Aβ toxicity and treat AD. In this study, GMC101 was selected as an AD model to explore the anti-AD activity of MFB. The results showed that MFB slowed down the paralysis rate of C. elegans and prolonged the life span of C. elegans, which may be related to the reduction of Aβ protein toxicity. In the oxidative stress and heat stress test, MFB slowed down the death of C. elegans caused by stress injury. It is speculated that MFB can alleviate stress injury, have the effect of antioxidation, and enhance the ability of antistress.
To clarify the pharmacodynamic material basis and mechanism of action of MFB, this study used UHPLC-Q-Orbitrap-MS/MS technology to identify its ingredients and combined network pharmacology to predict the active ingredients and mechanism of action.
A total of 45 compounds were tentatively identified from the MFB, mainly polyphenols. A network pharmacological analysis revealed that 39 of these compounds could act on targets related to neurological diseases, among which rutin, neochlorogenic acid, chlorogenic acid, and hyperoside may be the core components that exert neuroprotective effects. It has been shown that rutin can exhibit antidepressant effects by decreasing antioxidant activity and acetylcholinesterase, 42 Sun et al 43 reported that rutin can regulate tau hyperphosphorylation, inhibit gliosis and neuroinflammation, reduce synaptic loss, and improve the cognitive ability of Alzheimer mice. Lim et al 44 found that chlorogenic acid reduces oxidative stress and thus exerts antidepressant effects by inhibiting MAOB in hippocampal astrocytes. In addition to this, there is a large body of literature reporting that rutin,45-47 chlorogenic acid, 48 neochlorogenic acid, 49 and hyperoside50,51 can exert therapeutic neuroprotective effects by reducing the toxicity of Aβ protein, inhibiting neuroinflammation and antioxidation. In this study, the neuroprotective effects of the active ingredients rutin, neochlorogenic acid, and chlorogenic acid obtained from network pharmacological screening were verified using a transgenic model of the C. elegans, as a result of which they all slowed down the rate of paralysis of the nematode and showed different degrees of anti-AD effects, which suggests that they may be the basis of the pharmacological substances for the neuroprotective effects exerted by MFB.
Network pharmacology analysis revealed 116 potential targets, among which GAPDH, TNF, IL6, INS, and TP53, may be the core targets for the neuroprotective effect of MFB, of which TNF, IL6, and INS are all inflammatory factors, and some studies have shown that neuroinflammation and the related pro-inflammatory factors TNF and IL6 can contribute to depression by affecting HPA axis function and reducing hippocampal nerves. 52 It has also been shown that lowering TNF-α levels can reduce Aβ plaque deposition and tua tangles in the brain.53,54 TP53 is involved in regulating apoptosis as a transcription factor 55 and can interact with tau oligomers. 56 GAPDH is a key enzyme in the process of glycolysis. It has been claimed that GAPDH can interact with neurodegenerative disease-related proteins such as APP to promote Aβ amyloid production, which can also further lead to mitochondrial dysfunction and neuronal cell death, ultimately promoting the formation of AD. 57 Therefore, MFB may exert neuroprotective effects by regulating targets such as GAPDH, TNF, IL6, INS, and TP53; reducing inflammatory responses; decreasing Aβ production; reducing neuronal apoptosis; and regulating HPA axis function. Kyoto Encyclopedia of Genes and Genomes pathway analysis revealed that MFB exerts neuroprotective effects mainly involving pathways of neurodegeneration, AD, IL-17 signaling pathway, MAPK signaling pathway, cancer pathways, and lipid and atherosclerotic pathways. Studies have shown that IL-17, which can act directly on neurons and indirectly on glial cells to cause apoptosis, can also affect synaptic plasticity and be involved in the development of AD 58 ; in addition, it has been reported in the literature that dysregulation of MAPK signaling pathway is associated with AD, and inhibition of MAPK overactivation can significantly reduce Aβ deposition and improve cognitive function. 59 In summary, abnormalities in core targets such as GAPDH, TNF, IL6, and MAPK signaling pathway obtained from network pharmacology can affect Aβ amyloid plaque deposition, in order to further clarify whether active ingredients such as rutin plays a role by affecting Aβ deposition, in this study, different groups of nematodes were stained with Thioflavin T staining to observe the deposition of Aβ in the body of the nematodes, and the results showed that the deposition of Aβ in the body of the nematodes was significantly reduced by the administration of the drug, which suggests that rutin and other active ingredients may play a role in decreasing the expression of Aβ.
Conclusion
In conclusion, in this study, we revealed the neuroprotective effects of MFB using the C. elegans model of AD; 45 chemical components were tentatively identified by UHPLC-Q-Orbitrap-MS/MS technology, among which rutin, neochlorogenic acid, and chlorogenic acid were the main active components of MFB exerting neuroprotective effects, which could delay nematode paralysis and reduce Aβ deposition, and might target GAPDH, TNF, IL6, INS, and TP53 to regulate AD, neurodegeneration, IL-17, and MAPK signaling pathways, playing an important role in ameliorating neurological diseases. It reveals the potential mechanism of MFB in treating nervous system diseases through multicomponents, multitargets, and multichannels, which provide a theoretical basis for further experimental study on the mechanism of Mume Flos in treating nervous system diseases.
Materials and Methods
Instrument
Motic K-400C stereo microscope (Motic China Group Co., Ltd), HC-3018 high-speed centrifuge (Anhui Zhongke Zhongjia Scientific Instrument Co., Ltd), SW-CJ-1FD single person clean vertical console (Shanghai bosun Industrial Co., Ltd), HPX-9082MBE electrothermal constant temperature incubator (Shanghai bosun Industrial Co., Ltd), YXQ-SG46-280S portable steam sterilizer (Shanghai bosun Industrial Co., Ltd), BS-300+ electronic balance (Shanghai Yousheng weighing instrument Co., Ltd), Milli-Q integrated ultrapure water system (Merck Millie), BSP-150 biochemical incubator (Shandong Boke Biological Industry Co., Ltd), YCD-El259 Meiling refrigerator (Zhongke Meiling Cryogenic Technology Co., Ltd), and Thermo Vanquish Ultra High Performance Liquid Tandem Thermo ScientificTM Orbitrap Exploris 120 Electrostatic Field Orbitrap High Resolution Mass Spectrometer (Thermo Fisher Scientific, Inc.), Inverted Fluorescence Microscope DMi8 (Leica Camera AG).
Materials
Tryptone (Shanghai Bioengineering Co., Ltd, batch number: D406BA0046), Agar powder (Shanghai Bioengineering Co., Ltd, batch number: E418BA0027), Yeast powder (Shanghai Bioengineering Co., Ltd, batch number: E418BA0027), sodium chloride (Sinopharm Group Chemical Reagent Co., Ltd, batch number: C616BA0015), 5-fluorouracil (Shanghai Yuanye Biological Co., Ltd, batch number: Z08S7S20661, purity 98%), cholesterol (Sinopharm Group Chemical Reagent Co., Ltd, batch number: 69008214), magnesium sulfate heptahydrate (Sinopharm Group Chemical Reagent Co., Ltd, batch number: 20160406, analytical grade), potassium dihydrogen phosphate (Sinopharm Group Chemical Reagent Co., Ltd, batch number: 20151201, analytical grade), streptomycin sulfate (Shanghai Bioengineering Co., Ltd, batch number: B326BA3416), juglone (Shanghai Yuanye Biological Co., Ltd, batch number: M19J10E93478, purity >98%), chlorogenic acid (Chengdu Lemeitian Pharmaceutical Technology Co., Ltd, batch number: DST200520-021, purity ≥98%), rutin (Chengdu Lemeitian Pharmaceutical Technology Co., Ltd, batch number: DSTDL001701, purity ≥98%), hyperoside (Chengdu Lemeitian Pharmaceutical Technology Co., Ltd, batch number: DST200628-023, purity ≥98%), acetonitrile, formic acid, methanol (All were purchased from Fisher Company of the United States, chromatographically pure), Thioflavine T (Aladdin), TritonX-100 (Beijing Solarbio Science & Technology Co., Ltd) Tris-HCl (Beijing Solarbio Science & Technology Co., Ltd), and β-mercaptoethanol (Shanghai Macklin Biochemical Technology Co., Ltd).
Caenorhabditis elegans strains and culture conditions
The nematode strains used in this study were N2, GMC101 [unc-54p::A-beta-1-42::unc-54 3′-UTR + mtl-2p::GFP], CL4176 (smg-1(cc546ts);dvIs27) and nematodes were obtained from the Caenorhabditis Genetics Center. All nematodes were cultured on standard nematode growth medium with Escherichia coli OP50 as food, N2 and GMC101 were incubated at 20 °C and CL4176 at 16 °C.
Methods
Solution Preparation
Take 50.00 g of crude powder of Mume Flos, add 1500 ml of 50% methanol and extract with ultrasound for 30 min, concentrate until there is no alcohol taste, extract with petroleum ether, ethyl acetate and n-butanol in turn to obtain n-butanol part solution of Mume Flos, and concentrate to get n-butanol part extract 319.00 mg.
Weigh an appropriate amount of MFB extract and dissolve it in water to prepare a 2 mg/mL stock solution, filter it with a 0.22-μm microporous membrane, and dilute it with E. coli to 1 mg/mL, 0.5 mg/mL, 0.25 mg/mL, 0.1250 mg/mL, and 0.0625 mg/mL as dosing solution.
Take an appropriate amount of MFB extract and dissolve it with 50% methanol ultrasonically to 0.1 mg/mL, then filter it with a 0.22-μm microporous membrane to obtain the test solution for component analysis.
The Protective Effect of MFB on AD C elegans Model
Paralysis assay
The transgenic C. elegans GMC101 in the spawning stage were picked for synchronization treatment, cultured at 20 °C for 12 h, and then divided into administration groups of different concentrations and blank groups, incubated at 20 °C for 24 h, and then warmed up to 25 °C to induce paralysis, count the number of paralyzed C. elegans every 10 h, transfer the plate once a day until all C. elegans are paralyzed (touch the C. elegans lightly with a picking needle, if the worms keep their heads moving but their bodies do not move, it is judged as paralysis).
Life Span Assay
The transgenic C. elegans GMC101 in the spawning stage were picked for synchronization and incubated at 20 °C for 12 h, and then divided into administration groups of different concentrations and blank groups, incubated at 20 °C, and the growth of C. elegans was observed daily and the number of dead, lost and surviving C. elegans were recorded until all died.
Oxidative Stress Test
The synchronized N2 C. elegans were divided into administration groups of different concentrations and blank groups. After culturing at 20 °C for 3 d, they were transferred to a medium containing juglone (200 μM), and the survival of C. elegans was observed every 1 h until all C. elegans died.
Heat Stress Test
The synchronized N2 C. elegans were divided into administration groups of different concentrations and blank groups. After incubation at 20 °C for 3 d, the incubator temperature was raised to 35 °C, and the survival of C. elegans was observed every 1 h until all died.
Statistical Methods
Graphpad Prism 8 software was used for statistical analysis. The measurement data were expressed as mean ± standard deviation. ANOVA analysis was used for comparison between groups. P < .05 indicates that the difference is statistically significant.
MFB Components Analysis
Chromatographic conditions
Thermo Syncronis C18 chromatographic column (100 mm × 2.1 mm 1.7 μm), flow rate: 0.2 mL/min, column temperature: 30 °C, mobile phase: 0.1% formic acid aqueous solution (A) – acetonitrile (B), elution gradient: 0 to 5 min, 10% → 15% B; 5 to 15 min, 15% → 20% B; 15 to 20 min, 20% → 25% B; 20 to 21 min, 25% → 90% B; 21 to 25 min, 90% → 90% B; 25 to 26 min, 90% → 10% B; 26 to 31 min, 10% B; injection volume: 2 μL.
Mass spectrometry conditions
Ion source type: HESI; scan mode: Full MS/dd MS2; Positive ion electrospray voltage: 3.5 kV, negative ion electrospray voltage: 3.0 kV; Sheath gas flow rate: 45 Arb, aux gas flow rate: 7 Arb; Ion transfer tube temperature: 320 °C; Vaporizer temperature: 320 °C; Scan range: m/z 100 to 1500; Full MS resolution: 60,000, dd-MS2 resolution: 15,000; and HCD Collision Energies (%): 20, 40, 60.
Components Analysis
The reference sample and the test sample solution were taken for injection analysis under the conditions of liquid chromatography, and mass spectrometry and the total ion flow chromatograms under the positive and negative ion mode were obtained. combine the PubChem, ChemicalBook and other databases, and reference literature, establish the chemical composition information database. The mass spectrometry data processing was implemented using Xcalibur software (Thermo Fisher Scientific) and Compound Discoverer 3.1 software (Thermo Fisher Scientific). LC-MS data were imported into Compound Discoverer 3.3 software to quickly match the chemical components by using mzCloud, ChemSpider, and Mass Lists. The excimer ion peaks were identified by primary mass spectrometry, the molecular formulae were calculated by Xcalibur fitting, and the structures of chemical components were inferred based on the secondary mass spectrometry cleavage fragment information.
Network Pharmacology Analysis
Collection and treatment of potential active ingredient targets of MFB
Obtain the CAS numbers and SMILES files of potential active ingredients tentatively identified by UHPLC-Q-Orbitrap-MS/MS from the PubChem (https://pubchem.ncbi.nlm.nih.gov/) database, and import the CAS numbers and SMILES files into TCMSP (https://old.tcmsp-e.com) and SwissTargetPrediction (http://www.swisstargetprediction.ch/) databases to search and download potential active ingredient targets, using the Uniprot database (https://www.uniprot.org/) maps the target information of potential active ingredients to human gene names.
Gene chip and screening of differentially expressed genes
Searching related gene expression profile data in GEO database (https://www.ncbi.nlm.nih.gov/geo/) with “Alzheimer's Disease, Depression” as key words, and setting “Organism” as “Homo sapiens” to find eligible gene chip data sets, DEGs is screened by using the GEO2R function of GEO database, and the screening conditions are P < .05, |log2FC|>1, and volcanic maps and thermal maps are drawn for visualization.
Collection and processing of targets related to neurological diseases
Enter the keywords “Alzheimer's disease, Depression” in the genecards database (https://www.genecards.org/), the OMIM database (http://www.omim.org/), pharmgkb database (https://www.pharmgkb.org/), and the drugbank database (https://go.drugbank.com/) to obtain relevant targets, and screen, merge, and remove duplicate the targets obtained from the 4 databases to obtain relevant targets of AD and depression, and the targets obtained from the 4 databases were merged with the genes screened by GEO chip to remove duplicates to obtain AD and depression-related targets.
Acquisition of co-acting targets of “Potential Active ingredients of MFB-Disease”
Import the potential active ingredient targets and the neurological disease-related targets nto online Venny 2.1.0 (https://bioinfogp.cnb.csic.es/tools/venny/), the intersection part is the common target of “potential active ingredients of MFB-disease.”
Construction and analysis of the network diagram of “MFB—Active Ingredients—Related Targets—Disease”
Import the data files of potential active ingredients and common action targets into Cytoscape3.9.1, build the network diagram of “MFB—active ingredients—related targets—disease,” and use the “Analyze Network” function to conduct topology analysis.
PPI Network Construction
The co-interacting targets of “MFB-active ingredient-disease” were imported into the String database (https://string-db.org/) to obtain the protein interaction data file, which was imported into the Cytoscape 3.9.1 software to obtain the PPI network diagram, and topology analysis was performed to select the core targets based on the degree value, BC, and CC to select core targets.
Gene Ontology Functional Annotation and KEGG Pathway Enrichment Analysis
The “potential active ingredient-disease” co-interacting targets were imported into the Metascape database (https://metascape.org/), and the “sapiens” species were selected for GO functional annotation and KEGG pathway enrichment analysis.
Effect of Active Ingredients on C elegans Paralysis
Caenorhabditis elegans transgenic strain CL4176 in the spawning stage were picked for synchronization and incubated at 16 °C for 48 h. After that, the C. elegans were divided into administration groups of different concentrations and blank groups, and the temperature was raised to 25 °C to induce paralysis. After 36 h of culture, the paralysis of C. elegans was observed, and the number of paralyzed C. elegans was counted every 2 h.
Thioflavin T Fluorescence Staining of CL4176 C elegans
After synchronization, the transgenic C. elegans of CL4176 were cultured at 16 °C for 48 h, and the C. elegans were picked to each group of administration plates and blank control plates, and cultured at 25 °C for 36. The C. elegans in each group were washed into 1.5 mL centrifuge tubes with M9 buffer, fixed with 150 µL of 4% paraformaldehyde at 4 °C for 24 h, and then washed repeatedly with PBS buffer for 3 times. Add 150 µL of 5% β-mercaptoethanol, 125 mM Tris-HCl(pH = 7.4) and 1% TritonX-100, transfer to 37 °C and incubate for 24 h, rinse with PBS buffer for 3 times, discard the supernatant, stain with 0.125% Thioflavine T for 2 min, decolorize with 50% ethanol, rinse with PBS buffer, and make slices for observation.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241235749 - Supplemental material for Integrated UHPLC-Q-Orbitrap-MS/MS Method and Network Pharmacology for Exploring the Active Components and Potential Mechanisms of Neuroprotective Effect of the n-Butanol Part of Mume Flos
Supplemental material, sj-docx-1-npx-10.1177_1934578X241235749 for Integrated UHPLC-Q-Orbitrap-MS/MS Method and Network Pharmacology for Exploring the Active Components and Potential Mechanisms of Neuroprotective Effect of the n-Butanol Part of Mume Flos by Xue Wang, Hongsu Zhao, Mengying Wu, Yijie Gan, Shurong Zhang, Deling Wu, Jinmei Ou, Chuanshan Jin and Wei Zhang in Natural Product Communications
Footnotes
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Anhui Province (2008085QH394), and the Jiangsu Province Graduate Research Innovation Program in 2022 (KYCX22_2029).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
