Abstract
Objective
This study assesses the anti-inflammatory and therapeutic effects of Qianghuo Shengshi Tang (QHSST) on rheumatoid arthritis (RA) through network pharmacology and experimental validation and explore the underlying mechanisms.
Methods
Complete composition analysis of QHSST using UPLC-Q-Orbitrap-MS. And collection potential targets using relevant databases. Protein–protein interaction (PPI) networks were constructed and Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) enrichment analyses were conducted. Anti-inflammatory activity of QHSST was evaluated in RAW264.7 cells by assessing apoptosis, NO, interleukin (IL)-6, IL-10, and tumor necrosis factor-α (TNF-α). In rats with wind-cold-dampness paralysis-RA, efficacy of QHSST was confirmed by evaluating foot volumes, arthritis scoring index (AI), blood routine, rheumatoid factor (RF), anti-cyclic citrullinated peptide (CCP) antibody, and hematoxylin and eosin (H&E) staining and inflammatory factors. Enzyme-linked immunosorbent assay (ELISA) and immunohistochemistry were used to examine protein expression in the ankle joints to elucidate the mechanism of QHSST action.
Results
We identified 44 compounds using UPLC-Q-Orbitrap-MS. Additionally, network pharmacology revealed five active ingredients and seven key targets. GO and KEGG analyses highlighted the involvement of PI3K-Akt signaling and tyrosine kinase inhibitor resistance pathways. Cell studies showed that QHSST reduced apoptosis, NO, IL-6, and TNF-α levels, while increasing IL-10 level in the dexamethasone and 3.125 µg/mL groups, indicating its anti-inflammatory effects. Animal experiments revealed that QHSST and methotrexate significantly decreased AI scores, foot volumes, and blood routine. H&E staining revealed varying degrees of joint harm, accompanied by a decrease in RF, anti-CCP antibody, IL-6, and TNF-α levels, and an increase in IL-10 level, underscoring the efficacy of QHSST in treating RA. ELISA and immunohistochemistry suggest that QHSST may exerts its effects by modulating the EGFR/SRC/PI3 K/AKT/NF-κB pathway.
Conclusion
Our findings provide a scientific foundation for therapeutic benefits of QHSST in alleviating RA by confirming the anti-inflammatory and RA-relieving properties of the supplement.
Keywords
Work Flowchart

Introduction
Rheumatoid arthritis (RA) is an autoimmune disease characterized by chronic, symmetrical, and inflammatory features that are modulated by genetic, epigenetic, and environmental factors. 1 The hallmark of RA pathogenesis is the inflammation of joints, 2 which progresses from small to large joints and can lead to infection, osteoporosis, cancer, and respiratory and cardiovascular diseases. 3 RA also involves the destruction of articular bones and cartilage, as well as the weakening of tendons and ligaments. 4 The global prevalence of RA is five per 1000 individuals, with a morbidity rate of 0.5–1.0% in North America and Europe. Although RA can manifest at any age, it most frequently arises after the age of fifty. 5 The clinical diagnosis of RA has evolved to include diagnostic imaging and serological tests. A comprehensive assessment of the disease now includes specific markers such as RF and anti-CCP antibodies produced by B cell-activated plasma cells, which are crucial for serological diagnosis.6,7 Unfortunately, RA has no cure. Modern drug therapy remains the mainstay in treatment, which has led to substantial progress in achieving disease remission without the occurrence of joint deformities. 8 These drugs have great therapeutic benefits, however, there are adverse reactions, including stomach hemorrhage, osteoporosis, stomatitis, fatigue, and liver damage. 9 The effectiveness of traditional herbal remedies in decreasing side effects and alleviating RA symptoms has been demonstrated. Therefore, these remedies should be considered as a supplemental medical strategy. 10
Since the launch of “100 ancient classic prescriptions” in China, Traditional Chinese medicine (TCM) research has reached a climax, with the availability of the material basis of classic prescriptions, dosage form modification, and studies on efficacy, mechanisms of action, and other basic research transforming the available knowledge. We identified the classic formula Qianghuo Shengshi Tang (QHSST) in the Treatise on Internal and External Injuries for the treatment of RA, which is categorized as a “double syndrome,” according to TCM theory, and involves a lack of life energy in the body, along with the presence of wind, cold, dampness, and heat evils, which lead to obstruction of blood in the meridians. 11 QHSST has mostly been used in clinical studies, with a few laboratory studies. Consequently, we investigated its mechanism of action; however, as QHSST has many active substances and multiple targets of action, 12 the mechanism of its therapeutic effect on RA remains unclear. Network pharmacology techniques 13 were employed to predict the active components, core targets, and signaling pathways of QHSST in RA. Its therapeutic effect was then verified via a collagen-induced arthritis (CIA) model, and the mechanism of action was explored.
Materials, Reagents, and Animals
The following materials and instruments were used in this study: Notopterygii Rhizoma Et Radix (Anhui Yukang Traditional Chinese Medicine Drinking Tablets Co. Nanchong, Sichuan 220301); Angelicae Pubescentic Radix, Chuanxiong Rhizoma (Anhui Yukang Traditional Chinese Medicine Drinking Tablets Co. Nanchong, Sichuan 220501); Saposhnikoviae Radix (Anhui Youxin Pharmaceutical Co. Shijiazhuang, Hebei. 220901); Ligustici Rhizoma Et Radix (Anhui Youxin Pharmaceutical Co. Fushun, Liaoning 210401); Viticis Fructus, Glycyrrhzae Radix Et Rhizoma Prparata Cum Melle (Anhui Huadingtang Chinese Medicine Drinking Tablets Technology Co. Wuwei, Gansu 20220701); methotrexate (MTX; Shanghai Xinyi Pharmaceutical Co. National Drug Code H31020644); collagen type II (Shanghai Maokang Biotechnology Co. C875812); complete Fischer's adjuvant (Shanghai Haoran Biotechnology Co. F850325); Ulatan (Shanghai Haoran Biotechnology Co. M89996 - 500 G), 4% paraformaldehyde (Shanghai Maokang Biotechnology Co. MM1504); hematoxylin stain (Wuhan Sevier Biotechnology Co., Ltd G1004); eosin stain (Hefei Bomei Biotechnology Co. YE2080), RF (F3667-A); anti-CCP antibody (F1023-A), interleukin (IL)-6 (F3066-A), IL-10 (F3071-A); tumor necrosis factor (TNF)-α (F40565-A); EGFR (F3646-B); SRC (F21353-B); PI3 K (F3303-B); AKT (F21165-B); NF-κB (F8592-B) kits (Shanghai Kexing Trading Co., China); Toe Volumeter (Shandong Sentec Hydraulic Co.); automatic biochemical analyzer (Shenzhen Boxing Biotechnology Co.); tissue embedding machine (Changzhou Suburb Zhongwei Electronic Instrument Factory); RAW264.7 (iCell Bioscience Inc. Shanghai, China. iCell-m047); minimum essential medium (MEM, 11095080); fetal bovine serum (FBS, 10099141C); phosphate-buffered saline (PBS, 10010023); penicillin/streptomycin (P/S, 15070063) (Gibco BRL Gaithersburg, MD, USA); and cell counting kit 8 (CCK-8) (Elabscience Biotechnology Hubei, China. E-CK-A362).
Male Sprague–Dawley rats were used in the experiments. All animals were raised in a regulated environment at 22 ± 2 °C and a humidity of 55 ± 2%. All animal procedures were approved by the Animal Ethics Committee of Qinghai University, and the provisions of the General Requirements for Laboratory Animals (GB/T 35823-2018) were strictly implemented.
Methods
Active Ingredient Screening of QHSST
Seven herbs in QHSST were screened to identify active ingredients using the traditional Chinese medicine systems pharmacology database and analysis platform (TCMSP). Screening of Notopterygii Rhizoma Et Radix, Angelicae Pubescentic Radix, Chuanxiong Rhizoma, Saposhnikoviae Radix, Ligustici Rhizoma Et Radix, Viticis Fructus, Glycyrrhzae Radix Et Rhizoma Prparata Cum Melle was centered on oral bioavailability (OB > 30%) and drug-likeness (DL > 0.18), using Lipinski's rule of five. Given the lack of an active ingredient in Ligustici Rhizoma Et Radix., its screening criteria were OB > 20% and DL > 0.10; the rest remained unchanged.
Acquisition of QHSST and RA-Related Targets
The compounds were retrieved from the PubChem database using their corresponding Simplified Molecular Input Line Entry System (SMILES) names. The SMILES were searched using the SwissTargetPrediction database to find the relevant targets corresponding to the relevant compounds with a “probability > 0” and combined with the TCMSP to screen for relevant targets. Duplicate proteins were excluded, and any duplicates were eliminated from the vast databases of DisGeNET and GeneCards using the search term “rheumatoid arthritis” to find pertinent targets.
Drug–Disease Intersection Target Preparation, Protein-–Protein Interaction (PPI) Network Construction, and Enrichment Analysis
The objectives of QHSST and RA were uploaded into the Venn 2.1 platform to determine potential intersecting targets. The intersecting objectives for the STRING were uploaded, and analysis was performed by selecting “Multiple proteins,” restricting the species to a specific limit to “Homo sapiens,” establishing the “minimum required interaction score” to “high confidence (0.700),” and selecting “hide disconnected nodes in the network.” The preliminary PPI interaction maps were obtained, and the results were imported into Cytoscape and sorted by degree to filter and identify the top seven main targets. Weisheng Xin Online Platform was used for Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) analyses, and the results were visualized using the Microbiotics online tool and represented as bar graphs.
Drug–Active-Ingredient–Target-Pathway–Disease Diagram
Drugs, active ingredient, intersecting targets, pathways, and diseases were organized into network and type files, and the top 20 pathways with significant KEGG signaling pathways were selected to establish a visual drug–active-ingredient–target-pathway–disease diagram in Cytoscape.
Molecular Docking
The top seven core targets obtained from screening were verified by molecular docking with the corresponding active ingredients. The target protein IDs were searched in the PDB database, and the water molecules were deleted using PyMOL. The two-dimensional structures of the corresponding components were downloaded from the PubChem database and optimized for transformation using Chem3D 19.0. The receptor and ligand information was imported into AutoDock Tools 1.5.6 for molecular docking analysis.
Preparation of QHSST Extract
A total of 12.39 g of Notopterygii Rhizoma Et Radix and Angelicae Pubescentic Radix, 6.18 g of Chuanxiong Rhizoma, Saposhnikoviae Radix, Ligustici Rhizoma Et Radix, Glycyrrhzae Radix Et Rhizoma Prparata Cum Melle, and 3.72 g of Viticis Fructus officinale were weighed. For the extraction, the mixture was heated at reflux for 24 min (boiling at 100 °C, then reduced to low heat) four times (the same amount of ultrapure water was added each time) and then filtered through gauze. The filtrate from each cycle was combined. After concentrating to a specific volume, the QHSST extract was stored at 4 °C.
Ultra-High-Performance–Liquid-Chromatography (UPLC)-Q-Orbitrap-Mass-Spectrometry (MS) Compositional Analysis
The QHSST extract was separated on a CORTECS® UPLC® T3 column (2.1 mm × 100 mm, 1.6 μm) at a separation rate of 0.2 mL/min and a separation temperature of 35 °C. The injection volume was 3 μL. The mobile phase was acetonitrile (A) - 0.2% formic acid aqueous solution (B). The gradient conditions were as follows: 0–40 min, 2–25% A; 40–70 min, 25–45% A; 70–80 min, 45–60% A; 80–90 min, 60–100% A; and 90–100 min, 100% A. Electrospray ionization was used in both positive and negative ion modes, and the MS - ddMS2 scanning mode was used for the analysis. The mass spectrometry conditions were as follows: sheath gas flow rate, 48 Arb; auxiliary gas flow rate, 11 Arb; capillary temperature, 256 °C; gas heater temperature, 300 °C.
RAW264.7 Cell Culture and Transfection
RAW264.7 cells were cultured in Dulbecco's Modified Eagle Medium with 10% FBS and 1% penicillin-streptomycin at 37°C in a 5% CO2 incubator. The cells were passaged at a 1:3 ratio, and cells at the logarithmic growth phase in the 3–eighth generation were selected for subsequent experiments.
Effects of Drugs and Lipopolysaccharides (LPS) on Viability Assessed by CCK-8 Assay
RAW264.7 cells in the logarithmic growth phase were added to 96-well plates at 1 × 105 cells per well and incubated in an incubator at 37 °C with 5% CO2 for 24 h. The supernatant was discarded. Cell culture medium with different concentrations of QHSST (100, 50, 25, 12.5, 6.25, 3.125, 1.563, and 0.783 µg/mL) and LPS (100, 50, 25, 12.5, 6.25, 3.125, 1.563, and 0.783 µg/mL) was added (100 μL), and six replicates were used for each concentration. These samples were then incubated for 24 h. Subsequently, 10 μL of CCK-8 reagent was added to each well and incubated for 0.5 h. The absorbance was measured at 450 nm.
Cell Grouping and Drug Delivery
The cells were split into four groups: control (CON); model (MOL); dexamethasone (DXM); and YT groups, based on the optimal concentration of LPS. With the exception of the CON group, the cells were treated with the ideal concentration of LPS for modeling, and after a 24-h incubation period, the supernatant was removed. The medication was added at a specific concentration to the DXM and YT groups, while the CON and MOL groups received the complete medium. Following a 24-h incubation period, the pertinent indices were measured.
Flow Cytometry to Assess Apoptosis
RAW264.7 cells were inoculated into 6-well plates. Cells were collected 24 h after drug administration, and 500 μL of Annexin V Binding Buffer (1×), 2.5 μL of Annexin V-FITC, and 2.5 μL of PI were added. Samples were protected from light for 15–20 min, and apoptosis was detected and assessed using flow cytometry.
ELISA for Determining of NO, IL-6, IL-10, and TNF-α Levels
RAW264.7 cells in logarithmic growth phase were used for these ELISAs. Cell supernatants were collected, and the levels of NO, IL-6, IL-10, and TNF-α were determined following the instructions of the respective ELISA kits.
Establishment of a CIA, Wind-Cold-Dampness-Paralysis rat Model and Subsequent Experimental Grouping and Drug Administration
After a one-week feeding adaptation period, 40 SD rats were randomly divided into four groups (n = 10). Rats were randomly grouped according to their body weight into CON, MOL, MTX (0.89 mg/kg/biw), and YT (9.50 g/kg/qd) groups. For the modeling procedure, type II collagen (CII) was solubilized in a solution containing 0.1 mol/L acetic acid overnight at 4 °C to create an initial 0.8 mg/mL CII acetic acid immunization solution. An equal volume of complete Fischer's adjuvant was added for emulsification, and a total of 0.2 mL of the resulting solution was then injected at multiple points in the caudal root and stumped hindfoot of the rats. From the second day of the initial immunization onwards, all groups, except the CON group, were subjected to the induction of the wind-cold-damp paralysis phenotype. This protocol involved standing on ice twice a day while being exposed to an electric fan blowing at speed level 3 for 45 min each time, until the end of the experiment. On day 7 since the initial immunization, a secondary immunization was performed using 0.4 mg/mL CII acetic acid solution, using a preparation method similar to that of the initial immunization solution (Figure 1). After successful modeling for 28 consecutive days, gastric administration of MTX and QHSST was initiated for the MTX and QHSST treatment groups, respectively. Equal volumes of saline were gavaged into CON and MOL groups.

CIA model building process.
Normal condition and body weights of the rats, AI scoring, left and right foot volume measurements
Changes in the activity, coat, diet, and body weight of the rats were documented before and after modeling and drug administration. The rat AI scoring was blindly evaluated by individuals unrelated to the experimental procedures using the criteria shown in Table 1. Evaluations were conducted on the right and left hind limbs independently, with a maximum score of eight. The volumes of the left and right toes were measured using a volumetric tester. The ankle joints of the rats were labeled before measurement, placed in the tester, and an average value obtained by repeating the measurement three times. All of the above metrics were evaluated every 3 days before and every 4 days after successful modeling.
Scoring Criteria.

Determination of Blood Indices in Rats
The rats were anesthetized 24 h after the last drug administration (fasting) via abdominal cavity injection of urethane, and blood samples were obtained via the abdominal aorta for analysis of blood indices (n = 6), including white blood cell (WBC), neutrophil (NEU), C-reactive protein (CRP), neutrophil/lymphocyte ratio (NLR), hemoglobin (HGB), and platelet (PLT) levels.
Toxicity Assessment
The rats were anesthetized 24 h after the last drug administration (fasting) via abdominal cavity injection of urethane. Rat organs (heart, liver, spleen, lungs, and kidneys) were obtained and measured (n = 6), and the organ index (W) was determined through weight calculation. Blood samples were obtained via the abdominal aorta and centrifuged at 1500 × g for a total of 10 min to obtain the serum. Biochemical analyses of alanine transaminase (ALT), aspartate transaminase (AST), UREA, and creatinine (CREA) were performed to evaluate the effect of the treatments on the liver and kidney functions of the rats.
W = organ weight/body weight × 100%.
H&E Staining
The ankle joints of the rats were fixed in 4% paraformaldehyde for 24 h, decalcified with 15% ethylenediaminetetraacetic acid, embedded in paraffin wax, sectioned, and stained with H&E. The fibrous tissues and synovial cells of the ankle joints of the rats in different groups were observed under a microscope to determine the presence of hyperplasia or inflammatory cell infiltration.
ELISA to Determine serum Anti-CCP Antibody, RF, IL-6, IL-10, and TNF-α Levels
The blood samples were left to clot and then centrifuged for 10 min at 1500 × g. The upper serum layer was collected and used for the ELISA assay. Serum anti-CCP antibody, RF, IL-6, IL-10, and TNF-α levels were determined using the appropriate ELISA kits, following the manufacturer's directions, and the absorption at 450 nm was measured.
ELISA to Determine Expression of EGFR-, SRC-, PI3K-, AKT-, and NF-κB-Related Proteins in Ankle Joints
Rat ankle joint tissues excluding the muscle were obtained, and the level of EGFR, SRC, PI3 K, AKT, and NF-κB in the ankle joints was determined using the appropriate ELISA kit, following the manufacturer's directions, and the absorption at 450 nm was measured.
Immunohistochemical Determination of SRC and AKT Expression
The ankle joints of rats were fixed in 4% paraformaldehyde for 24 h, decalcified with 15% EDTA, embedded in paraffin, and sectioned. Immunohistochemical assays were performed to observe the expression of SRC and AKT in the ankle joints.
Data Analysis
Data analysis was conducted utilizing GraphPad Prism 8.0 and SPSS 28.0. One-way analysis of variance was used to test the homogeneity of variance; two-by-two comparisons were performed using t-tests; and one-way analysis of variance was utilized to compare the variations among groups. Statistical significance was determined by observing differences with P < 0.05.
Results
Screening of QHSST Active Ingredients
QHSST contained 137 active ingredients from seven herbs (Figure 2): Notopterygii Rhizoma Et Radix, Angelicae Pubescentic Radix, Chuanxiong Rhizoma, Saposhnikoviae Radix, Ligustici Rhizoma Et Radix, Viticis Fructus, and Glycyrrhzae Radix Et Rhizoma Prparata Cum Melle; each contained 9, 8, 12, 3, 20, 81, and 4 active ingredients, respectively. Euphorbiacein, Nodakenin, Coralloidin, Kaempferol, and Quercetin were the shared active ingredients of QHSST.

Venn diagram of active ingredients (QH: notopterygii rhizoma et radix, DH: Angelicae pubescentic radix, CX: chuanxiong rhizoma, FF: saposhnikoviae radix, GB: ligustici rhizoma Et Radix, MJZ: Viticis Fructus, GC: glycyrrhzae radix et rhizoma prparata cum melle).
Acquisition of QHSST and RA Related Targets
The SMILES of the relevant active ingredients were identified using the SwissTargetPrediction database to predict the relevant targets and integrated with the TCMSP, and 845 relevant targets were obtained. Relevant targets were acquired from the DisGeNET and GeneCards databases to obtain 4082 relevant targets.
Drug–Disease Intersection Target Preparation, PPI Network Construction and Enrichment Analysis
The targets of the active ingredients in QHSST were plotted on a Venn diagram with the targets of RA (Figure 3A), which revealed 427 potential intersecting targets. These intersecting targets were imported into STRING to obtain a PPI network, which had 425 nodes, 2803 edges, an average node degree of 13.2, and an average local clustering coefficient of 0.462 (Figure 3B-a). In Fig. 3B-b, targets with a higher degree are represented by larger shapes, thicker interconnecting lines, and darker colors. Furthermore, targets with a degree > 70 were selected as core targets (Figure 3B, C), which may be core targets for QHSST therapy to alleviate RA.

A:venn diagram of QHSST active ingredient–RA intersection targets; B: PPI network diagram of common targets of action of QHSST in RA treatment. (a) Original PPI chart, (b) Cytoscape software is optimized to obtain the chart. (c) Core target chart; C: Visualization results of GO functional enrichment analysis (BP: biological process; CC: cellular component; MF: molecular function.); D: Visualization of the top 20 KEGG pathways.
The GO analyses results yielded 3907 GO entries (P < 0.05), including 3428 biological processes (BP), 167 cellular components (CC), and 312 molecular functions (MF). To obtain a clearer picture of their functionality, the p- and count values were used as references for ranking, and the top 20 entries were selected for each section to generate a histogram (Figure 3C). The KEGG enrichment results yielded a total of 178 pathways (P < 0.05). The entries ranked in the top 20 were selected depending on the p- and count values, and a bar chart was generated (Figure 3D).
Drug–Active-Ingredient–Target-Pathway–Disease Graph
A drug–active-ingredient–target-pathway–disease diagram was constructed (Figure 4). The EGFR/PI3K-AKT signaling pathway containing a high number of core targets, such as EGFR, SRC, and AKT1, was selected for use in the animal experiments to verify the expression of the relevant pathway proteins and determine the mechanism of action of QHSST in RA.

Drug–active material–target-pathway–disease graph (blue: drugs; orange: active ingredients; purple: intersecting targets; green: relevant pathways; and red: diseases).
Molecular Docking
The five active components ammidin, quercetin, kaempferol, isoimperatorin, and phellopterin and seven core targets SRC (2SRC), HSP90AA1 (3O0I), STAT3 (6TLC), AKT1 (1H10), MAPK3 (2ZOQ), MAPK1 (2OJG), and EGFR (5UG9) were evaluated using molecular docking (Figure 5A) (for graphical purposes, all data in Figure 5A are absolute values of their binding energies). SRC and AKT were selected for visualization based on the magnitude of the binding energy and core targeting degree values as well as the number of proteins contained in the relevant pathways (Figure 5B, C).

A: molecular docking binding energy diagram; B: molecular docking visualization for SRC (a: ammidin, b: quercetin, c: kaempferol, d: isoimperatorin, e: phellopterin); C: molecular docking visualization for AKT (a: ammidin, b: quercetin, c: kaempferol, d: isoimperatorin, e: phellopterin).
UPLC-Q-Orbitrap-MS Compositional Analysis
The components in QHSST were identified by UPLC-Q-Orbitrap-MS (Figure 6A). The chemical composition was determined based on the literature, databases, and relative molecular mass. A total of 44 compounds (Table 2) were identified, including 14 coumarins, 15 flavonoids, 3 phenolic acids, 3 phthalides, 2 polyphenols, 1 organic acid, 1 triterpene, 1 diterpene, 1 lignan glycoside, and 3 other compounds, in both positive and negative ion modes. The QC constituents of each herb (Figure 6B) were Notopterol and Isoimperatorin (a QC constituent of Notopterygii Rhizoma Et Radix), Osthole and Columbianadin (a QC constituent of Angelicae Pubescentic Radix), 5-O-Methylvisamminol (a QC constituent of Saposhnikoviae Radix), Ferulic acid (a QC constituent of Chuanxiong Rhizoma and Ligustici Rhizoma Et Radix), Casticin (a QC constituent of Viticis Fructus), Liquiritin (a QC constituent of Glycyrrhzae Radix Et Rhizoma Prparata Cum Melle). The identification of chemical constituents can be used as a basis for quality control.

Results of the QHSST component analysis (upper A: total ion flow diagram in QHSST positive ion mode, lower A: total ion flow diagram in QHSST negative ion mode, B: structure of QC components).
UPLC-Q-Orbitrap-MS Compositional Analysis Identification Results.

Optimization of Drug Concentration
Figure 7A demonstrates the effect of different drug concentrations on cell viability. The cell viability remained stable when the drug concentration reached 3.125 µg/mL but decreased to less than 50% when the drug concentration reached 25 µg/mL, indicating a damaging effect on cells. Therefore, 3.125 µg/mL was selected for subsequent experiments.

A: effect of the drug concentration on cell viability; B:effect of different LPS concentrations on cell viability.
Optimization of LPS Concentration
Figure 7B demonstrates the effects of different LPS concentrations on cell viability. The cell viability remained stable at LPS concentrations of 0.783–12.5 µg/mL and decreased when the LPS concentration reached 25 µg/mL, indicating damaging effects on cells. Therefore, 0.783 µg/mL was selected as the modeling concentration for subsequent experiments.
Apoptosis Rate
Rates of apoptosis in various groups are summarized in Figure 8A, B, C, and D. As shown in Figure 8E, the rate of apoptosis was significantly higher in the MOL group than in the CON group (P < 0.05), while DXM and 3.125 µg/mL groups showed a reduction in apoptosis compared with that in the MOL group. These results indicate that QHSST has a certain inhibitory effect on inflammatory cell apoptosis.
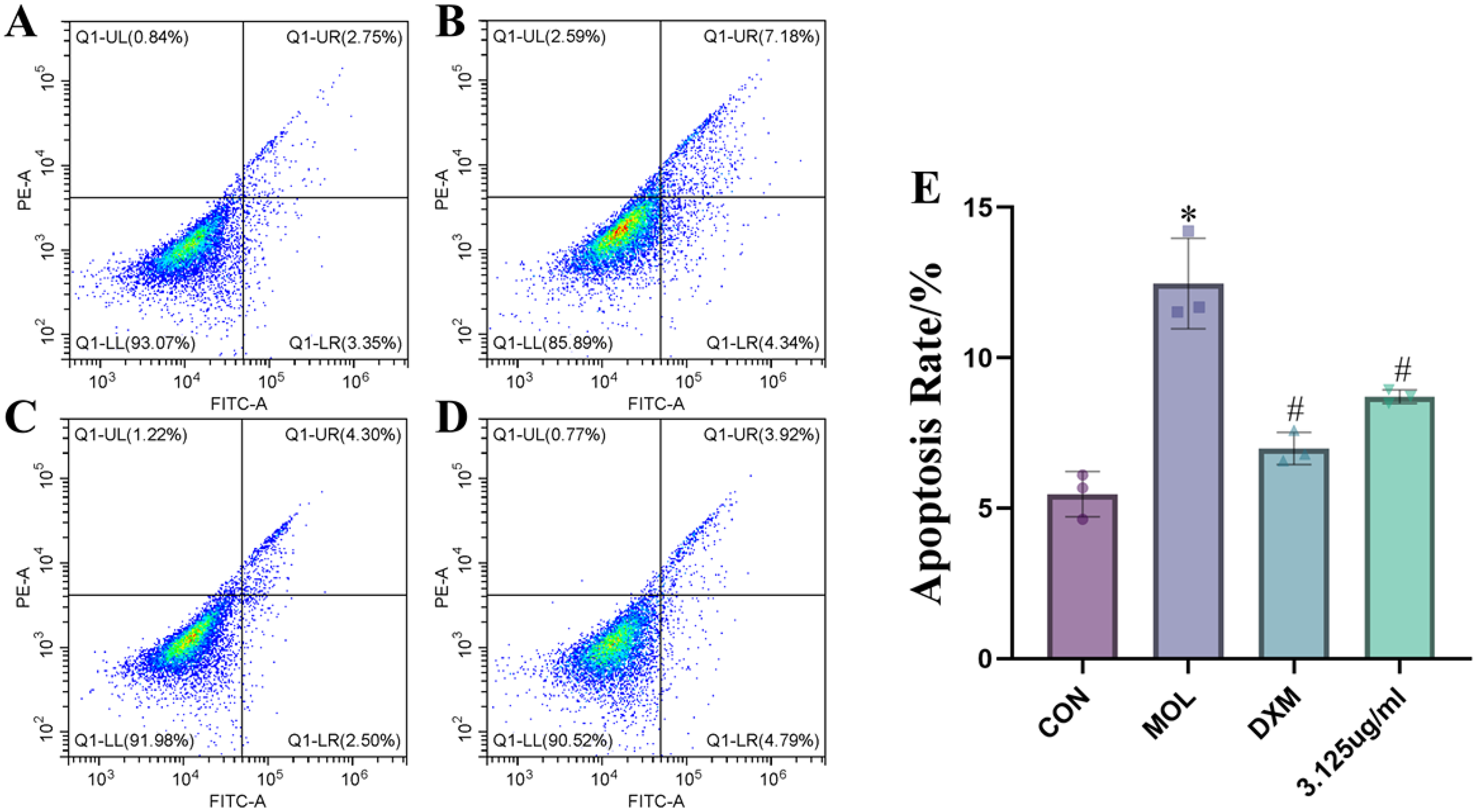
Apoptosis (A: CON group, B: MOL group, C: DXM group, D: 3.125 µg/ml, E: statistical analysis of apoptosis in each group) *P < 0.05 versus CON group; #P < 0.05vs MOL group.
NO, IL-6, IL-10, and TNF-α Levels
NO, IL-6, IL-10, and TNF-α are the most important indicators for assessing inflammation; therefore, they were measured using ELISA kits, and the results are shown in Figure 9. The NO, IL-6, IL-10, and TNF-α contents in the MOL group were significantly different from those in the CON group (P < 0.05). The level of these indicators also differed significantly between the DXM and 3.125 µg/mL groups and the MOL group (P < 0.05). These findings indicate that QHSST has a certain therapeutic effect on inflammation.

Levels of NO, IL-6, IL-10, and TNF-α *P < 0.05 versus CON group; #P < 0.05vs MOL group.
Modeling Success Indicators
On day 20, a trend toward significantly higher (P < 0.05) WBC, HGB, PLT, CRP, NEU, and NLR blood indices and anti-CCP antibody levels was observed (Figure 10A). AI scores and left and right foot volumes in the modeling groups (MOL, MTX, and YT) were compared with those in the CON group and were found to be statistically different (Figure 10B). In particular, the rat ankle joints on days 0 and 20 in the physical picture (Figure 10C) had significant swelling, indicating the successful establishment of the CIA model.

A: model validation metrics for blood indices, WBC, NEU, CRP, NLR, HGB, PLT, and the anti-CCP antibody ; B: changes in ai scores, left and right foot volumes, and body weights of rats over time ; C: physical picture of rat ankle joint swelling degree *P < 0.05 versus CON group; #P < 0.05vs MOL group.
Normal Condition and Body Weights of the Rats
During the modeling process, the rats in the CON group were in a good mental state, with a normal diet, water intake, feces, ankle joints, and lip color, whereas the rats in the modeling groups had poorer mental status, reduced food and water intake, dry and dull fur, thinner feces, swollen ankle joints, lavender lips, and vesicles in severe cases. After drug administration, the MTX and YT group rats showed a reduction in joint swelling, increased activity, increased diet and water intake, and an upward trend in body weight. On the 48th day after the end of treatment, an improving trend was observed in the AI score, body weight, left and right foot volumes, and rat ankle joint swelling (Figure 10B, C); the differences were found to be significant (P < 0.05), indicating that QHSST may alleviate RA.
Determination of Blood Routine Indices in Rats
At the end of the experiment, blood was collected from the abdominal aorta to determine the WBC, NEU, CRP, NLR, HGB, and PLT levels (Figure 11). Compared with the CON group, significant differences were found in WBC, NEU, NLR, and CRP in the MOL group (P < 0.05). A certain trend of changes in HGB and PLT was observed; however, the differences were not significant. Compared with the MOL group, significant differences were found in PLT, NEU, NLR, and CRP levels in the MTX group and in PLT and CRP levels in the YT group (P < 0.05). The remaining indices showed some trends; however, no significant differences were observed. The results showed that the symptoms of RA were effectively alleviated in the YT group, which could be attributed to the reducing WBC, PLT, NEU, NLR, and CRP levels.

Blood test indices, WBC, NEU, CRP, NLR, HGB, and PLT, in rats on day 48 *P < 0.05 versus CON group; #P < 0.05vs MOL group.
Toxicity Assessment
A preliminary evaluation of the toxic effects on rats in the administration groups was based on the organ indices of the heart, liver, spleen, lungs, and kidneys (Figure 12A). Compared to the CON group, no significant differences were observed in the heart, liver, spleen, lungs, and kidneys in the MOL, MTX, and YT groups, except for the kidneys in the YT group (P < 0.05). However, an increasing tendency was observed, suggesting that the administration may have slight toxic side effects on rats. The effects on the liver and kidney functions in the administration groups were evaluated in conjunction with the analysis of the organ, liver, and kidney indices (Figure 12B). The ALT, AST, urea, and CREA levels in the MOL, MTX, and YT groups did not differ significantly from those in the CON group. No significant increasing trend was observed, indicating that the administration had less effect on the liver and kidney functions of the rats.

A: effect of different treatments on the organ indices of the heart, liver, spleen, lungs and kidneys in rats; B: liver and kidney function indicators in rats (ALT, AST, urea, and CREA) *P < 0.05 versus CON group; #P < 0.05vs MOL group.
H&E Staining
H&E staining of rat ankle joint tissues showed that the CON group had no obvious changes in the joint tissues, whereas the MOL group showed more proliferated fibrous tissue and local narrowing of the joint cavity (Figure 13). In the MTX and YT groups, fibrotic hyperplasia was observed in the soft tissues around the joints and the lower layer of the synovial lining. An increased number of fibroblasts with long elliptical nuclei was also observed, accompanied by inflammatory cell infiltration. In summary, pathological changes were observed in the MOL group compared with the CON group. Compared with the MOL group, the MTX and YT groups showed a reduction in the extent of these lesions.

H&E staining of ankle joint tissue (×200).
Comparison of Anti-CCP Antibody, RF, IL-6, IL-10, and TNF-a Levels in serum of Rats in Each Group
Figure 14 shows the results for the anti-CCP antibody, RF, IL-6, IL-10, and TNF-a levels in the serum. These levels were significantly different (P < 0.05) between the MOL and CON groups and between the MTX and MOL groups (P < 0.05). Between the YT and MOL groups, the anti-CCP antibody, IL-6, and IL-10 levels were all significantly different (P < 0.05), with no significant difference for the RF and TNF-a levels; however, a decreasing tendency was observed. This demonstrated that QHSST can alleviate RA by lowering anti-CCP antibody, RF, IL-6, and TNF-a levels and elevating IL-10 levels.

Analysis of rat serum IL-6, IL-10, TNF-a, anti-CCP antibody and RF levels *P < 0.05 versus CON group; #P < 0.05vs MOL group.
Expression of EGFR-, SRC-, PI3K-, AKT-, and NF-κB-Related Proteins in Ankle Joints of Rats
Figure 15 shows the results of EGFR-, SRC-, PI3K-, AKT-, and NF-κB-related protein expression in rat ankle joints. EGFR, SRC, PI3 K, AKT, and NF-κB were significantly different between the MOL and CON groups (P < 0.05). EGFR, SRC, PI3 K, AKT, and NF-κB were significantly different between the MTX and YT groups and the MOL group (P < 0.05). This demonstrates that QHSST can alleviate RA by upregulating EGFR and downregulating SRC, PI3 K, AKT, and NF-κB expression.

Expression of EGFR, SRC, PI3 K, AKT, and NF-κB proteins in rat ankle joints *P < 0.05 versus CON group; #P < 0.05vs MOL group.
Immunohistochemical Determination of SRC and AKT Expression
We selected the proteins with the largest core target degree among the validated pathway proteins (SRC, AKT) for immunohistochemical validation (Figure 16A). Levels of SRC and AKT differed significantly between the MOL and CON groups (P < 0.05). Additionally, compared with levels in the MOL group, SRC and AKT levels were significantly different in the MTX and YT groups (P < 0.05) (Figure 16B, C).

Immunohistochemical analysis of SRC, and AKT. (A: Immunohistochemistry (40X); B: Percentage of area positive for SRC; C: Percentage of area positive for AKT) * P < 0.05 versus CON group; # P < 0.05 versus MOL group.
Discussion
Diseases of all types are driven by inflammation and are inextricably linked to organismal function, making inflammation a popular research topic. This study investigates whether QHSST can alleviate inflammation-induced RA by resisting inflammation. Through the QHSST treatment of the LPS-induced inflammation model of RAW264.7 cells, we found that QHSST can effectively inhibit apoptosis and NO production as well as reduce the expression level of inflammatory factors, thus indicating that QHSST has certain anti-inflammatory effects. Animal experiments (RA model) were further conducted to validate the anti-inflammatory effects of QHSST and preliminarily explore the mechanism of action of QHSST in RA treatment.
RA was chosen for this study because, in Chinese medicine, it is categorized under syndromes such as “cold-heat staggering,” “rheumatic-heat,” “toxic-heat stasis,” “prolonged illness,” and “wind-cold-damp paralysis.” Most RA patients in China live in humid, cold regions like coastal areas, Northeast China, and the Qinghai-Tibetan Plateau, where wind-cold-damp paralysis is common. Clinical studies14–18 have shown that QHSST dispels wind, eliminates dampness, and relieves pain, making it effective for treating paralytic rheumatism. Despite this, there are few studies on the mechanism of QHSST in RA pathogenesis. Therefore, we investigated RA pathogenesis and explored the pharmacological effects of QHSST via gavage in rats, using network pharmacology predictions and animal experiments.
The core target prediction of QHSST using network pharmacology yielded 427 targets, of which the seven core targets were SRC, HSP90AA1, STAT3, AKT1, MAPK3, MAPK1, and EGFR. KEGG enrichment analysis identified signaling pathways, such as MAPK, PI3K-AKT, EGFR tyrosine kinase inhibitor resistance, and others, that may be relevant to its mechanism of action and the treatment of RA. For the integration of core targets as well as related pathways, we focused on the inflammatory response, including core targets such as SRC, PI3 K, AKT1, EGFR, and NF-κB. Among these, EGFR is an important mediator of the pathogenesis of RA and synovial hyperplasia. 19 EGFR may be involved in immune cell regulation in RA synovial tissue, 20 promote endothelial cell proliferation and cytokine production in synovial fibroblasts, and inhibit osteoclast formation.21,22 SRC plays a pivotal role in bone metabolism by regulating growth, proliferation, adhesion, and other important processes. 23 SRC activation leads to cytoskeletal reorganization and reduced bone resorption activity.24–26 Activated SRC activates downstream P13K, 27 which in turn activates Akt via phosphatidyinositol-4,5-bisphosphate. Akt then releases cytoplasmic NF - κB, which regulates osteoblastic precursor cell differentiation. 28 PI3 K/Akt is a well-recognized inflammation-related pathway that regulates NF-κB stimulation of B cells to release inflammatory factors, such as IL-6, IL-10, and TNF-a. These inflammatory factors participate in osteoclast differentiation and converge on osteoclast migration, destroying the bone and articular cartilage, ultimately leading to joint deformity. 29 Therefore, it can be inferred that QHSST can modulate the EGFR/SRC/PI3 K/AKT/NF-κB signaling pathway to alleviate RA.
We established a rat CIA model to verify the core targets predicted by network pharmacology. We used the AI score, left and right foot volume measurements, anti-CCP antibodies, routine blood tests, and other related indices to evaluate the success of the CIA model and the therapeutic effect of QHSST. The experimental results showed that QHSST had a therapeutic effect. Our results show that QHSST upregulates EGFR, which inhibits SRC expression and subsequently the PI3K-AKT pathway proteins. This leads to the downregulation of NF-κB, reducing osteoclast migration, inflammatory factors in the bone, anti-CCP antibody levels, and RF, ultimately alleviating RA symptoms (Figure 17).

Schematic diagram of the pathway of the QHSST therapy in RA.
This study had some limitations. We focused on inflammation to determine the possible mechanism of action of QHSST; therefore, our results do not provide a complete understanding of the mechanisms that may be involved in the treatment of RA. In subsequent experiments, we aim to use a multi-omics approach, with liquid-quantity coupling, high-throughput screening, and other methods, to identify specific components and more comprehensively illustrate the related mechanisms of action.
Conclusion
By combining network pharmacology with in vitro and in vivo experiments, we were able to predict key core targets and associated pathways. The anti-inflammatory effect of QHSST has been confirmed by cellular experiments; in animal experiments, it has been shown that QHSST can alleviate RA by altering the pathways associated with inflammation. This will provide a new approach for the treatment of RA and help to elucidate the mechanism of action and pharmacological effect of QHSST.
Footnotes
Acknowledgments
The authors would like to thank the Department of Pharmacy, Qinghai University and the State Key Laboratory of Plateau Ecology and Agriculture for their support and assistance.
Author Contributions
JH, MW, YL, LT, GL, QW, DD, JZ, QJ, YD, CN and XL made substantial contributions to conception and design, and revised the manuscript critically for important intellectual content. CN and XL revised the manuscript and gave final approval of the version to be published. All authors read and approved the final manuscript.
Data Availability
The study is available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Approval
Compliance with the Ethical Review of Discipline Management Research at Qinghai University School of Medicine, No. 2022-56.
Funding
This study was supported by the Key Research and Development and Transformation Project of Qinghai Province (2021-SF-158).
Key Research and Development and Transformation Project of Qinghai Province, (grant number 2021-SF-158).
Informed Consent Statement
Not applicable. No human subjects were used in this article.
