Abstract
Introduction
Natural dyes have become very important for decades based on their extensive properties compared to synthetic dyes. Pima cotton was dyed with natural dyes derived from plants using mordants, a crucial step in modern industry.
Objective
This study explores the efficacy of banana leaf-extracted natural dyes with mordants in dyeing Pima cotton, examining color properties, chemical attributes, and fastness while exploring the potential for industrial applications.
Methods
In this study, banana leaf extracts were utilized for dyeing Pima cotton with mordants. The investigation assessed the multifunctional properties of alkaline and acetone using UV-Vis and FTIR or analyzed the dyed fabric's UPF. Applied to pre-mordanted Pima cotton, our dye interacts with various mordants like ferrous sulfate (Fe2SO4), copper sulfate (CuSO4), and potassium dichromate (K2Cr2O7). Factors influencing dye fixation on Pima cotton, including cellulose and mordants, were examined. Treated and untreated fabric samples were tested for tensile strength and elongation. Dyeing performance was assessed using color parameters (K/S, L*, a*, b*, ΔE) using the Kubelka–Munk equation and fastness properties, with consistent results compared to standard protocols.
Results
The study on natural dye extraction from banana leaves has yielded promising outcomes, showcasing the effectiveness of mordants in dye fixation on cotton. The results demonstrate high tensile strength, color strength, and fastness properties in the dyed Pima cotton samples. Statistical analysis highlighted the nuanced relationship between mordants and dyes. The study's comprehensive approach, UV-Vis and FTIR analyses, dyeing temperature optimization, and phytochemical screening, confirming the presence of flavonoids, saponins, alkaloids, and other bioactive compounds, positions banana leaf extracts as a sustainable and versatile resource for eco-friendly textile practices, offering a compelling alternative to synthetic dyes.
Conclusion
The study reveals Pima cotton's promising potential for eco-friendly dyeing, optimizing cost, time, and space for future sustainable textile applications.
Introduction
Natural dyes have long been valued for their ecological and sustainable properties in textile dyeing processes. Recently, much interest has been placed on circumventing the environmental problems associated with manufactured coloring agents using natural dyes. Natural dyes are becoming more critical because they are considered environmentally friendly.1,2 Pollution reduction is not the only central aspect that makes them attractive.. 3 A natural dye is extracted from turmeric and used to dye cotton at different dyeing conditions.4–6 Despite synthetic dyes, natural dyes are obtained from plants and minerals, 7 where wide-ranging plants, including monocots, a dicot, legumes, cereals, and fruit,8,9 are extensively used for natural dye extraction. Most natural dyes’ primary sources are vegetables, 10 plants 11 ; roots, 12 fruit, 12 bark, 13 leaves, 14 wood 15 ; and other organic sources, such as fungi 16 and lichens. 17 Food coloring is often used in natural dye fabrics such as cotton, linen, silk, jute, etc. 18 However, very little data is accessible on artificial coloring fibers such as polyester, nylon, acrylic, etc. 19 The archaeological exhumation of the colored content has been found worldwide. 20 It is an open affirmation of the old coloring of the past. Before the nineteenth century, natural colors were the principal sources of colored textures. 21 As the old standard colors were usually used for food shading, cowhide colors and common fabrics including fleece, silk, and cotton were colored. 22 This is a reflection of ecological issues relating to the usage of Azo dyes as a key photolytic degradation agent for prohibited carcinogenic amines and has prompted scientists worldwide to explore new environmentally sustainable alternatives for minimizing their harmful environmental impacts. 23 Natural colors cover shades and tints. 24 These tend to provide an incredible, irregular assortment of rich colors, which upgrade adhere plants to texture or scrub squeeze shades into cloth.
According to earlier researchers,25–27 dyed products contrast more with natural colors. Artificial colors are cheaper and available with various magnificent shades, 28 used to improve pace shading properties. Artificial colors are available in a wide range and can be used as basic materials. 29 However, using artificial colors induces genuine medical issues and has a detrimental effect on the environmental equalization of design. 26 The colors prepared from synthetic mixtures often include sodium chloride toluene and copper azide precursor, 30 and these types of synthetic compounds can be particularly risky to the human body. 31 Traditional dyes are thus gaining fame in many nations. 32 In addition, several nations have only forced implications of synthetic dyes over specific colors based on conditional versions and urgent needs. 33 This way, the greener and horticultural development, expressed in various publications, is more attractive. 34 With the swift desire for eco-friendly conditions, most companies and commodity fare companies seek to make the best use of the characteristic colors and color a printed material that aims at the specialty market. 35 Popular dyes are isolated in three classes from plants such as indigo, 36 minerals such as ocher, 32 and plants.37,38 The third group of plants is nontoxic, non-cancerous, and biodegradable, as cited in multiple research papers.39–41 There is a need to make economical use of popular dyes and to adopt a suitable and institutionalized coloring strategy without deciding the appropriate nature of colored materials.42,43 Pakistan is a horticultural country, and its content industry is the basis of its fare; therefore, it is necessary to convince content owners to use suitable dyes to protect the environment from hazards. 44 Here, to support the industry, experts can create three major types of natural dyes: substantive, vat, and mordant dyes; other less common forms of natural dyeing include rust dyeing, dye painting with earth oxides, and mud dyeing.
Due to the increasing awareness of sustainable resources, the value of environmentally safe materials has recovered. 45 The same concept goes with herbal dyes; consumers are becoming more conscious of fabrics and textiles. Natural colorants have some benefits because they are usually delicate, radiant, and are human eye reflectors. 46 Blending and matching frames may render various colors. 47 A little difference in the process or modification of a bite with a similar color may create new colors or a broad range of colorants. 48 Natural coloration renders unusual coloring and concurrently blends. 49 The traditional coloring is sustainable, agro-inexhaustible, and organic. 26 The failure from the thinning process transforms into major compost for farming fields like complex, indigo, and many dye plants that can be grown on squandered soil. Using wasteland is another value of natural coloration, such as tea garden dyes. 24 In the same context, the unemployment problem can be solved based on lunching dyeing industries because usual dye production requires much effort.50,51 By manufacturing standard dyes, oil derivatives can also be reduced because the natural coloration is acceptable for human skin and well-being in contrast to different allergens.32,52
There are many wells with signature dyes, such as herbs, minerals, and animal sources. Shading is different from usual outlets and threatens the unit of experts and scientists. 53 Characteristic dyes are extracted from various plants’ roots, stems, and blooms, such as onion strips and turmeric skin in some queries. 54 Natural dyes are ideal for natural fiber teething to enhance their eco-friendly properties.48,55 Textiles such as thread, yarn, or garments may be dyed through natural dyes like synthetic dyes. Appropriate or natural dyes are the resources used for the color. 56 Only some natural tints can be added directly to textiles, but in most situations, a different phase is needed.32,57 Although many different kinds of green dye exist, none can produce a deep, rich green in a dye pot. According to researchers who work with dye plants (Prabhu & Bhute, 2012; Samanta & Konar, 2011), yellow is the most prevalent hue easily accessible in the natural world.
In the past, green dyes were generated by combining yellow and blue or vice versa.58,59 Textiles would have been first dyed in blue, followed by a yellow over-dye. Over-dyeing involves dying fiber in one color and then dying it again in another. 60 The most famous natural approach to creating green is mixing a yellow and blue dye or adding iron to a yellow dye, which dulls down the yellow and shifts it through the mustard toward green. 61 Copper can be converted into nanoparticles as per earlier researchers 62 and used to modify yellow, frequently producing a neon-like green that is more vivid. Iron may be used safely with proper handling, but it is pretty hard on fibers, especially the more delicate protein fibers like wool or silk. Both copper and iron are heavy metals 63 ; however, copper may be risky and difficult to dispose of securely, whereas iron can be utilized and disposed of safely. Blending a yellow dye with a blue dye or vice versa is the kindest fabric choice. 64
This study enhances existing knowledge by investigating the color characteristics of Pima cotton fabrics dyed with alkaline and acetone banana leaf extractions and exploring mordant-dye interactions. We comprehensively assess the impact of extraction methods and mordants on mechanical and color properties. Choosing banana leaves for their eco-friendly attributes and as an ideal dye source, we aim to contribute to sustainable dyeing practices. Beyond textiles, banana components play versatile roles, from flame retardant finishing for cotton to mordants and potential dye adsorbents. Our research introduces a novel exploration into the coloration potential of banana blossom stem sap, broadening understanding. Examining the influence of mordants on natural dye color fixation, we propose an innovative method, exemplified by green dye extraction from banana leaves. Integrating mordants into Pima cotton fabric dyeing, our study explores the phytochemical screening of banana leaf extract, providing a comprehensive analysis through UV-VIS spectroscopy and FTIR. The addition of the ultraviolet protection factor assessment enriches our study by evaluating the potential impact of UV radiation on the skin. Our research contributes to understanding sustainable dyeing practices and the multifunctional aspects of banana plant derivatives in textiles.
Material and Methods
Data Sets
We extract natural dyes from waste materials of banana leaves under optimized conditions to get a high yield and then dye the Pima cotton with extracted dyes to analyze the affinity of dyes to the fabric. After that, dyed fabric is characterized to check color strength and washing fastness. We conduct all Overall work in the Green Chemistry Lab, Physical Lab, and Hi-Tech Laboratories of Government College Women's University Faisalabad. We used banana (Musa paradisiaca Linn) leaves to extract dye. All the samples were collected from Government College Women's University Faisalabad, University of Agriculture Faisalabad, and the other cooperative places of Faisalabad, where high-quality banana plants were found. The leaves from the Banana plant were manually cut out from plants and then thoroughly rinsed with tape water to remove atmospheric contaminants.
Moreover, these leaves were crushed in the laboratory using hydrogen liquid as a quick dryer to crush the leaves with the freezing condition to make a fine powder of leaves, as shown in Figure 1, where we added in three different bottles with different quantities (250, 500 and 750 g). The samples were kept from sunlight, made into powder using hydrogen liquid, and then crushed; the banana plant leaves were for further extraction. The crushed material was kept from moisture and prevented in an air-tight jar with proper labeling.

Crushed leaves of banana plant using liquid nitrogen.
In our study, the Pima cotton fabric, renowned for its exceptional quality, featured a meticulously selected plain weave pattern. The weight per area of the fabric was precisely measured at 180 g/m², and the density in both the warp and weft directions was determined to be 60 threads/cm, ensuring consistency and accuracy in our experimental setup. These values were carefully chosen to represent typical characteristics of Pima cotton fabric and provide a solid foundation for our dyeing and mordanting processes. The fabric's structural details, including the weave type, weight per area, and density, are fundamental in evaluating its response to the experimental treatments and contribute to the robustness and reproducibility of our research outcomes.
Detail Methods
Our experimental approach employed a strategic combination of mixed solvents to optimize the dye extraction process. Hydrochloric acid (HCl) was meticulously utilized to fine-tune the pH level, ensuring precise control over the chemical environment. Sodium hydroxide (NaOH) played a pivotal role as the basic solvent, skillfully neutralizing acidic components and contributing to the overall effectiveness of the extraction process. Additionally, ethanol (C2H5OH) served as the alcoholic solvent, leveraging its expansive solubility range to extract dyes from the raw materials efficiently. This meticulous selection of mixed solvents reflects our commitment to achieving a harmonious and controlled extraction environment, where each component contributes to the success of the dye extraction process. To avoid any chemical toxicity and synthesis, an eco-friendly natural dye product with less or non-hazardous effects, distilled water, was used as a solvent as per the methods of earlier researchers. 65 We used a volumetric Solution, where solutes add volume to a quantity of solvent to ensure the accuracy of the desired concentration as per the investigation of the earlier researcher. 66 We combined a predetermined amount of water and powdered medium to identify precise concentrations. 67
Further, we used the weight-to-volume technique to form our desired solution. We took the 1000 ml distilled water and heated it for 80 min at 60–70 °C, then took the 50 g of sample powder, added it to the preheated distilled water, and mixed it by shaking. This solution was used to extract dye. 68 The mordants employed in this study, namely ferrous sulfate (Fe2SO4), copper sulfate (CuSO4), and potassium dichromate (K2Cr2O7), ensure clarity and precision. We use pre-mordanting in our experimental design with different concentrations like ferrous sulfate at 5%, copper sulfate at 3%, and potassium dichromate at 2% weight of the fabric, optimizing conditions for effective dye fixation. We applied mordanting process parameters to control and standardize the experimental conditions, including a mordanting time of 2 h, a temperature of 60 °C, and a liquor-to-fabric ratio of 1:20.
Extraction
We used conventional techniques of hydro distillation and the Soxhlet apparatus to extract distilled water and other techniques like ultrasound, pulsed electric field, enzyme digestion, and microwave heating to operate more quickly and produce extracts with higher yields and higher quality as per the investigation of earlier researchers.69,70 We used rollers to squeeze out liquids as one option per the investigation of (Wu et al, 2015), and our methodology is shown in Figure 2. Aqueous, organic solvent, acid, alkali, PH of the media, and conditions of extraction, such as temperature, duration, and material to the liquid ratio particle size of the substrate, all affect how well colorants contained in natural plant sources are extracted as per the methods of earlier researchers. 71

Methodology for extraction.
In the dyeing process, we utilized a state-of-the-art continuous dyeing machine (K.M.Ashraf & Sons (Pvt) Ltd) with a liquor-to-fabric ratio of 20:1. The Pima cotton, weighing 200 grams, underwent dyeing with our extracted natural dyes at a concentration of 5 grams per liter. The exhaust dyeing process ensured optimal absorption and fixation of the natural dyes onto the fabric. Figure green indicates that the applied methodology from wide-ranging existing techniques remains for multiple purposes to target dye extraction, as we also indicated the plant selection methods via using dried plant and applying aqueous extraction method for final liquid dyes outcomes as per the investigation of the earlier researcher. 72 This meticulous approach enhances the clarity of our experimental methodology and provides essential insights for reproducibility and further research.
Methods to Obtain Dye
In the Pima cotton dyeing process with extracted natural dyes, plant materials such as leaves, roots, or fruits are sourced and subjected to microwave heating at 60 °C alongside a 30-min sonication process to enhance extraction efficiency as per the method of Bashir, Adeel, Aftab, Mia, Imran, and Pharmacy. 70 The solution underwent filtration post-microwave heating and sonication, separating solid components and yielding the desired green dye in the filtrate. Modulating pH conditions for optimal dye extraction and subsequent Pima cotton dyeing is crucial in the experimental protocol. Hydrochloric acid (HCl), functioning as the acidic solvent, facilitated pH adjustment, and its use in the range of approximately 2 to 4 enhanced the solubility of specific dye compounds. Sodium hydroxide (NaOH), acting as the basic solvent, neutralized acidic components in the dye solution, with pH values around 9 to 11 establishing an alkaline environment. Ethanol (C2H5OH), the alcoholic solvent, served as a versatile medium for dye dissolution, with its neutral pH maintaining a stable solution. The meticulous control of pH parameters, ranging from acidic to alkaline conditions, underscored the importance of solvent selection in tailoring the dyeing process, ultimately influencing the solubility, reactivity, and overall color vibrancy of the natural dyes on the Pima cotton. The resulting dye solution is filtered, and acetone contributes to the dyeing phase. The Pima cotton, pre-mordanted with substances like ferrous sulfate (Fe2SO4), copper sulfate (CuSO4), and potassium dichromate (K2Cr2O7), undergoes dye application. Mordants are crucial in fixing the dye, determining color, and enhancing properties like UV protection. Rigorous testing follows, encompassing tensile strength, elongation, and assessments of color parameters and fastness properties. The results showcase the effectiveness of this eco-friendly Pima cotton dyeing approach, promising sustainability and vibrant outcomes for various industrial applications in the realm of eco-textile garments.
Phytochemical Screening
We check the phytochemical screening as per the investigation of, 73 where several green agents are studied, such as phenolic, tannin, carotenoids, alkaloids, and flavonoids, which play a significant role as reducing agents; it is crucial to identify the medicinally active species in plant extracts. Therefore, this research found these are primary components in the banana leaves based on the phytochemical test as per the investigation. 74 About 5 ml of banana leaf extract was vigorously shaken into 5 ml of distilled water to create foam. Methods demonstrate that the extract from banana leaves contains Saponins. 75 About 5 ml of banana leaf extract received 2 ml of hydrochloric acid. The mixture was heated for approximately 30 min, cooled, and then treated with Hager's reagent in the filtrated solution. Consequently, a yellow tint develops, indicating that the banana leaf extract contains alkaloid groups. 76 Ten drops of ferric chloride (FeCl3) solutions were added to 5 ml of an extract of banana leaves. The bluish hue of the banana leaf extract indicates the presence of phenolic chemicals. 77 After treating 5 ml of banana leaf extract with 10% sodium hydroxide, the solution becomes a bright yellow, indicating that the extract contains flavonoids. 78 To the 2 ml of banana leaf extract, we added 1 ml of strong sulphuric acid, 2 ml of chloroform, and 2 ml of acetic acid. A blue-green indicates the existence of steroids, which were verified as per the method of earlier researchers. 79
Characterization
We used characterization methods accomplished in the Green Chemistry lab, GC Women University, Faisalabad, where ultraviolet-visible spectroscopy (UV-Vis spectroscopy) and Fourier transform-infrared spectroscopy (FTIR) were used for dye extraction from banana leaves.
UV-Vis and FTIR Details
Ultraviolet-visible spectroscopy has wide-ranged features; we use this technique for sample identification and quantitative measurements, where we analyze the concentration in solution to determine specific wavelength by measuring the absorbance as per the investigation of earlier researchers.80–82 We prepared our samples for taking absorbance through a UV-Vis spectrophotometer per the range. 83 A total of 1.0 ml of natural dye was dissolved in 99.0 ml of water, and the pH of the solutions was adjusted to 2.5, 3.5, 5.5, 7.0, 9.0, 10.0, and 11.0 by acetic acid, sodium carbonate, and sodium hydroxide solutions. The cuvettes were washed with distilled water and dried with clean tissue paper. The reaction mixture was taken in the cuvette, and absorbance was taken from 300 to 600 nm by a UV-2100 spectrophotometer with a path length of 10. We used FTIR to analyze chemical parameters that compare infrared intensity to the number of waves or light wavelengths. Using FTIR, we find the vibrational characteristics of the functional groups in the research material and check how much functional groups absorb infrared light as per the investigation of the earlier researcher. 84
Ultraviolet Protection Factor (UPF) Value
To identify average UV irradiance, we use the UPF (ultraviolet protection factor) value and then test UVA and UVB transmission as per the methods of ATTCC183.
85
A Perkin–Elmer double beam spectrophotometer of type Lambda 35 with an incorporated globe connection and a Schott glass UG-11 filter was used for the measurements. We used the (400,
Determination of Color Strength and Fastness
We used scattering coefficient and color strength (K/S) for determining a sample's quality; how intense the color is in terms of the profundity of the color-dyed cloth is the tinctorial value or color strength (K/S). We used parametric analysis as per the earlier researcher Kubelka–Munk theory investigated by,87,88 where data color 650 TM spectrophotometer was used to measure the reflectance (%) of the dyed cloth samples. We use color difference by CMC DE, where L* denotes brightness, C* denotes chroma, and h denotes hue angle in this color space. Chroma C* has a value of 0 in the middle and is measured as a displacement from the lightness axis (L*). We measure the Hue angle in degrees and begin at the + a* axis (0° is + a*, red, and 90° is + b, or yellow). L* = luminance, C* = chroma, and h = hue as per the investigation of. 89
We used color fastness parameters based on ISO procedures and t for colorfastness testing. Then, the ISO-105-C03 wash durability test was used to identify a composite sample of 10 cm by 4 cm and soap solutions in a washing machine as indicated. 90 After that, we assessed the speed rating utilizing the color fading and color staining gray scales in the color-matching cabinet, and the ISO-105-X12 technique was used to test the rubbing fastness using a sample size of 5 cm to 14 cm. The hand crank is spun ten times at a speed of one turn per second, and the fastness rating is then assessed using a color-stained gray scale. The ISO-105-E04 technique was also used to assess a color's resistance to sweat as per the earlier researcher investigations. 91
Results and Discussion
We optimized natural dye extraction from banana leaves using mixed solvents (hydrochloric acid, sodium hydroxide, ethanol, and distilled water). Mordants (ferrous sulfate, copper sulfate, potassium dichromate) ensured adequate dye fixation on cotton under standardized conditions. Various techniques (hydro distillation, ultrasound) were employed. Banana leaves were screened for medicinal components. UV-Vis and FTIR characterized the material, and UPF values assessed fabric UV protection. ISO procedures evaluated fabric quality, durability, and colorfastness. Focusing on eco-friendly synthetic dyes, our study utilizes banana leaf pigments for Pima cotton dyeing. Alkaline and acetone extracts were applied to pre-mordanted cotton, and UV-Vis and FTIR analyses were conducted. We investigated factors influencing dye fixation, Pima cotton mechanical properties, and dyeing performance. Results revealed treated fabrics with high tensile strength, color intensity, and fastness. Phytochemical screening identified flavonoids, saponins, and alkaloids, suggesting industrial applications for banana leaf dye.
Statistical Analysis and Identification of Color Parameters
As mordants ferrous sulfate (Fe2SO4), copper sulfate (CuSO4), and potassium dichromate (K2Cr2O7) were employed. The variables influencing the extracted dye's fixation with cellulose were investigated. 92 The tensile strength and elongation percent of the treated and non-treated cloth samples were evaluated to determine their mechanical characteristics. We investigated coloring effectiveness in terms of color parameters (K/S, L*, a*, b, and E) and fastness characteristics (wash, sweat, and light fastness) as per the investigation of earlier researchers. 93 The samples exhibit fastness, high color strength, and high tensile strength. The Color measurements of Pima cotton fabrics dyed with alkaline and acetone Banana leaf extraction are mentioned in Table 1. These findings are significant for industrial applications since they may be used to produce natural color from banana leaves at a low cost. Another goal is to boost the manufacture of reasonably priced eco-textile clothing. 94
Colour measurements of Pima cotton fabrics dyed with alkaline and acetone banana leaf extraction.

Table 1 presents Pima cotton fabrics dyed with alkaline and acetone banana leaf extract color measurements. These include critical parameters for assessing the fabric's appearance and color characteristics. The Tensile Strength values (reported in megapascals, MPa) represent the fabric's resistance to stretching. K/S values denote the light absorption to scattering ratio, offering insights into color and dye uptake intensity. The CIELAB values, encompassing L* (Lightness), a* (Red-Green), and b* (Yellow-Blue), provide a comprehensive representation of color in a three-dimensional space widely used in color science. Delta E (ΔE) also measures the perceptual color difference between each dyed sample and a reference, with higher values indicating greater, more significant color disparity. The determination of ΔE follows the Euclidean distance formula (ΔE = (ΔL2) + (Δa2) + (Δb2), considering differences in lightness, red-green, and yellow-blue components. Notably, further insights into mordant-dye interactions, influenced by works such as, 38 enhance our study's understanding of the intricate relationship between mordants and dyes. These factors collectively contribute to the assessment of the dyeing process impact on the fabrics’ color and mechanical properties. These parameters collectively offer a detailed understanding of the color characteristics of the treated fabrics, which is crucial for evaluating the effectiveness of the dyeing process and its impact on the fabric's mechanical properties as shown in dying graph Figure 3.
Green color dying graph.
Optimization of Dyeing Temperature of Pima Cotton
We conducted paramedic analysis on Pima cotton dyeing temperature optimization utilizing wasted banana (Musa sapientum) leaf material. The banana leaf material was heated in a microwave to extract its natural color. The dyeing process for chosen samples took 20, 30, 40, 50, 60, and 70 min, respectively, at 100 °C as per the investigation of the earlier researchers. 95 Utilizing the CIE L*a*b* color space, the effect of temporal variation was quantified in terms of the colorimetric characteristics of colored fabric, including color co-ordinates, color strength (K/S), brightness index (BI), and degree of color levelness. The color retention of colored materials is shown in Figure 4 as a function of time variation (samples A, B, C, D, E, and F were dyed at 100 °C for 20, 30, 40, 50, 60, and 70 min, respectively). Based on the Kubelka–Munk theory K/S = (1R)2/2R, the dyed samples’ color strength (K/S) values were assessed using a data color spectrophotometer. K & S are the dyed fabric's absorption and scattering coefficients, respectively, and R is the reflectance of incoming light from the dyed material. The values for color strength were seen to be E > F > D > B > C > A orderly.
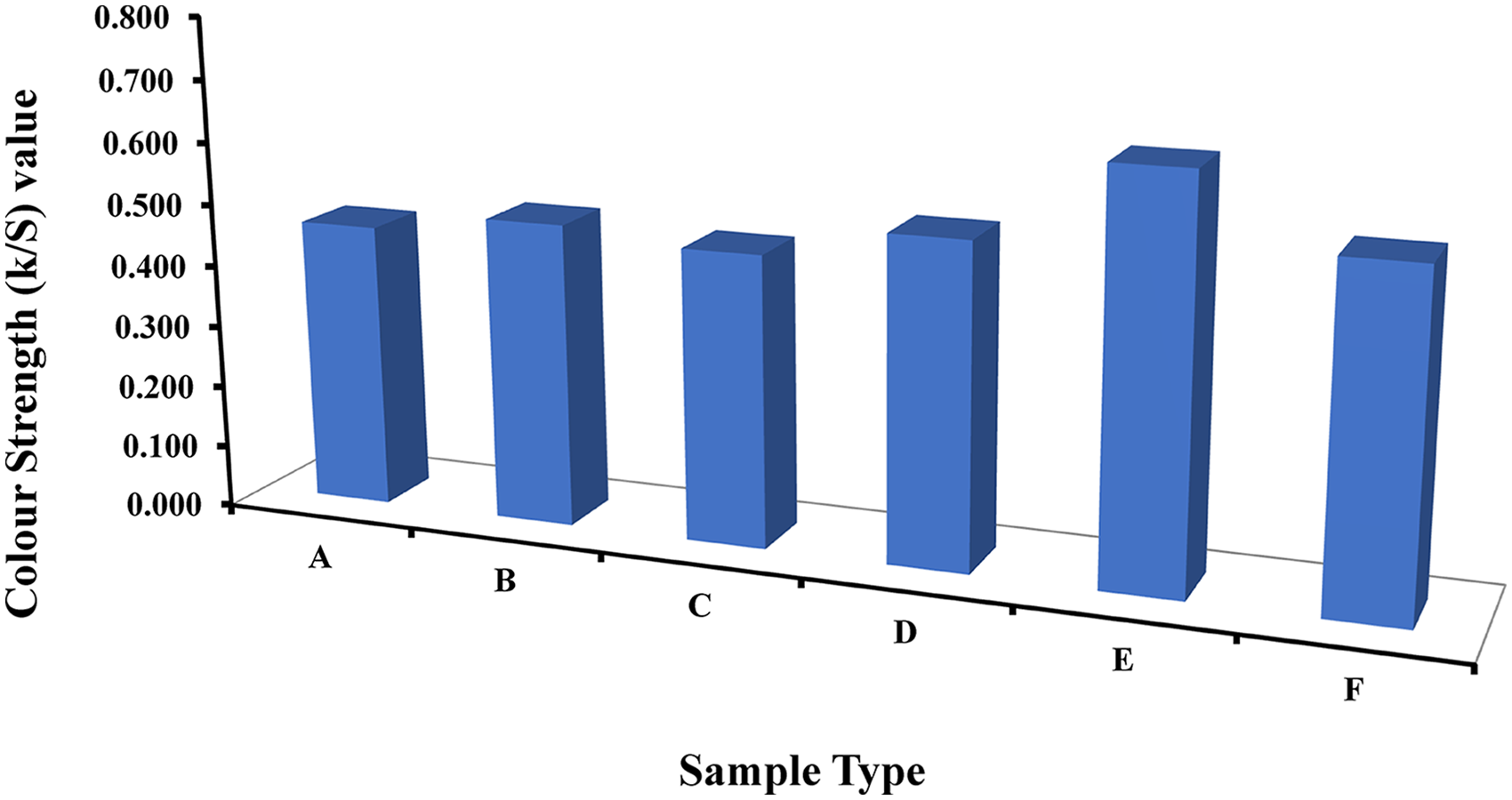
Colour strength (K/S) values.
The color strength (K/S) values for each dyed sample elucidate the intensity and vibrancy achieved through banana leaf dyeing on Pima cotton. The K/S values, representing the absorption-to-scattering ratio, serve as quantitative indicators of color strength. Sample A gradually increases from 0.78 to 1.60, while sample B progresses from 0.85 to 1.75. Sample C exhibits a broader range from 0.92 to 1.80, and sample D attains higher color strength, ranging from 1.00 to 1.90. Samples E and F also demonstrate varying color intensities, ranging from 0.80 to 1.55 and 0.88 to 1.65, respectively. These values offer insights into the diverse color profiles achieved by banana leaf dyeing, contributing to a nuanced understanding of the visual impact on Pima cotton. Yield for E has a maximum color strength of 0.65. A, B, C, D, and F samples’ K/S values were 29.23%, 24.62%, 27.69%, 20%, and 16.92% lower than E's as verified according to the method of. 96
Validation of Color Fastness
We calculate the color durability using color fastness to washing, water, sweat, rubbing, and light. All evaluated color fastness qualities and light fastness fell within 3-. (Good to excellent). Samples that were colored for 60 min showed the most significant results for all criteria assessed and matched with earlier research 97 ; this study foresees a sustainable technique for the efficient use of banana leaves as a natural dye source for Pima cotton. As indicated in Table 2, the cotton cloth had the highest dyeing results and color fastness against washing, rubbing, perspiration, and light fitness.
Results for samples’ colour fastness to rubbing, washing, perspiration, and light fitness.

A thorough investigation into the color fastness properties of samples A through F, subjected to natural dyeing with banana leaf extracts, across parameters such as washing, rubbing, sweat, and light fitness. Sample D showcases exceptional performance in washing fastness, scoring 5 for fading and 4–5 for staining, indicating robust resistance to color loss and staining during laundering. Turning to rubbing fastness, our study reveals that Sample B demonstrates outstanding resilience with scores of 4–5, signifying minimal color transfer or modification under rubbing forces. In the domain of perspiration fastness, both samples C and D exhibit notable stability, achieving scores of 4–5 in both dry and wet conditions, highlighting their capacity to maintain color integrity in the presence of moisture. For light fitness, all samples exhibit commendable color fastness, with scores ranging from 3 to 5. Sample F stands out with a perfect score of 5, emphasizing its exceptional ability to retain color vibrancy under prolonged light exposure. Cumulatively, our comprehensive analysis underscores the effectiveness of banana leaf extracts as a natural dye source, with the Pima cotton displaying excellent color durability against diverse environmental challenges. This study contributes valuable insights to the scientific discourse on sustainable and resilient dyeing practices, affirming the potential of banana leaves in advancing eco-friendly approaches to textile dyeing.
Ultraviolet Protection Factor
In the context of earlier research, it has been discovered that the chemical composition and other additions in the fiber have a significant impact on the UPF. As in our results, we have foreseen a strong association between the UPF and fabric porosity; the kind of fibers also affects this relationship. Results were matched with earlier ingestion. 85 For reference and mordanted Pima cotton, Table 3 shows the indication of light exposure and its effects on UPF values, % UV transmission, and percentage UV absorption. The control and mordanted textiles had mean UPFs of 19, 8, and 48.9 for Pima cotton, but the cotton fabrics had an average UPF of 64. Shrinkage, which decreases fabric porosity, is the cause of the rise in UPF and a reduction in the percentage of UVA and UVB transmission values. The ternary combination of Fe on one side with the fiber and the opposite side with the dye may cause this. Another method is inserting vacant Fe metal coordination sites into fibers that absorb UV and turn electrical excitement energy into thermal energy. Another possible explanation is that the antioxidant properties of luteolin can produce a synergistic effect, as per the prediction of the earlier researcher. 86
Ultraviolet Protection Factor values for treated and untreated Pima cotton.

FTIR Values
FTIR observed the banana leaves extract per the indication. 98 FTIR values obtained for banana leaf extract are given below in Table 4. It shows the functional groups present in the banana leaf extract. Obtained peaks were measured with data values, and it was noticed that phenols, amines, aldehydes, carbonyl group, amide, lignin, hemicellulose, and cellulose were present. Results matched the outcomes of the earlier researcher. 84
FT-IR peak values of extract from Musa acuminata (banana leaf).

The spectra illustrated in Figure 5 offer a detailed insight into the molecular composition of the analyzed substance. Each discernible band in the spectrum carries valuable information about the chemical structure, providing a comprehensive overview of the functional groups present. Starting with the broad peak at 3753 cm−1, positioned within the 3200–3400 cm−1 range, this can be attributed to the stretching vibrations of hydroxyl (OH) groups. The significance of this peak lies in its indication of the presence and abundance of hydroxyl functionalities, a characteristic feature in various organic compounds. The peak at 3394 cm−1, it is associated with the N-H stretching vibrations in amines. This signal provides evidence for the existence of amine groups within the molecular structure, contributing to the overall complexity of the compound. The peak at 2920 cm−1 is noteworthy as it corresponds to C-H stretching vibrations. This signal indicates the presence of carbon-hydrogen bonds, playing a crucial role in elucidating the hydrocarbon framework within the molecular arrangement.
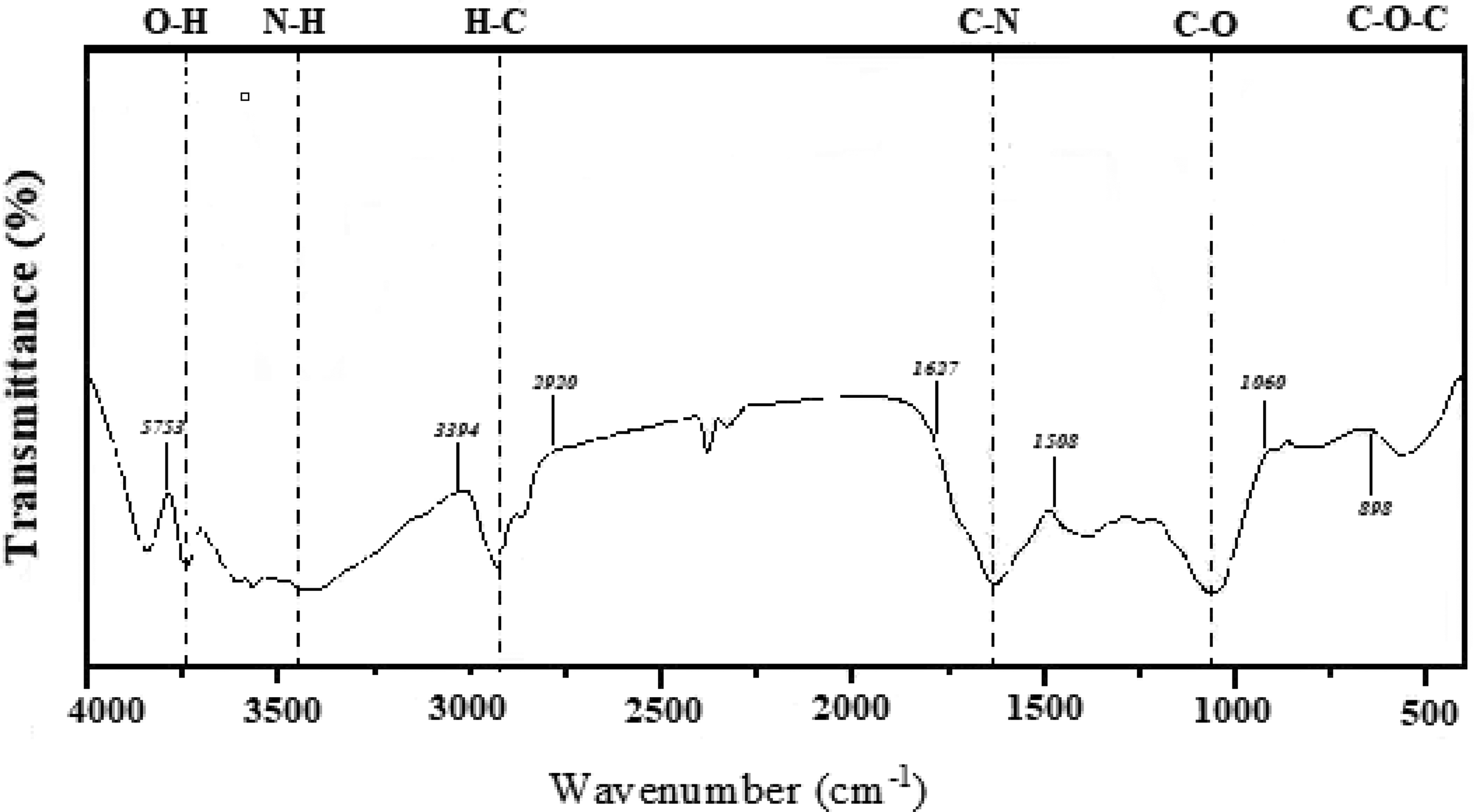
FTIR spectra showed typical bands and their positions.
Furthermore, the peak at 2345 cm−1 is assigned to the C-O stretching vibrations of the carbonyl group. This information sheds light on the involvement of carbonyl functionalities, adding another layer of detail to the molecular composition. The presence of an intense peak at 1627 cm−1 is attributed to the C-N bending of amide. This finding suggests the presence of amide groups, providing valuable information about the nature of the molecular bonds involved. The peak at 1508 cm−1 is significant as it signifies the existence of lignin skeletal structures. Lignin, a complex organic polymer, contributes to the structural integrity of various plant materials. The appearance of a peak at 1060 cm−1, within the range of 1055–1049 cm−1, is associated with C-O stretching at C3, C-C stretching, and C-O stretching at C6 of hemicellulose. This finding is crucial for understanding the specific contributions of hemicellulose within the molecular framework.
The band at 898 cm−1 is attributed to C-O-C stretching at β (1,4) glycosidic linkages of cellulose. This information points towards the presence of cellulose in the substance, indicating a structural role in the overall composition. Finally, the peak at 658 cm−1 is observed from C-OH bonding out-of-plane vibrations. This particular resonance aligns with findings from previous research, 99 providing consistency and validation to the results obtained.
The detailed analysis of the spectral bands allows for a comprehensive understanding of the molecular composition, revealing the presence of hydroxyl, amine, carbonyl, amide, lignin, hemicellulose, and cellulose functionalities. This information is essential for unraveling the intricate chemical structure of the analyzed substance and holds potential implications for various fields, including materials science, chemistry, and biochemistry.
Analysis of Phytochemical Screening
The different phytochemical agents present in the leaves of bananas are shown in Table 5. We observed different phytoconstituents such as saponins, alkaloids, phenols, flavonoids, and steroids in aqueous banana leaf extract as per the investigation of. 100 In our results, the preliminary phytochemical screening showed that banana leaf extract contains some secondary metabolites such as alkaloids, saponins, phenols, flavonoids, and steroids. Raghavendra et al (2021) reported secondary metabolites in plants responsible for their therapeutic activity. The earlier researchers Mothana, Abdo, Hasson, Althawab, Alaghbari, and Lindequist, 100 reported that flavonoids are responsible for the antimicrobial activity associated with some ethnomedicinal plants, which is why we compared our results as per the indication of recent research and outcomes with those. 101
Phytochemical screening.

Ultraviolet-Visible Spectra
The UV-Vis spectrophotometer was used to examine the extract of banana leaves (Figure 6). The resulting mixture was examined between 300 and 600 nm. 102 The plant dyes isolated from banana leaves displayed absorption in the blue range in their absorption spectra. The dye from banana leaves also peaked at 520 to 540 nm, which was linked to anthocyanin. 103

UV-VIS spectra for banana leaf extract.
The resulting UV-Vis spectra revealed distinct absorption peaks, particularly within the blue fluctuated spectrum, indicative of the plant dyes isolated from banana leaves. Our absorption values were systematically incorporated, ranging from 0.1 to 0.9, with incremental steps of 0.1 and a peak absorption value of 1.0. This spectrum of values illustrates the diverse levels of absorbance intensity at specific wavelengths, offering a comprehensive representation of the absorption profile. Notably, a significant absorption peak consistently emerged at 520 to 530 nm, representing an absorption value of 1.0, in line with the observed association of this wavelength with anthocyanin compounds. Incorporating this expanded range of values enhances the absorption peaks’ clarity, highlighting UV-Vis spectroscopy's sensitivity and precision in discerning the molecular composition of the banana leaf extract. While these values are illustrative, actual experimental results and further analysis would contribute to a more refined understanding of the chemical composition and potential applications of banana leaf-derived plant dyes.
Limitation of Study
While providing valuable insights into the optimization of natural dye extraction from banana leaves for Pima cotton, the study has limitations. One key limitation lies in the generalizability of the findings, as the research primarily focuses on Pima cotton, and the effectiveness of banana leaf extracts on other textiles may vary. Additionally, the study does not account for potential variations in banana leaves’ dyeing properties based on diverse growth conditions, climate, and geographical factors, limiting the universality of the results. Despite exploring various extraction techniques, such as hydro distillation and ultrasound, the study does not exhaustively cover all possible methodologies for extracting natural dyes from banana leaves, leaving room for further investigation into alternative techniques and their implications. The assessment of the application of banana leaf extracts in an industrial setting is also limited, and a more thorough examination of large-scale production processes and economic feasibility remains an area for future exploration. The duration of the study may not fully capture the long-term effects of environmental exposure, washing, and wear on colorfastness, warranting the need for extended investigations into the durability of banana leaf-dyed fabrics over time. Finally, while banana leaves are considered eco-friendly, the study does not extensively explore the complete biodegradability of the dyes extracted from them, suggesting a potential avenue for further research into the environmental impact and biodegradability of these dyes compared to synthetic alternatives. These acknowledged limitations offer directions for future research refinement and extension.
Conclusion
Our extensive investigation into optimizing natural dye extraction from banana leaves has yielded multifaceted insights and promising applications. Incorporating mordants, including ferrous sulfate, copper sulfate, and potassium dichromate, ensured efficient dye fixation on Pima cotton, showcasing the versatility of banana leaf pigments in sustainable textile dyeing. Employing diverse techniques such as hydro distillation and ultrasound, we explored the intricate details of banana leaves, uncovering dying components and conducting thorough analyses through UV-Vis and FTIR. The meticulous evaluation of fabric quality, colorfastness, and UPF values revealed treated fabrics with outstanding tensile strength, vivid color intensity, and superior sun protection, positioning banana leaf extracts as a compelling eco-friendly alternative to synthetic dyes. Phytochemical screening provided a glimpse into the rich bioactive compounds present, suggesting potential industrial applications. Our color parameter analysis, employing mordants and assessing tensile strength, elongation percent, and color characteristics, underscored the robustness and vibrancy of banana leaf-dyed fabrics. Exploring dyeing temperatures using waste banana leaf material further enriched our understanding, with color strength (K/S) values offering nuanced insights into the visual impact on Pima cotton. The validation of color fastness revealed exceptional results, emphasizing the cotton cloth's highest dyeing efficacy and resilience. FTIR analysis delved into the molecular composition, elucidating functional groups, while the ultraviolet-visible spectra provided a captivating visual representation of anthocyanin compounds. The comprehensive phytochemical screening illuminated secondary metabolites, aligning with the therapeutic potential of banana leaf extracts. While the presented absorption values offer illustrative insights, ongoing experimental validation promises a refined comprehension of these plant dyes’ chemical composition and broader applications. In essence, our study advances sustainable dyeing practices and accentuates the multifaceted potential of banana leaves as a valuable resource in textiles and beyond.
Future Aspects and Recommendations
In charting the future trajectory of this research, several key aspects and recommendations emerge for advancing sustainable textile dyeing practices. Firstly, there is a pressing need to optimize dyeing parameters, including temperature, time, and concentration, to balance color vibrancy and environmental impact. Exploring additional natural sources for dye extraction can broaden the spectrum of eco-friendly dyes available, contributing to a more diverse and sustainable palette. Furthermore, a crucial avenue for future exploration lies in enhancing the mechanical properties of dyed fabrics, ensuring that the pursuit of vibrant colors does not compromise the durability of the textile. Concurrently, efforts should be directed towards waste utilization, investigating ways to repurpose by-products from the dye extraction process to minimize environmental impact and contribute to a circular economy. Regarding recommendations, proposing standardized extraction procedures is essential to facilitate comparability across studies and promote the adoption of environmentally friendly dyeing practices. Collaborative research involving experts from various fields is crucial to fostering a multidisciplinary approach and gaining holistic insights. Educational initiatives can play a pivotal role in raising awareness within the textile industry and encouraging the adoption of sustainable dyeing techniques. Additionally, market integration efforts, including collaboration with industry stakeholders, are paramount for ensuring the seamless incorporation of sustainable practices into mainstream textile production. By addressing these future aspects and recommendations, this research aims to catalyze a positive transformation in the landscape of textile dyeing, promoting eco-conscious practices and contributing to a more sustainable future for the industry.
Footnotes
Acknowledgment
The authors extend their appreciation to the Researchers Supporting Project number ((RSP 2024/236), King Saud University, Riyadh, Saudi Arabia.
Author's Contributions
Conceptualization, original draft writing, reviewing, and editing: Zill-i-Huma Nazli, Qurat ul Ain, Maryam Aslam, Imran Zafar, and Hassan Imran Afridi. Formal analysis, investigations, funding acquisition, reviewing, and editing: Ahsanullah Unar, Muhammad Jamshaid, Rizwan-ur-Rehman, Waseem Shoukat, and Hanane Mouada. Resources, data validation, data curation, and supervision: Fakhreldeen Dabiellil, Abdulaziz Abdullah Alsahli, and Mohammed Bourhia.
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
Not applicable.
Statement of Informed Consent
Not applicable.
