Abstract
Introduction
Traditional medicine remains an essential part of healthcare, particularly in regions with limited access to modern medical facilities. Hagenia abyssinica and Vernonia amygdalina are widely used in Ethiopia for managing infectious diseases, but comprehensive studies on their phytochemical composition and antibacterial activity are limited.
Objective
This study aimed to evaluate the phytochemical constituents and antibacterial activities of stem bark extracts of H. abyssinica and V. amygdalina to validate their traditional uses and identify potential bioactive compounds.
Methods
Stem bark samples were collected, dried, and sequentially extracted with petroleum ether, chloroform, acetone, and methanol. Standard qualitative phytochemical screening tests were performed on the crude extracts. Antibacterial activity was assessed against Staphylococcus aureus (ATCC 25923) and Escherichia coli (ATCC 25922) using the agar well diffusion method at concentrations of 50, 100, and 150 mg/mL. Ciprofloxacin was used as a positive control, while dimethyl sulfoxide (DMSO) served as a negative control.
Results
Phytochemical screening revealed the presence of saponins, tannins, glycosides, terpenoids, flavonoids, and alkaloids, depending on the solvent and plant species. Methanol extracts yielded the highest percentages (3.32% for H. abyssinica and 3.48% for V. amygdalina). The acetone and methanol extracts exhibited the strongest antibacterial effects, with inhibition zones ranging from 24.67 ± 1.53 to 30.33 ± 0.58 mm against both bacterial strains, in some cases comparable to or greater than ciprofloxacin (25.00 ± 0.00 mm). Non-polar extracts displayed weak or no activity. Statistical analysis (ANOVA, p < 0.05) confirmed significant differences between extracts.
Conclusion
The results confirm that H. abyssinica and V. amygdalina stem barks are rich in phytochemicals with significant antibacterial potential. These findings support their traditional applications and suggest that polar extracts, particularly acetone and methanol fractions, may provide promising leads for the development of new antimicrobial agents.
This is a visual representation of the abstract.
Introduction
Traditional medicine plays a crucial role in healthcare, especially in developing countries where access to modern medical facilities is limited. The World Health Organization reports that approximately 80% of the global population relies on plant-based remedies for their primary healthcare needs, highlighting the importance of traditional medicine in global health systems. 1 In recent years, there has been a growing interest in scientifically validating traditional medicinal practices and exploring the bioactive compounds in medicinal plants, particularly in the context of rising antimicrobial resistance.2,3
Hagenia abyssinica (Bruce) J.F. Gmel., known as “Kosso,” is traditionally used in East Africa for treating gastrointestinal disorders and bacterial infections. 4 While existing research has indicated its antimicrobial potential, study has primarily focused on specific extracts without a comprehensive analysis of its phytochemical profile across various solvents. 5 This lack of detailed phytochemical characterization limits our understanding of its efficacy against resistant strains of bacteria, which is increasingly pertinent given the global rise in antibiotic resistance. 6
Vernonia amygdalina, or “Girawa,” is widely recognized for its medicinal properties across sub-Saharan Africa, particularly for treating malaria and gastrointestinal infections.7,8 Although some studies have documented its antimicrobial activities, there is a significant gap in systematic evaluations of its stem bark extracts against clinically relevant pathogens, particularly multidrug-resistant strains. 9 Furthermore, many studies have focused on leaf extracts, neglecting the potential therapeutic benefits of stem bark, which may contain different phytochemical profiles. 10
Despite the traditional significance of both H. abyssinica and V. amygdalina, comprehensive data on their phytochemical compositions and antibacterial efficacy against common pathogens like E. coli and S. aureus are sparse. This study aims to address these gaps by systematically evaluating the phytochemical constituents and in vitro antibacterial activity of crude extracts from the stem bark of these plants. By doing so, this research seeks to validate traditional uses and identify potential bioactive compounds that could contribute to the development of new antimicrobial therapies, thus offering an innovative approach to combating bacterial infections.
Materials and Methods
Chemicals and Apparatus
The extraction procedures utilized solvents of analytical grade purity, including petroleum ether (boiling range 40-60 °C), chloroform, acetone, and methanol, all procured from certified suppliers. Dimethyl sulfoxide (DMSO, ≥ 99.5% purity) was used to prepare stock solutions of the crude extracts. Ciprofloxacin (standard reference antibiotic) was employed as a positive control. Nutrient agar and Mueller-Hinton agar (Oxoid, UK) were used for culturing and susceptibility testing. A rotary evaporator (Büchi R-300) was employed to concentrate the extracts under reduced pressure at 40–45 °C. All glassware and laboratory apparatus were of borosilicate grade and thoroughly sterilized before use. Phytochemical screening involved standard reagents including concentrated sulfuric acid (H2SO4), hydrochloric acid (HCl), ferric chloride (FeCl3), and Draggendorff's reagent, all of analytical grade.
Plant Material Collection and Preparation
The study focused on two medicinal plants, V. amygdalina and H. abyssinica. Fresh stem barks were collected during the dry season (December 2023 to January 2024), specifically in the early morning, from the South Ari district in the South Omo Zone of the Southern Nations, Nationalities, and Peoples Region (SNNPR), Ethiopia. The plant materials were identified and authenticated at the Jinka University Herbarium Laboratory. Voucher specimens were collected and deposited. After collection, the stem barks were washed thoroughly with tap water to remove adhering soil and dust particles. They were then chopped into small pieces and left to air dry under shade at ambient temperature (25-27 °C) in a well-ventilated environment to prevent direct exposure to sunlight. The drying process continued for two weeks until constant weight was achieved. Subsequently, the dried materials were pulverized into a fine powder using a mortar and pestle and stored in airtight containers at room temperature until further extraction and analysis.
Extraction of Plant Material
The air-dried and powdered stem bark materials of H. abyssinica and V. amygdalina were subjected to sequential solvent extraction using petroleum ether, chloroform, acetone, and methanol. For each extraction, 200 grams of powdered plant material were soaked in 2 liters of solvent, maintaining a 1:10 (w/v) ratio to ensure optimal solvent penetration and efficient extraction of phytochemicals.
The powdered materials were soaked in each solvent for 72 h at room temperature (25-27 °C) with occasional agitation to enhance extraction efficiency. The mixtures were filtered first through Whatman No. 1 filter paper, followed by cotton wool to remove particulate matter. The filtrates were concentrated using a rotary evaporator (Büchi R-300) under reduced pressure at a bath temperature of 40–45 °C to obtain crude extracts. The concentrated extracts were then dried further by exposing them to a stream of cold, dry air in a fume hood, ensuring that the relative humidity was maintained below 50% to prevent fungal contamination and degradation of sensitive bioactive compounds. The dried crude extracts were transferred into pre-weighed, airtight amber glass containers and stored at 4 °C until further antibacterial assays. The percentage yield of each extract was calculated based on the dry weight of the extract relative to the initial weight of the plant material.
Phytochemical Screening
Preliminary phytochemical screening of the crude extracts was performed to qualitatively identify major classes of secondary metabolites, including alkaloids, flavonoids, saponins, tannins, terpenoids, glycosides, and anthraquinones. Standard procedures were followed as described by Harborne, 11 with adaptations from Sofowora 12 and Trease and Evans 13 where applicable.
Test for Alkaloids (Dragendorff's Test):
Approximately 0.5 g of each extract was dissolved in 10 mL of 1% hydrochloric acid, heated gently, and filtered. A few drops of Dragendorff's reagent were added to 2 mL of the filtrate. The formation of an orange or reddish-brown precipitate indicated the presence of alkaloids.
Test for Flavonoids (Ammonia Test):
An aqueous portion of the extract was treated with 5 mL of dilute ammonia solution. Concentrated sulfuric acid (1 mL) was then added. The appearance of a yellow coloration, which disappeared upon standing, confirmed the presence of flavonoids.
Test for Saponins (Froth Test):
About 0.5 g of each extract was vigorously shaken with 5 mL of distilled water in a test tube. Persistent frothing that lasted for at least 15 min indicated the presence of saponins. The froth was then mixed with three drops of olive oil, and the formation of a stable emulsion confirmed the result.
Test for Tannins (Ferric Chloride Test):
Approximately 0.5 g of the extract was boiled in 10 mL of distilled water and filtered. A few drops of 0.1% ferric chloride solution were added to the filtrate. A blue-black or greenish-brown precipitate indicated the presence of tannins.
Test for Terpenoids (Salkowski Test):
About 0.5 g of the extract was dissolved in 2 mL of chloroform. Concentrated sulfuric acid (3 mL) was carefully added along the sides of the test tube to form a distinct layer. A reddish-brown coloration at the interface indicated the presence of terpenoids.
Test for Anthraquinones (Borntrager's Test):
About 0.5 g of the extract was boiled with 10 mL of sulfuric acid (H2SO4) and filtered while hot. The filtrate was shaken with 5 mL of chloroform, and the chloroform layer was separated. After adding 1 mL of dilute ammonia solution to the chloroform layer, a pink or red color in the aqueous phase indicated the presence of free anthraquinones.
Test for Cardiac Glycosides (Keller-Killiani Test):
Approximately 0.5 g of each extract was dissolved in 5 mL of distilled water. To this, 2 mL of glacial acetic acid containing a trace amount of ferric chloride (FeCl3) was added. The mixture was then carefully under-layered with 1 mL of concentrated sulfuric acid by tilting the test tube. A brown ring at the interface indicated the presence of cardiac glycosides. A violet ring below the interface and a greenish layer above were additional confirmatory indications of cardiac glycosides.
Antimicrobial Assay
Preparation of Different Concentrations of Crude Extracts
To evaluate the antibacterial properties, stock solutions of the crude extracts were prepared at a concentration of 200 mg/mL by dissolving 2 grams of each dried extract in 10 mL of dimethyl sulfoxide (DMSO). The DMSO used for this purpose was of analytical grade (≥99.5% purity) and was sterile-filtered using a 0.22 μm membrane filter to eliminate potential microbial contaminants.
The resulting stock solutions were thoroughly vortexed to ensure complete dissolution of the extracts. From these stock solutions, working concentrations of 50 mg/mL, 100 mg/mL, and 150 mg/mL were prepared by serial dilution using sterile DMSO. All prepared solutions were stored in sterile, airtight containers and maintained at 4 °C until use in antimicrobial assays to preserve their stability and prevent microbial contamination.
Collection of Test Organisms
The bacterial strains used in this study were obtained from the Ethiopian Public Health Institute (EPHI), Addis Ababa, Ethiopia. The selected organisms were S. aureus (ATCC 25923) and E. coli (ATCC 25922), which are standard reference strains widely used for antimicrobial susceptibility testing. Upon collection, the bacterial strains were sub-cultured on nutrient agar to ensure purity and viability. These standard strains were chosen to enable reproducibility and comparison of antibacterial activity with global reference data. Ciprofloxacin (5 μg/disc) served as the positive control, while sterile DMSO (used as a solvent for extract preparation) functioned as the negative control to validate the specificity of antibacterial effects.
Culturing and Standardization of Inoculum
Each bacterial strain was freshly cultured on Mueller-Hinton agar plates and incubated at 37 °C for 18–24 h to obtain isolated colonies. Well-isolated colonies were then picked using sterile inoculating loops and suspended in 5 mL of sterile 0.85% saline solution in sterile test tubes. The bacterial suspension's turbidity was adjusted to match that of the 0.5 McFarland Standards, which corresponds to approximately 1.5 × 108 CFU/mL. 14 The McFarland Standard was prepared by mixing 0.5 mL of 1.175% (w/v) barium chloride dihydrate (BaCl2·2H2O) with 99.5 mL of 1% (v/v) sulfuric acid (H2SO4), resulting in a fine barium sulfate precipitate that provided the desired turbidity.
The turbidity of the bacterial suspension was visually compared against the McFarland Standard tube held against a white background with contrasting black lines. When necessary, adjustments were made by adding more bacterial colonies or sterile saline until the turbidity matched the standard. The standardized inoculum was used within 15 min of preparation for the antibacterial susceptibility tests to maintain cell viability and concentration consistency.
Antibacterial Activity of Crude Extracts
The antibacterial activities of the crude extracts were assessed using the agar well diffusion method, following standardized protocols described by the Clinical and Laboratory Standards Institute 15 with necessary modifications. Mueller-Hinton agar plates were uniformly inoculated with the standardized bacterial suspension (adjusted to 0.5 McFarland standard, ∼1.5 × 108 CFU/mL) using a sterile cotton swab to ensure an even lawn of bacterial growth. 16
Using a sterile cork borer of 6 mm diameter, wells were aseptically punched into the agar. Each well was filled with 100 µL of the respective plant extract solution at concentrations of 50 mg/mL, 100 mg/mL, and 150 mg/mL. The same volume (100 µL) of DMSO (negative control), prepared at the highest concentration used in extract dilutions, was also loaded into wells to assess any potential solvent-related inhibition. Ciprofloxacin (5 µg/disc) was included on each plate as the positive control to benchmark the antibacterial efficacy of the plant extracts.
The plates were allowed to stand at room temperature (25 °C) for 30 min to enable diffusion of the test solutions into the agar matrix. Subsequently, the plates were inverted and incubated at 37 °C for 24 h. After incubation, the diameters of inhibition zones around the wells (including the diameter of the well) were measured in millimeters (mm) using a digital caliper. 17 All experiments were performed in triplicate, and the mean zone of inhibition was calculated for each concentration. 18 Inhibition zones measuring greater than 15 mm were classified as exhibiting strong antibacterial activity, zones between 10 mm and 15 mm were considered to have moderate activity and those less than 10 mm were categorized as having weak activity.
Statistical Analysis
All experiments were performed in triplicate, and the results are presented as mean ± standard deviation (SD). Data were analyzed using one-way analysis of variance (ANOVA) to determine significant differences between groups. When the ANOVA indicated significance, Tukey's post hoc test was applied for multiple comparisons. Statistical analyses were performed using SPSS software (version 25.0; IBM Corp., Armonk, NY, USA). A p-value < 0.05 was considered statistically significant.
Results and Discussion
Extraction Yield
The percentage yields of crude extracts obtained from the stem barks of H. abyssinica and V. amygdalina using petroleum ether, chloroform, acetone, and methanol are summarized in Table 1 and 2. For both plants, extraction yield increased with the polarity of the solvent, indicating a higher abundance of polar secondary metabolites. The highest yields were recorded for methanol extracts: 3.32% for H. abyssinica and 3.48% for V. amygdalina. Conversely, petroleum ether extracts yielded the lowest quantities, suggesting limited non-polar constituents in the stem barks.
Percentage Yield of Crude Extracts of H. abyssinica Stem Bark.
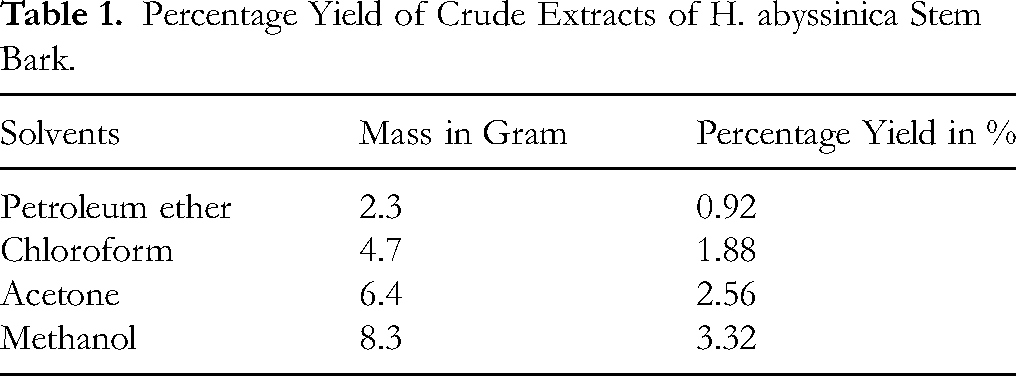
Percentage Yield of Crude Extracts of V. amygdalina Stem Bark.
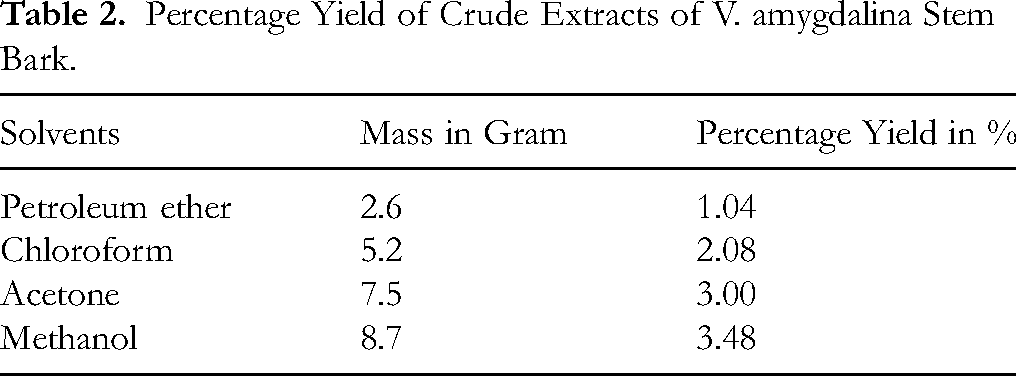
Phytochemical Screening
Preliminary phytochemical screening results of the two medicinal plants conducted with different reagents were recorded (Table 3).
Phytochemical Constituents of H. abyssinica and V. amygdalina Stem Bark Extracts.
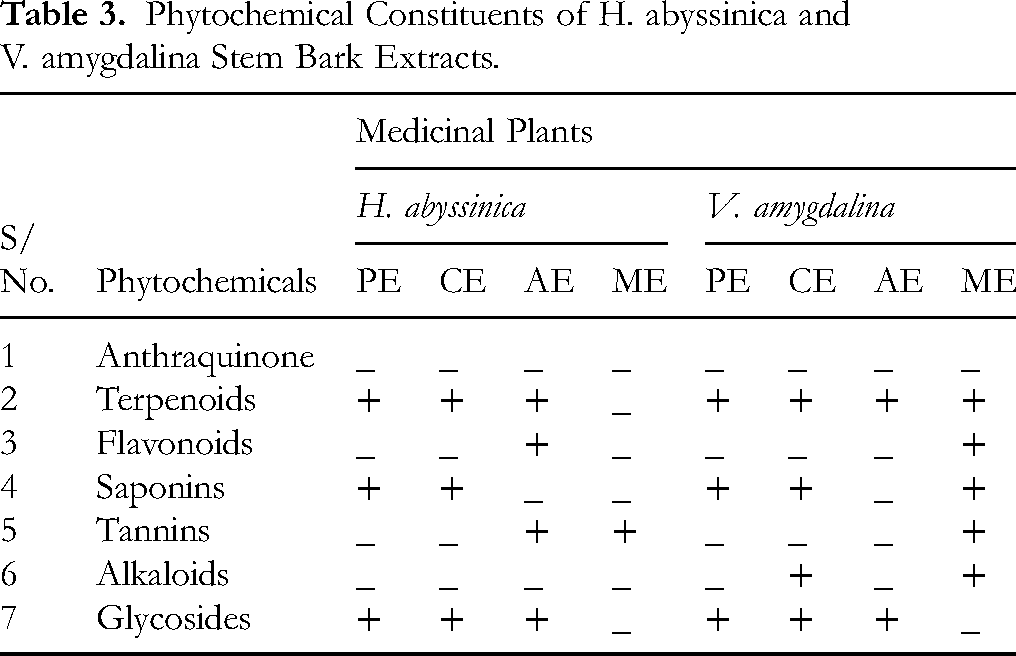
The preliminary phytochemical screenings was conducted on four different crude extracts (petroleum ether, chloroform, acetone, and methanol extract) of H. abyssinica and V. amygdalina stem barks. In the current study, terpenoids, saponins, and glycosides were revealed in the petroleum and chloroform extracts of H. abyssinica stem bark. The acetone extracts contained terpenoids, flavonoids, tannins, and glycosides, while the methanol extract contained only tannins. Anthraquinone and alkaloids were absent in all extracts of H. abyssinica stem bark.
In the present study, the preliminary screening of V. amygdalina stem barks showed the existence of terpenoids, saponins, and glycosides in the petroleum ether extracts. The chloroform extract contained terpenoids, saponins, alkaloids and glycosides. In acetone extracts, only the presence of terpenoids and glycosides were revealed, while more of secondary metabolites such as terpenoids, flavonoids, saponins, alkaloids and tannins were found in the methanol extracts. Terpenoids were found in all extracts of V. amygdalina stem barks while anthraquinones were not present in all extracts (Supplementary Figure S1 and S2).
Antimicrobial Activity Results
The antibacterial activity of the crude extracts was evaluated against S. aureus (ATCC 25923) and E. coli (ATCC 25922) using the agar well diffusion method. The mean diameters of inhibition zones (in mm, expressed as mean ± SD, n = 3) are summarized in Table 4.
The in-Vitro Antibacterial Activities of Crude Extracts and References.

(Key: PE: Petroleum ether Extract, CE: Chloroform Extract, AE: Acetone Extract, ME: Methanol Extract, CI: and Ciprofloxacin).
A study by Wolde 19 indicated that H. abyssinica extract has a significant antibacterial effect against S. aureus and S. typhi. Kifle 20 also noted considerable antidiarrheal properties in mice from the leaf solvent fractions of H. abyssinica. Additionally, Biruktayet 21 reported its extensive use for strong anti-tapeworm activity.
In the present study, concentrations of 50 mg/mL, 100 mg/mL, and 150 mg/mL were used to assess the antibacterial activity of crude extracts from various medicinal plants (Supplementary Figure S3, S4, and S5). Among the extracts tested, the acetone extract of H. abyssinica stem bark exhibited the highest activity against E. coli, with inhibition zones of 28.33 ± 1.15 mm, 30.33 ± 0.58 mm, and 24.67 ± 1.53 mm at 50, 100, and 150 mg/mL, respectively, which were comparable to or greater than the standard antibiotic ciprofloxacin (25.00 ± 0.00 mm). Similarly, the methanol extract of H. abyssinica displayed strong inhibition, particularly against S. aureus, producing inhibition zones of 24.67 ± 0.58 mm, 24.67 ± 0.58 mm, and 28.33 ± 1.15 mm across the three concentrations.
Previous research suggests that V. amygdalina contains various bioactive compounds that contribute to its pharmacological effects, including antimicrobial, antimalarial, antioxidant, and anti-inflammatory properties.10,22 In the current study, the acetone extracts showed the strongest antibacterial activity against S. aureus, with inhibition zones of 15.33 ± 0.58 mm, 16.67 ± 0.58 mm, and 29.00 ± 1.00 mm, while the methanol extract also exhibited substantial effects (20.33 ± 0.58 mm at 150 mg/mL). In contrast, petroleum ether extracts of both plants generally showed weak or no activity, with values often below 10 mm.
Moderate antibacterial activity was observed with chloroform extracts of both species, such as 15.67 ± 0.58 mm for H. abyssinica against S. aureus at 100 mg/mL, and 14.67 ± 0.58 mm for V. amygdalina against S. aureus at 150 mg/mL.
Statistical analysis using one-way ANOVA followed by Tukey's post hoc test confirmed that these differences were significant (p < 0.05). Extracts prepared with polar solvents (acetone and methanol) consistently exhibited stronger antibacterial effects compared to non-polar solvents (petroleum ether and chloroform), supporting the view that polar phytochemicals such as flavonoids, tannins, and glycosides contribute substantially to antibacterial activity.
Limitations of the Study
This study was limited to in vitro antibacterial assays using only two reference bacterial strains (S. aureus ATCC 25923 and E. coli ATCC 25922). The results may not fully represent the activity of the extracts against a wider range of clinically relevant or multidrug-resistant pathogens. Further studies involving broader microbial panels, advanced chromatographic techniques, and in vivo models are necessary to validate the therapeutic potential and safety of these plant extracts.
Conclusions
The present in vitro experimental study investigated the antibacterial effectiveness of stem bark extracts of Hagenia abyssinica and Vernonia amygdalina against Staphylococcus aureus (ATCC 25923) and Escherichia coli (ATCC 25922). Extraction yields increased with solvent polarity, with methanol providing the highest yields (3.32% for H. abyssinica and 3.48% for V. amygdalina). Phytochemical analysis confirmed the presence of secondary metabolites including saponins, tannins, glycosides, terpenoids, flavonoids, and alkaloids.
Antibacterial testing using the agar well diffusion method revealed that polar solvent extracts (acetone and methanol) demonstrated the strongest inhibitory activity, with inhibition zones ranging from 24.67 ± 1.53 mm to 30.33 ± 0.58 mm. These values were in some cases equal to or greater than the inhibition zone of ciprofloxacin (25.00 ± 0.00 mm), the standard reference drug. In contrast, non-polar extracts (petroleum ether and chloroform) showed weak or no activity, typically below 10 mm. Statistical analysis (ANOVA, p < 0.05) confirmed significant differences among extracts, underscoring the greater antibacterial efficacy of polar solvent fractions.
Overall, the findings validate the traditional medicinal use of H. abyssinica and V. amygdalina in the treatment of bacterial infections. The results suggest that polar phytochemicals, particularly flavonoids, tannins, and glycosides are likely responsible for the observed antibacterial effects. These extracts represent promising sources of natural antibacterial agents and warrant further investigation for isolation of active compounds, mechanistic studies, and in vivo validation to establish their therapeutic potential.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251387880 - Supplemental material for Phytochemical Screening and Antibacterial Assay of the Stem Bark of Hagenia abyssinica and Vernonia amygdalina
Supplemental material, sj-docx-1-npx-10.1177_1934578X251387880 for Phytochemical Screening and Antibacterial Assay of the Stem Bark of Hagenia abyssinica and Vernonia amygdalina by Tadasa Hailu, Mesfin Tadesse, Yeshambel Birhanu and Misganew Desta in Natural Product Communications
Footnotes
Acknowledgements
We are thankful to Jinka University for their generous financial support. We also appreciate the Chemistry and Biology department of Jinka University for granting us access to their laboratory facilities. Additionally, we sincerely thank the Ethiopian Public Health Institute of South Omo Zone for their valuable assistance in providing laboratory facilities for antimicrobial assay.
Ethical Approval
To conduct this study, ethical clearance was obtained from the Ethical Review Committee of the College of Natural and Computational Science, University of Jinka.
Author Contributions
Tadasa Hailu, Mesfin Tadesse, Yeshambel Birhanu, and Misganew Desta contributed equally to this work. Tadasa Hailu and Mesfin Tadesse conducted the phytochemical analysis. and antibacterial assays. Yeshambel Birhanu and Misganew Desta conducted the antibacterial assays. All authors participated in data analysis, interpretation, writing and revision of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Jinka University.
Disclosure Statement
The authors report there are no competing interests to declare.
Statement of Human and Animal Rights
There are no animal subjects in this article and informed consent is not applicable.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplementary Figure S1 - Phytochemical screening of Hagenia abyssinica stem barks
Supplementary Figure S2 - Phytochemical screening of Vernonia amygdalina stem barks
Supplementary Figure S3 - Antibacterial activity of H. abyssinica and V. amygdalina stem barks crude extracts with conc. of 50 mg/mL
Supplementary Figure S4 - Antibacterial activity of H. abyssinica and V. amygdalina stem barks crude extracts with conc. of 100 mg/mL
Supplementary Figure S5 - Antibacterial activity of H. abyssinica and V. amygdalina stem barks crude extracts with conc. of 150 mg/mL
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

