Abstract
Keywords
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, and irreversible fibrotic interstitial lung disease, clinically characterized by dyspnea, inspiratory crackles, and coughing bouts lasting more than 6 months. 1 IPF usually presents as typical interstitial pneumonia both radiologically and histopathologically.2,3 High-resolution computed tomography of the chest reveals honeycomb changes to the lung tissue, as well as peripheral, subpleural, and bibasilar reticular opacities.1,4 IPF is most common in men aged > 50 years with a history of smoking, and the average life expectancy after diagnosis is approximately 3 years, with a very poor prognosis. 5 Most patients diagnosed with IPF die of the disease, mainly because of the progression of respiratory failure. 6
The currently approved drugs for the treatment of IPF are the antifibrotic agents: pirfenidone and nintedanib, both of which have been reported to delay the decline in lung function and reduce mortality in IPF patients. 7 However, the drugs neither can restore the condition of fibrotic lungs or stop the progression of the disease nor do they show consistent effects on related symptoms. 8 In addition, 20% to 30% of patients taking these drugs cannot continue their use for a long term because of side effects. 9 The side effects of pirfenidone include photosensitivity, dyspepsia, and decreased appetite, 10 whereas nausea and diarrhea have been reported with the use of nintedanib. 11 Owing to the limitations of the existing treatments, there is growing interest in the development of more effective and safe treatments.
IPF has long been treated in traditional Korean medicine under the category of illnesses known as “stagnated cough” and “lung atrophy” because dry cough and progressive dyspnea are its main symptoms. This disease progresses pathologically by damaging “fluid and humor,” leading to weakness of the lungs and a pattern of “lung atrophy.” 12 A screening study was conducted using Farfarae Flos, Trichosanthis Semen, Lilii Bulbus, Adenophorae Radix (ADR), Asteris Radix, and Scrophulariae Radix, which are all lung-moistening herbal medicines with proven anti-inflammatory effects; the efficacy of ADR was reportedly higher than that of the other remedies. 13
In traditional Korean medicine, ADR has been widely used to treat respiratory diseases, such as colds, asthma, and bronchitis, owing to its efficacy in “nourishing yin, engendering fluid, clearing lung fire, dispelling phlegm, and suppressing cough.” 14 ADR has been reported to have pharmacological effects, including antiasthmatic, 15 antioxidant, 16 and antitumor 17 effects. Hu et al, 18 demonstrated dose-dependent antitussive, expectorant, and anti-inflammatory effects of ADR. However, there are few published reports on the effects of ADR on IPF. Therefore, we investigated the antifibrotic efficacy and mechanism of action of ADR in a mouse model of bleomycin-induced pulmonary fibrosis.
Materials and Methods
Animals
Male ICR mice (7 w old, 31 ± 2 g; Daehan Biolink, Seoul, Korea) were used after a week of adaptation in a constant temperature and humidity chamber (22 ± 1 °C, 55 ± 10%) with a 12-h light/dark cycle and ad libitum access to food and water. The experimental animal protocol was approved by the Kyung Hee University Institutional Animal Care and Use Committee (KHUASP(SE)-18-177).
Sample Preparation
ADR, a root of Adenophora triphylla (Thunb.) A. DC collected in October from Hunan Province, China, was obtained from Kyung Hee Herb Pharm. Specimen HLL-004 was stored at the Department of Herbal Pharmacology, College of Korean Medicine, Kyung Hee University. A 200-g sample of ADR was placed in a reflux extractor with 1500 mL of water and heated for 2 h at a temperature just below the boiling point, to obtain 240 mL of ADR water extract. The extract was then filtered through a filter paper and concentrated using a rotary vacuum evaporator. The concentrate was then dried in a freeze dryer (FD-5508, IlShin Lab Co.) to obtain 41.75 g of dried extract (yield 20.88%).
Pulmonary Fibrosis Mouse Model Induction and Sample Administration
Pulmonary fibrosis was induced by direct intratracheal injection of bleomycin (Donga-ST) into the bronchi of each mouse via intratracheal instillation (i.t.). 19 Briefly, after each animal was anesthetized with 2% isoflurane (O2:N2O = 30:70), the throat skin was incised to expose the trachea, and 2 mg/kg bleomycin was directly injected through the trachea using an insulin syringe, after which the incision was sutured.
ADR was orally administered twice daily for 10 d after bleomycin administration. The mice were divided into the following groups (8 animals each): The normal group (NOR) consisted of mice without instillation or sample administration; the sham group (SHAM) received only saline instillation; the control group (CON) received bleomycin instillation and distilled water (untreated control). The 3 treatment groups (ADR 30, ADR 100, and ADR 300) received bleomycin instillation and sample administration of 30, 100, or 300 mg/kg of ADR.
Isolation of Bronchoalveolar Lavage Fluid and Lung Tissue
On the 11th day after induction, the mice were euthanized with urethane (1.2 g/kg, i.p.), and an incision was made from the anterior neck to the anterior chest to expose the lungs and trachea. The trachea was incised and a 24G Angiocath catheter was inserted. Bronchoalveolar lavage fluid (BALF) was collected by infusing the bronchus 3 times with 0.5 mL of phosphate-buffered saline and recovering it using a syringe. Lung tissues were isolated, fixed with 4% paraformaldehyde, and used for tissue staining.
Measurement of Cell Counts in BALF
The collected BALF samples were centrifuged at 4 °C for 15 min at 67.5 g. Subsequently, the precipitate was resuspended in phosphate-buffered saline, and the total cell count was measured using an automatic hemocytometer. The precipitate was then placed in a Cytospin III and centrifuged again at 8.4 g for 3 min. After confirming that the cells had been collected on a cytospin-only slide, they were fixed in ethanol, stained with H&E, and trypan blue staining was performed. Thereafter, the ratios of total cells, macrophages, lymphocytes, and granulocytes among 200 cells were randomly measured using a microscope (200×).
Histological Analysis of Lung Tissue
The lungs were extracted from the euthanized mice and fixed in 4% paraformaldehyde to prepare paraffin blocks. Lung slices (5-μm thick) were then stained with Masson's trichrome to investigate collagen deposition due to fibrosis in the lung tissue. To evaluate the degree of pulmonary fibrosis, 2 observers quantified, analyzed, and measured the degree of fibrosis in each tissue using the Ashcroft scale. 20
Measurement of Transforming Growth Factor-β and α-Smooth Muscle Actin in Lung Tissue
The levels of transforming growth factor-β (TGF-β) and α-smooth muscle actin (α-SMA) in lung tissue were measured using enzyme-linked immunosorbent assay (ELISA). The collected lung tissue was homogenized using a Bullet Blender Tissue Homogenizer (NextAdvance), with a radioimmunoprecipitation assay as lysis buffer. Further, the sample was placed on ice and allowed to react for 30 min. Centrifugation was performed at 4 °C for 30 min at 2823 g, and the supernatant was stored at −80 °C after centrifugation. The supernatant was analyzed using a TGF-β ELISA Kit (R&D Systems) and an α-SMA ELISA Kit (Novus Biologicals) according to the manufacturer's protocol. The absorbance was measured at 450 nm using a microplate reader (SpectraMax® Plus, Molecular Devices Corp.).
Statistical Analysis
All data are expressed as mean ± standard error (SEM). One-way analysis of variance followed by Dunnett's test using GraphPad Prism version 5.0 (GraphPad Inc.) was used to compare each treatment group with the control group.
Results
Changes in Pulmonary Fibrotic Histology and Body Weight
The body weights of the CON mice were significantly different from those of the NOR and SHAM groups, with a weight loss of more than 10% at 4 days after bleomycin administration (Figure 1D). Additionally, alveolar wall thickening, alveolar collapse, and massive deposition of inflammatory cells and collagen in the lung interstitium were evident in the lungs of the CON group, and fibrosis was observed in lung tissues. In contrast, the ADR groups showed a decrease in histological fibrosis indicators compared with the CON group. Particularly, in the ADR 300 group, a notable decrease was observed in the degree of alveolar and bronchial wall thickening, inflammatory cells, and collagen deposition (Figure 1A and B). In terms of the Ashcroft scale, a pulmonary fibrosis indicator, lungs from the CON group showed a significant increase compared to those from the NOR and SHAM groups. In the lungs of the ADR groups, a decreasing trend in fibrosis was evident compared to that observed in the CON group; in particular, a significant reduction in fibrosis was observed in the ADR 300 group (P < .05) (Figure 1C). However, there was no significant increase in body weight in the ADR groups compared with the corresponding values in the CON group (Figure 1D).

Effects of ADR on pulmonary fibrotic histological change and body weight in a bleomycin-induced pulmonary fibrosis mouse model. (A, B) Lung sections stained with Masson's trichrome, the scale bar in A and B is 400 and 40 μm, respectively. (C) Ashcroft scores. (D) Changes in body weight. After the induction of pulmonary fibrosis using bleomycin, body weight was measured every morning before ADR administration. Ten days after ADR administration, the left lung was excised and stained with Masson's trichrome for histological analyses. *** P < .001 versus CON, ** P < .01 versus CON, * P < .05 versus CON. Abbreviations: ADR, Adenophorae Radix; NOR, normal group; SHAM, sham group; CON, control group; ADR groups, groups treated with water extract of ADR at different concentrations.
Inflammatory Cells in BALF
Compared to the results from the SHAM group, the number of total cells, macrophages, lymphocytes, and granulocytes increased in the BALF of CON mice. The ADR groups showed a decrease in the number of total cells, macrophages, lymphocytes, and granulocytes in BALF compared to the CON group. ADR 300 caused a significant decrease in the total number of cells, macrophages, lymphocytes, and granulocytes compared with the CON group (Figure 2).
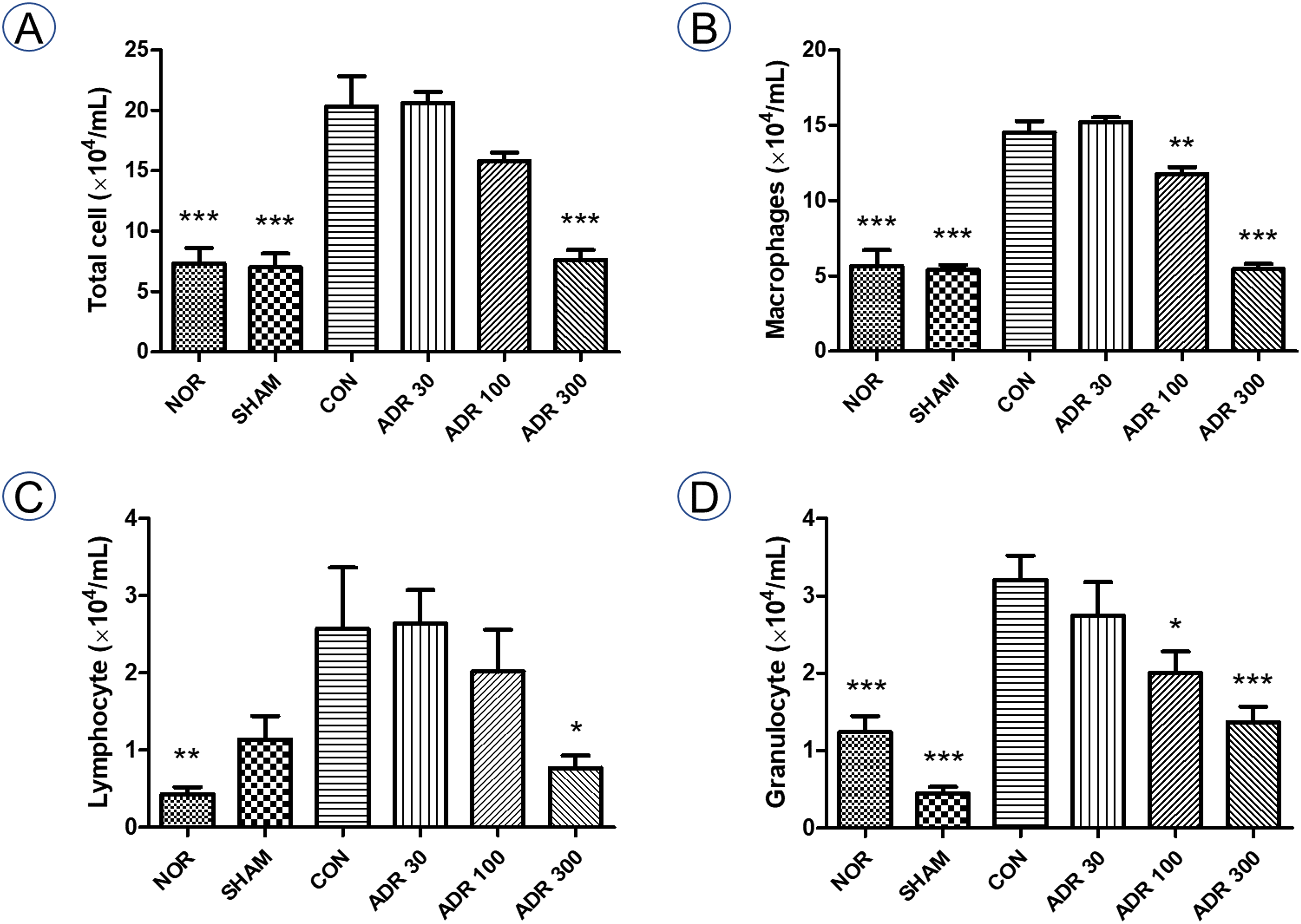
Effects of ADR on inflammatory cells in BALF in a bleomycin-induced pulmonary fibrosis mouse model. After 10 d of ADR administration in a bleomycin-induced pulmonary fibrosis mouse model, the BALF was analyzed. Data are shown as mean ± SEM. *** P < .001 versus CON, ** P < .01 versus CON, *P < .05 versus CON. Abbreviations: BALF, bronchoalveolar lavage fluid; ADR, Adenophorae Radix; NOR, normal group; SHAM, sham group; CON, control group; ADR groups, groups treated with water extract of ADR at different concentrations.
TGF-β and α-SMA Levels in Lung Tissue
Lungs from the CON group exhibited significantly increased levels of TGF-β and α-SMA (35.35% and 65.48%, respectively), compared with the corresponding values in the SHAM group. However, the ADR groups showed a trend of decreasing TGF-β and α-SMA levels compared to the corresponding values in the CON group. ADR 300 administration resulted in significant reductions (17.12%) in α-SMA levels compared with those in the no treatment (CON) group (Figure 3).

Effects of ADR on TGF-β and α-SMA production in lung tissue in a bleomycin-induced pulmonary fibrosis mouse model. After 10 d of ADR administration in a bleomycin-induced pulmonary fibrosis mouse model, the left lung was extracted and homogenized and the TGF-β and α-SMA levels were analyzed. Data are shown as mean ± SEM. *** P < .001 versus CON, ** P < .01 versus CON, * P < .05 versus CON. Abbreviations: ADR, Adenophorae Radix; TGF-β, transforming growth factor-β; α-SMA, α-smooth muscle actin; NOR, normal group; SHAM, sham group; CON, control group; ADR groups, groups treated with water extract of ADR at different concentrations.
Discussion
In this study, we used a bleomycin-induced pulmonary fibrosis mouse model to investigate the effects of ADR on IPF. ADR was found to decrease the level of histological indicators of fibrosis (including the Ashcroft score values) and the number of inflammatory cells in the BALF, as well as TGF-β and α-SMA levels in the lung tissue.
In the currently recognized pathogenic model of IPF, symptoms arise from repeated injury to the lung epithelial cells and repair dysfunction.21,22 Although the exact mechanism of fibrosis in IPF is not yet fully understood, it is established that fibrosis is induced when various microinjuries occur in pulmonary epithelial cells and that growth factors secreted from damaged epithelial cells promote fibroblast proliferation, activation, and differentiation into myofibroblasts. Myofibroblasts are resistant to cell death, have a stronger proliferation ability, and contribute to collagen deposition by secreting extracellular matrix (ECM). 23 In addition, when the epithelial cells of the alveoli are damaged, an exudate is formed in the alveoli and destruction of the alveolar wall commences, resulting in the loss of surfactants and collapse of the alveoli. Despite this damage, alveolar re-epithelialization does not form properly in IPF, and mesenchymal cells migrate from the interstitial tissue to the alveoli because of basement-membrane damage. Excessive accumulation of ECM components, such as fibroblasts or collagen, prevents collapsed alveoli from expanding. When ECM deposition occurs between alveolar epithelial cells and vascular endothelial cells, destruction of lung tissue and honeycomb-shaped sites of damage are evident.24,25 When these changes cause chronic and repetitive damage to the alveolar epithelium, fibrosis and remodeling occur within the alveoli. 21 Thus, inflammatory cells, fibroblasts, myofibroblasts, and alveolar epithelial cells play important roles in fibrosis progression. Therefore, drugs that prevent inflammation, inhibit the proliferation of fibroblasts or their differentiation into myofibroblasts or inhibit collagen synthesis may be potential candidates for the treatment of IPF.26,27
To investigate the effect of ADR on lung tissue, the degree of collagen deposition and histological changes in the cells were examined using Masson's trichrome staining of the extracted lung tissue sections. The sections were evaluated using the Ashcroft scale, the most commonly used scoring system (scores of 0-8) for assessing pulmonary fibrosis in bleomycin-induced animal models. 28 The experimental results showed that ADR reduced the degree of pulmonary fibrosis, as reflected by the Ashcroft scale scores. A significant reduction was observed in samples from the lungs treated with ADR 300, confirming the inhibitory effect of ADR on pulmonary fibrosis. No significant differences in body weights were observed between the ADR and CON groups. This may be due to the relatively short duration of the experiment (11 days); a longer period of ADR administration might be required to induce significant changes in body weight.
The inflammatory response plays a pivotal role in nearly all wound healing and fibrotic processes. 29 Especially, bleomycin induces acute injury to endothelial cells, leading to the release of inflammatory mediators, resulting in leukocyte infiltration. 30 The excessive accumulation of inflammatory cells within the vascular, interstitial, and alveolar compartments of the lung may precipitate vascular and parenchymal damage. 31 Furthermore, the early recruitment of leukocytes (granulocytes, lymphocytes, and macrophages) may contribute to the localized upregulation of TGF-β, ultimately leading to pulmonary fibrosis.30,32 Consequently, we investigated the count of leukocytes in BALF, and confirmed that ADR reduced leukocytes which are related to pulmonary fibrosis, and the most significant reduction was observed in ADR 300.
When pulmonary fibrosis progresses and TGF-β is activated, TGF-β inhibits the proliferation of alveolar epithelial cells, differentiates fibroblasts into myofibroblasts, and promotes epithelial-mesenchymal transition, exacerbating pulmonary fibrosis. 33 In normal cases, myofibroblasts in damaged tissue are removed and recovery occurs; however, in the case of IPF, myofibroblasts are not removed, and fibrosis progresses.34,35 Myofibroblasts are differentiated fibroblasts that express α-SMA; when α-SMA is activated, myofibroblasts stop proliferating and produce a large amount of ECM-related proteins, leading to the accumulation of ECM, which plays an important role in the progression of fibrosis. 36 Therefore, the expression of α-SMA is closely related to the progression of pulmonary fibrosis, 37 and it has been reported that there is an increase in epithelial cells expressing α-SMA in the lungs of animals treated with bleomycin 38 and patients with IPF. 39 Therefore, we examined the degree of pulmonary fibrosis through the expression levels of TGF-β and α-SMA. We found that ADR treatment tended to decrease the expression levels of TGF-β and α-SMA, with a significant decrease of α-SMA level occurring in lung tissue of the ADR 300 group.
An effective ADR dose of 300 mg/kg in mice corresponds to 18 g for a 60 kg human (or 86.2 g of raw material). Given the approximately 10 times faster metabolism rate in mice compared to humans, the clinical dosage can be estimated at 8.62 g of raw material, aligning with the typical clinic dosage range in Korean medicine for lung diseases, which is 10-20 g of raw material.40–42 In the current results, the protective effect of ADR at the dose of 300 mg/kg was around 30%, whereas both nintedanib (28 d, 120 mg/kg) and pirfenidone (42 days, 300 mg/kg) were reported to show efficacy of more than 50%.43,44 Despite the difference in expiration dates, the protective efficacy of ADR may be around 30% to 50% compared with that of conventional drugs. In addition, ADR has been reported as nontoxic, and a clinical study has also shown no adverse effects in humans. 45 Therefore, ADR might gently reduce pulmonary fibrosis without side effects. While we did not specifically investigate the effective compounds of ADR, we assumed that water-soluble compounds, encompassing polysaccharides with reported antiaging, 46 memory impairment reversal, 47 immune-regulating, 48 and free radical scavenging effects, 49 could constitute the primary group of effective compounds in the current findings. Furthermore, triterpenoids like lupenone, lupeol, and taraxerol, which exhibit pharmacological activities such as anti-inflammatory and hepatoprotective effects,50,51 despite their limited solubility in water, may contribute partially to the observed results.
Taken together, the results of this study demonstrated the antifibrotic effects of ADR, especially at a dose of 300 mg/kg. We examined the mechanism of efficacy by assessing the levels of inflammatory cells, TGF-β, and α-SMA. Further studies are needed to ascertain the toxicity of each concentration of the drug and the specific mechanisms of action, such as the TGF-β/Smad3 pathway and verify the synergistic effect of ADR co-administration with pirfenidone, which is already known to be a therapeutic agent.
Conclusion
ADR is a lung-moistening herbal medicine with proven anti-inflammatory effects. In a previous screening study, ADR was reported to have a higher antifibrotic effect than other herbal medicines. Therefore, this study was conducted to investigate the antifibrotic efficacy and mechanism of action of ADR in a mouse model of bleomycin-induced pulmonary fibrosis. This study showed that ADR decreased the degree of histological fibrosis (based on the Ashcroft score), number of inflammatory cells in the BALF, and the TGF-β and α-SMA levels in lung tissue. ADR at a dose of 300 mg/kg resulted in the most significant reduction in the measured indicators. These results indicate that the administration of 300 mg/kg ADR attenuates pulmonary fibrosis in an animal model of IPF.
Footnotes
Acknowledgments
Author Contributions
Conceptualization was done by BY and LB-J. Methodology was designed by KE-J and BY. Validation was done by KE-J and BY. Formal analysis was done by KE-J and PJ. Investigation was carried out by KE-J, PJ, YC-H, PJ-W, KK-I, and KJ-H. Resources were handled by PJ-W and BY. Data curation was done by PJ and KK-I. Writing-Original Draft was done by KE-J and PJ. Writing-Review and Editing were done by KE-J, PJ, KJ-H, and LB-J. Supervision was done by Jung H-J and Lee B-J. Project administration was done by BY and LB-J. Funding acquisition was handled by LB-J.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research: This research was supported by the Basic Science Research Program of the National Research Foundation of Korea [grant number: NRF-2018R1A2B6009621] and the Korean Health Technology R&D Project through the Korea Health Industry Development Institute [grant number: HI20C1205].
Ethical Approval
This study was approved by the Kyung Hee University Institutional Animal Care and Use Committee, Seoul, Republic of Korea (KHUASP(SE)-18-177).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Kyung Hee University Institutional Animal Care and Use Committee's (KHUASP(SE)-18-177) approved protocols.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
