Abstract
Objective:
Sijunzi decoction (SJZD) has been used for alleviating peptic ulcer or gastric discomfort, and treating spleen disorders since the Song Dynasty, but its pharmacological effect on human gastric cancer (GC) is still unclear. In this research, a network pharmacology-based strategy was applied to explore active ingredients, potential targets, and molecular mechanisms of SJZD against GC.
Methods:
The active compounds and potential targets of SJZD, as well as GC-associated gene targets, were retrieved from publicly available databases. Bioinformatics approaches were used to assess the network interaction, functional regulation, and signaling pathways between SJZD ingredients and GC targets. The anticancer effects of SJZD against GC were verified in vivo by a mouse subcutaneous model.
Results:
The results of network analysis showed that quercetin was the most active ingredient in SJZD. Several prominent target genes of SJZD were identified, such as AKT1 and STAT3. Gene ontology analysis revealed that the core anti-GC targets of SJZD included transcription factor activity and kinase activity. Pathway enrichment analysis indicated that GC patients could be benefited from SJZD treatment via modulation of signaling pathways related to endocrine system, cancer, and infectious disease. Furthermore, in vivo experiments showed that high-dose SJZD could inhibit GC xenograft tumor growth, reduce GC cell proliferation, induce GC cell apoptosis, and decrease the expression of p-AKT1 and p-STAT3.
Conclusions:
Taken together, our results suggest that SJZD can serve as an effective adjuvant therapeutic agent for GC patients.
Introduction
Gastric cancer (GC) is the fifth most frequently diagnosed cancer and the third leading cause of cancer-attributable mortality, which has accounted for more than 1 000 000 new cases in 2018 and 783 000 deaths (equating to 1 in every 12 deaths) worldwide. 1 Recent evidence illustrates that several risk factors, such as Helicobacter pylori infection, alcohol assumption, and smoking, may influence GC tumorigenesis.2,3 Besides, abnormal activation of some genes, including HER-2, can contribute to GC progression.4‐6 However, the exact mechanisms of GC are still unclarified. Although chemotherapy, radiotherapy, molecular targeted therapy, and immunotherapy have emerged as the main treatment regimens for patients with advanced stage GC,2,3,7‐9 the overall survival rate is still poor and these therapies can lead to serious adverse events and affect the quality of life. Hence, the prognostic and quality-of-life scores of advanced stage GC are still below expectations.7,9
Traditional Chinese medicine (TCM) is the world's oldest healthcare tradition practiced for treating cancer since ancient times. 10 It has been proved that TCM could increase the effectiveness of chemotherapy, reduce the toxic and side effects of chemotherapy and radiotherapy, prolong the survival rate, and increase the quality of life of cancer patients.11‐16 Sijunzi decoction (SJZD), a classic formula for nourishing the spleen and “reinforcing asthenia qi,” has been used to alleviate peptic ulcer or abdominal discomfort and treat spleen disorders since the Song Dynasty. 17 It consists of ginseng root (Panax ginseng C.A. Mey), Poria cocos, white atractylodes (Atractylodes macrocephala) rhizome, and locorice (Glycyrrhiza uralensis) root. Clinical studies have shown that SJZD improves the postoperative survival, cellular function, and nutritional status of GC patients after surgical operation.18,19 Zhao et al 20 found that SJZD markedly inhibited the growth of GC tumors in SGC-7901 xenografts-bearing nude mice. Similarly, another study demonstrated that SJZD inhibited GC cell lines growth and colony formation. 21 Further analysis showed that SJZD induced the apoptosis of side population cells in GC cell lines via upregulation of PARP, caspase-3 and Bax, and downregulation of bcl-2.21,22 However, the potential targets and active ingredients of SJZD are still unelucidated.
TCM formulas are characterized by the synergistic effects of poly-targets, poly-pathways, and multiple components, 23 which also play considerable roles in regulating biological functions and pathophysiological processes.15,24,25 With the advent and rapid rise of bioinformatics, a novel TCM network pharmacology-based approach was established. It can be used to systemically assess the interrelationship between the active components of a TCM and the molecular targets of human cancers at the network level and beyond. 26 This can help to elucidate the mechanisms of action and uncover the scientific basis of TCM formula in a complex biological system.27‐29 Hence, the application of network pharmacology on TCMs may ultimately contribute to the future development of TCM formulas for cancer treatment.
In this work, a network pharmacology-based approach was employed to unravel the molecular mechanisms underlying the therapeutic effects of SJZD on GC. Bioinformatics analyses were conducted to explore the network interaction, functional regulation, and key signaling pathways between SJZD ingredients and GC targets. Furthermore, the potential anti-GC properties and molecular targets of SJZD were validated in vivo.
Results
SJZD-GC Targets
To obtain GC-related targets, we firstly identified 200 active ingredients and 2094 target genes of these ingredients. The results showed that these candidate ingredients can regulate various targets, and multiple targets could also connect to a single ingredient. For example, stigmasterol was associated with 30 potential targets, such as nuclear receptor coactivator 2, progesterone receptor, and so on. In addition, nuclear receptor coactivator 2 could be modulated by 52 different ingredients, such as hederagenin, sitosterol, and lupiwighteone. Then, 2264 GC-related genes were collected from Online Mendelian Inheritance in Man (OMIM; http://www.omim.org/) 30 and GeneCards-The Human Gene Database (http://www.genecards.org/) databases. 31 After eliminating the redundancy, a total of 149 known therapeutic targets were identified in this study, which were all related to GC treatment (Figure 1A).
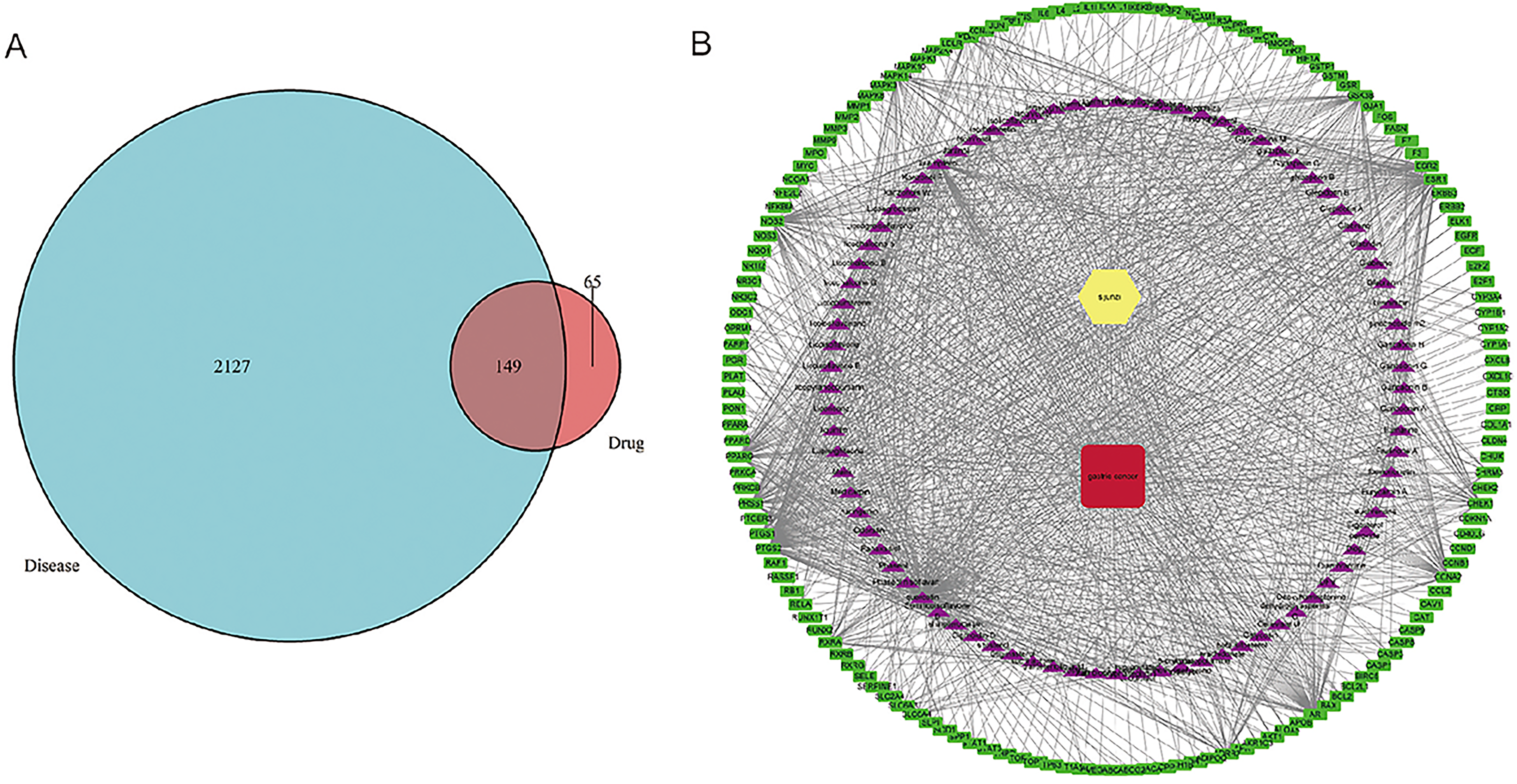
Construction of SJZD-GC targets network. (A) Venn diagram of the intersection of SJZD ingredients and GC-related genes. (B) Network diagram of the SJZD ingredients target genes of GC. The green panel represents gene, the purple triangle represents SJZD ingredient. SJZD, Sijunzi decoction; GC, gastric cancer.
SJZD Ingredients-GC Targets Network Analysis
The network of SJZD ingredients-GC targets was established with a total of 262 nodes and 1430 edges, including 149 GC target genes as anticancer targets (Figure 1B). The purple node represents the active ingredients of SJZD, the green node represents the GC target genes, the red node represents GC, the yellow node represents SJZD, and the 1430 edges represent the interaction between the chemical components and the GC target genes (Figure 1B). As can be seen from this figure, 111 SJZD main ingredients act on the same or different GC target genes, reflecting its multicomponent and multitarget anticancer action characteristics. According to the degree of connectivity of the nodes, the SJZD compounds were ranked as follows: quercetin (degree = 118), kaempferol (degree = 37), naringenin (degree = 23), 7-Methoxy-2-methyl isoflavone (degree = 20), 2-[(3R)-8,8-dimethyl-3,4-dihydro-2H-pyrano[6,5-f]chromen-3-yl]-5-methoxyphenol (degree = 20), licochalcone A (degree = 20), and shinpterocarpin (degree = 20), which might serve as the main anticancer substance.
Protein–Protein Interaction (PPI) Network of Anti-GC Targets Regulated by SJZD Treatment
To reveal the potential mechanisms underlying the therapeutic effects of SJZD on GC, the PPI network of targets for SJZD against GC was constructed based on the results of 2.2.3, which also delineated the intersection of SJZD-related drug targets in 2.1 and GC-related targets in 2.2 (Figure 2A). Next, the downloaded string-interactions.tsv file was imported into R to obtain barplot figure (Figure 2B). There was a total of 149 nodes and 361 edges, with an average node degree of 4.85 and a PPI enrichment p-value of <1.0 × 10−16. Among them, AKT1 (degree = 38), STAT3, (degree = 37), Jun proto-oncogene (JUN; degree = 36), mitogen-activated protein kinase 1 (MAPK1; degree = 35), and mitogen-activated protein kinase 3 (MAPK3; degree = 35) were of high degree values and exhibited strong interaction with other target genes, which might play key roles in this PPI network.

PPI network of anti-GC-related protein. (A) The interactive network of SJZD-related gene targets for GC therapy. (B) Bar plot of the PPI network. PPI, protein–protein interaction; SJZD, Sijunzi decoction; GC, gastric cancer.
Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analysis
To further unravel the molecular mechanisms of SJZD against GC, GO enrichment analysis was carried out to determine the biological process, cellular component, and molecular function of these gene targets. These results demonstrated that the targets of SJZD were positively associated with “transcription factor activity,” “kinase activity,” “MAPK signal transduction,” “phosphatase binding,” and “receptor binding” (Figure 3A).

Functional analysis for the major targets of SJZD. (A) GO and (B) KEGG enrichment analyses for the major targets of SJZD. (C) Representative KEGG pathway (hsa05226) of the major targets of SJZD. SJZD, Sijunzi decoction; GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes.
With regard to KEGG pathway analysis, the significantly enriched KEGG pathways by the targets were revealed (Figure 3B). The top 5 enriched pathways were “Lipid and atheroscierosis” (hsa05417), “Hepatitis B” (hsa05161), “Kaposi sarcoma-associated herpesvirus infection” (hsa05167), “Human cytomegalovirus infection” (hsa05163), and “AGE-RAGE signaling pathway in diabetic complications” (hsa04933). The representative KEGG pathway is illustrated in Figure 3C (GASTRIC CANCER, hsa05226). These results suggest that GC patients probably benefit from SJZD treatment via modulation of signaling pathways related to endocrine system, cancer, and infectious disease.
Validation of Molecular Targets
The tumor volume of the high-dose group was smaller than that of the control group, and the weight of GC tumor in the high-dose group was also lower than that of that in the control group. These findings indicate that the administration of high-dose SJZD could suppress GC xenograft tumor growth in vivo (Figure 4A and B).

SJZD treatment inhibitS GC cell growth in vivo. (A) Compared to the control group, high-dose SJZD treatment significantly decreased GC xenograft tumor volume. (B) Compared to the control group, high-dose SJZD treatment significantly reduced GC xenograft tumor weight. (C) Immunohistochemical staining revealed that high-dose SJZD treatment significantly reduced Ki-67 expression in xenografted tissues (magnification: × 200. Scale Bars: 100 μM). (D) Terminal deoxynucleotidyl transferase dUTP nick end labeling assay indicated that SJZD treatment significantly induced GC cell apoptosis. (E) Western blot analysis demonstrated that SJZD treatment decreased the expression levels of p-AKT1 and p-STAT3 in GC xenograft tumor model. Values represent mean ± SD from three independent experiments. ***P < .001, *P < .05. SJZD, Sijunzi decoction; GC, gastric cancer; SD, standard deviation.
Ki-67 is related to cell proliferation and can be used as an indicator to detect cell proliferation activity. The higher the positive rate of Ki-67, the higher the proportion of proliferating cell, and the faster the tumor growth. The nucleus is bluish purple and the Ki-67 protein is brownish red. As shown in Figure 4C, the color of MKN45 cells was darker in control group and lighter in SJZD treatment groups, respectively. The medium- and high-dose groups were the lightest, followed by the low-dose group, suggesting that SJZD could inhibit the growth of GC cells in a dose-dependent fashion.
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay is commonly used to detect cell apoptosis. As shown in Figure 4D, the normal nuclei are labeled with blue fluorescence, while the apoptotic cells are signaled by green fluorescence. Notably, the green fluorescent area was expanded along with the increase of SJZD doses. After fusion, the blue and green fluorescence were completely overlapped, indicating that SJZD could induce the apoptosis of GC MKN45 cells, and the effect was more obvious with the increase of SJZD doses, while the apoptosis of the control cells was not obvious.
The AKT1 signaling pathway is activated by the phosphorylation of phosphatidylinositol 4,5-bisphosphate 3-kinase (PI3K), which plays the major role in regulating cancer cell proliferation, invasion, and apoptosis. As to STAT3, aberrant STAT3 signaling can promote carcinogenesis not just by inducing cell proliferation but also by suppressing apoptosis. p-AKT1 is an active form of AKT1, p-STAT3 also is an active form of STAT3, all of which play critical roles in cancer initiation and progression. In this study, we found that SJZD could decrease the expression levels of p-AKT1 and p-STAT3 (Figure 4E).
Discussion
TCM cures the symptoms of an illness in a holistic manner. Network pharmacology-based approach has been used to the molecular mechanisms of TCM at the network level and beyond, which helps to discover active components and elucidate the mechanisms of action of herbal formula. In this work, we ultimately identified 6 active compounds, 20 kernel target genes, and key signaling pathways.
A growing body of literature has highlighted that these 6 compounds are closely related to antitumor and correlated signal pathways. Quercetin has been demonstrated to suppress GC cell growth; induce apoptosis, necrosis, and autophagy; and enhance the efficacy of other anticancer drugs.32,33 Kaempferol can induce cancer cell apoptosis and death, reduce cancer cell viability, modulate DNA methylation, and inhibit epithelial–mesenchymal transformation. 34 Naringenin is reported to have a chemopreventive action against N-methyl-N′-nitro-N-nitrosoguanidine (MNNG) induced gastric carcinoma in experimental rats. 35 In addition, naringenin enhances the anticancer effect of ABT-737 (a Bcl-2 inhibitor) on GC cells. Further study found that naringenin increased the cleavage of PARP and caspase-3, decreased Akt activation, and upregulated p53 expression, which may be involved in the suppression of GC cell growth. 36 Similarly, licochalcone A inhibited the proliferation of GC cells by inducing cell cycle arrest and cell death.37‐40
With regard to the result of PPI analysis, AKT1 ranked first in the network. AKT1, as a downstream effector of PI3K signaling, is evolved in the invasion, migration, apoptosis inhibition, autophagy regulation, and epithelial-to-mesenchymal transition (EMT) of GC cells.41‐48 For STAT3, its dysregulation may initiate tumorigenesis by suppressing cell death and promoting tumor cell growth angiogenesis and EMT.49,50 Thus, it is speculated that AKT1 and STAT3 may be the key targets modulated by SJZD in GC patients. Further in vivo experiment confirmed our prediction.
GO and KEGG enrichment analysis showed SJZD could regulate various signals and cancer-related pathways. Among them, “AGE-RAGE signaling pathway in diabetic” is reported to be associated with cancer. Aberrant expression of this pathway could lead to carcinogenesis, tumor cell growth, proliferation, apoptosis inhibition, and metastasis.51‐53 However, whether SJZD could modulate these pathways remains to be validated in vitro and in vivo.
Conclusions
In conclusion, our results suggested that SZJD treats GC by regulating its molecular targets, thus suppressing cell proliferation and inducing apoptosis. Network pharmacology can serve as a promising approach for studying the potential effects of TCM against GC.
Materials and Methods
Data Preparation
Active ingredients in SJZD
To retrieve the active ingredients of SJZD, Traditional Chinese Medicine Systems Pharmacology Database (TCMSP, http://tcmspw.com/tcmsp.php) was searched. 54 This database contains a complete platform for TCM analysis. In total, 613 active ingredients were identified, including 190 in Panax ginseng C.A. Mey, 55 in Atractylodes macrocephala, 88 in Poria cocos, and 280 in Glycyrrhiza uralensis.
ADME (Absorption, Distribution, Metabolism, Excretion) screening
To explore the therapeutic role of SJZD active compounds in GC, 2 ADME-related models, namely, drug-likeness (DL) and oral bioavailability (OB), were utilized to examine the potential active components of SJZD.
DL
The concept of DL has been used to assess whether a chemical entity can be considered as a drug substance and its “drug-like” properties (eg, pharmacokinetic and pharmacodynamic parameters). To screen potential drug-like compounds, a database-dependent model was applied to calculate the DL of each compound in SJZD based on Tanimoto similarity and molecular descriptors.55,56 The values of DL were calculated as follows:
OB
OB, which is an important pharmacokinetic parameter in drug screening cascades, is a critical indicator of the efficiency of active drug delivery to the systemic circulation. In the present study, the OB screening is calculated via a powerful internal system, OBioavail1.1, 58 and the OB value was set according to the following basic principles. Firstly, the data extracted from the TCM needed to be as comprehensive as possible with a minimum of molecular compounds. Secondly, the model derived from the available pharmacological data should be reasonably explained. 59 The bioactive ingredients with an OB value of ≥30% were selected for subsequent analysis.
As mentioned above, the potential bioactive ingredients were screened according to the following selection criteria: DL ≥ 0.18 and OB ≥ 30%. Eventually, 200 ingredients were obtained with 2 repetitions: 61 in Panax ginseng C.A. Mey, 10 in Atractylodes macrocephala, 25 in Poria cocos, and 104 in Glycyrrhiza uralensis.
Target Prediction
SJZD-related drug targets
“Baizhu” (Atractylodes macrocephala), “fuling” (Poria cocos), “gancao” (Glycyrrhiza uralensis), and “renshen” (Panax ginseng C.A. Mey) were, respectively, searched in TCMSP database based on the related targets. The information of the targets was collected separately, and then converted to official symbols on the UniProt database (http://www.uniprot.org/). 60
GC potential targets
A variety of candidate genes associated with GC were gathered from GeneCards. 31 The keyword “gastric cancer” was used, and 2126 genes were collected with relevance ≥5. Another database, OMIM, 30 was also been searched with the keyword “gastric cancer,” from which we collected 138 genes with no duplication of GeneCards. The official symbols of all the potential targets were converted from the UniProt database (http://www.uniprot.org/). 60
SJZD gastric cancer targets
Perl script (http://www.perl.org/) was employed to calculate the intersection of SJZD-related drug targets in 5.2.1 and gastric cancer targets in 5.2.2 to identify the drug targets of SJZD acting on GC. A Venn diagram was drawn by R language (http://www.r-project.org/, version 3.6.4).
Network Visualization
Through the use of Cytoscape software (http://www.cytoscape.org), an interaction network between the active ingredients in 5.1.2 and the drug target in 5.2.3 was visualized. Specifically, the ingredients and targets were illustrated by nodes, and the association between 2 nodes was signified by an edge.
PPI Network Construction
To construct a PPI network, the SJZD anti-GC target proteins obtained in 5.2.3 was imported into the STRING platform (http://string-db.org). The lowest interaction threshold was fixed at 0.95, the species was set to “Homo sapiens,” and the remaining parameters were left to the default setting. The size of PPI network was indicated by the strength of the interaction force, and the degree of the connection was denoted by the number of the interacting targets. Hub genes were identified according to the degree of connectivity.
GO and KEGG Enrichment Analysis
“DOSE,” “clusterProfiler,” and “pathview” (http://bioconductor.org/biocLite.R) installation packages were imported in R language for GO and KEGG enrichment analysis. For GO analysis, OrgDb was set as “org.Hs.egdb” (http://www.bioconductor.org/), with p-value and q-value thresholds of .05 each. For KEGG analysis, organism was set as “has,” with p-value and q-value thresholds of .05 each.
Experimental Validation
GC cell culture and preparation of SJZD
MKN45 cells (a human gastric cancer cell line) were purchased from Shenzhen Otwo Biotech Inc. and verified by a short tandem repeat analysis. The cells were cultivated in RPMI-1640 medium containing 10% fetal bovine serum (JRH Biosciences) and 1% penicillin–streptomycin and then maintained under a humidified atmosphere at 37 °C with 5% CO2.
SJZD was prepared as previously described with minor modifications. 61 Gancao, Renshen, Fuling, and Baizhu were mixed at the ratio of 2:3:3:3:3 in weight for Sijunzi decoction preparation. Briefly, the above 4 herbs were processed into coarse powder through pulverization, mixed and soaked in 12 volumes of distilled water for 30 min, followed by decoction for 90 min. The total extracts were filtered, and the supernatants were collected. The herb pellets were subjected to the second round of extraction by the addition of 5 volume of water, decoction for 60 min and filtration. Afterward, 2 parts of supernatants were combined and concentrated by rotary evaporation. The extracts were then precipitated with ethanol (final concentration: 50%, vol/vol) at 4 °C overnight. On the following day, the ethanol mixture was filtered and precipitated; while the pellets were dissolved in ultrapure water, followed by centrifugation (8400 rpm, 15 min). The collected supernatants were lyophilized and store at −80 °C before use. Finally, the total productive rate was 26.55%, with a total of 330 g herbs yielded 87.62 g Sijunzi decoction. The obtained SJZD was dissolved in phosphate-buffered saline (PBS) and subjected to 0.22-µM filtration prior to cell treatment.
Animal experiments
Twenty-four male BALB/C nude mice (weighted 19-25 g and aged 4-6 weeks) were procured from Silaike Laboratory Animal Co. LD (animal certificate No. SCXK 2019-0004). The mice were housed in well-ventilated sterile polypropylene cages equipped with sterile bedding, maintained under a 12:12 light-dark cycle at 25 °C, and supplied with complete formula feed and sterilized water. The procedures of mouse handling in compliance with the National Institutes of Health Guidelines for Care and Use of Laboratory Animals. The animal study protocol was approved by the Animal Ethics Committee of Hunan University of Chinese Medicine.
Animal groups and treatment
After 7 days of acclimatization, MKN45 cells were adjusted to 1.5 × 108/mL, and then 0.2 mL of cell solution was injected subcutaneously into the armpit of the mice to generate a GC model. After 8 days of inoculation, the tumor volume of mice reached about 100 mm3, which indicated that the xenograft model was successfully constructed. According to the weight, all mice were divided into 4 groups: control group, low-dose group, median-dose group, and high-dose group. Each group had 6 mice. For drug administration, the control group was given 10 mL/kg of normal saline every day. Corresponding to the biologically equivalent dose of 50%, 100%, and 200% of the human dose, the mice in SJZD groups was given 2.3 g, 4.6 g, and 9.2 g/kg of SJZD by gavage for low-, medium-, and high-dose groups, respectively. Mice weight, tumor length (L), and tumor width (W) were measured every 2 days. The tumor volume was calculated as follows: L × W2 × 0.5 (mm3). Following treatment for 24 days, the mice were euthanized and their tumors were carefully removed and measured.
Immunohistochemistry
To evaluate tumor cell proliferation, the level of Ki-67 was detected using an immunohistochemistry assay. After fixing in 10% paraformaldehyde for 12 h, the mouse tumors were embedded in paraffin, dewaxed in xylene and then rehydrated in ethanol. With the use of a pressurized cooker, antigen retrieval was conducted with 0.01 M citrate buffer (pH 6.0) for 3 min. After washing 3 times with PBS, the samples were fixed in 95% ethanol for 30 min. Finally, the tumor cells were stained with Ki-67 antibody (ab15580, Abcam) using a streptavidin-peroxidase detection kit (ZB2301, ZSGB-BIO) by following the kit's protocol.
TUNEL assay
Xenograft cancer cell apoptosis was examined by TUNEL assay with reaction mixture (C1088, Beyotime) as previously described. 62 Briefly, paraffin sections were prepared by dewaxing in dimethylbenzene, dehydrating in ethanol, and treatment with DNase-free protease K for 15 to 30 min at 37 °C. After washing twice with PBS, the paraffin sections were incubated with 50 µL of TUNEL working solution for 1 h at 37 °C, followed by visualization under a fluorescence microscope (Olympus). GC cell apoptosis was measured as the percentage of TUNEL-positive cells (green) over total cells (blue). For each sample, at least 300 cells were counted independently by 2 observers in 5 random fields.
Western blotting
The following primary antibodies were used for western blotting: phosphorylation-AKT serine/threonine kinase 1 (p-AKT1, rabbit, bs-0876R, Bioss, 1:500), phosphorylated-signal transducer and activator of transcription 3 (p-STAT3, rabbit, AF3293, Affinity, 1:500), and GAPDH (TA-08, ZSGB-BIO, 1:2000). Total protein was isolated from tumor samples using RIPA lysis buffer (CWBIO). After 10% SDS-PAGE electrophoresis, the protein samples were transferred onto a 0.45-μm PVDF membrane. Later, the membrane was inhibited with 5% nonfat milk in PBST (PBS + 0.1% Tween 20) for 60 min. After incubation with the abovementioned primary antibodies at 4 °C for overnight, the protein samples were incubated with horseradish peroxidase-conjugated goat antirabbit secondary antibody (ZB-2301, ZSGB-BIO, 1:2500) for 2 h at ambient temperature. Lastly, the protein bands were visualized by enhanced chemiluminescence and recorded using a Bio-Rad ChemiDoc XRS Imaging system. The gray-scale intensity values of the protein bands were estimated using an Image J software, and GAPDH was employed as an internal control for normalization.
Statistical Analysis
SPSS software version 21.0 was used to perform the statistical test. All data were presented as mean ± SD. Statistical differences between groups were compared using one-way ANOVA. P < .05 was deemed to be of statistical significance. All experiments were conducted in triplicate.
Footnotes
Acknowledgments
Author Contributions
(I) Conception and design: Junfeng Yan, Qinghua Peng; (II) Collection and assembly of data: Yuejun Li, Hong Wang, Linli Liao; (III) Data analysis and interpretation: All authors; (IV) Manuscript writing: All authors; (V) Final approval of manuscript: All authors. Yuejun Li, Hong Wang, and Linli Liao contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hunan Province, National Natural Science Foundation of China (grant number 2018JJ6042, 81703916).

