Abstract
Background
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, and fibrotic interstitial lung disease. Trillin, an active saponin component isolated from Trillium tschonoskii Maxim, exhibits various pharmacological properties. This study aimed to investigate the preventive effects of Trillin on bleomycin (BLM)-induced IPF and elucidate its underlying mechanisms.
Methods
BLM was administered via intratracheal injection to C57BL/6J mice to establish an IPF model. Different concentrations of Trillin were then administered as treatments. Network pharmacology and molecular docking approaches were employed to predict potential Trillin targets against IPF. A combination of histopathological examination, immunohistochemical analysis, Western Blotting, and other molecular biological techniques were utilized to assess Trillin's protective effect on BLM-induced IPF and to investigate underlying pathways.
Results
Trillin effectively inhibited lung tissue injury induced by BLM in mice, moderated inflammation and oxidative stress-related signaling pathways, and reduced the expression of IPF-associated proteins such as TGF-β1 and α-SMA. Network pharmacology analysis identified 18 core Trillin-IPF target genes. Molecular docking analysis suggested that pro-inflammatory cytokines IL-1β, IL-2, and TNF-α have high binding affinity with Trillin, suggesting their potential as anti-IPF targets. Additionally, experimental verification confirmed that Trillin suppressed NLRP3 inflammasome activation by reducing IL-1β release.
Conclusions
This study demonstrates that Trillin effectively reduces oxidative stress and blocks the IL-1β/NLRP3 inflammasome positive feedback loop, providing protection against BLM-induced IPF. Consequently, our research underscores the potential of Trillin as a promising therapeutic agent for IPF and lays a theoretical foundation for its future clinical development and application in treating BLM-induced IPF.
Keywords
Introduction
Idiopathic pulmonary fibrosis (IPF) is an interstitial lung disease characterized by chronicity, progressiveness, and fibrosis. 1 Unfortunately, IPF is often misdiagnosed and treated improperly with immunosuppressive therapy. This leads not only to increased hospitalizations and mortality but also to a poor prognosis, with a median life expectancy of only 3 to 5 years post-diagnosis in the absence of treatment. 2 Current guidelines recommend pirfenidone and nintedanib as treatments for IPF due to their pleiotropic mechanisms of action. 3 However, these medications have shown limited success in slowing IPF progression or enhancing patients’ quality of life and are associated with significant tolerability issues. 4 Moreover, lung transplantation remains the only alternative therapy for IPF patients, yet it is suitable for only a small subset of patients. 5 Therefore, there is an urgent need for the development of safe and efficient treatments for IPF.
Trillin (Tr) is an active saponin component derived from Trillium tschonoskii Maxim, characterized as a sterol 3-β-

The chemical structure and dosing regimen of Trillin. (A) The molecular structure of Trillin. (B) Animal experiment model diagram.
While the exact cause of IPF remains unknown, it is widely accepted that a combination of genetic and environmental factors contributes to its development. Repeated local micro-injuries are identified as playing a central role in triggering the disease in senescent alveolar epithelium. This results in abnormal interactions between epithelial cells and fibroblasts, activation of matrix-producing myofibroblasts, accumulation of extracellular matrix (ECM), and remodeling of the interstitial pulmonary matrix. 11 Moreover, the pro-inflammatory cytokine interleukin (IL)-1β is known to contribute to the development and progression of pulmonary fibrosis by promoting collagen expression and inflammation. 12 The significance of inflammasomes in IPF has been highlighted in numerous studies, with the NOD-like receptor protein 3 (NLRP3) being an extensively studied component. The NLRP3 inflammasome, comprising NLRP3, ASC, and pro-caspase-1, has been shown to play a crucial role in the fibrotic process.13‐15 Clinical research has also revealed the significant role of the NLRP3/IL-1β pathway in IPF progression.16,17 Generally, IL-1β maturation and secretion are regulated by NLRP3 inflammasomes. This means that both internal and external stimuli can activate these inflammasomes, leading to the activation of caspase-1 and subsequently IL-1β. However, several recent studies have indicated that IL-1β can also promote the expression of NLRP3 in a positive feedback manner.18‐21 Additionally, an imbalance in oxidation and antioxidants contributes to the aging of myofibroblasts, and oxidative damage to ECM proteins can result in their fragmentation or stabilization, leading to inflammation and fibrosis. 22 In IPF patients, an increase in the production of lipid peroxides, DNA oxidation, and protein carbonyl species in lung tissue and biological fluids has been observed, along with significantly reduced levels of various antioxidant scavengers.23,24
Network pharmacology, an approach within systems biology, employs network analysis of biological systems to elucidate the relationships among drugs, targets, and diseases. Its primary aim is to identify specific signal nodes for designing multi-target drug molecules. 25 In recent years, network pharmacology has been extensively used to explore the pharmacological effects of herbal formulations and individual herbal components on diseases, their underlying mechanisms, and to assess network toxicity. 26 Molecular docking, a theoretical simulation method, is utilized to study intermolecular interactions and predict binding modes and affinity. This method is commonly applied in drug development and the exploration of potential drug action targets. 27 To enhance the prediction of potential targets for Trillin's effect on IPF, we utilized both network pharmacology and molecular docking.
In this study, our objective was to investigate the effects of Trillin on bleomycin (BLM)-induced IPF and to explore the potential underlying mechanisms using network pharmacology, molecular docking, and experimental validation. Our findings demonstrate that Trillin can inhibit fibrosis, reduce oxidative stress, and suppress inflammation in vivo. Through network pharmacology and molecular docking analysis, 18 core target genes associated with Trillin and IPF were identified, all showing strong binding affinity to Trillin. Notably, pro-inflammatory cytokines such as IL-1β, IL-2, and TNF-α exhibited robust interactions with Trillin, indicating their potential as anti-IPF targets. Further, our experimental validation confirmed that Trillin effectively decreases the expression of IL-1β, NLRP3, and caspase-1 in a mouse model of IPF. Thus, our study demonstrates that Trillin can effectively alleviate BLM-induced IPF by interrupting the positive feedback loop of the IL-1β/NLRP3 inflammasome. Consequently, our research positions Trillin as a promising therapeutic agent for IPF, providing a theoretical foundation for its future clinical application in treating BLM-induced IPF.
Materials and Methods
Network Pharmacology
Access to Trillin Target Genes and IPF-Related Genes
To identify Trillin target genes, we utilized the Bioinformation Analysis Tool for Molecular Mechanism of Traditional Chinese Medicine (BATMAN-TCM, http://bionet.ncpsb.org.cn/batman-tcm/) and SwissTargetPrediction (http://www.swisstargetprediction.ch/). After removing duplicates, gene symbols were annotated using the UniProt database (https://www.uniprot.org/) to compile the Trillin target gene set. Similarly, the DisGeNET (https://www.disgenet.org/) and GeneCards (https://www.genecards.org/) databases were scanned for targets related to IPF. These IPF-related targets, once duplicates were removed, were imported and normalized in the UniProt database to finalize the IPF target list. To identify common targets between the drug (Trillin) and the disease (IPF), both sets of targets were uploaded to the Venny 2.1.0 online platform for analysis.
Protein–Protein Interaction (PPI) Network and Core Genes
To acquire the original PPI data, the common targets were imported into the STRING database (https://string-db.org/cgi/input.pl), setting the species to “Homo sapiens” and adjusting the Confidence Score to 0.7. Subsequently, the PPI network was constructed and visualized using Cytoscape software. Core genes were further screened by CytoNCA software. Additionally, the construction of the “Trillin-target-IPF” network was accomplished using the same software.
Enrichment Analysis
Enrichment analysis, which includes gene ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis, was performed using R version 4.2.0 and the following R packages: “colorspace,” “stringi,” “DOSE,” “ggplot2,” “clusterProfiler,” and “enrichplot.”
Molecular Docking
The crystal structures of the targets were obtained from the RCSB Protein Data Bank (RCSB PDB, https://www.rcsb.org/) and modified with the Pymol program. The two-dimensional structure of Trillin was acquired from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/). The software ChemBio3D Ultra was used to minimize energy and convert the two-dimensional structure to a three-dimensional one. The receptors and Trillin ligand were then imported into AutoDockTools to add hydrogens, define rotatable bonds, and configure docking parameters. Finally, the saved PDBQT files of the proteins and Trillin were used for docking and computing binding affinity in Vina, and the docking models were visualized using Pymol (3D).
Animals and Reagents
Forty female C57BL/6J mice were purchased from the Laboratory Animal Center at the Xiangya School of Medicine, Central South University (Changsha, China). The mice, aged 6 to 8 weeks, were housed in SPF-grade facilities and had ad libitum access to water and food. Prior to the experiment, they underwent a week of acclimation to laboratory conditions. All experiments adhered to the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 8023). The Ethics Committee of Experimental Animal Welfare at Central South University approved the experimental protocols (ethics code: 2020sydw0969).
Trillin (C33H52O8, CAS 14144-06-0, purity >98%) was sourced from Nanjing Spring & Autumn Biological Engineering Co., Ltd (Nanjing, China). It was dissolved in 0.5% carboxymethylcellulose sodium (CMC-Na; Chengdu Chron Chemicals Co., Ltd, China) for intragastric administration. BLM (bleocin vials, 15 mg potency) was purchased from Nippon Kayaku Co. Ltd – Japan. It was dissolved in freshly prepared saline, and injected into the trachea.
Establishment of BLM-Induced IPF Model
All animals were randomly assigned to one of four groups (n = 6-12): control group (Con, n = 6), BLM group (n = 12), BLM + Trillin 50 mg/kg/day group (BLM-Trillin (L), n = 11) and BLM + Trillin 100 mg/kg/day group (BLM-Trillin (H), n = 11). 10 To induce IPF, mice were anesthetized with sodium pentobarbital and received intratracheal administration of BLM at a dose of 3.5 mg/kg on day 0.28,29 The control group received an equal volume of saline. 30 To ensure uniform distribution of BLM in the lungs, the mice were promptly rotated, and the skin incision on the neck was sutured. Trillin was administered for 15 consecutive days starting the day after model preparation. On day 16, the mice were anesthetized with 50 μg/g pentobarbital (National Pharmaceutical Group Chemical Reagent Co., Ltd) for blood collection from the orbital venous plexus (Figure 1B). Lung tissues were harvested by flushing with cold saline and divided into two parts: one for histopathological analysis fixed in 4% paraformaldehyde, and the other snap-frozen in liquid nitrogen and stored at −80 °C for biochemical studies.
Measurement of TNF-α, IL-1β, and IL-6 Levels
We centrifuged mouse blood samples at 4000 rpm for 15 min at 4 °C to extract serum. The levels of TNF-α, IL-1β, and IL-6 in the serum were then measured using ELISA kits from BOSTER Biological Technology (Wuhan, China), following the manufacturer's instructions.
Histopathological Examination
Lung tissues were treated with 4% paraformaldehyde and subsequently embedded in paraffin to generate lung slices of 4 μm thickness. These slices were then used for pathohistological analysis. To assess the severity of lung damage, the level of inflammatory cell infiltration, and the degree of IPF, sections were stained using hematoxylin and eosin (H&E) to visualize cellular and structural details, and Masson's trichrome staining to highlight fibrotic changes.
Immunohistochemistry Analysis
Immunohistochemistry labeling was used to identify changes in α-SMA, TGF-β1, 8-OHdG, and 4-HNE in the lung tissues. For antigen retrieval, 4 μm thick paraffin-embedded lung tissue sections were first deparaffinized in xylene and then dehydrated in graded ethanol. They were heated in citrate buffer (pH 6.0) and allowed to cool to room temperature. The sections were then treated with 3% H2O2 for 10 min to quench endogenous peroxidase activity. To block non-specific binding, the tissue sections were incubated with 3% BSA for 30 min at room temperature. After removing the blocking solution, they were incubated with specific primary antibodies overnight at 4 °C. Following this, the sections were treated with secondary antibodies and washed with PBS. They were then counterstained with hematoxylin to induce diaminobenzidine-induced chromogenic reactions. Finally, the expression levels of α-SMA, TGF-β1, 8-OHdG, and 4-HNE in each lung tissue sample were examined using a light microscope.
Western Blot Analysis
The total protein concentration in fresh lung tissue samples was evaluated using the BCA method. Samples were lysed on ice with ice-cold RIPA buffer containing 0.1% PMSF (BOSTER Biological Technology; Wuhan, China). For protein separation, SDS-PAGE was performed at either 10% or 12% concentration, transferring 20 μg of protein onto 0.45 μm polyvinylidene difluoride (PVDF) membranes. To block non-specific binding, the blotted membranes were incubated with 5% skim milk at room temperature for 1 h. The membranes were then incubated overnight at 4 °C with primary antibodies, including anti-SMA (1:2000, Abcam; Cambridge, UK), anti-4-HNE (1:1000, Abcam; Cambridge, UK), anti-NLRP3 (1:1000, AdipoGen; San Diego, USA), anti-Caspase-1(p20) (1:1000, Affinity Biosciences; OH, USA), anti-IL-1β (1:1000, ProteinTech; Wuhan, China), and anti-GAPDH (1:1000, ProteinTech; Wuhan, China). Following washing, the membranes were treated with peroxidase-labeled secondary antibodies for one hour at 37 °C. Band intensity was visualized using ECL reagent (US Everbright Inc., Suzhou, China), and the results were analyzed using the ChemiDoc imaging system and Image Lab 6.0 software (Bio-Rad Laboratories Inc., Hercules, CA, USA).
Statistical Analysis
The data were analyzed using the GraphPad Prism version 8.0 (GraphPad Software, Inc.) software. The results were shown as the mean ± standard error of the mean (SEM). One-way ANOVA and the Student–Newman–Keuls were used to analyze the statistics. Results for all analyses were deemed significant when P < .05.
Results
Trillin Suppressed BLM-Induced Fibrosis and Inflammation in Lung Tissue
H&E and Masson's trichrome staining were employed to assess the impact of Trillin on BLM-induced IPF. Compared to the control group, lung tissue sections from mice treated solely with BLM showed prominent infiltration of inflammatory cells, as well as fibroblast and myofibroblast infiltration and accumulation in the alveoli, as revealed by H&E staining. However, these aforementioned lesions were alleviated after Trillin intervention (Figure 2A, H&E staining). Collagen deposition, commonly observed in IPF, was also investigated. Lung tissue stained with Masson's trichrome revealed that Trillin-treated mice exhibited reduced collagen deposition and fewer fibrotic lesions in the pulmonary interstitium compared to the BLM group (Figure 2A, Masson staining). Furthermore, we established a BLM-induced early-stage inflammation mouse model to evaluate Trillin's anti-inflammatory effects. As depicted in Figure 2B to D, Trillin significantly decreased the elevated levels of inflammatory cytokines TNF-α, IL-1β, and IL-6 induced by BLM, suggesting that Trillin alleviates BLM-induced IPF by attenuating the inflammatory process.

Trillin suppresses BLM-induced IPF and inflammation. (A) Representative lung micrographs of the lung tissue specimens stained with H&E staining and Masson's Trichrome staining. Magnification, 200×. Bar scale: 100 μm. (B, C, D) TNF-α, IL-1β, and IL-6 concentration in serum were examined. Con: Control group; BLM: bleomycin group; Tr: Trillin; BLM-Tr(L), BLM + Trillin 50 mg/kg/day group; BLM-Tr(H), BLM + Trillin 100 mg/kg/day group. Data are mean ± SEM, n = 6-12. **P < .01 versus Con; ##P < .01 versus BLM.
Trillin Inhibited TGF-β1 and α-SMA Expression in BLM-Induced IPF
TGF-β1 is a crucial pro-fibrotic factor that can induce fibroblast proliferation and differentiation into myofibroblasts, leading to excessive production and deposition of ECM and promoting the development of IPF.31,32 Myofibroblasts can be distinguished from fibroblasts by the presence of the marker α-SMA, which is also a biomarker of myofibroblast activation. 33 Immunohistochemical analysis revealed that the BLM group had higher levels of TGF-β1 and α-SMA expression compared to the control group, which were reduced by Trillin treatment (Figure 3A and B). Western blotting studies further demonstrated that Trillin can inhibit the increase in α-SMA protein expression induced by BLM (Figure 3C and D). These findings provided further evidence that Trillin can protect mice from BLM-induced IPF.

Trillin inhibits TGF-β1 and α-SMA expression in BLM-induced IPF (A and B) The TGF-β1 and α-SMA immunohistochemistry in lung tissues. Magnification, × 200. Bar scale: 100 μm. (C) The representative result of α-SMA expression in lung tissues was calculated by Western blotting. (D) Quantification of results from B. Data are mean ± SEM, n = 6-12. **P < .01 versus Con; ##P < .01 versus BLM.
Trillin Inhibited BLM-Induced Oxidative Stress Imbalance in Lung Tissue
In addition to inflammation, an imbalance of oxidative stress is one pathway that contributes to the development of IPF. Several investigations have demonstrated that lipid peroxidation and DNA damage can lead to excessive production of ROS in lung tissue, which in turn, contributes to the progression of IPF. 34 4-HNE is a marker of oxidative stress and a product of lipid peroxidation, while 8-OHdG is a biomarker of ROS-induced oxidative DNA damage. To assess the effectiveness of Trillin as an antioxidant in preventing BLM-induced IPF, we examined the changes in 4-HNE and 8-OHdG levels in lung tissue. Immunohistochemistry results revealed that the levels of 8-OHdG and 4-HNE were increased in mice treated with BLM. However, treatment with Trillin inhibited this upregulation. Furthermore, immunoblotting analysis confirmed that Trillin could prevent the BLM-induced increase in 4-HNE levels (Figure 4A-D). These results indicated that Trillin has a protective effect against lipid peroxidation and DNA damage caused by oxidative stress in BLM-treated mice.

Trillin inhibits BLM-induced oxidative stress imbalance in lung tissue. (A, B) The 8-OHdG and 4-HNE immunohistochemistry in lung tissues. Magnification, 200×. Bar scale: 100 μm. (C) The representative result of 4-HNE expression in lung tissues was calculated by Western blotting. (D) Quantification of results from B. Data are mean ± SEM, n = 6-12. **P < .01 versus Con; ##P < .01 versus BLM.
Trillin and IPF-Related Target Screening
302 Trillin target genes were identified in the BATMAN-TCM and SwissTargetPrediction databases. 924 and 6286 IPF-related target genes were screened from DisGeNET and GeneCards, respectively. After removing 777 duplicate genes, a total of 6433 IPF target genes were identified (Figure 5A). The intersection of Trillin and IPF targets resulted in 207 co-targeted genes (Figure 5B).

Trillin and IPF-related target screening. (A) IPF-related target genes. (B) Co-targeted genes of Trillin and IPF.
Construction of Trillin-Targets-IPF Network
To better demonstrate the interaction between Trillin and IPF target genes, we constructed a Trillin-targets-IPF network, consisting of 209 nodes and 414 edges. In this network, blue elliptical nodes represented target genes, green V-shape nodes represented Trillin, and yellow diamond nodes represented IPF (Figure 6).

Construction of Trillin-targets-IPF network. Blue elliptical nodes represent target genes, green concave quadrilateral node represents Trillin, and yellow diamond node represents IPF.
Enrichment Analysis
To investigate the underlying mechanisms of Trillin against IPF, GO and KEGG enrichment analyses were performed on a total of 207 co-target genes of Trillin and IPF.
The GO enrichment analysis revealed that BP were mainly involved in the positive regulation of transcription by RNA polymerase II, signal transduction, and positive regulation of cell population proliferation; CC were mainly involved in the membrane, cytoplasm, and plasma membrane; and MF were mainly involved in protein binding, metal ion binding, and identical protein binding (Figure 7A and C). According to the KEGG enrichment analysis, it revealed that the pathways affected significantly were Estrogen signaling pathway, EGFR tyrosine kinase inhibitor resistance and Jak-STAT signaling pathway (Figure 7B and D).
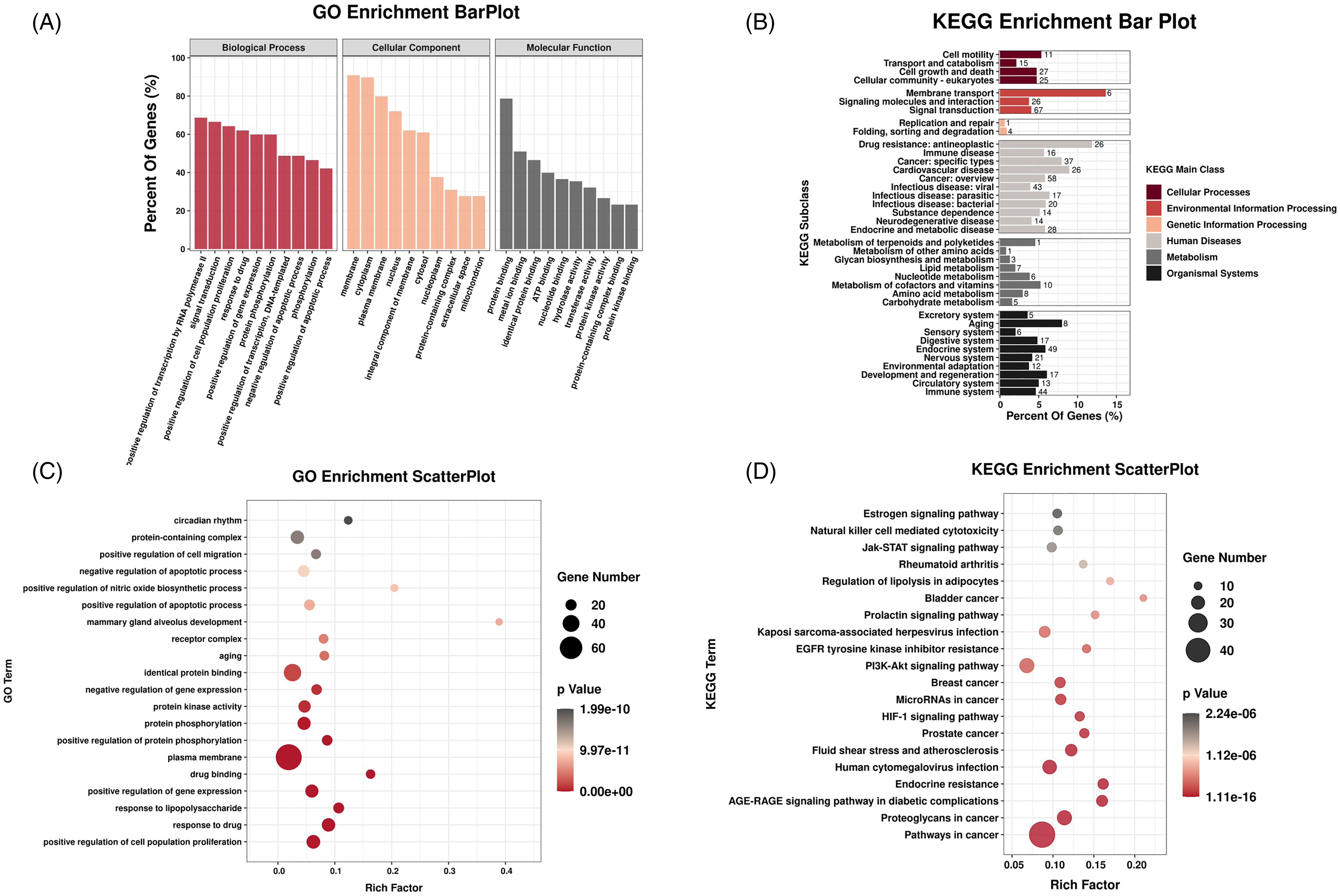
Enrichment analysis. (A) GO Enrichment BarPlot. (B) KEGG Enrichment Bar Plot. (C) GO Enrichment ScatterPlot. (D) KEGG Enrichment ScatterPlot.
Protein–Protein Interaction (PPI) Network and Core Target
The PPI network was constructed by importing 207 co-targeted genes into the STRING database, resulting in a network with 187 nodes and 820 edges (Figure 8A). Subsequently, the PPI network was visualized by Cytoscape software, and the hub genes (represented by the yellow node in Figure 8B) were analyzed. Using the CytoNCA software, 18 core target genes were further identified (depicted as yellow nodes in Figure 8C). These core target genes included IL-1β, IL-2, TNF-α, and other relevant genes.

Protein–protein interaction (PPI) network and core target. (A) Co-targeted genes PPI network of Trillin and IPF. (B) PPI network visualization. The yellow nodes were hub genes. (C) Core target genes (yellow nodes).
Molecular Docking
Molecular docking was used to investigate the interaction between 18 core targets and Trillin. Typically, a lower affinity score suggests a more stable ligand-receptor binding arrangement. Based on the network pharmacology findings and previous studies, IL-1β, IL-2, TNF-α, STAT3, CASP3, and AKT1 were selected from the NCA-PPI network for molecular docking (Figure 9A-F).

Images of Molecular docking between Trillin and the following targets. (A) IL-1β. (B) IL-2. (C) TNF-α. (D) STAT3. (E) CASP3. (F) AKT. Hydrogen bonds are indicated by dashed lines.
For IL-1β protein as shown in Figure 9A, the structure of Trillin could interact with TYR24 (length = 2.5) and GLN81 (length = 2.2) through one hydrogen bond, respectively; and SER84 (length = 2.3, 2.4) through 2 hydrogen bonds.
For IL-2 protein as shown in Figure 9B, Trillin could form one hydrogen bond with HIS79 (length = 2.9) and LYS76 (length = 2.6), respectively.
For TNF-α protein in Figure 9C, Trillin could form a hydrophobic bind with GLU135 (length = 2.7) and ASN46 (length = 2.3), respectively.
For STAT3 protein in Figure 9D, Trillin could form a hydrophobic bind with LYS20 (length = 2.2).
For CASP3 protein in Figure 9E, Trillin could form a hydrophobic bind with ARG81 (length = 2.4), CYS170 (length = 2.2), and LEU168 (length = 2.5), respectively; and ARG144 (length = 2.3, 2.3) through 2 hydrogen bonds.
For AKT1 protein in Figure 9F, Trillin could form a hydrophobic bind with ARG48 (length = 2.4) and PRO51 (length = 2.4), respectively; and ASN54 (length = 2.2, 2.3, 2.5) through 3 hydrogen bonds.
Docking affinity data in Table 1 show that Trillin effectively binds to the active sites of protein targets, with an average binding energy of −8.2 kcal/mol. Notably, the docking analysis revealed that the interaction between Trillin and IL-1β exhibited the lowest binding energy (−8.9 kcal/mol), indicating a highly favorable and robust binding interaction.
Molecular Docking Results of 6 Core Co-Targeted Genes with Trillin.

Trillin Reduced BLM-Induced Activation of NLRP3 Inflammasome
The activation of the NLRP3 inflammasome and IL-1β is a partial cause of IPF, according to previous studies. 35 Additionally, the molecular docking result showed that IL-1β had the lowest binding energy with Trillin and it could regulate the expression of NLRP3 in a positive feedback manner. 36 Therefore, we speculated that Trillin may alleviate IPF by acting on the NLRP3 inflammasome. To test this, we used Western blotting to detect the expression levels of NLRP3, Caspase-1 (p20), and IL-1β. As shown in Figure 10A to F, the expressions of IL-1β, NLRP3 and p20 were considerably higher in the BLM-treated group than in the control group. However, Trillin significantly reduced the expression levels of these proteins in the BLM group. These results suggest that Trillin may inhibit the activation of the NLRP3 inflammasome by reducing IL-1β levels.
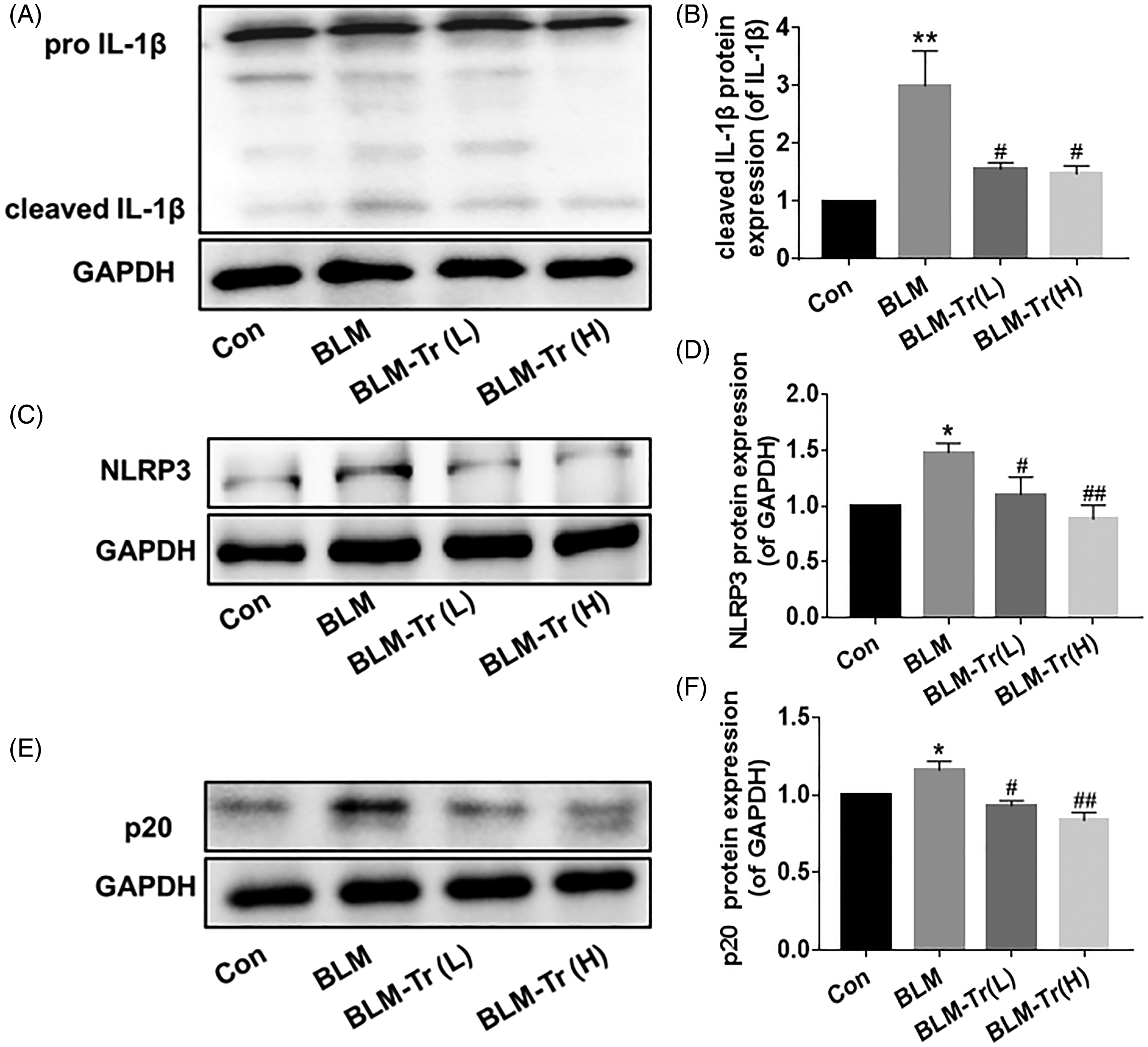
Trillin reduced BLM-induced activation of NLRP3 inflammasome. (A, C, E) The representative results of pro-IL-1β, cleaved IL-1β, NLRP3 and p20 expression in lung tissues were calculated by Western blotting. (B, D, F) Quantification of results from A, C, E. Data are mean ± SEM, n = 6-12. *P < .05, **P < .01 versus Con; #P < .05, ##P < .01 versus BLM.
Discussion
In this study, we found that Trillin positively impacted a BLM-induced IPF mouse model. Trillin reduced BLM-induced lung tissue injury in mice, ameliorated oxidative stress-related signaling pathway activation, inhibited inflammation, and decreased TGF-β1 and α-SMA expression. Utilizing network pharmacology, we identified 18 core Trillin and IPF targets, with molecules like IL-1β, IL-2, and TNF-α showing strong binding affinity during molecular docking. Experimental verification confirmed Trillin's effects on IL-1β, and its role in preventing NLRP3 inflammasome activation by reducing pro-inflammatory cytokine IL-1β release. These findings suggest Trillin's potential effective anti-fibrotic and anti-inflammatory effect for IPF treatment.
BLM, a chemotherapy drug used in various cancer treatments, is known to cause IPF, and the severity and distribution of fibrosis depend on the drug's dosage. 37 It is widely recognized that lung epithelial injury triggers healing pathways involving growth factors, cytokines, and ECM-driven signaling. This process recruits inflammatory cells, promotes fibroblast proliferation, and expands the ECM, ultimately leading to tissue fibrosis.38,39 Trillin, a bioactive compound isolated from Trillium tschonoskii Maxim., has shown promise in treating diabetic cardiomyopathy by inhibiting pro-inflammatory cytokines like IL-1β, IL-6, and TNF-α. Its neuroprotective effects involve antioxidant activity regulation, apoptosis prevention, and autophagy modulation. Trillin also exhibits anti-tumor effects, inducing apoptosis and necrosis, and inhibiting cervical cancer cell proliferation.7,8,40,41 Moreover, it has alleviated CCl4-induced hepatotoxicity and LPS-induced acute lung injury.9,10 However, its protective effects against BLM-induced IPF remain unexplored. This study aims to evaluate Trillin's therapeutic potential in BLM-induced IPF.
Although the mechanisms of IPF are not fully understood, the proliferation, migration, and differentiation of fibroblasts into myofibroblasts, forming fibroblastic foci, are known factors in IPF onset. 42 Cytokines and growth factors, especially TGF-β1, play a critical role in this process by promoting fibroblast activation and differentiation into α-SMA-expressing myofibroblasts, leading to fibrosis.43,44 Bergenin has been noted to reduce TGF-β1-induced fibroblast activation and ECM accumulation, and decrease BLM-induced fibrosis in IPF mice.45,46 Furthermore, reducing myofibroblast markers α-SMA and COL-I can inhibit fibroblast-myofibroblast differentiation. 47 Our findings indicate Trillin's potential in reducing lung histopathy, collagen deposition, and fibrosis-related markers like TGF-β1 and α-SMA in BLM-induced IPF.
One of the main pathogenic factors in IPF is oxidative stress. Studies have shown that TGF-β1 interacts with both oxidants and antioxidants in the lungs. TGF-β1, on one hand, promotes the differentiation of myofibroblasts, which generate oxidants. On the other hand, oxidants can enhance the release and activation of TGF-β1, thereby contributing to IPF.48,49 Oxidative stress induces lipid peroxidation and oxidative DNA damage. 4-HNE, a marker of lipid peroxidation, is involved in various mechanisms of IPF development, including inflammation, myofibroblast differentiation, and ECM remodeling.50,51 Furthermore, the level of 8-OHdG, a product of oxidative DNA damage, serves as an indicator of oxidative stress. Its expression has been significantly increased in silica-induced IPF.52,53 Therefore, we employed immunohistochemistry and immunoblotting techniques to assess the expression of 8-OHdG and 4-HNE in lung tissue. Our research indicates that BLM increases the expression of these proteins, causing lipid peroxidation in lung tissue and oxidative damage to lung DNA. However, treatment with Trillin decreases the expression of these proteins. In conclusion, our findings suggest that the therapeutic impact of Trillin on BLM-induced IPF is partly due to its ability to reduce lipid peroxidation and inhibit DNA oxidative damage.
Network pharmacology and molecular docking are often combined to analyze and predict drug action targets. In our study, we employed this approach to identify and analyze the targets of Trillin and IPF, discovering 207 common targets. This was followed by GO analysis and KEGG enrichment analysis. Notably, we found that the Estrogen signaling pathway, EGFR tyrosine kinase inhibitor resistance, and the Jak-STAT signaling pathway, among others, might play crucial roles in Trillin's potential therapeutic effects against BLM-induced IPF. We further identified 18 core targets and performed molecular docking to pinpoint those with high affinity, including IL-1β, IL-2, and TNF-α. Subsequently, our results showed that Trillin down-regulated IL-1β expression and affected NLRP3 activation. It is recognized that the inflammatory and fibrotic phases are two consecutive stages in IPF development. 50 Pro-inflammatory cytokines like IL-1β, IL-6, and TNF-α can contribute to IPF development by activating fibrotic pathways mediated by TGF-β1. 51 Numerous studies showed that inflammasomes played a critical role in the inflammatory process. 52 NLRP3, extensively studied, consists of the NLRP3 protein, ASC, and the inflammatory protease caspase-1. NLRP3 is crucial in fibrosis development, and ROS activation of NLRP3 leads to cytokine precursor cleavage and mature cytokine production via Caspase-1, inducing IPF.31,53,54 Our investigation revealed that Trillin significantly reduces IL-1β, IL-6, and TNF-α production induced by BLM, demonstrating potent anti-inflammatory effects. Additionally, IL-1β can activate NF-κB by binding to the IL-1R receptor, promoting NLRP3 transcription. 19 Inhibition of the IL-1β/NF-κB pathway has been shown to alleviate BLM-induced IPF. 20
In a previous study, we demonstrated that Trillin could mitigate LPS-induced acute lung injury by inhibiting NF-κB. 10 In this study, we found that Trillin directly targets IL-1β, as identified through network pharmacology. BLM significantly increased NLRP3 expression and enhanced Caspase-1 (p20) and active IL-1β expression in lung tissue, which was reversed by Trillin treatment. In summary, our findings suggest that Trillin alleviates BLM-induced IPF by disrupting the IL-1β/NLRP3 inflammasome positive feedback loop.
In conclusion, our study demonstrated that Trillin exhibits a protective effect against BLM-induced IPF by attenuating oxidative stress and inhibiting the positive feedback loop between IL-1β and the NLRP3 inflammasome (Figure 11). These findings offer valuable insights into the therapeutic potential of Trillin for the prevention and treatment of lung diseases and other inflammatory disorders.

Schematic of Trillin's mechanism in attenuating bleomycin-induced pulmonary fibrosis. Trillin exerts anti-inflammatory, antioxidant and anti-Fibrotic effects by reducing IL-1β levels to decrease NLRP3 inflammasome activation,diminishing lipid peroxidation and DNA damage caused by oxidative stress and interfering with the transformation of fibroblasts into myofibroblasts by downregulating myofibroblast markers like α-SMA.
Footnotes
Author Contributions
The experiments were conceived and designed by W.Q.L. and F.L. and were carried out by X.Y.L. and S.S.W. The data were collected by S.L., S.F.X., Y.Y.Y., J.Q.L., and Y.C., and were analyzed by W.Q.L. and X.Y.L. The manuscript was written and revised by F.L. and X.Y.L. The experiment was administered and overseen by B.K.Z., W.Q.L., and F.L. Each author read the final draught and gave their consent.
Data Availability
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Experimental Animal Welfare of Central South University, Hunan Province, China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Project established by Chinese Pharmaceutical Association Hospital Pharmacy department, Hunan Provincial Natural Science Foundation of China, National Natural Scientific Foundation of China, Scientific Research Project of Hunan Provincial Health and Family Planning Commission (grant numbers CPA-Z05-ZC-2021-002, 2020JJ5821, 82173911,81973406, and 202113050843).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with The Ethics Committee of Experimental Animal Welfare of Central South University approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
