Abstract
Objectives
Peimine (PM), derived from Fritillaria thunbergii Miq, has been demonstrated with protective effects on pulmonary fibrosis (PF). However, the detailed mechanisms of PM on PF remain unknown. The aim of current study was to assess the therapeutic effects and the possible mechanism of PM on PF.
Methods
In this study, mice received bleomycin (BLM) injection to induce PF and then received the treatment of PM orally. The therapeutic effects of PM on PF were firstly assessed through histopathological staining (hematoxylin and eosin and Masson staining) and measuring hydroxyproline (HYP) level in lung. Then, we measured the levels of epithelial-mesenchymal transition (EMT)-related markers (vimentin and E-cadherin), pro-inflammatory factors (interleukin [IL]-1β, IL-6, and tumor necrosis factor alpha [TNF-α]), and oxidative stress-related indicators (superoxide dismutase[SOD], malondialdehyde [MDA] and glutathione peroxidase [GSH-Px]) in lung. Furthermore, untargeted metabolomics were employed to explore the effects of PM on metabolites in lung.
Results
PM treatment improved the pathological changes including reducing the infiltration of inflammatory cells and decreasing collagen deposition, and decreasing the HYP level in lung in PF mice. Moreover, PM treatment up-regulated E-cadherin and down-regulated vimentin, decreased IL-1β, IL-6 and TNF-α expression, increased SOD and GSH-Px activities, and decreased MDA level in lung. Untargeted metabolomics analysis showed that PM altered the metabolites in lung of mice with BLM-induced PF. The differential metabolites were mainly associated with tryptophan metabolism, nicotinate and nicotinamide metabolism, alanine, aspartate and glutamate metabolism, arginine biosynthesis, and D-glutamine and D-glutamate metabolism.
Conclusion
PM exhibited therapeutic effects on BLM-induced PF mice including reducing collagen deposition, inhibiting EMT and reducing inflammation and oxidative stress. The mechanism of PM on PF may be associated with regulating tryptophan metabolism, nicotinate and nicotinamide metabolism, alanine, aspartate and glutamate metabolism, arginine biosynthesis, and D-glutamine and D-glutamate metabolism in lung.
Keywords
Introduction
Pulmonary fibrosis (PF) is a disease characterized by diffuse alveolitis and structural disorders of the alveoli that eventually lead to interstitial fibrosis of the lung. Patients with PF often present with respiratory distress, which can lead to respiratory failure and death when not treated promptly and effectively. 1 The median survival time after the diagnosis of PF is approximately 2-3 years, 2 with a 5-year survival rate of approximately 50%. 3 After 2019, the antifibrotic drugs nintedanib and pirfenidone are widely used in patients with progressive PF.4–6 However, these drugs could not completely prevent the progression of PF. Currently, the only available effective therapy for progressive PF is lung transplantation but limited survival rate, high cost, and few organ donors limit its scope of application. 7 Therefore, it is urgent to explore efficient methods and medications that can downregulate the progression of PF.
Natural products derived from plants have been demonstrated with significant therapeutic effects on PF. 8 A clinical study showed that combination treatment of green tea extract and nintedanib exhibited higher clinical efficacy on PF than nintedanib treatment alone. 9 Metaanalysis revealed that combination treatment of herbal medicines and pirfenidone significantly improved cough and enhanced the quality of life in patients with PF. 10 Furthermore, natural products such as isoglycyrrhizin 11 and quercetin 12 have been demonstrated with protective effects on bleomycin (BLM)-induced PF model. Elucidating the protective mechanisms of natural products on PF can contribute to their clinical application.
Untargeted metabolomics is a technical tool capable of determining the disturbances in an organism during a specific physiological or pathological period by examining the small molecules, and it can intuitively characterize the overall metabolic profile in a biological system. 13 Owing to the widespread applications of untargeted metabolomics, many studies have confirmed that changes in the metabolic levels of the human body are closely correlated with the development of PF. The metabolomic study of clinical serum samples from 2 groups of patients with idiopathic PF (IPF) and healthy volunteers revealed the upregulation of triglycerides and ceramides in IPF patients that might be closely associated with epithelial pathology and could be used as a diagnostic marker for IPF. 14 The study of the regulation of metabolite levels provides new ideas to elucidate the mechanisms underlying the medical treatment of PF. An in vivo study revealed that the BLM can alter the distribution of metabolites in mice, while terrestrosin D reversed this change by regulating amino acid and lipid metabolism. 15 Baicalin can effectively inhibit the pathological changes of PF caused by BLM. Additionally, baicalin may improve PF by modulating 4 key pathways, including taurine and hypotaurine metabolism, glutathione metabolism, and glycerophospholipid metabolism. 16
Fritillaria thunbergii Miq has been used as a medication in China for a long history. Peimine (PM) is one of the main active constituents of Fritillaria thunbergii Miq. and has antibacterial, anti-inflammatory, analgesic, and antitussive effects. 17 These effects suggest that PM can potentially treat PF. An in vivo study confirmed that PM could prevent PF by inhibiting macrophage M2 polarization. 18 However, the detailed mechanism of PM on PF remains unclear. To explore the therapeutic effect of PM on PF and its mechanism of action, this study first used BLM to induce PF in mice and then treated the mice with PM. The therapeutic effects of PM on mice with PF were firstly identified. Then, the effects of PM on epithelial-mesenchymal transition (EMT), inflammatory response and oxidative stress were investigated. Finally, untargeted metabolomics was employed to explore the effects of PM on metabolites in the lung.
Methods
Animals and Reagents
C57BL/6 mice were purchased from SPF biotechnology. PM (B20080, CAS: 23496-41-5, purity: ≥ 98%, formula: C27H45NO3, molecular structure: Figure S1), BLM (B73357) and dexamethasone (DXM, S17003) were purchased from Yuanye Bio-Technology. Assay kits for Hydroxyproline (HYP) concentration (A030-2-1), superoxide dismutase (SOD) activity (A001-1-2), malondialdehyde (MDA) concentration (A003-1-2), glutathione peroxidase (GSH-Px) activity (A005-1-2), and total protein concentration (A045-4-2) were purchased from Jiancheng Bioengineering institute. Enzyme-linked immunosorbent assay kits for interleukin (IL)-6 (EK206), IL-1β (EK201B), and tumor necrosis factor (TNF)-α (EK282) were obtained from Multi sciences.
Animal Experiments
In this study, 75 male 8-week-old C57BL/6 mice (20-22 g) were housed at 25 °C, 45% relative humidity. Based on previous studies, DXM was selected as a positive control drug. 19 The mice were grouped into control, model, DXM, PM low dose (LPM), and PM high dose (HPM) groups randomly. Anesthesia was performed by intraperitoneally injecting sodium pentobarbital and mice were then intratracheally injected with BLM (3 mg/kg) or saline. Drug treatment was started on the second day of intratracheal injection. For the treatment, mice in the DXM, LPM, and HPM groups were orally treated with 1 mg/kg DXM, 20 15 mg/kg PM, and 30 mg/kg PM per day, 21 respectively. The control and model groups were administered with vehicle. After 21 days of drug treatment, all mice were euthanized. The lung tissue was collected and frozen or fixed for further study. This study was approved by the Animal Ethics Committee at Jiaxing Hospital of Traditional Chinese Medicine (approval no. 2021-0016).
Lung Tissue Biochemical Test
HYP concentration, SOD activity, MDA concentration, GSH-Px activity, and total protein concentration in lung tissue were measured using biochemical assay kits. These assays were performed by following the procedure mentioned in the kit. Briefly, lung tissues were prepared into homogenates according to the instructions firstly. The homogenates were added to the 96-well plate along with the reaction substrate and the reaction was carried out according to the guidelines of instructions. The color development reaction was then carried out by adding the corresponding chromogenic reaction substrate. After color development, the absorbance was measured at a specific wavelength to calculate the content of the target component.
Histopathological Testing
The fixed lung tissues were transferred to different concentrations of ethanol, dehydrated and embedded in paraffin to obtain 3-μm sections. Then, the sections were stained using hematoxylin and eosin (HE) or Masson's trichrome stain. The severity of alveolitis was quantified and analyzed by determining the Szapiel score of the HE-stained samples. 22 The severity of fibrosis was analyzed using the modified Ashcroft score of the Masson-stained samples. 23
ELISA
IL-1β, IL-6, and TNF-α in lung tissue homogenates were measured using a double-antibody one-step sandwich ELISA. The thresholds for IL-6, IL-1β, and TNF-α test kits were 23.44-1500, 7.81-500, 10.938-700 pg/mL, respectively. In brief, the test sample, standard, and detection antibody were added to the microplate wells coated with the captured antibody. Next, the plate was incubated in a water bath and then thoroughly washed. Further, the reaction substrate was added and the absorbance of the developed color was measured using a microplate reader. The standard curve was plotted using the results of the standard and the concentrations of the analytes were calculated in each sample. The concentration of lung tissue protein was used for normalization of the results.
Reverse Transcription Quantitative Polymerase Chain Reaction
Total RNA of the lung tissue was extracted using TRIzol. After normalization, cDNA was prepared using a reverse transcription kit (KR116, TIANGEN biotec Co., Ltd). All cDNA samples were subjected to quantitative polymerase chain reaction (qPCR) by following the procedure mentioned in the qPCR kits (FP205, TIANGEN biotec Co., Ltd) to determine the mRNA expression levels of Il1b, Il6, Tnfa, Vim, and Cdh1. Primer sequences have been mentioned in Table 1. The data were analyzed using 2−ΔΔCT method.
Primer Sequence.

Western Blot
The total protein of lung tissue was extracted from the lung tissue using RIPA lysis buffer. After standardizing the protein concentration, the samples were mixed with the loading buffer and denatured in a water bath at 98 °C for 7 min. After performing electrophoresis on a 10% sodium dodecyl sulfate-polyacrylamide gel, the separated proteins were transferred onto the polyvinylidene fluoride membrane. Antibodies for vimentin (10366-1-AP, Proteintech, 1:5000) or epithelial cadherin (E-cadherin, 20874-1-AP, Proteintech, 1:5000) were added to the membrane after blocking and it was then incubated overnight at 4 °C. Next, the membranes were washed thrice and incubated with relative secondary antibodies at room temperature. The membranes were washed again and then developed by enhanced chemiluminescence. Quantitative analysis of the captured bands was performed using Image J software.
Lung Tissue Metabolomics
Lung tissues were homogenized using 80% methanol in the ratio of 1:5. The suspension was centrifuged and the supernatant was diluted with water to reduce the methanol concentration to 53%. Next, the suspension was recentrifuged and the supernatant was collected for untargeted metabolomics analysis that was performed as previously described. 24 Quality control (QC) samples and QC matrices, metabolite analysis, data processing, and other details can be found in the Supplemental Material.
Statistical Analysis
All data are expressed as mean ± standard deviation (SD) and compared by one-way analysis of variance followed by Tukey honest significant difference (HSD) test as post hoc analysis. P-value of <.05 was considered to be statistically significant.
Results
Therapeutic Effects of PM on PF Mice
After 21 days treatment, the numbers of survived mice in control, model, DXM, LPM, HPM were 15, 7, 10, 9, 11, respectively (survival rate 100%, 46.67%, 66.67%, 60%, 73.33% respectively, Figure S2). The HE staining results showed that the mice in the control group had normal lung tissue structure, clear and intact alveolar structure, and no apparent inflammatory cell infiltration. Compared to the control group, lung tissues of the model mice showed dilated alveolar septum, collapsed or even occluded alveolar cavity, and significant inflammatory cell infiltration in lung tissues. Compared to the model group, the structure of lung tissue of mice in the DXM, LPM, and HPM groups was improved and lesser inflamed (Figure 1a). The results of Szapiel score were consistent with the HE results; higher scores in the model group were observed than the control group, whereas significantly lower scores were obtained in the DXM, LPM, and HPM groups than the model group (Figure 1c). Masson staining results showed that the collagen deposition was not significant in the control group of mice. On the other hand, the model group showed significant collagen deposition in the airways, around the blood vessels, and in the consolidation areas. Compared to the model group, collagen deposition in the lung tissue of mice in the DXM, LPM, and HPM groups was significantly reduced (Figure 1b). The Ashcroft scoring results were consistent with the Masson staining results, wherein more severe fibrosis was observed in the model group, and relatively less severe fibrosis was seen in the DXM, LPM, and HPM groups than model group (Figure 1d). Similarly, higher HYP levels were observed in the lung tissues of the model group. Compared to the model group, the HYP levels in the lung tissues of the mice in the DXM, LPM, and HPM groups were considerably lower (Figure 1e).
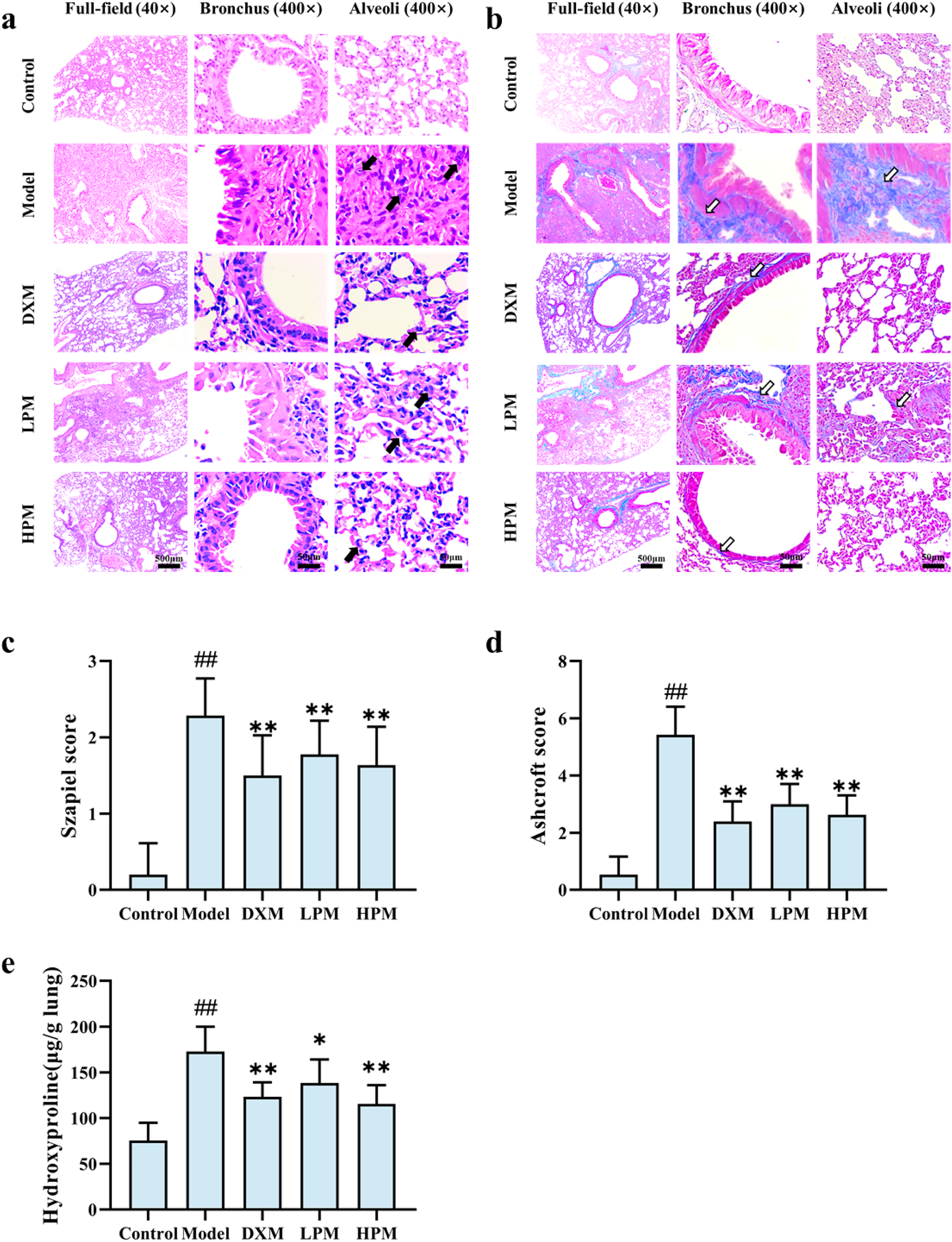
PM has a therapeutic effect in mice with BLM-induced PF. Mice received intratracheal injections of BLM (3 mg/kg) on day 0 to establish PF model. Different doses of PM were given to mice by gavage daily starting on day 1. All mice were euthanized on day 21. (a-d) HE staining and Szapiel scores (a, c) and Masson trichrome staining and Ashcroft scores (b, d) of lung tissue showed that both inflammation and collagen deposition in lung tissue were alleviated after receiving PM treatment. (e) Elevated HYP concentrations in fibrotic lung tissue also decreased after PM treatment. All the data are presented as mean ± SD (n = 15, 7, 10, 9, 11 for 5 groups respectively). ##: P < .01, compared with control group; *: P < .05, compared with model group; **: P < .01, compared with model group, all determined by one-way ANOVA with post hoc Tukey HSD test. Abbreviations: DXM, dexamethasone group; PM, peimine; LPM, peimine low dose group; HPM, peimine high dose group; PF, pulmonary fibrosis; BLM, bleomycin; HYP, hydroxyproline; HE, hematoxylin and eosin; HSD, honest significant difference. Black arrows indicated inflammatory cells. White arrows indicated fibrosis.
Effects of PM on EMT in PF Mice
The reverse transcription quantitative polymerase chain reaction (RT-qPCR) results showed that the mRNA expression levels of Vim were lower and Cdh1 mRNA expression levels in the lung tissues of mice from the model group were higher than those of the control group. Compared to the model group, the mRNA levels of Vim were significantly higher in the DXM, LPM, and HPM groups, whereas Cdh1 mRNA relative expression level was relatively lower (Figure 2a, b). Western blot results showed that the relative protein expression of vimentin in the lung tissue of the model group was remarkably lower and E-cadherin protein was considerably higher than that in the control group. The relative expression of vimentin in the DXM, LPM, and HPM groups was higher and the relative expression of E-cadherin was lower than in the model group (Figure 2c-e).

PM treatment reduced EMT in pf model mice. (a-e) The mRNA levels of Vim (a) and Cdh1 (b) were measured using RT-qPCR, and the expression of EMT-related marker proteins vimentin (c, d) and E-cadherin (c, e) was examined by western blot. PM treatment decreased gene and protein expression of the marker of mesenchymal cell (vimentin) and increased the marker of epithelial cell (E-cadherin) in lung. n = 3 per group). ##: P < .01, compared with control group; *: P < .05, compared with model group; **: P < .01, compared with model group, all determined by one-way ANOVA with post hoc Tukey HSD test. Abbreviations: DXM, dexamethasone group; PM, peimine; LPM, peimine low dose group; HPM, peimine high dose group; EMT, epithelial-mesenchymal transition; RT-qPCR, reverse transcription quantitative polymerase chain reaction; HSD, honest significant difference.
Effects of PM on Inflammatory Response and Oxidative Stress in PF Mice
The IL-1β, IL-6, and TNF-α in the lung tissue of mice in model group were higher than those in the control group. The levels of IL-1β, IL-6, and TNF-α in DXM, LPM, and HPM groups were considerably lower compared to the model group (Figure 3a-c). The mRNA levels of Il1b, Il6, and Tnfa were higher in the mice with PF compared to those of the control group. The mRNA levels of Il1b, Il6, and Tnfa were lower in DXM, LPM, and HPM groups than those in the model group (Figure 3d-3f).

PM has anti-inflammatory and oxidative stress-reducing effects in mice with BLM-induced PF. (a-c) ELISA results showed that the elevated IL-1β (a), IL-6 (b) and TNF-α (c) in fibrotic lung tissue were significantly reduced after getting PM treatment. (d-f) RT-qPCR results for mRNA levels of Il1b (d), Il6 (e) and Tnfa (f) were down-regulated after PM treatment. (g-i) Levels of oxidative stress-related indicators including SOD (g), MDA (h), GSH-Px (i) in lung tissue were measured. PM treatment significantly improved oxidative stress. All the data are presented as mean ± SD (n = 15, 7, 10, 9, 11 for 5 groups respectively in ELISA and biochemical assay; n = 3 per group in RT-qPCR). ##: P < .01, compared with control group; *: P < .05, compared with model group; **: P < .01, compared with model group, all determined by one-way ANOVA with post hoc Tukey HSD test. Abbreviations: DXM, dexamethasone group; PM, peimine; LPM, peimine low dose group, HPM, peimine high dose group; PF, pulmonary fibrosis; BLM, bleomycin; SOD, superoxide dismutase; MDA, malondialdehyde; GSH-Px, glutathione peroxidase; TNF-α, tumor necrosis factor alpha; RT-qPCR, reverse transcription quantitative polymerase chain reaction; HSD, honest significant difference.
In addition, the antioxidative markers in the lung tissue of each group were measured by performing biochemical test. The results showed that SOD and GSH-Px activities were lower, while MDA level was higher in the lung tissues of the mice in the model group than those in the control group. Compared to the model group, SOD and GSH-Px activities were significantly higher and MDA levels were significantly lower in the lung tissues of the mice in DXM, LPM, and HPM groups (Figure 3g-i).
In summary, DXM or PM treatment improved histopathological changes, inhibited EMT, lowered inflammatory responses, and alleviated oxidative stress in the lungs of mice with PF. This suggested that PM could be served as a potential therapeutic agent for PF. Between the 2 PM groups, more significant improvement can be found in the HPM group. Therefore, the high-dose group of PM was selected for the follow-up experimental study.
Effects of PM on Lung Metabolites in PF Mice
Multivariate analysis (principal component analysis, PCA) was firstly conducted to reflect the general differences in metabolites among each group. 25 Figure 4a shows that the samples of the control and model groups were clustered separately in the PCA score plot, and a clear separation in the dispersion area was observed, indicating that the lung metabolic profile of model group mice was changed. Similarly, the samples of the model and HPM groups were also clustered separately in the PCA score plot with a clear separation in the dispersion area, indicating that the lung metabolic profiles of the mice in the HPM group were changed. To obtain better separation and reduce confounding factors and to screen the differential metabolites, a partial least squares-discriminant analysis (PLS-DA) supervised model was established to evaluate the explanatory and predictive power of the model. The results showed that the lung metabolic profiles of the control and model groups and the model and HPM groups were well separated and the PLS-DA model had high explanatory and predictive powers. To verify the validity of the PLS-DA model, permutation tests were performed. According to the criteria of permutation tests, the analysis model should be considered to be valid when the values of R2-points are higher than the values of Q2-points and the intercept of the regression line of the Q2-points to the vertical axis are at, or below zero. 26 The results suggested that the PLS-DA model is reliable in terms of explanatory and prediction powers (Figure 4b-e).

Pm alters the metabolites of lung tissue in mice with BLM-induced PF. (a) Score plots of principal component analysis among control, model and HPM groups. (b-e) Score plots and permutation tests of partial least squares discriminant analysis between the control and model groups (b, c), and between the model and HPM groups (d, e). Results of pathway analysis between control and model groups (f) and between model and HPM groups (g). Heat map of the common pathway related differential metabolites (`). Common pathways are marked in red. Pathways indicated by “a-t” can be found in legend of Table 2. Control, Model and HPM groups (n = 6 per group). Abbreviations: C: control group; M, model group; P, HPM group; PF, pulmonary fibrosis; HPM, peimine high dose group; BLM, bleomycin
Differential Metabolites in Lung After PM Treatment.
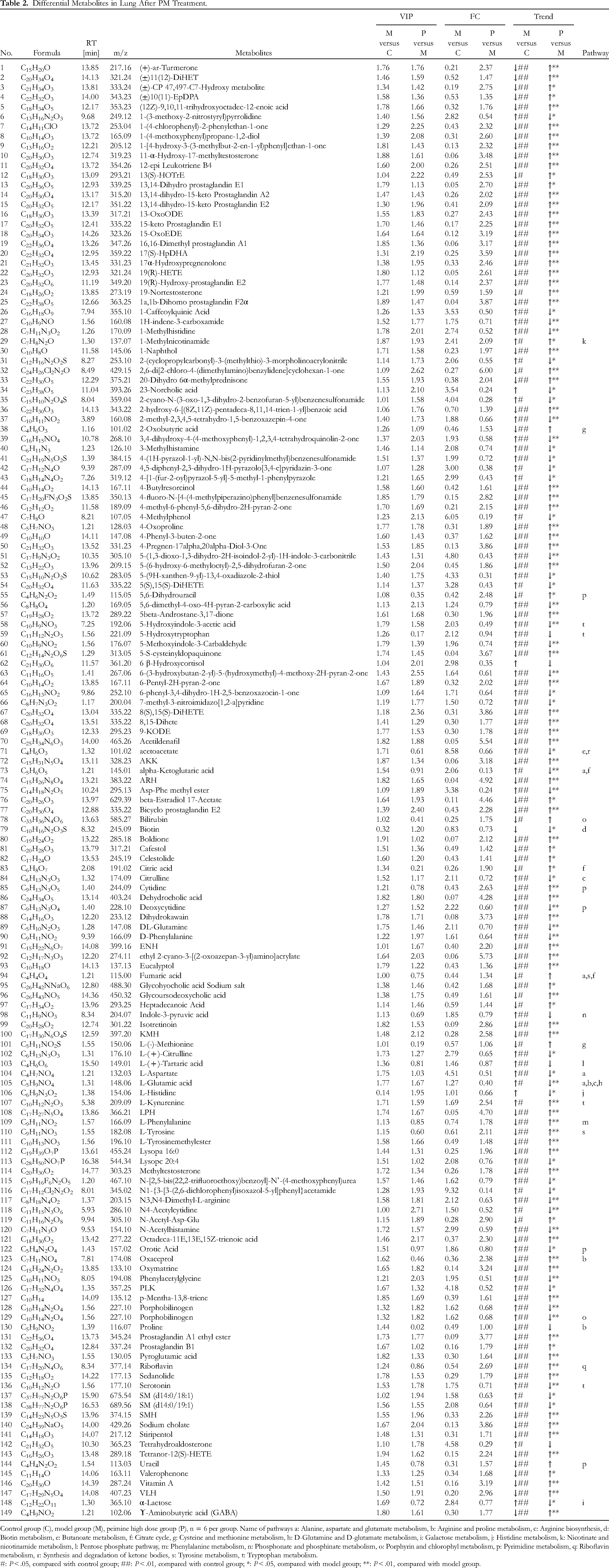
Control group (C), model group (M), peimine high dose group (P), n = 6 per group. Name of pathways a: Alanine, aspartate and glutamate metabolism, b: Arginine and proline metabolism, c: Arginine biosynthesis, d: Biotin metabolism, e: Butanoate metabolism, f: Citrate cycle, g: Cysteine and methionine metabolism, h: D-Glutamine and D-glutamate metabolism, i: Galactose metabolism, j: Histidine metabolism, k: Nicotinate and nicotinamide metabolism, l: Pentose phosphate pathway, m: Phenylalanine metabolism, n: Phosphonate and phosphinate metabolism, o: Porphyrin and chlorophyl metabolism, p: Pyrimidine metabolism, q: Riboflavin metabolism, r: Synthesis and degradation of ketone bodies, s: Tyrosine metabolism, t: Tryptophan metabolism.
#: P < .05, compared with control group; ##: P < .01, compared with control group; *: P < .05, compared with model group; **: P < .01, compared with model group.
The following 3 criteria were used to screen for differential metabolites: P < .05, fold change >1.2 or < .8, and VIP > 1.0. A total of 149 differential metabolites were obtained from screening (Table 2).
Having screened the differential metabolites, we further analyzed the pathway related these metabolites using MetaboAnalyst platform (https://www.metaboanalyst.ca/) and Kyoto Encyclopedia of Genes and Genomes. The MetaboAnalyst platform is commonly used in analyzing the pathway of metabolites, 27 Differential metabolic pathways were selected based on a pathway impact >.05 and P < .05. The metabolic pathways that were altered in the model group compared with the control group mainly included tryptophan metabolism, nicotinate and nicotinamide metabolism, alanine, aspartate and glutamate metabolism, arginine biosynthesis, D-glutamine and D-glutamate metabolism, arginine and proline metabolism, porphyrin and chlorophyl metabolism, pyrimidine metabolism, galactose metabolism, cysteine and methionine metabolism, synthesis and degradation of ketone bodies, butanoate metabolism, tyrosine metabolism, citrate cycle (TCA cycle), phosphonate and phosphinate metabolism, pentose phosphate pathway, riboflavin metabolism, and phenylalanine metabolism. The metabolic pathways that were significantly altered in PF mice after PM treatment included tryptophan metabolism, nicotinate and nicotinamide metabolism, alanine, aspartate, and glutamate metabolism, arginine biosynthesis, D-glutamine and D-glutamate metabolism, histidine metabolism, and biotin metabolism, of which the tryptophan metabolism, nicotinate and nicotinamide metabolism, alanine, aspartate and glutamate metabolism, arginine biosynthesis, and D-glutamine and D-glutamate metabolism were common pathways between the control group and model groups as well as between the model and HPM groups (Figure 4f, 4g). To improve the visualization, the relative contents of pathway-related metabolites in the 3 groups were displayed by a heatmap (Figure 4h).
Discussion
In this study, we first constructed a BLM-induced PF mouse model and evaluated the therapeutic effect of PM. Pathological staining results revealed a significant proliferation of interstitial lung tissue with inflammatory cell infiltration in the model group, which was accompanied by a significant increase in collagen deposition in the lung tissue. In addition, the level of HYP was elevated in the lung tissue of the model group. The level of HYP could reflect the degree of fibrosis, and these changes were in agreement with the pathological features of PF. The mice with PF showed improved histopathological features and less HYP levels after treatment with PM.
In addition, we evaluated the effect of PM on EMT by detecting the expression of the epithelial cell marker (E-cadherin) and mesenchymal cell marker (vimentin) in lung tissue. The results showed that the expression of E-cadherin and vimentin in the model group was downregulated and upregulated respectively, suggesting the occurrence of EMT. PM treatment inhibited EMT in PF model mice. EMT is crucial for the production of myofibroblasts in PF. 28 EMT promotes the transdifferentiation of epithelial cells into fibroblasts that are then activated and transformed into α-SMA-expressing myofibroblasts. 29
Furthermore, our results revealed the anti-inflammatory and anti-oxidative potentials of PM on PF. Pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α play an important role in the development of PF. These cytokines have been shown to be elevated in PF mice. 30 Some drugs targeting inflammatory cytokines have entered phase II or III trials, such as IL-1 inhibitor (NCT01538719) for systemic sclerosis, 31 IL-6 inhibitor (NCT02453256) for scleroderma, 32 and anti-TNF antibody (adalimumab; NCT03180957) for the treatment of fibroproliferative disorders of the hands. 33 Besides, oxidative stress has equal importance in the development of PF. SOD and GSH-Px, which are major antioxidant enzymes, play a crucial role in maintaining the oxidant/antioxidant balance, whereas MDA is the lipid peroxidation end product in the body, and its content is indicative of the degree of cellular oxidative injury. 34 Several anti-oxidative agents such as quercetin (NCT02874989) 35 and inhibitors of the oxidant species such as GKT137831 (NCT03865927) 36 have entered clinical trials for the treatment of PF.
The pharmacodynamic studies on PM above have proved that it has good therapeutic effects on PF, particularly at high dose. DXM has significant anti-inflammatory effects and is commonly used clinically as a standard of care in the treatment of PF.37,38 Therefore, it was chosen as the positive control in this study and no significant difference was found between DXM and HPM groups in therapeutic effects according to the studies above. Based on this, we selected the HPM group for metabolomic analysis to explore the potential therapeutic pathways. The results showed that alanine, aspartate, and glutamate metabolism; arginine biosynthesis; D-Glutamine and D-glutamate metabolism; nicotinate and nicotinamide metabolism; and tryptophan metabolism are common metabolic pathways among the control, model, and HPM groups. The obtained results suggested that these pathways might be the potential pathways for the therapeutic effects of PM.
Alanine, Aspartate, and Glutamate Metabolism; Arginine Biosynthesis; and D-Glutamine and D-Glutamate Metabolism Pathways
Alanine, aspartate, and glutamate metabolism; arginine biosynthesis; and D-glutamine and D-glutamate metabolism metabolic pathways had great relations. In the present study, glutamate, α-ketoglutaric acid (α-KG), citrulline and aspartate levels were decreased in PF mice after PM treatment. Glutamate and α-KG are intermediates and end products of glutamine catabolism respectively, while citrulline is the intermediate product of the urea cycle. Glutamine catabolism has an important role in the pathology of PF. 39 For ECM, fibroblasts require large amount of energy in order to synthesize collagen. Besides relying on glycolysis, glutamate produced by the glutamine catabolic pathway is also converted by glutamate dehydrogenase to α-KG that enters the TCA cycle function to provide energy. 40 Moreover, glutaminolysis also reduces apoptosis and increases collagen stability, thus contributing to the development of PF.41,42 When glutamate is converted to α-KG, ammonia is also produced in large quantity as a by-product of the reaction and subsequently excreted out of the body through the urea cycle. Ammonia undergoes a 2-step reaction, wherein ornithine synthesizes the first product, followed by aspartate that synthesizes argininosuccinic acid, which is then broken down into fumarate and arginine. Arginase is highly expressed in PF-affected lung tissue and converts arginine to ornithine and urea. 43 However, further studies are necessary to identify if this effect was due to the direct intervention of PM on the urea cycle or indirect intervention due to the modulation of glutamine catabolism caused by PM.
Nicotinate and Nicotinamide Metabolism
Our results found that elevation of 1-Methylnicotinamide (MNAM), a metabolite involved in nicotinate and nicotinamide metabolism, was observed in model group, and it increased after the treatment with PM. MNAM is produced from nicotinamide, catalyzed by nicotinamide N-methyltransferase (NNMT), and can selectively block the interaction of Smad3 with TGF-β receptor I, which in turn exerts antifibrotic and anti-inflammatory functions. 44 It has been found that NNMT expression is elevated in a compensatory manner during the fibrosis process to increase the level of MNAM. 45 It has also been found that supplementation of nicotinic acid, the precursor of MNAM, ameliorates fibrosis, which may be related to the increase in the levels of reactive substrates as well as alleviating nicotinamide adenine dinucleotide depletion.46–48 However, further studies are required to confirm if PM is directly involved in niacin-associated pathways.
Tryptophan Metabolism
In this study, PM treatment significantly reduced the levels of serotonin (5-hydroxytryptamine, 5-HT) and 5-hydroxyindole-3-acetic acid (5-HIAA) and elevated the level of kynurenine in the lung tissue of the mice with PF. L-Kynurenine in tryptophan metabolism belongs to the kynurenine pathway, while 5-HIAA, 5-hydroxytryptophan, and 5-HT all belong to the 5-HT pathway. Interestingly, these 2 pathways affect the pathology of PF in opposite ways. Kynurenine has an antagonistic effect on the development of PF. Supplementation with kynurenine can alleviate a variety of fibrotic diseases, including PF.49,50 5-HT is a neurotransmitter. 5-HT upregulates the expression of TGF-β1 and increases the nuclear localization of phosphorylated Smad3, which in turn promotes myofibroblast differentiation and ECM deposition, exacerbating fibrosis. 51 In contrast, treatment of human lung fibroblasts with inhibitors of the 5-HT transporter 5-HT2A/2B inhibited TGF-β expression and reduced collagen deposition. 52 In addition, 5-HT promotes the release of the inflammatory cytokines including IL-1β, IL-6, and TNF-α that can also be inhibited by 5-HT transporter inhibitors.53–55 5-HT is converted to 5-HIAA catalyzed by monoamine oxidase and is excreted in the urine ultimately. Notably, the precursor of both the kynurenine pathway and 5-HT pathway is tryptophan, suggesting the correlation between the 2 pathways may due to an underlying homeostatic mechanism. It suggests that the antifibrotic effect of PM may be potentially associated with the regulation of 5-HT level by enhancing 5-HT catabolism. Although this study did not find any notable changes in melatonin levels before and after the PM treatment, it is still a factor that needs further study.
There are also some limitations in our study. A direct effect of PM on EMT, inflammatory response, and oxidative stress were evaluated using animal model. However, all of these effects were not examined in vitro using cultured cells. Interestingly, with the increase in understanding the roles of metabolites in physical and pathological conditions, endogenous metabolites have been shown to be involved in various pathological processes in PF including EMT, 56 inflammatory response, 57 and oxidative stress. 58 In vitro models such as EMT of alveolar epithelial cells (TGF-β-induced EMT in A549 cell), 59 M1/M2 macrophage polarization (LPS-induced M1 and IL-4-induced M2 polarization in RAW264.7 cell) 60 and oxidative stress-mediated alveolar epithelial cell death (H2O2-induced oxidative stress in A549 cell) 61 can be used in our future studies to further verify the effects and deeply elucidate the mechanisms of PM on PF based on modulating host metabolism. Moreover, according to the development of pathology in BLM-induced PF mouse model, mice undergo inflammatory phase from day7-10 after BLM injection. 62 The effects of PM on cytokine levels in the inflammatory phase of BLM-induced PF mouse model could also be studied in our future research work to make a comprehensive evaluation of PM on different stages of PF.
Conclusion
This study found that PM treatment can ameliorate PF by decreasing collagen deposition, inhibiting EMT, and reducing inflammatory response and oxidative stress. The mechanism of PM in the treatment may be related to regulating the tryptophan metabolism, nicotinate, and nicotinamide metabolism, alanine, aspartate, and glutamate metabolism, arginine biosynthesis, and D-glutamine and D-glutamate metabolism pathways (Figure 5).
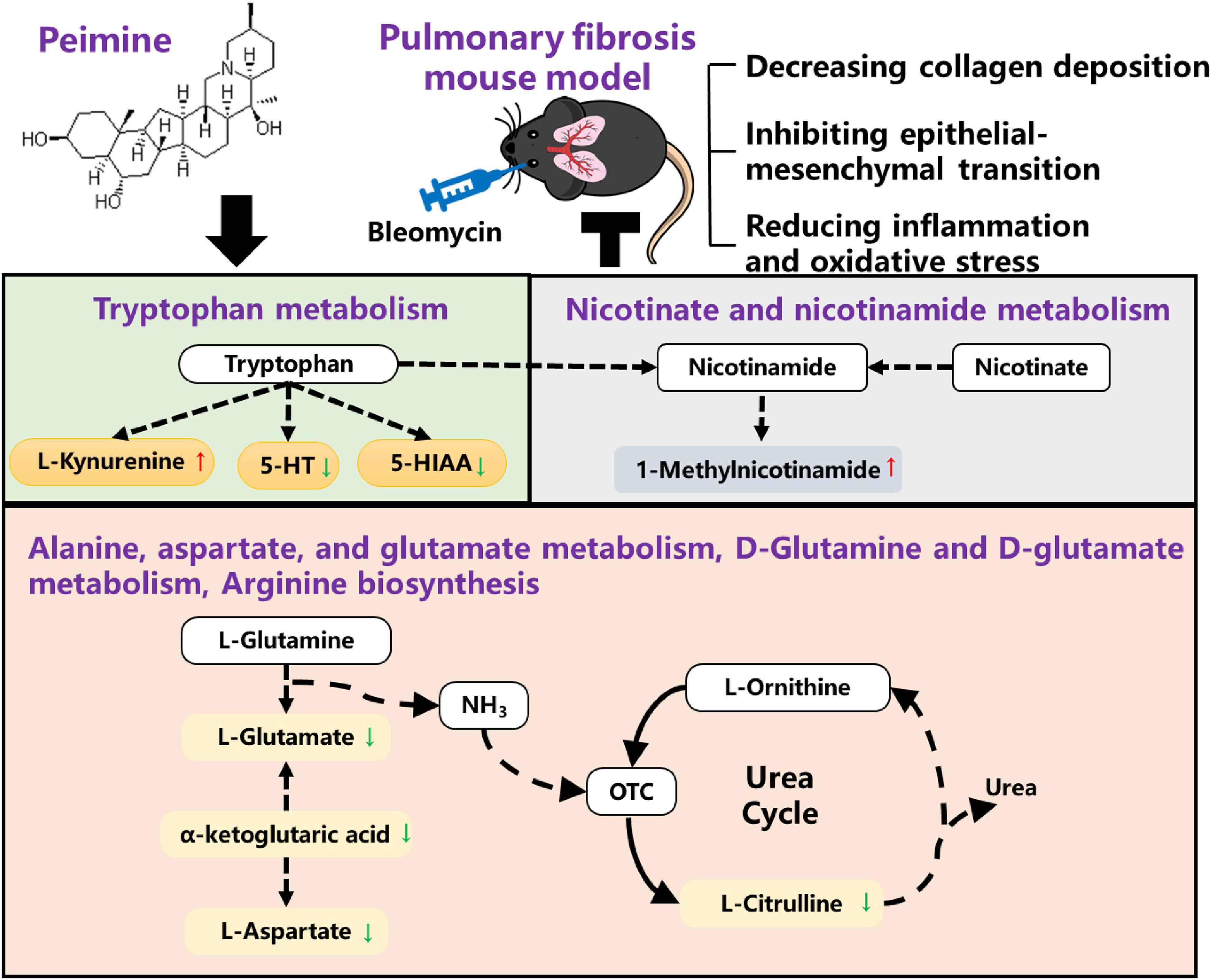
Graphical abstract.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231214947 - Supplemental material for Peimine Alleviated Bleomycin-Induced Pulmonary Fibrosis in Mice Through Reducing Epithelial-Mesenchymal Transition, Inflammation and Oxidative Stress and Regulating Host Metabolism
Supplemental material, sj-docx-1-npx-10.1177_1934578X231214947 for Peimine Alleviated Bleomycin-Induced Pulmonary Fibrosis in Mice Through Reducing Epithelial-Mesenchymal Transition, Inflammation and Oxidative Stress and Regulating Host Metabolism by Xiaojuan Li, Feng Zhang, Aijuan Shen, Jian Hu, Minjin Chen and Huiqun Yang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zhejiang Chinese Medicine University joint project (grant number 2022FSYYZZ23).
Ethical Approval
This study was approved by the Animal Ethics Committee at Jiaxing Hospital of Traditional Chinese Medicine (approval no. 2021-0016).
Statement of Human and Animal Rights
All procedures conducted in this study were in accordance with the Institutional Animal Care guidelines of Jiaxing Hospital of Traditional Chinese Medicine, China and approved by the Animal Ethics Committee of Jiaxing Hospital of Traditional Chinese Medicine (approval no. 2021-0016).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
