Abstract
Introduction
Nonalcoholic fatty liver disease (NAFLD) is a liver disease characterized by abnormal lipid accumulation and steatosis in liver parenchymal cells caused by nonalcoholic factors. 1 NAFLD encompasses the liver disease process of simple fatty liver, non-alcoholic steatohepatitis (NASH), and liver fibrosis, and can even progress to cirrhosis or liver cancer. 2 NAFLD is a multifactorial disease, the development of which is influenced by various factors such as metabolism, environment, genetics, and intestinal microorganisms. It includes physiological and biochemical changes including aberrant lipid metabolism in the hepatocytes, insulin resistance, inflammatory response, and endoplasmic reticulum stress. 3 Its incidence and disease burden have recently increased, making it a public health problem worldwide.4,5 The prevalence of NAFLD has risen to about 29.8% worldwide, with South America and the Middle East having the highest frequency and Africa having the lowest. 6
No specific medication has been approved for NAFLD or NASH. Conventional treatment of NAFLD includes lifestyle interventions such as dietary management, exercise, and weight loss. Clinical therapy strategies primarily focus on reducing hepatic fat accumulation and metabolic load, oxidative stress, inflammation, gut microbe regulation, and anti-hepatic fibrosis. Whereas the efficacy of these therapeutic measures is not very pronounced (<50% histologic improvement in NASH), and bariatric surgery may be required to achieve and maintain therapeutic benefits when necessary.7–9 Therefore, it is of great significance to study the pathogenesis of fatty liver and explore effective intervention targets for developing targeted therapeutic drugs. NAFLD is a multisystem metabolic disease with complex pathogenesis. Early theories for the development of NAFLD were described as the “two-hit hypothesis”, where the accumulation of hepatic fat due to obesity and insulin resistance constituted the “first hit” while oxidative stress and inflammatory reactions constituted the “second hit”. 10 Subsequent studies have proposed a “three-hit hypothesis” that includes fat accumulation, oxidative stress, inflammation, impaired hepatocyte regeneration, and proliferation. 11 Nowadays, the widely accepted theory is the “multiple-hit model”, 12 which involves a wider range of metabolic processes. However, steatosis, oxidative stress, and inflammatory responses remain the primary drivers of NAFLD.13–15
Western treatment is unsatisfactory with highly toxic side effects, it has been a hot topic in recent years to explore the treatment options for this disease from the perspective of traditional Chinese herbs. It has been shown that natural extracts of herbs such as Safflower yellow, Honokiol, Zanthoxylum bungeanum amides, Quercetin, and Hesperidin, have a mitigating effect on lipid accumulation and oxidative stress caused by NAFLD.16–19 The holistic effect of multi-target therapy and the advantages of fewer toxic side effects give Chinese herbs and other natural ingredients a wide range of potential clinical applications in improving NAFLD. Sericin is a natural protein secreted by the domestic silkworm that encapsulated on the surface of the silk, which is rich in serine, aspartic acid, threonine, and many other amino acids. 20 The complex and varied structure of the sericin protein and its rich amino acid moiety gives it a wide range of biological activities and properties, such as whitening and moisturizing, antioxidant, anti-inflammatory, antimicrobial, hypoglycemic, and pro-cellular value-added.21–23 Previous studies showed that wound dressings made of sericin protein have excellent breathability, in addition to pH-responsive degradation, softness, super-absorbency, as well as good cytocompatibility. 24 It is shown that sericin has a wide range of applications and commercial value due to its antioxidant activity. 25 Another study showed that 250 mg/kg sericin was effective in inhibiting the expression of inflammatory factors in the hippocampus and improving the antioxidant capacity in aged mice after 21 days of treatment. 26 Other studies have demonstrated the anti-inflammatory and antioxidant effects of sericin on various disease.27–30
In our preliminary experiments, sericin was given to hyperglycemic mice by gavage at doses of 250, 500, and 1000 mg/kg body weight for 30 days. After sericin intervention, mice in the hyperglycemic model showed a decrease in blood glucose and a decrease in area under the curve (AUC). 31 It showed that sericin had an auxiliary hypoglycemic effect on the hyperglycemic model mice. 31 These results suggest that sericin may have a positive effect on symptom relief in NAFLD. Previous studies have shown that NAFLD leads to elevated levels of oxidative stress and inflammation in vivo; while nuclear factor-erythroid 2-related factor 2 (Nrf2) and its downstream proteins, such as kelch-like Epichlorohydrin-associated protein 1 (keap1), heme oxygenase 1(HO-1), glutathione peroxidase 4 (GPX4), and nuclear factor kappa-B p65 (NF-κB p65), are implicated in this physiological change.32–35 Thereby, the objective of this study was to evaluate the effects of sericin on NAFLD model mice and to explore its mechanism of action from the perspective of Nrf2-mediated antioxidant and anti-inflammatory pathways. The results of this study were expected to provide new insights into the pharmacological effects of sericin.
Methods and Materials
Sericin Extraction and its Characterization
Fresh Bombyx mori cocoons were provided by the Guangxi Institute for Product Quality Inspection (Nanning, China). In the present study, sericin was extracted by methods described previously. 20 Briefly, dried cocoons were weighted, cut into pieces, and degummed in deionized water at a ratio of 1:30 (w: v) at 100 °C for 3 h twice. Insoluble components of the soaking solution was filtered and the solution was freeze-dried to obtain sericin powder. Sericin extracts were analyzed with an amino acid analyzer (Hitachi L-8500A, Tokyo, Japan) and the major components were serine (26.0%), aspartic acid (16.5%), and threonine (9.8%). 20
Experimental Animals
60 healthy adult male specific-pathogen-free C57BL/6 mice (6-8 weeks of age), weighing 24-28 g, were purchased from Hunan Silaike Jingda Laboratory Animal Co., Ltd, and reared in the Laboratory Animal Center of Guangxi University of Traditional Chinese Medicine. Mice were reared in a barrier system with constant temperature (22-25 °C) and humidity (55%–70%). The indoor light was provided for 12 h a day. The normal diet was provided by the animal experimental center. In contrast, the high-fat diet was formulated according to a specific formula shown in Table 1. All animal experimental procedures were approved by the Animal Care Committee of Guangxi University of Chinese Medicine (No.DW20220408-060).
High-Fat Feed Formulations.

Establishment of NAFLD Model Mice, Experimental Grouping, and Sample Collection
After one week of adaptive feeding, 8 SPF-healthy male C57BL/6 mice (6-8 weeks old) were randomly selected as the control group and fed with a normal diet. The remaining mice were fed with a high-fat diet for modeling. 8 weeks later, 4 mice fed with the high-fat diet were randomly selected for liver pathology sections to observe whether NAFLD models were formed. The successfully modeled mice were randomly divided into the model group, 1000 mg/kg sericin group, 500 mg/kg sericin group, 250 mg/kg sericin group, 1000 mg/kg sericin + ML385 group, Zhibitai Capsules group, with 8 mice in each group. The experimental dose was selected based on the results of our previous 90-day subchronic toxicity study and hypoglycemic experiment, which indicated that the NOAEL of sericin was > 1000 mg/kg in SD rats and Kunming mice by gavage.20,31 ML385 (MedChemExpress, New Jersey, USA) was chosen as a protein inhibitor of Nrf2. Zhibitai Capsules (Chengdu Diao Group, China), a proprietary Chinese medicine that has been shown to have good therapeutic effects on NAFLD rats, 36 was chosen as the positive medicine. Mice in the sericin dose groups were given a corresponding dose of sericin every day by intragastric administration for 4 weeks. Mice in the 1000 mg/kg sericin + ML385 inhibitor group were treated with 1000 mg/kg sericin by gavage for four weeks, along with 10 mg/kg ML385 once every two days for two weeks by intraperitoneal injection. Mice in the Zhibitai Capsules group were treated with 62.4 mg/kg Zhibitai by intragastric administration. The dosing volume of sericin and Zhibitai Capsules was 0.2 ml/10 g body weight. Mice in the control group and model control group were given an equal volume of pure water by intragastric administration.
During the experiment, body weight, food intake, physiological activity status, responsiveness, and skin breakage of mice were observed and recorded. At the end of the experiment, mice were sacrificed after anesthesia with sodium pentobarbital, and blood and liver tissue were collected. The plasma was extracted after blood centrifugation (3500 rpm/min for 15 min) and placed in a −80 °C refrigerator. After being weighed, part of the liver was fixed in 4% paraformaldehyde, and the rest was preserved in cryopreservation tubes and stored in a −80 °C refrigerator for subsequent testing.
Determination of serum TC, TG, LDL-C, HDL-C, ALT, AST in Mice
The levels of total cholesterol (TC), total triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were detected according to the specifications of commercial reagent kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Determination of MDA, GSH, and SOD Content in the Liver of Mice
The liver tissue was weighed and homogenated with normal saline (w:v = 1:9), and the supernatant was collected for the assays. The levels of Malondialdehyde (MDA), reduced glutathione (GSH) and superoxide dismutase (SOD) were determined with commercial assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Determination of TNF-α and IL-6 Content in the Liver of Mice
The contents of tumor Necrosis Factor-α (TNF-α) and interleukin −6 (IL-6) were determined using commercial assay kits (Shanghai Enzymotec Biotechnology, Shanghai, China). Briefly, liver tissue was homogenized in phosphate buffered saline (PBS) to prepare a 10% liver homogenate. The homogenate was then centrifuged and the supernatant was collected and used for the assay according to the instructions of the commercial assay kits.
Hematoxylin-eosin (HE) Staining
Liver tissue fixed with 4% paraformaldehyde was dehydrated by gradient ethanol. After dehydration, paraffin embedding was performed in an integrated paraffin embedding machine (Leica Arcadia) to obtain tissue wax blocks. Slices (4 μm) were then prepared with a pathology sectioning machine (Leica RM2245). It is subsequently dewaxed and then stained with HE Stain Kit (Solarbio, Beijing, China). Slices were stained with hematoxylin and eosin and observed by a light microscope (Leica DM3000).
Oil red O Staining
The assay was conducted using an Oil Red O Stain Kit (Solarbio, Beijing, China). Briefly, liver tissues were embedded and sliced into sections of 4 μm, and fixed with fixative. The slices were immersed in an oil red dye solution, and then background differentiation and staine were performed with 60% isopropanol and hematoxylin successively. The pathological changes in liver tissue were observed under an optical microscope (Leica DM3000).
Western Blot Analysis of Nrf2, Keap1, HO-1, GPX4 and NF-κB p65 in Liver Tissues of Mice
The liver tissues were weighed and mixed with radio immunoprecipitation assay lysate containing phenylmethanesulfonyl fluoride, homogenized and centrifuged. The protein concentration was quantified with a bicin-choninic acid assay protein assay kit (Solarbio, Beijing, China). The same amount of protein was isolated in 10% SDS-PAGE (150 V, 1 h), and the target protein was transferred to the polyvinylidene fluoride (PVDF) membrane at constant flow (400 mA, 35 min). The PVDF membrane was sealed with a commercial rapid sealing solution for 35 min and then incubated with the diluted Nrf2, Keap1, HO-1, NF-κBp65 (Proteintech Group, Wuhan, China) and GPX4 (Abcom, Cambridge, England) antibody (1:2000-1:5000), at 4°C for 8 h. After that, the PVDF membrane was washed with TBST (10 min/ time, 5 times). The membrane was then incubated with secondary antibodies conjugated to horseradish peroxidase (1:5000, yardage) for 1 h at room temperature. The PVDF membrane was washed with TBST again (10 min/ time, 5 times). Finally, enhanced chemiluminescent chemiluminescent substrate was added to the PVDF membrane and developed in the gel imager.
Immunohistochemistry Analysis of GPX4 in Liver Tissues of Mice
The liver tissues were fixed, embedded, and sliced into 4 μm thickness sections. The paraffin sections were deparaffinized and antigenically repaired. Endogenous peroxidase was then removed and serum sealing was performed. The sections were incubated with GPX4 antibody at 4 °C overnight, and then subjected to diaminobenzidine staining, and hematoxylin staining. The expression of GPX4 protein in liver tissues was observed under a light microscope (Leica DM3000).
Statistical Analysis
SPSS 22.0 statistical software was used for data analysis. Homogeneity of variances was tested by Bartlett's test. One-way analysis of variance was used to compare differences between quantitative variables. The least-significant difference method was used for pre-post comparisons, and the Waller-Duncan (W) and Bonferroni methods were utilized for post-post comparisons. P < .05 was considered to be statistically significant.
Result
General Condition of Mice and the Effect of Sericin on Liver index
All mice were in good condition during the experiment, with normal vital activities and no abnormal deaths. NAFLD mouse model was constructed after 8 weeks of high-fat diet feeding, as evidenced by the presentation of hepatic tissues. The livers of the high-fat diet mice were yellowish, with a tense pericardium and a greasy feeling on the cut surface compared to the control group. The results of HE staining demonstrated disordered liver lobules, excessive fat-containing vacuoles in the cytoplasm, and cellular swelling. The results of the Oil red O staining demonstrated the accumulation of fat in the liver of mice fed with a high-fat diet. Compared with the control group, the liver coefficient was increased in the model group (Figure 1). After 4 weeks of gavage with sericin or Zhibitai Capsules, the Liver Index of the NAFLD model mice in each group decreased, but the difference was not statistically significant. In contrast, the liver indices of the 1000 mg/kg sericin + ML385 group did not differ significantly from those of the model group.

Effect of sericin on liver Index in NALLD model mice. * indicates comparison with the model group, P < .05. ** indicates comparison with the model group, P < .01.
Effects of Sericin on serum TC, TG, LDL-C, HDL-C, ALT, and AST Levels
Serum lipid levels were measured in mice to examine the impact of sericin on hepatic steatosis and lipid deposition brought on by a high-fat diet. As shown in Figure 2, serum TC, TG, and LDL-C were significantly higher in the model group compared with the control group (P < .05). After sericin intervention, the levels of TC, TG, and LDL-C in all sericin dose groups and the Zhibitai Capsules group decreased significantly compared with the model group (P < .05). As a protective indicator, HDL-C levels were raised significantly in all dose groups of sericin and Zhibitai Capsules group (P < .05). On the other hand, TG and HDL-C levels did not significantly differ between the 1000 mg/kg sericin + ML385 group and the model groups (P > .05). Based on the data, sericin may alleviate steatosis and lipid deposition in NAFLD by reducing serum levels of TC, TG, and LDL-C lipids and raising HDL-C expression.

Effect of sericin on serum lipids and transaminase levels in NAFLD model mice. (A) serum TC level, (B) serum TG level, (C) serum LDL-C level, (D) serum HDL-C level, (E) serum ALT level, (F) serum AST level. * indicates comparison with the model group, P < .05. ** indicates comparison with the model group, P < .01. LDL-C, low-density lipoprotein cholesterol.
As shown in Figure 2E-F, after sericin treatment, ALT and AST levels increased in the model group and decreased in the dosage groups as compared to the control group; however, the difference was not statistically significant (P > .05). The trend of the Zhibitai Capsules on ALT and AST was similar to that of sericin.
Effect of Sericin on Liver MDA, GSH, and SOD Levels
To investigate oxidative stress changes within the mouse liver, mouse liver GSH, MDA, and SOD activities were assayed. As shown in Figure 3A, GSH levels were significantly decreased in the model group compared to the control group (P < .05). All dose groups and the Zhibitai Capsules group had a rising GSH level. When compared to the model control group, the difference in GSH levels between the 1000 mg/kg sericin group and the Zhibitai Capsules group was statistically significant (P < .05). Compared with the control group, the model group showed a significant increase in MDA levels and a decrease in SOD activity (Figure 3B-C, P < .05). Sericin significantly inhibited the expression of hepatic MDA in NAFLD mice (P < .05) and significantly increased SOD activity (P < .05). The changes in MDA and SOD in the Zhibitai Capsules group followed the same trend as in the sericin dose groups, whereas the MDA levels in the 1000 mg/kg sericin + ML385 group were close to those of the model group. The results suggest that sericin may improve the level of hepatic oxidative stress due to a high-fat diet.
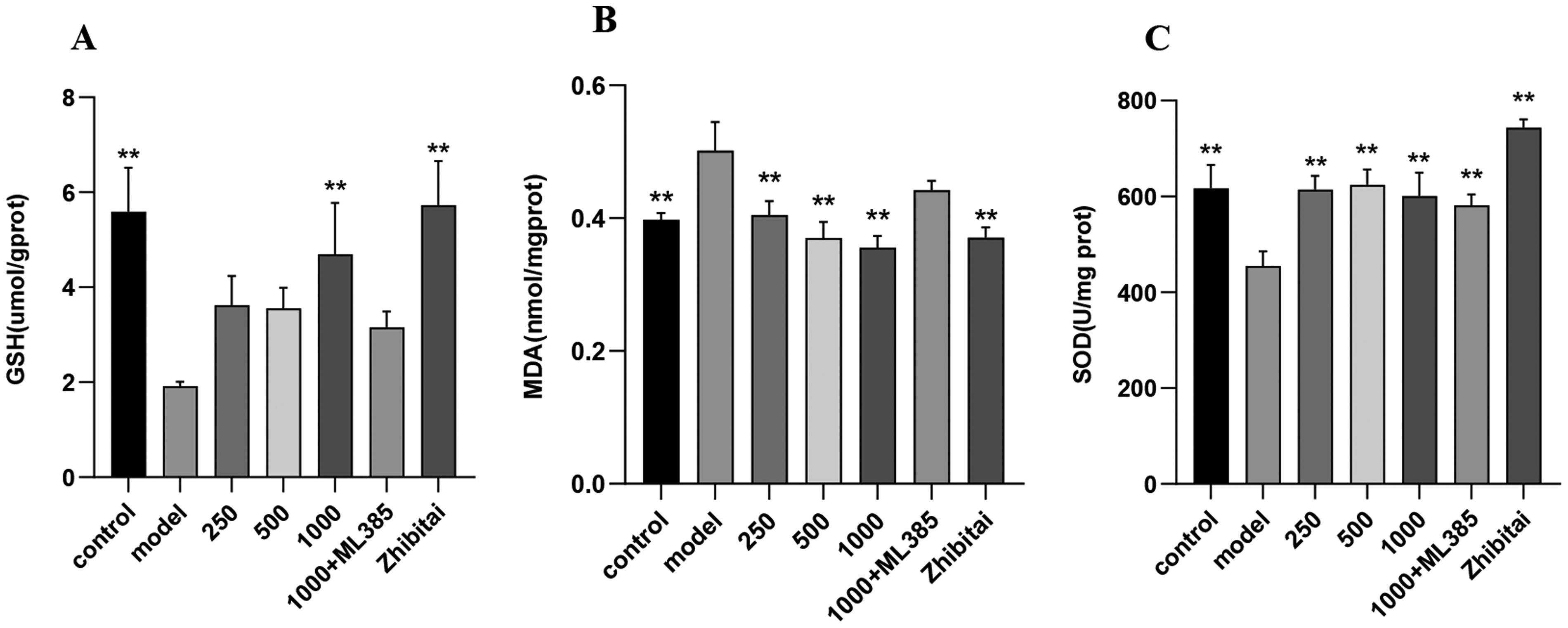
Effects of sericin on oxidative stress in liver tissue of NAFLD model mice in liver tissue of NAFLD model mice. (A) liver GSH level, (B) liver MDA level, (C) liver SOD level. *indicates comparison with the model group, P < .05. ** indicates comparison with the model group, P < .01.
Effect of Sericin on Liver TNF-α and IL-6 Levels
To investigate the effect of sericin on the levels of inflammatory factors in the liver of NAFLD mice, the levels of TNF-α and IL-6 in the liver of mice were detected. As shown in Figure 4A–B, the levels of TNF-α and IL-6 in liver were significantly higher in the model group compared to the control group (P < .05). The levels of TNF-α and IL-6 decreased after the treatment of sericin, with the levels in the 250 and 1000 mg/kg sericin groups statistically significant with those of the model group (P < .05). TNF-α and IL-6 levels were significantly decreased in the Zhibitai Capsules group compared with the model group (P < .05). The findings imply that sericin inhibited inflammatory expression in the NAFLD model as Zhibitai Capsule.

Effect of sericin on liver tissue inflammation in mice model of NAFLD. (A) liver LI-6 level, (B) liver TNF-α level. * indicates comparison with the model group, P < .05. ** indicates comparison with the model group, P < .01.
Hematoxylin-Eosin (HE) Staining
The results of the HE staining revealed that the control group's liver had a complete hepatic lobule structure as well as a complete and clear hepatocyte morphology (Figure 5A). Compared with the control group, the model group had disorganized hepatic lobules with more lipid droplet vacuoles accompanied by inflammatory cell infiltration and ballooning phenomenon (Figure 5B). In contrast, there was a significant improvement in hepatic histopathology after sericin intervention (Figure 5C-E). Mice treated with 250 and 500 mg/kg sericin showed significantly decrease in HE staining pathology scores when compared with the model group (Figure 5H, P < .05). A significant improvement in hepatic histopathology was observed in the Zhibitai Capsules group (Figure 5H and G). The 1000 mg/kg sericin + ML385 group still had many lipid droplet vacuoles, with no improvement in the inflammatory infiltrate or ballooning.

He staining of liver tissue, 40 × 10 (scale bar: 50 µm). Red arrows indicate steatosis and blue arrows indicate inflammatory necrosis. (A) control group, (B) model group, (C) 250 mg/kg sericin group, (D) 500 mg/kg sericin group, (E) 1000 mg/kg sericin group, (F) 1000 mg/kg sericin+2 mg/kgML385 group, (G) Zhibitai Capsules group. (H) HE staining score of liver tissue. * indicates comparison with the model group, P < .05. ** indicates comparison with the model group, P < .01.
Oil Red O Staining
Lipid accumulation was observed in the livers of mice by Oil red O staining. As shown in Figure 6, a large number of red lipid droplets was observed in the livers of the model group with granular accumulation when compared with the control. Red lipid droplet aggregation in the liver was significantly reduced after treatment with sericin intervention compared to the model group, especially in the 250 mg/kg sericin group. Meanwhile, Zhibitai Capsules intervention significantly reduced fat accumulation in the liver of mice, whereas the red lipid droplets of 1000 mg/kg sericin + ML385 group were still present in large quantities.

Liver tissue oil red O staining, 40 × 10 (scale bar: 50 µm). Red aggregates indicate lipid precipitation. (A) control group, (B) model group, (C) 250 mg/kg sericin group, (D) 500 mg/kg sericin group, (E) 1000 mg/kg sericin group, (F) 1000 mg/kg sericin+2 mg/kgML385 group, (G) Zhibitai Capsules group.
Western Blot
As shown in Figure 7, the expression of Nrf2, Keap1, HO-1, and GPX4 was significantly decreased in the model group compared with the control group, while the level of NF-κBp65 was significantly increased (P < .05). Nrf2 levels were elevated in all dosage groups compared to the model group after sericin intervention, and Nrf2 levels were significantly higher in the Zhibitai Capsules group compared to the model group (P < .05). The levels of Keap1, HO-1, and GPX4 were significantly higher in all dose groups of sericin and the Zhibitai Capsules group compared with the model control group (P < .05). In contrast, NF-κBp65 protein expression in the livers of NAFLD mice was significantly down-regulated in all dose groups of sericin and the Zhibitai Capsules group (P < .05). Furthermore, the 1000 mg/kg sericin + ML385 group's treatment reduced the expression of Nrf2, Keap1, HO-1, and GPX4, and the 1000 mg/kg sericin + ML385 group's Nrf2 level was statistically different from that of the sericin dose groups (P < .05). These results indicate sericin may improve the antioxidant defense system by upregulating pathway proteins such as Nrf2, Keap1, HO-1, and GPX4, and inhibit inflammation by suppressing the expression of NF-κBp65 protein in the liver of mice.
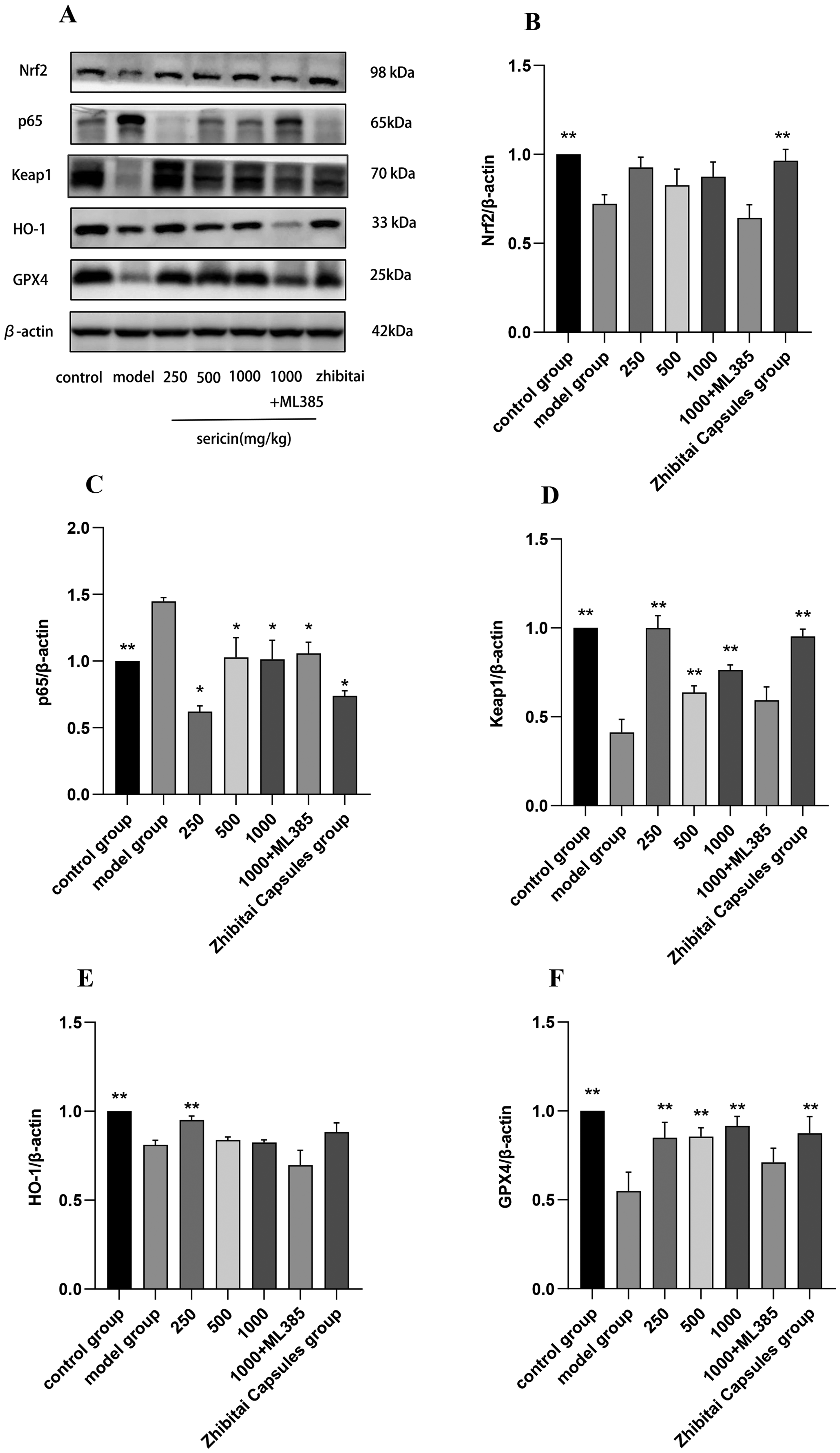
Effect of sericin on the expression of proteins. (A) The expression of Nrf2, Keap1, HO-1, GPX4, and NF-κB p65 in liver tissues was examined by western blot analysis. (B) Nrf2 proteins level, (C) NF-κB p65 proteins level, (D) Keap1 proteins level, (E) HO-1 proteins level, (F) GPX4 proteins level. The relative expressions of these proteins were normalized to β-actin. * indicates comparison with the model group, P < .05. ** indicates comparison with the model group, P < .01.
Immunohistochemistry
GPX4 plays a critical role in the removal of hydrogen peroxide products from cells. As shown in Figure 8, a large amount of positive expression product was deposited in the control group. Compared with the control group, the expression of GPX4 was reduced in the model group, and no obvious brownish-yellow deposition products were observed. However, protein expression of GPX4 was up-regulated to varying degrees in all sericin dosage groups, especially in the 500 mg/kg sericin group, as well as in the Zhibitai Capsules group. The 1000 mg/kg sericin + 2 mg/kg ML385 group was similar to the model group, with no obvious areas of brownish-yellow positive reaction.

Effect of sericin on GPX4 expression in liver tissues of NAFLD model mice (20 × 10, scale bar: 75 µm). The yellowish color indicates a positive reaction. (A) control group, (B) model group, (C) 250 mg/kg sericin group, (D) 500 mg/kg sericin group, (E) 1000 mg/kg sericin group, (F) 1000 mg/kg sericin+2 mg/kgML385 group, (G) Zhibitai Capsules group.
Discussion
NAFLD is a chronic disease with a high prevalence globally, which is associated with oxidative stress, inflammation, lipid accumulation, and many other factors. 37 In this study, a mice model was used to investigate the alleviating effect of sericin on symptoms caused by NAFLD and its mechanism.
Modern lifestyles and dietary habits tend to lead to steatosis and inflammation in the body, which in turn cause fat deposition and accumulation as well as lipid metabolism disorders, leading to the development of NAFLD. 38 NAFLD includes insulin resistance, steatohepatitis, fibrosis, and obesity features. 39 In this study, C57BL/6 mice were fed a high-fat diet for 8 weeks and the pathological sections showed that the liver lobules of the modeled mice were disorganized with a large number of fat vacuoles. After 4 weeks of gavage with sericin, serum lipid levels in high-fat diet mice were altered. TC, TG, and LDL-C lipid levels were significantly high in the model group fed with a high-fat diet, indicating that steatosis and damage had occurred within the liver tissue. After sericin intervention, TC, TG, and LDL-C levels decreased to different degrees in all dose groups as well as in the Zhibitai Capsules group when compared with the model group. HDL-C is a “good cholesterol”, 40 inhibits inflammation, and prevents cell damage and necrosis, which is a protective indicator. 41 HDL-C levels increased after the intervention of sericin and Zhibitai Capsules. The results of the lipid quartet in the study were consistent with the results of histological analysis of liver tissues, indicating that after the sericin intervention, steatosis and lipid deposits in the mice's livers were decreased. These results indicate that sericin may improve hepatocellular steatosis and lipid deposition in mice by down-regulating serum TC, TG, and LDL-C levels and raising HDL-C levels.
ALT and AST are mainly located in hepatocytes and hepatocyte mitochondria, respectively. They can be released into the bloodstream when the liver parenchyma undergoes cellular necrosis and extensive damage, resulting in elevated serum levels of ALT and AST, which are the key indicators for evaluating liver injury. 42 In this study, a high-fat diet increased serum ALT and AST levels in the model group, and serum ALT and AST levels decreased after the intervention of sericin and the Zhibitai Capsules, although the difference was not statistically significant. This may be due to the relatively short duration of feeding and treatment. A previous study has indicated that ALT and AST levels decreased significantly after 34-36 weeks of feeding. 38 Oxidative stress is a series of adaptive responses caused by a dysregulation of the balance between the reactive oxygen components of the body and the antioxidant system, which leads to problems in the redox signaling system, interferes with the normal redox state of the cell, thus damaging the cellular macromolecules, such as proteins, lipids, as well as nucleic acids (which are associated with the scavenging of free radicals), and ultimately leads to hepatotoxicity. 43
GSH, a crucial intracellular regulating molecule engaged in biotransformation in vivo, has an integrative detoxifying impact and can bind to various toxins and medications. 44 In addition, GSH is one of the most important antioxidants in the cellular antioxidant system and plays an important role in the fight against reactive oxygen species (ROS). The lack of cellular GSH leads to the accumulation of ROS, which in turn disrupts cellular redox homeostasis, ultimately triggering hepatocyte injury or even death. 6 SOD is a class of free radical scavengers that also plays a role in scavenging lipid peroxides and oxygen free radicals to maintain metabolic homeostasis in the body and protect liver cells. 45 Under oxidative stress, intracellular lipids are oxidized, generating large amounts of MDA, which leads to increased oxidative stress in the cells. 46 A previous study showed that a high-fat diet decreased oxidative stress in C57BL/6 mice in vivo. After treatment with 5 nmol/g TBE-31 (an Nrf2 activator), hepatic GSH levels and SOD activity were increased, and MDA levels were decreased. 47 Results of the current study were consistent with the fact that intervention with sericin down-regulated MDA levels and increased GSH abundance as well as SOD activity in high-fat diet mice. In summary, sericin counteracts oxidative damage caused by a high-fat diet by ameliorating oxidative stress and increasing the activity of hepatic antioxidant enzymes. These results are consistent with the effects of sericin on serum lipid indexes and liver function levels described above.
In addition to oxidative stress, hepatic inflammatory response is another major feature of NAFLD. Inflammatory factors IL-6 and TNF-α are involved in the development and progression of NAFLD, with increasing concentrations of them being significantly associated with an increased risk of NAFLD.48,49 With the expression and infiltration of inflammatory factors, NAFLD can be driven to produce fibrosis and eventually hepatocellular carcinoma. 50 Previous studies have shown that medicines may improve NAFLD in rats by modulating liver IL-6 and TNF-α levels.51,52 In this study, a high-fat diet increased the expression of hepatic inflammatory factors IL-6 and TNF-α in mice, while the intervention of sericin significantly down-regulated the expression levels of IL-6 and TNF-α. It is suggested that sericin may ameliorate liver injury in NAFLD by counteracting cellular inflammatory factors, which is consistent with previous findings on the anti-inflammatory effects of filaggrin.53–55
We further investigated the mechanism of anti-oxidative stress and anti-inflammatory effects of sericin in ameliorating NAFLD in mice, by examining the expression of Nrf2, NF-κBp65, Keap1, HO-1, and GPX4 proteins in the liver of mice. Nrf2 is an important transcription factor that regulates cellular redox homeostasis in vivo. It can regulate redox homeostasis, energy metabolism, macromolecule repair, and mitochondrial physiology, as well as mitochondrial inflammation.56–58 Keap1 is an old partner of Nrf2, which is a sensor of electrophiles and Nrf2 is an effector of synergistic activation of cytoprotective genes, where overexpression of Keap1 has been shown to inhibit the nuclear accumulation and transcriptional activity of Nrf2. 59 Under steady-state/normoxic conditions, Nrf2 is ubiquitinated, binds to the Keap1 complex in the cytosol, and is degraded; under oxidative stress conditions, Nrf2 dissociates from the complex and subsequently enters the nucleus, where it dimerizes with the sMAF protein and induces the expression of a series of antioxidant response element-containing antioxidant genes and II-phase detoxification enzymes to enhance the body's resistance to oxidative stress.60,61 GSH is a tripeptide composed of glutamate, cysteine, and glycine which is one of the most important components of the intracellular antioxidant defense system. As mentioned previously, GSH counteracts ROS accumulation and maintains redox homeostasis. GPX4, a downstream protein of Nrf2, is an antioxidant enzyme with the ability to scavenge membrane lipid hydroperoxide products, thus conferring cellular protection. 62 GSH can be used by GPX4 to reduce lipid peroxides to their alcoholic forms, thus GSH and GPX4 are considered to be two key factors in the elimination of lipid peroxides as well as in the inhibition of iron death. 45 Various proteins of the GSH synthesis pathway are regulated by Nrf2, including the subunit of System Xc- (SLC7A11), the catalytic and regulatory subunit of glutamate-cysteine ligase (GCLC/GCLM), glutathione synthetase (GSS), and γ-glutamylcysteine synthetase (GCS).61,63 HO-1, a downstream target factor of Nrf2, is an inducible heme-degrading enzyme that plays a key role in redox homeostasis. 64 In the presence of oxidative stress and cellular damage in the organism, HO-1 mRNA and protein expression levels increase in vivo, whereas Nrf2 can directly regulate HO-1 promoter activity. 65
The results of the current study showed low expression of Nrf2, Keap1, HO-1, and GPX4 protein levels in the livers of mice in the model group fed with a high-fat diet. The levels of oxidative stress-related proteins Nrf2, Keap1, HO-1, and GPX4 were up-regulated in the livers of mice in all dose groups after sericin intervention. The level of Nrf2 in the 1000 mg/kg sericin + ML385 group was significantly lower than that in the sericin intervention group. In the 1000 mg/kg sericin + ML385, Keap1, HO-1, and GPX4 protein expression were lower than that of sericin at all sericin dosages. This result is consistent with the changes in the contents of the oxidation-related products GSH, SOD, and MDA in the present experiments, which suggests that the oxidative defense of sericin against NAFLD may be related to the activation of Nrf2 and the up-regulation of the Nrf2 downstream proteins Keap1, HO-1, and GPX4.
The NF-κB family is a group of transcription factors that have long been recognized as having a role in promoting the expression of inflammatory factors, whereas NF-κB p65 is a key factor in the classical pro-inflammatory signaling pathway. 66 There is a close interaction between Nrf2 and NF-κB. Previous studies have shown that upregulation of Nrf2 inhibits IL-1β-induced activation of the NF-κB p65 signaling pathway in human chondrocytes. 67 Activation of NF-κB p65 by external stimuli induces the production of many inflammatory factors such as driving fibrosis in NAFLD. 68 In our study, a high-fat diet increased hepatic NF-κB p65 protein expression in mice, and the intervention of sericin inhibited hepatic NF-κB p65 protein expression in mice in all dose groups. This was consistent with the changes in the expression of inflammatory factors IL-6 and TNF-α observed in the current study. These results indicate that sericin may ameliorate the inflammatory response in NAFLD caused by a high-fat diet by regulating the NF-κB signaling pathway.
It should be noted that there are limitations in the current study. First of all, the etiology of NAFLD in humans is complex and the mice model used in the current study did not fully recapitulate the human conditions. Secondly, the dose-dependent effects of sericin was not very clear and the inhibitory effect of the inhibitor on Nrf2 was not very effective in this study, possibly due to the relatively short treatment time or insufficient dose range. Further work is needed to explore the effects of sericin on NAFLD model using a broader range of doses and a longer duration of treatment duration. Although sericin was observed to affect the expression of several proteins related to the Nrf2- GPX4/ NF-κB p65 pathway, the impact of sericin on this signaling pathway needs further evaluation. Moreover, a multiple signaling pathway approach is needed to provide more information for evaluating the mechanism of action of sericin.
Conclusion
In the present experiment, sericin had a palliative effect on symptoms induced by high-fat diet-induced NAFLD in mice. Treatment of sericin was observed to effectively alleviate steatosis and lipid deposition in livers of mice. It is possible that the oxidative stress in the liver was alleviated by up-regulating the expression of key proteins of the oxidative defense system relating to the Nrf2 signaling pathway, which led to an increase in the GSH level and SOD activity, and a decrease in the MDA level of the liver in mice. In addition, sericin may decrease the secretion of TNF-α, IL-6, and other inflammatory factors in NAFLD mice by down-regulating the signaling expression of NF-κB p65, a key mediator of the inflammatory pathway. The above results indicate that sericin has an ameliorative effect on liver injury in NAFLD mice by alleviating oxidative stress and inflammatory responses. It remains to be explored whether this effect is regulated through the Nrf2- GPX4/ NF-κB p65 signaling pathway.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241287061 - Supplemental material for Exploring the Mechanism of Action of Sericin in Ameliorating non-Alcoholic Fatty Liver Disease in Mice Based on the Nrf2- GPX4/ NF-κB p65 Pathway
Supplemental material, sj-docx-1-npx-10.1177_1934578X241287061 for Exploring the Mechanism of Action of Sericin in Ameliorating non-Alcoholic Fatty Liver Disease in Mice Based on the Nrf2- GPX4/ NF-κB p65 Pathway by Jinyue Li, Dan Yi, Yuqiu Gao, Lihong Zhou, Pingjing Wen and Guangqiu Qin in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank the staff of the Preventive Medicine Laboratory of Guangxi University of Traditional Chinese Medicine for their technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Experimental Animal Welfare Ethics Committee of Guangxi University of Traditional Chinese Medicine (Approval Code: DW20220408-060, Approval Date: April 15, 2022).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Innovation Project of Guangxi Graduate Education (YCSW2023372), the Innovation Project of Guangxi Graduate Education of GXUCM (YCSY2023020), and Guangxi University of Chinese Medicine (S202310600106, S202310600142, 2022A009).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
