Abstract
Introduction
Osteosarcoma, the most common primary malignancy of the bone, often occurs in young people and children. Although its incidence is only 1% of all cancers, it has a high case fatality rate owing to high incidence rate of metastasis. 1 Since the 1970s, several approaches have been used to treat osteosarcoma, such as surgery and multiagent regimens, including methotrexate, doxorubicin, cisplatin, and bleomycin. 2 With combined treatments of chemotherapy, surgery, emerging targeted therapy, and immunotherapy, the 5-year survival rate of osteosarcoma has increased to approximately 70%. However, the 5-year survival rate after metastasis remains as low as 20% to 30%.3,4 Current standard treatments are usually insufficient to control recurrent or metastatic osteosarcoma. 5 Although systemic treatment options for advanced sarcoma are increasing, it is still vital to find new compounds for the treatment of metastatic osteosarcoma.
Epithelial–mesenchymal transition (EMT) is an intricate process in which epithelial cells downregulate intercellular adhesion structure and polarity, obtain mesenchymal properties, rearrange the cytoskeleton, and enhance motility.6,7 EMT is regulated by several signaling pathways, such as Wnt, TGF-β, and Notch-mediated signaling, as well as several cytokines, including epidermal growth factor, hepatocyte growth factor, insulin growth factor, and fibroblast growth factor.8,9 During EMT, transcription factors such as Slug, Snail, Twist, and Zeb1 constitute a core transcriptional network that promotes cancer cell migration and invasion, thereby conferring metastatic potential. 10 During EMT, E-cadherin expression is inhibited, and cell adhesion is reduced. 11 Simultaneously, the expression of N-cadherin and fibronectin increases to remodel the cytoskeleton for migration.12,13 Moreover, EMT can be regulated by STAT3 to enhance cell migration in many tumors, including esophageal squamous cell carcinoma, non-small cell lung cancer, and gastric cancer.14‐16
STAT3 is a key regulator of the IL-6-mediated signaling pathway 17 in which the tyrosine and serine residues of STAT3 are phosphorylated and activated by JAK1 and JAK2. 18 STAT3 activation not only promotes the proliferation of cancer cells, but also enhances angiogenesis and metastasis.19,20 Lots of plant extracts have been proved to exert antitumor effects by inhibiting STAT3 activation.18,21 For example, celastrol inhibits proliferation, invasion, migration, and angiogenesis in various tumors. 22 Therefore, targeting STAT3 activation is a promising approach for treating cancer metastasis.
Oxyresveratrol (ORES), trans-3,5,2’,4’-tetrahydroxystilbene, is a phytochemical found in mulberry twigs and fruits with a structure similar to that of resveratrol.23,24 ORES has DNA-damaging activity and antitumor effects,25,26 but it remains unknown whether ORES inhibits human osteosarcoma migration. Here, we found that ORES inhibited U2OS migration in osteosarcoma cells and attenuated the EMT process, in which the phosphorylation of STAT3 was decreased. Our findings indicate that ORES is a potential therapeutic agent for osteosarcoma.
Materials and Methods
Compound and Reagents
ORES, WP1066, and dimethyl sulfoxide were purchased from Sigma (purity ≥ 98%, analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAG)E gel and high performance liquidchromatography), Dulbecco's modified Eagle's medium (DMEM), enhanced chemiluminescence substrates, and fetal bovine serum (FBS) from Thermo Fisher Scientific (Grand Island, NY, USA), IL-6 from PeproTech, Inc. (NJ, USA), primary antibodies against GAPDH, E-cadherin, N-cadherin, Twist, phophorylation-STAT3 (p-STAT3), and STAT3 from Cell Signaling Technology (Danvers, MA, USA), the osteosarcoma cell U2OS (HTB-96) from the American Type Culture Collection (Manassas, Virginia, USA), and horseradish peroxidase (HRP)-conjugated anti-rabbit antibody from Jackson ImmunoResearch Inc. (West Grove, PA, USA).
Wound Healing Assay
Human osteosarcoma cancer cell U2OS was cultured in DMEM supplemented with 10% FBS and maintained at 37 °C in an incubator with 5% CO2. U2OS cells in good logarithmic growth phase were seeded into a 6-well plate at 4 × 105/well and cultured overnight. The next day, a 200 μL pipette tip was used to scratch the bottom of the 6-well plate with a ruler as the standard. Each well was washed 3 times with phosphate buffered solution (PBS) to aspirate the scratched cells. Serum-free DMEM and different concentrations of ORES were added to each well. The well plate was then placed under the X40 microscope to take pictures at 0 h and 16 h. ImageJ software was used to calculate the scratch area of the images. The calculation formula was scratch healing rate (%) = (0 h scratch area−16 h scratch area)/0 h scratch area.
Cell Viability Assay
U2OS cells were seeded into 96-well plates at 1 × 105 cells per well in DMEM medium containing 10% FBS and 1% penicillin. Cell viability was measured using a CCK-8 assay kit (Beyotime Biotechnology) according to the manufacturer's instructions. Briefly, 10 μL of the CCK-8 solution was added to the wells and incubated with cells for 1.5 h. The cell viability was then measured at 450 nm.
Transwell Assay
The U2OS cells in the logarithmic growth phase under good conditions were counted. The cell concentration was adjusted to 1 × 103 cells/mL. Then, 100 µL of the cells was added to the upper compartment of the chamber, then treated with ORES (0, 1, 3, and 10 μM) in serum-free DMEM, while the lower compartment was filled with 600 µL of 10% FBS DMEM. After incubation for 16 h, the migrated cells were fixed in methanol and glacial acetic acid buffer (methanol:glacial acetic acid = 3:1) for 30 min and stained with crystal violet solution for 30 min. After removing the non-adherent cells, the migrated cells were captured using a microscope, and the No. of cells was calculated using Image J software.
Western Blot Analysis
U2OS cells were seeded into a 6-well plate at a density of 1 × 106 cells/well and cultured overnight. When the cell coverage was approximately 80% to 90% of the well area, the DMEM was changed with 1 mL of FBS-free DMEM containing ORES (0, 1, 3, and 10 μM), and the culture was continued for another 24 h. The 6-well plate was removed from the incubator and placed on ice, and then pre-cooled PBS was gently added along the wall of the well to wash the cells. The PBS was discarded and 1 × loading buffer was added to lyse the cells. Equal amounts of lysate supernatant were added to the prepared SDS-PAGE gel, and the voltage for electrophoresis was set to 80 V for 30 min. After 30 min, the samples were compressed to the boundary line of the separating and concentrated gels, and the voltage set to 110 V. Electrophoresis was completed when the sample reached 3/4 of the way up the gel plate. After electrophoresis, the proteins were transferred to a nitrocellulose membrane at 110 V for 90 min in an ice water bath. The NC membrane was then incubated with 5% w/v nonfat dry milk for 1 h at room temperature. The primary antibodies were added and incubated at 4 °C for overnight. The NC membranes were then washed 3 times and incubated with HRP-conjugated secondary antibody for 1 h at room temperature. After washing 3 times, specific proteins were detected using an enhanced chemiluminescence detection system (Pierce ECL, ThermoFisher Scientific).
Statistical Analysis
The experiments were independently repeated at least 3 times and are displayed as the mean ± SEM. One-way analysis of variance (ANOVA) and Tukey test were used to compare the indicated values of multiple experimental groups using GraphPad Prism 8 software, in which P <.05 indicated statistical significance.
Results
ORES Attenuates the Migration of U2OS Cells
Although ORES has anti-inflammatory and anti-metabolic effects, its effect on cancer cell migration remains unclear. Therefore, wound healing and transwell assays were performed to determine whether ORES affects tumor cell migration. The wound healing assay is a general approach for evaluating cell migration and wound repair. 27 As shown in Figure 1, 1 μM ORES inhibited the wound healing and migration of U2OS cells after 16 h compared to 0 μM ORES. As the concentration of ORES increased (3 and 10 μM), the migration of U2OS cells was significantly inhibited (Figure 1A). As shown in Figure 1B, with 1 μM ORES treatment, the healing area of U2OS cells decreased by approximately 20%. With 3 μM ORES treatment, the healing area decreased by approximately 35%, and by approximately 40% with 10 μM ORES treatment. Although 10 μM ORES treatment inhibited U2OS cell proliferation, it did not reduce cell viability at 3 μM and10 μM (Figure 1C). These results indicate that ORES inhibited U2OS cell migration.

Oxyresveratrol (ORES) inhibits the wound healing of U2OS cells. U2OS cells were seeded in a 12-well plate and reached 95% confluency. The scratch was then made on cell monolayers. The floating cells were washed out by PBS. The U2OS cells were treated with 0, 1, 3, and 10 µM of ORES in serum-free Dulbecco's modified Eagle's medium (DMEM) for 16 h. (A) Images were captured using a microscope. (B) The healing area was measured by Image J software. (C) The cell viability was detected using a CCK-8 assay kit. The experiments were independently repeated at least 3 times. The data are shown as mean ± SEM. One-way analysis of variance (ANOVA) with post hoc Tukey test was used to determine the difference between indicated groups.
To determine further the effect of ORES on cell migration and metastasis, we performed a Transwell assay. As illustrated in Figure 2A, the No. of cells passing through the small holes in the transwell chamber was significantly decreased after treatment with ORES. Moreover, with 10 μM ORES, U2OS cell migration was completely inhibited (Figure 2B). Taken together, these results suggest that ORES can reduce cell migration in vitro, indicating that ORES may inhibit tumor metastasis in vivo.
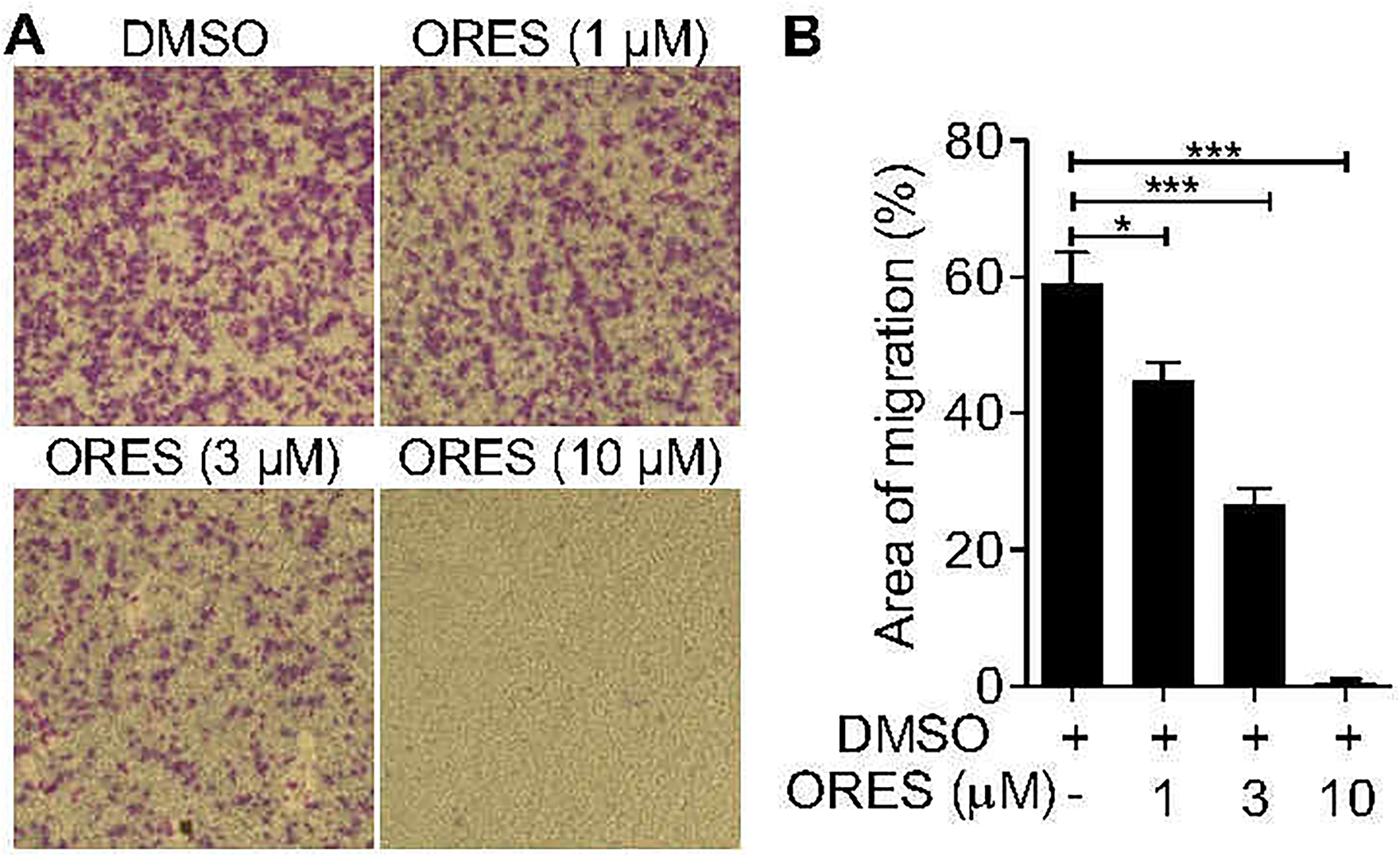
Oxyresveratrol (ORES) inhibits the migration of U2OS cells. The U2OS cells were incubated with 0, 1, 3, and 10 µM in serum-free Dulbecco's modified Eagle's medium (DMEM) for 16 h. Transwell migration assay was carried out to measure cell migration. (A) Images were captured using a microscope. (B) The area of migration was detected by Image J software. The experiments were independently repeated at least 3 times. The data are shown as mean ± SEM. One-way analysis of variance (ANOVA) with post hoc Tukey test was used to determine the difference between indicated groups.
ORES Inhibits EMT in U2OS Cells
E-cadherin, a critical adhesion molecule, is downregulated in the EMT process to promote degradation of adherens junctions.28,29 Meanwhile, the protein of adhering junctions could be replaced by N-cadherin, which provides greater connection flexibility and results in enhanced cell motility. 30 To determine the effect of ORES on EMT, we performed Western blotting to detect the expression of 2 critical EMT-related proteins, E-cadherin and N-cadherin. E-cadherin expression was obviously increased and N-cadherin expression was decreased after treatment of U2OS cells with 1, 3, and 10 μM ORES (Figure 3A). Furthermore, when treated with 3 μM ORES, E-cadherin expression was 8 times higher than that of without ORES. When U2OS cells were treated with 10 μM ORES, E-cadherin expression was 22 times higher than that of without ORES (Figure 3B). Additionally, in treatment with 1 μM ORES, the N-cadherin expression was decreased by 55% compared with no ORES. Moreover, in treatment with 3 μM ORES, the expression of N-cadherin was decreased by 40% compared to that of no ORES. When the dose of ORES reached 10 μM, the N-cadherin expression decreased by 30% compared to that of ORES (Figure 3C). In addition, ORES dose-dependently reduced the expression of vimentin (Figure 3D), another marker of EMT. Collectively, these data demonstrate that ORES downregulates the EMT of U2OS cells.

Oxyresveratrol (ORES) inhibits the U2OS cell epithelial–mesenchymal transition (EMT). The U2OS cells were treated with 0, 1, 3, and 10 µM of ORES for 24 h. (A) EMT-associated protein E-cadherin, N-cadherin, and vimentin were evaluated by Western blot assay. The expression of E-cadherin (B), N-cadherin (C), and vimentin (D) were quantified by Image J software. The experiments were independently repeated 3 times. The data are shown as mean ± SEM. One-way analysis of variance (ANOVA) with the post hoc Tukey test was used to determine the difference between indicated groups.
ORES Attenuates the STAT3 Phosphorylation in U2OS Cells
Twist is a critical transcription factor for inducing EMT31,32 and can be modulated by STAT3-mediated signaling.33,34 Therefore, we further determined whether ORES inhibits EMT by downregulating STAT3 phosphorylation and Twist expression. The expression of total STAT3 (t-satat3), phosphorylation of STAT3 (p-STAT3), and Twist was measured by Western blotting. In Figure 4A, we show that the STAT3 phosphorylation and Twist expression were significantly reduced upon treatment with ORES at 1, 3, and 10 μM, in which the STAT3 phosphorylation was decreased by 30%, 70%, and 75%, respectively. Meanwhile, the Twist expression was decreased by 30%, 60%, and 70% (Figure 4B and C). These results indicate that ORES attenuated STAT3 activation and Twist expression in U2OS cells.

Oxyresveratrol (ORES) attenuates the phosphorylation of STAT3 in U2OS cells. The U2OS cells were treated with 0, 1, 3, and 10 µM of ORES for 24 h. (A) The expression of p (phosphorylation)-STAT3 (p-STAT3), t (total)-STAT3 (t-STAT3), and Twist were detected by Western blot assay. The ratio of p-STAT3 to t-STAT3 (B) and the expression of Twist (C) were quantified by Image J software. The experiments were independently repeated 3 times. The data are shown as mean ± SEM. One-way analysis of variance (ANOVA) with the post hoc Tukey test was used to determine the difference.
IL-6-Mediated STAT3 Activation Rescues the Migration of ORES-Treated U2OS Cells
Given that ORES inhibited the phosphorylation of STAT3, we further determined whether the enhancement of STAT3 phosphorylation could reverse the effect of ORES on U2OS cell migration. IL-6 was used as an agonist of STAT3 to induce STAT3 phosphorylation. As shown in Figure 5A, 10 μM ORES inhibited the phosphorylation of STAT3, and the addition of IL-6 reversed this effect (Figure 5B). Transwell assays showed that IL-6 significantly reversed the inhibitory effect of ORES on U2OS cell migration (Figure 5C and D). However, WP1066, an inhibitor of STAT3, did not increase the inhibitory effect of ORES on U2OS cell migration (Figure 5E). Overall, these data suggest that IL6-induced STAT3 phosphorylation can rescue the migration of ORES-treated U2OS treated with ORES.

IL-6-induced STAT3 phosphorylation rescues oxyresveratrol (ORES) inhibited cell migration in U2OS cells. The U2OS cells were treated with 10 µM ORES and 20 ng/mL of either IL-6 or 10 µM WP1066 for 16 h. (A) Phosphorylation-STAT3 (p-STAT3) and total-STAT3 (t-STAT3) were measured by Western blot. (B) The ratio of p-STAT3 to t-STAT3 was quantified by Image J software. (C) Cell migration was detected by transwell assay and the images were captured by microscope. The effect of ORES (D) or WP1066 (E) on migration was calculated by Image J software. The experiments were independently repeated at least 3 times. The data are shown as mean ± SEM. One-way analysis of variance (ANOVA) with the post hoc Tukey test was used to determine the difference.
Discussion
In this study, ORES displayed an inhibitory effect on the migration of U2OS cells, which inhibited EMT and activation of STAT3. Therefore, ORES may be a potential agent for the inhibition of osteosarcoma.
EMT is originally known as part of morphogenesis during embryonic development and is also involved in pathological processes, such as wound healing, fibrosis, and cancer metastasis. 35 During tumorigenesis, tumor cells undergoing EMT have a greater chance of metastasis, which is related to their loose characteristics. 36 EMT can reduce cell adhesion ability, which is associated with decreased E-cadherin expression and increased fibronectin expression, resulting in enhanced cell motility and invasion. 37 Therefore, EMT plays an important role in enhancing cancer cell migration in lung cancer, breast cancer, and gastric cancer.38,39 Generally, tumor cells at the primary site can undergo EMT to gain locomotor and invasion abilities, evade blood or lymphatic drainage, and finally exude from blood vessels or lymphatic vessels, return to the state of epithelial cells, and then proliferate to form secondary tumors. 40 Twist is a vital transcription factor that regulates the EMT process. 41 Increased Twist expression inhibits E-cadherin expression but increases that of N-cadherin, which in turn activates EMT and accentuates cell metastasis and invasion. 42 In vivo, Twist could be regulated by TGF-Î2/Smad3 signaling to modulate the process of EMT 1 , which is regulated by the PI3K / AKT and p53 signaling pathways to mediate the metabolism of breast cancer. 43 Lim et al found that dioscin, a plant constituent, could suppress EMT by inhibiting TGF-β1-mediated signaling and reducing lung cancer cell migration. 44 Yang et al showed that Astragalus polysaccharide could regulate EMT through the Wnt/β-catenin signaling pathway to inhibit breast cancer cell migration. 45 EMT is also required to promote osteosarcoma metastasis. 46 Therefore, targeting EMT signaling is a promising treatment for metastatic tumors. In this study, ORES, a plant extract, inhibited osteosarcoma cell migration and metastasis by inhibiting EMT and suppressing the phosphorylation of STAT3.
STAT3 is highly expressed in many tumors, including prostate, lung, ovarian, and breast cancers, and is required for tumor growth, metastasis, and prognosis. 47 The activation of STAT3 induces abnormal expression of oncogenes, which are closely related to cancer cell proliferation, differentiation, and apoptosis, thereby promoting cell proliferation and malignant transformation, restraining cell apoptosis, and exhibiting carcinogenic effects. 48 Since the activation of STAT3 exists not only in tumor cells but also in several immune cells, 49 targeting this pathway is a prospective strategy for treating tumors. Clinically, enhanced phosphorylation of STAT3 is associated with poor prognosis in patients with osteosarcoma. 50 Therefore, targeting STAT3-mediated signaling is a promising strategy for treating osteosarcoma patients. Apatinib is a selective inhibitor of vascular endothelial growth factor receptor-2-mediated signal activation, which can attenuate the phosphorylation of STAT3 and promote the apoptosis of osteosarcoma. 51 ORES displays inhibitory effects on osteosarcoma metastasis and STAT3 phosphorylation. Its activation can be suppressed by ORES to inhibit further the process of EMT and attenuate osteosarcoma cell migration. IL-6 induces STAT3 phosphorylation. Our results confirmed that ORES reduced STAT3 activation and inhibited osteosarcoma cell migration. Additionally, STAT3 is involved in inflammation and plays a significant role in several diseases, such as inflammatory bowel disease, asthma, and psoriasis.52,53 Therefore, ORES may play a therapeutic role in inflammatory diseases.
ORES is a plant-derived chemical compound with various pharmacological properties. For example, ORES possesses anti-inflammatory activity. Hankittichai et al reported that ORES inhibited IL-1��-mediated signal transduction in human microglia by downregulating AKT and ERK phosphorylation. 54 Ashraf et al reported that ORES could also alleviate OVA-induced allergic airway inflammation by inhibiting the expression of IL-4, IL-5, and IL-13. 55 Additionally, ORES could induce apoptosis-like cell death in the triple-negative human breast cancer cell line MDA-MB-231 by inducing ROS instead of in a caspase-dependent manner. 26 These studies indicate that ORES could act on multiple targets and exert various effects. Therefore, ORES has great potential as a new antitumor small-molecule drug.
Our study confirms that ORES inhibits U2OS cell migration by suppressing STAT3 phosphorylation and Twist expression, which further inhibits EMT. Therefore, ORES is a potential agent for the treatment of metastatic osteosarcoma and provides a new option for cancer treatment.
Conclusions
Our study revealed that ORES inhibited osteosarcoma metastasis. Using the U2OS cell line, we demonstrated that ORES attenuated cell migration and EMT. Furthermore, ORES inhibited the expression of Twist and phosphorylation of STAT3. Our findings indicated that ORES is a potential candidate for treating metastatic osteosarcoma.
Footnotes
Acknowledgments
We appreciate the support provided by the Research Center of the Shanghai Pudong Hospital.
Author’ Contributions
LT and YX were responsible for the study design and data acquisition; JZ, ZZ, and ZC performed the data analysis and functional experiments. YB and YC undertook the project design and manuscript revisions. All authors have read and approved the final manuscript.
Availability of Data and Materials
The data of this study are available from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, the Program for the Outstanding Clinical Discipline Project of Shanghai Pudong, the Key Disciplines Group Construction Project of Pudong Health and Family Planning of Commission Shanghai, the Talents Training Program of Pudong Hospital Affiliated to Fudan University, the Project of Key Medical Specialty and Treatment Center of Pudong Hospital of Fudan University (grant Nos. 81971753, PWYgy2018-09, PWZxq2017-11, PJ201903, Tszb2020-15).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
