Abstract
Objective
To explore the intervention mechanism of acacetin combined with wortmannin on myocardial fibrosis in fructose-induced SHR rats.
Methods
Using fructose-induced SHR rats combined with HUVEC cell induced by high-sugar DMEM, the intervention mechanism of acacetin combined with wortmannin on myocardial fibrosis in rats was explored.
Results
In fructose-induced SHR rats, the intervention results of acacetin on myocardial fibrosis showed that acacetin significantly downregulated serum REN, Ang II, ET-1 levels, and upregulated serum PGE2 and NO levels (P < 0.05 or P < 0.01). The combination of wortmannin showed further enhanced effect on the level of ET-1 and PGE2 (P < 0.05). Acacetin and wortmannin significantly increased the expression of CD31 protein and CD31, VE-cadherin mRNA levels in the heart, decreased the expression of CVF, α-SMA protein and CVF, α-SMA, Vimentin and Fibronectin mRNA levels, and enhanced the improvement effect after the combination of acacetin and wortmannin (P < 0.05 or P < 0.01). Both acacetin and wortmannin significantly activated the level of PI3Kp85 in myocardium (P < 0.05 or P < 0.01), and wortmannin significantly inhibited PI3Kp110 (P < 0.01). While, acacetin combined with wortmannin, the activation effect on PI3Kp85 was enhanced, and the inhibitory effect on PI3Kp110 was weakened (P < 0.01). In HUVEC endothelial mesenchymal transformation model, wortmannin significantly inhibited the phosphorylation of IRS-1 serine (P < 0.01); After PI3Kp85 silencing, both acacetin and wortmannin inhibited the activation of PI3 K downstream protein AKT phosphorylation (P < 0.01).
Conclusion
Acacetin synergistic with PI3 K inhibitor wortmannin inhibits the induction effect of PI3Kp110 on IRS-1 serine phosphorylation, indirectly activates insulin signaling pathway, modulates vasoactive factors, alleviates cardiac endothelial mesenchymal transformation in fructose-induced SHR rats, and improves myocardial fibrosis.
This is a visual representation of the abstract.
Introduction
Hypertension is a clinical syndrome characterized by persistent arterial hypertension (systolic and/or diastolic blood pressure). Blood pressure is strongly associated with cardiovascular disease morbidity, with a 10% to 50% increased risk of coronary atherosclerotic heart disease or stroke in people with blood pressure (120 to 129)/(80 to 84) mm Hg compared with those with blood pressure below 120 to 80 mm Hg and a 50 to 100% increased risk in those with blood pressure (130 to 139)/(85 to 89) mm Hg.1,2 The recently published China Cardiovascular Health and Disease Report 2021 states that there are approximately 245 million hypertensive patients in China, and 435 million people have normal high blood pressure. The prevalence is as high as 27.5% in adults and up to 50% to 60% in patients with hypertension with insulin resistance (IR). 3
IR is a pathological condition in which the body's sensitivity to insulin at the physiological level decreases for various reasons, and is the basis for the pathogenesis of type II diabetes, metabolic syndrome and other diseases. In addition to regulating metabolism as the only hypoglycemic hormone in the body, insulin can also directly activate cell “survival signals” and exert cardiovascular protection, 4 while IR will cause insulin to fail to exert the above physiological effects. In addition, IR-induced metabolic disorders such as diabetes and metabolic syndrome further increase the risk of cardiovascular disease. 5 Particularly in hypertensive patients with IR, the risk of cardiovascular disease is greatly increased, for example, in patients with diabetes, the prevalence of cardiovascular disease increases by 20% to 30% for every 5 mm Hg increase in systolic or diastolic blood pressure. 6 In patients with such conditions, IR is aggravated by constant high pressure that reduces the sensitivity of the heart tissue to insulin.IR aggravates the cardiovascular burden by inducing glucose and lipid metabolism disorders, which is more likely to cause excessive cardiac pressure and myocardial hypertrophy, cardiomyocyte overload leading to mitochondrial damage, triggers apoptosis of cardiomyocytes, and then stimulates fibroblast synthesis and secretion of collagen to occur myocardial fibrosis.
Mitochondrial damage in heart parenchymal cells will trigger apoptosis, apoptosis-related signaling molecules stimulate cardiac parenchymal cells to secrete transformation growth factor β (TGF-β), etc., induce fibroblasts and even epithelial/endothelial cells in the heart to transform into active fibroblasts, which synthesize and secrete various fibrin to fill the gap left by apoptosis of parenchymal cells, and this process is cardiac fibrosis. The process of transforming epithelial/endothelial cells into fibroblasts is called epithelial/endothelial mesenchymal transformation (EMT/End MT). 7 By observing that markers of endothelial mesenchymal transformation, such as CD31 and α-SMA, change in endothelial cells, it can indicate the extent of endothelial mesenchymal transformation. Myocardial fibrosis is originally intended to repair itself, but because matrix components such as collagen do not have the physiological functions of heart parenchymal cells, continuous pathological stimulation will lead to excessive proliferation of cardiac fibroblasts, excessive deposition of collagen, myocardial remodeling, and eventually heart failure. Therefore, it is of great significance to find drugs for the treatment of myocardial fibrosis to prevent and control cardiovascular diseases.
Abnormalities in the insulin receptor and its mediated insulin signaling pathway are the molecular pathological basis of insulin resistance. Under normal circumstances, insulin binds as a signaling molecule to the glycoprotein insulin receptor located on the cell membrane of each target organ to form a mooring site, and the insulin receptor recruits and phosphorylates the tyrosine residue of the linker protein insulin receptor substrate 1 (IRS-1), which binds to and activates the subunit p85 with phosphatidylinositol 3 kinase (PI3 K) and its catalytic subunit p110, which stimulates the serine /threonine residue phosphorylation activation at a specific site of the downstream signal transduction molecule protein kinase B (AKT). Subsequently, AKT produces different physiological effects by phosphorylating different signal transduction proteins, participating in cell growth, metabolism, apoptosis, and other processes. There is also a negative feedback regulation mechanism inside this signaling pathway, and the serine kinase activity center of the PI3Kp110 subunit can phosphorylate the serine residue of IRS-1, and the serine phosphorylation of IRS-1 inhibits its tyrosine phosphorylation and thus inhibits the activation of the insulin signaling pathway. Abnormalities in any signal-transducing protein in the insulin signaling pathway can lead to insulin resistance and vice versa. Key proteins including IRS-1, PI3 K, and AKT are also considered candidates for the treatment of insulin resistance.
SHR is a genetically stable hypertensive rat that naturally develops symptoms of insulin resistance with age due to genetic characteristics. 8 In order to simulate the pathogenesis of most patients with hypertension with IR in the clinic, fructose was fed SHR to simulate the pathogenesis of the disease. This method is currently recognized as a relatively stable method for establishing a rat model of hypertension with IR. 9
Due to its complex composition, traditional Chinese medicine has the characteristics of multi-target therapy, and has natural advantages for the treatment of complex metabolic diseases. Therefore, we hope to find active compounds with clear structures in traditional Chinese medicine that can effectively treat heart disease in hypertensive patients with IR. Among the many natural product chemical components of traditional Chinese medicine, flavonoids are widely found in legumes, labial plants such as Thlaspi arvense Linn., 10 Chrysanthemum morifolium Ramat., 11 etc., which can not only relax blood vessels and lower blood pressure, 12 but also regulate glycolipid metabolism, 13 suggesting that flavonoids may have the potential to treat myocardial damage caused by hypertension with IR.
It has been reported that acacetin (AC) has the effect of lowering blood pressure, improving myocardial damage caused by ischemia-reperfusion,14,15 and improving lipid metabolism disorders. 16 At the same time, the previous research of our group has established the fructose-induced SHR model accompanied by metabolic syndrome successfully, and also found that acacetin improved myocardial mitochondrial dysfunction through regulating oxidative stress, mitochondrial fission and fusion, and mitochondrial pathway apoptosis mediated by PI3 K/AKT signaling pathway in hypertensive rats with IR.
However, there are few studies on the effect of acacetin on myocardial damage caused by myocardial fibrosis caused by hypertension combined with IR. Therefore, this project intends to use fructose-induced SHR model to clarify the anti-myocardial fibrosis effect of AC and explore its potential mechanism of action, so as to provide a reference for potential therapeutic drugs in patients with clinical metabolic syndrome.
Materials and Methods
Pharmaceuticals
 .
.
Acacetin (AC, 480-44-1, Source Leaf Biotechnology, China); Valsartan (H20050508, Huanglong Pharmaceutical, China); wortmannin (19545-26-7, MCE, USA).
Animals
8 seven-week-old male Wistar Kyoto Rats (WKY, Vital River, Beijing) and 56 seven-week-old male spontaneously hypertensive rats (SHR) were selected, animal license number: SCXK (Beijing) 2016-0006. Adaptive feeding for one week, during which time free diet and water are available. All the animal experiments involved have been approved by the Animal Care and Use Committee of Henan University of Chinese Medicine (DWLL201908112), Zhengzhou, China.
Cells
The Human Umbilical Vein Endothelial Cell Line (HUVEC) was purchased from the Shanghai Institute of Biological Sciences Cell Bank.
Reagents
PI3Kp110 (20584-1-AP, Proteintech Group, China); PI3Kp85 (60225-1-1 g, Proteintech Group, China); Human α smooth muscle actin (α-SMA, 55135-1-AP, Proteintech Group, China); Platelet-endothelial cell adhesion molecules (CD31, 66065-1-1 g, Proteintech Group, China); β-actin (AC026, ABclonal, China); TGF-β (ab92486, Abcam, USA); AKT (9272S), p-AKT (9271S) (CST, USA); Fluorescent secondary antibodies (C80911-11, C80816-10, LI-COR, USA); Iodine [125I] insulin radioimmunoassay kit (F01-INS, Beijing, China); ATP content test box (ATP-4-Y, COMIN, China); Polycyanoacrylate (BCA) protein quantitative kit (PC0020) (Solarbio, USA); Sodium dodecyl sulfate-polyacrylamide (SDS-PAGE) gel electrophoresis kit (131-0173, Bio-Rad USA); Total RNA Extraction Kit (R1200, Solarbio, USA); BeyoRT™ III cDNA First Strand Synthesis Kit (D7178 M) (Beyotime, China); ChamQ universal SYBR qPCR master mix (L/N8E271E8, Vazyme, China); Rapamycin target protein (mTOR, 2983S), p-mTOR (5S36S), p70 ribosomal protein S6 kinase (p70S6 K, 9202S), p-p70S6 K (9205S) (CST, USA); ET-1 (E-EL-R0167c), PGE2 (E-EL-0034c), REN (E-EL-R0030c), Ang II. (E-EL-R1430c) (Elabscience, China); NO (A013-2, Nanjing, China); NT-proBNP (E-EL-R0670c), myoglobin (MYO, 1E26R46LV3) (Elabscience, China); Insulin receptor substrate 1 (IRS-1, 2382), serine phosphorylated insulin receptor substrate 1 (p-IRS-1, 2381) (CST, USA); mannitol (Mannitol, Dingguo, Beijing, China); fructose (A100226-0005, Diamond, China); glucose (Sigma, USA); liposomes 2000 (Lipo-2000, 1952313, Invitrogen, USA); Lactate dehydrogenase (LDH, A020-2-2), lactic acid (LD, A019-2-1), citric acid (CA, A128) (Nanjing Jiancheng, China); Electron microscopy fixative solution (G1102, Servicebio, China); RIPA lysate (R0020);
Instruments
Centrifuge-5804R high-speed cryogenic refrigerated centrifuge (Eppendorf, Germany); Noninvasive tail artery blood pressure meter (Techman, China); −80 °C Ultra-low Temperature Freezer (Haier, China); −20 °C freezer (Haier, China); M-TURBO Ultrasound Diagnostic System (Sonosite, USA); Odyssey Two-Color Infrared Fluorescence Imaging System (LI-COR, USA); Mini-PROTEAN Tetra Electrophoresis Tank, the all-round protein rapid transfer instrument (Bio-Rad, USA); CO2 Incubators (Thermo, USA); Ultra-clean workbench (Sujing Group, China); Light microscope (TS100, Nikon, Japan); iMarkTM Microplate Absorbance Reader (Bio-Rad, USA); Transmission electron microscopy (HT7700, Hitachi, Japan); Real-time PCR (AB Applied Biosystems, USA).
Experimental Methods
Grouping and Administration of Rats
WKY rats were divided into normal WKY group and normal with wortmannin WKY + W group, and distilled water was given free to drink, and gavage distilled water at a dose of 1 mL/100 g per day. SHR was given 10% fructose solution for free drinking, and divided into 5 groups: model group (SHR (F)), wortmannin group (SHR (F) + W), gavage was given the same dose of distilled water as WKY group; the AC treatment group (SHR (F) + AC), AC + wortmannin group (SHR (F) + AC&W), 25 mg/kg acacetin was given by gavage; the positive control group (SHR(F)+Y), 30 mg/kg valsartan was administered by gavage. wortmannin is administered intraperitoneally at a dose of 1 mg/kg every 3 days.
Determination of SBP, HOMA-IR and Cardiac Function in Rats
Systolic blood pressure in rats was measured weekly by noninvasive tail artery blood pressure monitor; Every 2 weeks, blood was taken by orbital vein blood collection, each rat used the second drop of blood to detect fasting blood glucose value (FBG) by blood glucose meter, collected about 0.4–0.5 mL of blood, centrifuged at 4 °C, 3000 g for 20 min to obtain serum, and detected fasting insulin content (FINS) by radioimmunoassay, HOMA-IR = FBG × FINS/22.5. When the results of SBP and HOMA-IR detected at the end of the sixth week of the experiment showed that AC and TA could significantly improve the two indicators, rat cardiac function was detected and the rats were dissected. Fresh hearts are rapidly quenched in liquid nitrogen and subsequently transferred to a −80 °C freezer for long-term storage for use.
Before taking the material, remove the coat on the rat's chest facing the heart, completely exposing the skin. After anesthesia by intraperitoneal injection of 1% sodium pentobarbital, the M-TURBO ultrasound diagnostic system was used to detect the left ventricular ejection fraction (LVEF), left ventricular short axis shortening rate (LVFS), ventricular septal thickness (IVS) and left ventricular posterior wall thickness (LVPW) by echocardiography, and the average of the three cardiac cycles was measured.
Determination of Cardiac Collagen Volume Fraction (CVF) in Rats
Observation was made using Mason staining. First, fresh rat hearts were taken, mung bean-sized tissue at the apex of the heart was quickly cut and fixed in 3.7% formaldehyde for 24 h, dehydrated by ethanol gradient, and xylene, paraffin embedding, and cut into 4 mm thickness slices. Xylene dewaxing, ethanol gradient rehydration, potassium dichromate, hematoxylin, li chun red acid fuchsin, phosphomolybdenum acid, aniline blue staining, glacial acetic acid differentiation, absolute ethanol dehydration, xylene transparent, neutral gum seal, microscopic examination of myocardial morphological changes under light microscope. Collagen fibers are blue after dyeing; Muscle fibers, cellulose, and red blood cells are red.
Determination of Serum Biochemical Indexes in Rats
The serum ET-1, PGE2, REN, Ang II content of rats was detected by enzyme-linked immunosorbent assay (ELISA), and serum NO level was detected by colorimetric method, and the operation method strictly followed the instructions.
Determination of EMT Levels in Hearts
The assay was performed by immunofluorescence double staining. First, fresh rat hearts were taken, mung bean-sized tissue at the apex of the heart was quickly cut and fixed in 3.7% formaldehyde for 24 h, and after dehydration by ethanol gradient, xylene, paraffin embedding was performed, and sliced into 4 μm thickness sections. Dewaxing with xylene, ethanol gradient rehydration, antigen retrieval, 10% normal rabbit serum blocking, adding CD31 from different sources, α-SMA primary antibody dilution, incubating overnight at 4 °C, adding corresponding fluorescent secondary antibody protected from light incubation, DAPI counterstaining nuclei after sealing, observation under an inverted microscope and acquisition of pictures.
Determination of Protein Expression in Hearts
The WB method was used for the determination. The expression levels of total cardiac proteins CD31, α-SMA, PI3Kp85, and PI3Kp110 were detected.
Western blotting (WB) was used to determine rat cardiac protein expression. Freeze the heart tissue at −80 °C, extract the total protein with RIPA lysate, and use BCA method for protein quantification. Dilute each group into 1 × sample with 5× loading buffer solution and adjust to the same concentration. Each group took 50 μg of total protein for SDS-PAGE electrophoresis, transferred the separated protein bands to the polyvinylidene fluoride (PVDF) membrane by semi-dry method, incubated in PBS containing 5% skimmed milk powder for 1 h to block the non-specific binding site, added 5% skimmed milk powder diluted specific antibody TGF-β of the antigen to be tested, the antibody dilution ratio was 1:1000, and stood at 4 °C for 16 h to specifically bind the antigen and antibody to PBS containing 0.02% Tween20 (PBST) rinsed 5 times/5 min washing to remove unbound antibodies, added fluorescent secondary antibodies diluted with PBST containing 5% skim milk powder, incubated in a dark environment at room temperature for 1.5 h, washed again to remove unbound secondary antibodies, and after the PVDF film was dry, scanned and imaged with Odyssey two-color infrared fluorescence imaging system and quantified the data.
Determination of mRNA Levels in Rat Hearts
The determination was performed by RT-qPCR. The mRNA levels of human vascular endothelial cadherin, CD31, α-SMA, Vimentin, and fibronectin were detected by real-time quantitative polymerase chain reaction (RT-qPCR). First, rat frozen hearts were taken, three samples were randomly selected from each group, 100 mg of tissue was quickly weighed, total RNA was extracted by total RNA extraction kit, RNA bands were separated by agarose gel electrophoresis, and whether RNA was degraded by imaging. The RNA was then quantified with a limited protein nucleic acid analyzer. Each group took 4 μg of RNA to reverse transcription with BeyoRT™ III cDNA first-strand synthesis kit using a thermal cycler, and finally used ChamQ universal SYBR qPCR master mix to quantify the mRNA of VE-cadherin, CD31, α-SMA, Vimentin, and Fibronectin with a real-time PCR instrument, and the operation method was carried out in strict accordance with the kit instructions, and the primer sequences were shown in Table 1.
VE-Cadherin, CD31, α-SMA, Vimentin, Fibronectin Primer Sequences.

HUVEC Cell Culture
H9c2 cells are cultured in a CO2 incubator at 37 °C, 5% CO2, saturated humidity. The complete medium was a high-sugar DMEM formulated with 100 U/mL penicillin, 100 g/mL streptomycin, 10% FBS, pre-warmed at 37 °C before use. Experiments can be carried out when the cells grow to 70%–80% (logarithmic phase). Cell passage will be carried out on the cells that grow to 70% to 80%, one dish is divided into three dishes.
Determination of Protein Expression in HUVEC Cells
The WB method was used for the determination. First, normal (NC) and its PI3Kp85 silencing control (siRNA-NC) group, model (M) and its PI3Kp85 silencing control (siRNA-M) group, wortmannin (W) and its PI3Kp85 silencing control (siRNA-W) group, AC (AC) and its PI3Kp85 silencing control (siRNA-AC) group, AC plus wortmannin (AC&W) and its PI3Kp85 silencing control (siRNA-AC&W) group, AC low, medium, and high dose group, wortmannin low, medium and high dose group. HUVEC cells are seeded in Petri dishes at a density of 1 × 105 cells/mL, 5 mL per well, and 2 parallel dishes per set. The next day was given complete medium for NC and siRNA-NC groups, complete medium containing 80 mM glucose in the M and siRNA-M groups, complete medium containing 80 mM glucose and 1 μM wortmannin in the W and siRNA-W groups, complete medium containing 80 mM glucose and 5 μM AC in the AC and siRNA-AC groups, complete medium containing 80 mM glucose, 5 μM AC and 1 μM wortmannin in the AC&W groups. 1, 5, and 10 μM AC in the low, medium, and high AC groups and 80 mM glucose and 1, 5, and 10 μM wortmannin in the low, medium, and high dose groups. After 48 h of incubation in a CO2 incubator, total protein was extracted from RIPA lysate and protein quantification was performed using BCA method. The expression levels of total proteins IRS-1, p-IRS-1, AKT, p-AKT, CD31, and α-SMA in HUVEC cells were detected according to the method described in the above experimental WB.
Statistical Analysis
All experimental data were statistically analyzed using SPSS26.0 software, and the result data were represented in the form of mean ± standard deviation (
Results
Effect of AC Combined with Wortmannin on Blood Pressure, IR and Cardiac Function in Fructose-Induced SHR
Mitochondrial damage activates the mitochondria in the somatic pathway of cell apoptosis, apoptosis-related factors stimulate the generation of myofibroblasts, synthesis, and secretion of collagen by activating TGF-β proteins cause myocardial fibrosis. Compared with the WKY group, SBP and HOMA-IR were significantly increased in the SHR(F) group, and LVEF and LVFS were significantly reduced (P < 0.01), these results are shown in Fig. 1 B-E. Compared to the SHR(F) group, the AC group was able to significantly pull back the above indicators (P< 0.05 or P < 0.01). But unexpectedly, wortmannin did not antagonize the efficacy of AC, and even it could significantly reduce HOMA-IR (P < 0.01) when used alone; When combined with AC, LVFS (P < 0.05) was significantly improved compared with AC alone. These results are shown in Fig. 1 B-E.

Effects of AC plus wortmannin on the heart function in fructose-induced SHR.
Effect of AC Combined with Wortmannin on Cardiac CVF and serum Biochemical Indexes in Fructose-Induced SHR
Compared with the WKY group, the cardiac CVF of rats in the SHR(F) group was significantly increased (P < 0.01), these results are shown in Fig. 2 A-B, and myocardial fibrosis appeared in SHR & IR rats, the profibrotic factors REN, Ang II, and ET-1 in the serum of rats in the SHR(F) group were significantly increased (P < 0.01), and the inhibition of fibrotic factors PGE2 and NO were significantly reduced (P < 0.05 or P < 0.01), these results are shown in Fig. 2 C-G. Compared with SHR(F) group, both AC and wortmannin alone significantly reduced rat cardiac CVF (P < 0.01). When the two drugs are used in combination, CVF (P < 0.05 or P < 0.01) can be further elevated compared to use alone, these results are shown in Fig. 1 A-B. When AC alone was treated with REN, Ang II, ET-1, and NO, they could significantly recover (P < 0.05 or P < 0.01), and wortmannin treatment could only make these factors have a certain correction trend; After the combination of AC and wortmannin only ET-1, PGE2 and NO showed a further improvement trend compared with the single dosing group, especially PGE2 showed a significant (P < 0.05). These results are shown in Fig. 2 C-G. Therefore, it is speculated that the combination of AC and wortmannin can improve myocardial fibrosis in SHR & IR rats through endothelial cell-related factors instead of RAAS system, which may be related to EMT.

Effects of AC plus wortmannin on the heart function in fructose-induced SHR.
Effect of AC Combined with Wortmannin on Markers of Endothelial-Mesenchymal Transformation in Heart of Fructose-Induced SHR
Compared with the WKY group, the mRNA levels of endothelial cell markers CD31 and VE-cadherin in the SHR(F) group were significantly reduced, and the mRNA levels of mesenchymal cell markers α-SMA, Vimentin and Fibronectin were significantly increased, these results are shown in Fig. 3 E-I, and the expression levels of CD31 and α-SMA proteins were consistent with their mRNA (P < 0.05 or P < 0.01), these results are shown in Fig. 3 A-D, indicating endothelial-mesenchymal transformation in SHR & IR rat hearts. Both AC and wortmannin significantly improved endothelial-mesenchymal transformation, but the effect was more significant when the two drugs were combined (P < 0.05, P < 0.01).

Effect of AC combined with wortmannin on rat endothelial-mesenchymal transformation markers (WB and RT-qPCR), and the expression of PI3Kp85 and PI3Kp110 in fructose-induced SHR
Effects of AC Combined with Wortmannin on PI3Kp85 and PI3Kp110 in Heart of Fructose-Induced SHR
Compared with the WKY group, the p85 subunit of total cell protein PI3 K in the SHR(F) group was significantly reduced (P < 0.01), while the p110 subunit did not change significantly, only a downward trend. AC can significantly call back the p85 level (P < 0.01), while wortmannin can significantly inhibit p110 (P < 0.01) and activate p85 (P < 0.05). When the two drugs were combined, the p85 subunit was further retracted, and although the inhibited state of p110 was significantly recovered compared with the wortmannin alone group (P < 0.01), it was still in the inhibited state (P < 0.05) compared with the SHR(F) group. These results are shown in Fig. 3 J-K.
Effect of Different Doses of AC or Wortmannin on Markers of Fibrosis in High Glucose-Induced HUVEC
Compared with the NC group, the levels of α-SMA protein and CD31 protein in the M group were significantly increased, which confirmed that the high glucose-induced HUVEC under these conditions could successfully establish an endothelial-mesenchymal transformation cell model. Compared with the M group, both medium-dose and high-dose AC could significantly recall α-SMA and CD31, so 5 μM AC was used for follow-up experiments. Only low-dose wortmannin can improve both α-SMA and CD31, so 100 μM wortmannin was selected for follow-up testing. These results are shown in Fig. 4A-D.
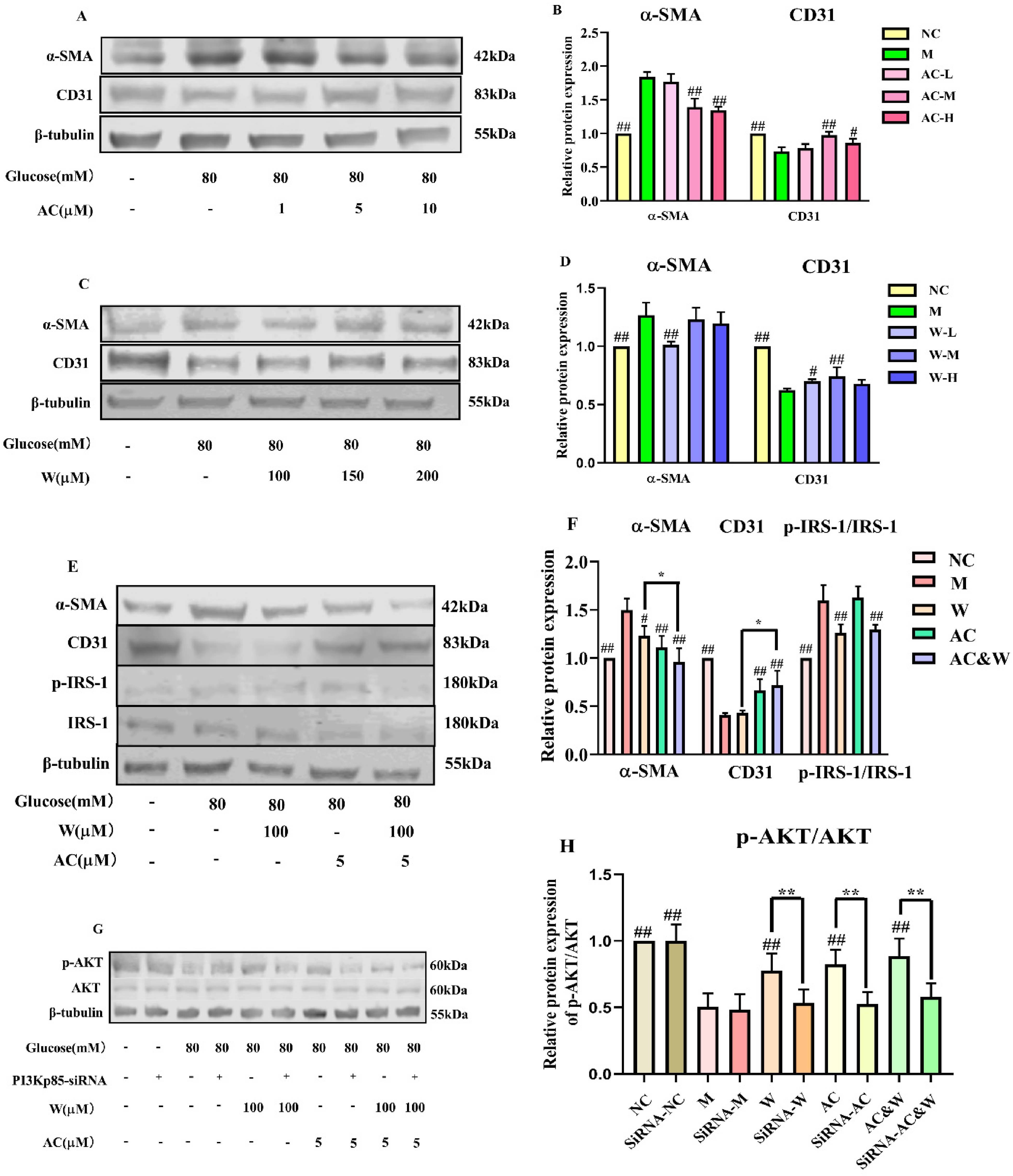
Effects of different concentrations of AC and wortmannin on HUVEC cells.
Effect of AC Combined with Wortmannin on Endothelial-Mesenchymal Transformation Markers in High Glucose-Induced HUVEC
Compared with the NC group, the level of α-SMA protein in the M group was significantly increased, and the level of CD31 protein was significantly reduced. Compared with M group, both AC and wortmannin can significantly retract α-SMA and CD31, but the combination of the two drugs improves the effect better. These results are shown in Fig. 4E-F.
Effect of AC Combined with Wortmannin on Phosphorylation of IRS-1 Serine in High Glucose-Induced HUVEC
Compared with the NC group, the phosphorylation of IRS-1 serine in the M group was significantly increased. Compared with group M, AC could not inhibit IRS-1 serine phosphorylation, while wortmannin could significantly reduce IRS-1 serine phosphorylation, and the improvement effect of wortmannin on IRS-1 de-inhibition became weaker after the combination of the two drugs, but there was still a certain pullback trend, suggesting that the improvement effect of wortmannin on HUVEC endothelial interstitial transformation may be by inhibiting the induction of IRS-1 serine phosphorylation by PI3Kp110, but AC did not have this effect. These results are shown in Fig. 4E-F.
Silent Effect of PI3Kp85 on AKT in High Glucose-Induced HUVEC
Compared with the NC group, the phosphorylation level of AKT in the M group was reduced; Compared with group M, both AC and wortmannin could significantly upregulate the phosphorylation level of AKT, and the combination of the two drugs could further strengthen the upward regulation trend. It suggests that either AC directly upregulates PI3Kp85 levels or wortmannin indirectly phosphorylates IRS-1 serine levels by inhibiting PI3Kp110, which ultimately activates downstream AKT and thus insulin signaling pathways. These results are shown in Fig. 4G-H.
Discussion
Metabolic syndrome (MS) is one of the key concerns in global public health, and it significantly increases the risk of cardiovascular disease, type 2 diabetes, nonalcoholic fatty liver disease, and other diseases, 17 mainly associated with abdominal obesity, insulin resistance, dyslipidemia, and increased blood pressure. 18 The main pathogenesis of MS is closely related to IR, which reduces the sensitivity of insulin-sensitive tissues to insulin signaling, resulting in an imbalance of glucose homeostasis in the internal environment. 19 Acacetin is a natural flavonoid widely present in a variety of plants,11,20 which has pharmacological activities such as lowering blood lipids, lowering blood sugar, antioxidant, and anti-tumor. 21 Our previous study also found that acacetin can reduce blood pressure and improve blood lipid disorders, insulin resistance, aortic fibrosis and endothelial dysfunction in fructose-induced spontaneous hypertensive rats. 15
The molecular mechanism of IR is the abnormality of insulin receptors and signal transduction proteins downstream of the receptor, and damage to any link of the entire insulin signal transduction pathway will cause IR. AC preferentially improves IR and then hypertension, suggesting that AC may improve hypertension and other complications by regulating insulin signaling.
Mitochondrial damage can induce myocardial fibrosis in a variety of ways.22,23 Taking oxidative stress as an example, abnormal function of the mitochondrial respiratory chain leads to excessive production of ROS, which can cause oxidative damage to intracellular biological macromolecules. Initial cells repair damage by initiating protein repair and protein degradation systems, but persistent accumulation of oxidative damage products ultimately triggers apoptosis. 24 This pathological apoptosis stimulates cell synthesis and secretion of a large number of chemokines, such as TGF-β, increased vascular permeability at the site of injury, exudation of immune cells from the blood, and chemokine recruitment infiltrates the affected area and breaks down the cells. TGF-β not only has chemokine activity, but also promotes the activation of fibroblasts in a resting state under normal physiological conditions into active fibroblasts,25,26 and can also stimulate the transformation of damaged endothelial cells and obtain the characteristics of fibroblasts (EMT), 27 fibroblasts repair tissues by synthesizing and secreting fibrin to fill the voids left by apoptosis, but these fillers do not have the physiological function of parenchymal cells. The continuation of this process can lead to fibrosis. Therefore, in the heart of SHR & IR rats, a large number of parenchymal cells were apoptosis due to mitochondrial dysfunction, which eventually led to decompensation of cell function and myocardial fibrosis.
Our previous studies confirmed that acacetin improved myocardial mitochondrial dysfunction by regulating the PI3 K/AKT signaling pathway-mediated oxidative stress, mitochondrial fission and fusion, and apoptosis of the mitochondrial pathway in fructose-induced SHR rats. 14 As shown in this study, AC can regulate RAAS system and endothelial cell correlated factors, activate the key protein PI3Kp85 subunit of the insulin signaling pathway, and improve SHR & IR cardiac endothelial mesenchymal conversion and myocardial fibrosis in rats. As an antagonist of PI3 K, wortmannin did not exert the expected antagonist effect on acacetin. After 7 weeks of intraperitoneal injection of 1 mg/kg dose of wortmannin every three days, wortmannin did not antagonize the improvement of AC on SBP, HOMA-IR, LVEF and LVFS, but further enhanced the effect of acacetin on endothelial cell-related factors, cardiac EMT and myocardial fibrosis after the combination with acacetin.
PI3 K is a family of kinases with a dimeric structure, including the regulatory subunit p85, which is responsible for binding to IRS-1 after tyrosine phosphorylation activation through the protein domain during signal transduction, and the catalytic subunit p110, with a serine/threonine kinase activity center, is responsible for catalytic activation of downstream proteins. 28 However, wortmannin almost completely binds to the protein of 110 kDa in vitro, but does not bind to the protein of 85 kDa, 29 indicating that the inhibitory effect of wortmannin on PI3 K is exerted by binding to p110, not p85, and its antagonist object is precisely the p110 subunit of PI3 K. The siRNA used in vitro is designed for the mRNA sequence of the PI3Kp85 subunit, so the conclusion obtained in vitro should be specifically described as AC can exert a protective effect by activating PI3Kp85, so it makes sense that it does not antagonize the efficacy of AC when using wortmannin to antagonize PI3Kp110.
The transduction process of signal transduction proteins in the body is subject to complex regulation, the hypoglycemic effect of insulin depends on the activation of the insulin signaling pathway, the activation of this pathway depends on the activation of PI3 K, and the activation of PI3 K depends on the activation of IRS-1 tyrosine phosphorylation, but IRS-1 serine/threonine phosphorylation will inhibit its own tyrosine phosphorylation. In fact, in addition to activating downstream AKT and other signal transduction proteins to activate the insulin signaling pathway, the activated PI3 K can also induce serine/threonine residue phosphorylation of IRS-1 through the serine/threonine kinase activity center on p110, which belongs to the negative feedback regulation mechanism of the insulin signaling pathway, thereby inhibiting the insulin signaling pathway. The inhibition of p110 by wortmannin is not only manifested in the inhibition of downstream signal transduction proteins such as AKT in insulin signaling pathway, but also in the inhibition of the above-mentioned negative feedback, thereby inhibiting or activating two diametrically opposed effects on insulin signaling pathway, respectively. Factors that determine the ultimate response of wortmannin may include mode of administration, dose of drug, duration of administration, etc. 30 In the case of intraperitoneal injection every three days at a dose of 1 mg/kg, wortmannin did not aggravate the HOMA-IR of SHR & IR rats, but improved, so it was speculated that wortmannin in this condition may activate the insulin signaling pathway and improve IR by inhibiting the negative feedback regulation with p110. With this hypothesis in mind, this study used wortmannin combined with AC to improve myocardial fibrosis in fructose-induced SHR rats, and explored the mechanism of wortmannin and AC respectively.
Abnormal activation of the RAAS system and impairment of endothelial cell function are involved in the pathogenesis of hypertension and insulin resistance. REN and Ang II are important components of the RAAS system. The REN synthesized and secreted by the kidneys enters into the blood circulation, catalyzes the conversion of angiotensinogen secreted by hepatocytes into angiotensin I, which is hydrolyzed to Ang II under the catalytic effect of angiotensin-converting enzyme. In addition to being a strong vasoconstriction effector, Ang II can also directly stimulate the proliferation of cardiac fibroblasts, participate in collagen synthesis, and is a profibrotic effector factor. 31 ET-1 synthesized and secreted by endothelial cells also has an effect similar to Ang II, while factors such as NO and PGE2 are also synthesized and secreted by endothelial cells, which have physiological activities to inhibit fibrosis while relaxing blood vessels. 32 The results of Fig. 2 showed that the volume fraction of cardiac collagen in SHR & IR rats was significantly increased, obvious myocardial fibrosis symptoms appeared, the levels of profibrotic effector factors REN, Ang II, and ET-1 increased, and the inhibition of fibrotic effector factors NO and PGE2 decreased. AC could downregulate collagen volume fraction and improve all of the above cytokine levels. Although wortmannin also had an effect on collagen volume fraction, it did not seem to have a significant improvement effect on cytokines, but only a certain improvement trend; When AC was combined with wortmannin, the effect of improving collagen volume fraction was enhanced, but only ET-1, NO and PGE2 secreted by endothelial cells among the five vascular factors were further improved. However, REN and Ang II did not receive further pullbacks after the combination of the two drugs. From this, we focus our attention on endothelial cells.
In the past, it was thought that the core event of myocardial fibrosis was the transformation of the heart into fibroblasts during in-situ fibroblasts under stress and the synthesis of extracellular matrices such as collagen. However, latest studies have shown that cardiac fibroblasts have multiple sources, including endothelial cells in the heart. When these cells were damaged, they were also induced to transform by various cytokines and lose their original characteristics,33–35 such as CD31, VE-cadherin, and obtain the characteristics of fibroblasts, such as α-SMA, Vimentin, and Fibronectin. This process is called EMT, and by detecting the changes in these markers in endothelial cells separately, it can indicate the degree of EMT. The results of Fig. 3 showed that SHR & IR rats showed obvious EMT characteristics at both mRNA level and protein level, that is, the level of endothelial cell markers decreased, and mesenchymal cell marker characteristics were occurred. Consistent with the results of collagen volume fraction, both AC and wortmannin can effectively improve EMT, and showed better effect in combination.
Through the establishment of HUVEC intracellular mesenchymal transformation model in vitro supplementary verification, it was found that the level of IRS-1 serine/threonine phosphorylation in the cells of the model group was significantly increased, and wortmannin could effectively inhibit it, but AC could not, confirming that wortmannin could indeed inhibit the negative feedback regulation of p110 to IRS-1 and indirectly activate the insulin signaling pathway. In Fig. 4, it was shown that by silencing p85, both AC and wortmannin were significantly inhibited in their activation of AKT downstream of PI3K.
Conclusion
AC can directly activate the insulin signaling pathway by activating the p85 subunit of PI3 K, while wortmannin can inhibit the negative feedback regulation effect of p110 on IRS-1 and indirectly activate the insulin signaling pathway, which together improve myocardial fibrosis in SHR & IR rats, and the improvement effect is nonlinear additive.
Supplemental Material
sj-pptx-1-npx-10.1177_1934578X231214920 - Supplemental material for Acacetin Combined with Wortmannin Activated Insulin Signaling Pathway by Inhibiting the Induction Effect of PI3Kp110 on IRS-1 Serine Phosphorylation to Improve Myocardial Fibrosis in Fructose-Induced SHR Rats
Supplemental material, sj-pptx-1-npx-10.1177_1934578X231214920 for Acacetin Combined with Wortmannin Activated Insulin Signaling Pathway by Inhibiting the Induction Effect of PI3Kp110 on IRS-1 Serine Phosphorylation to Improve Myocardial Fibrosis in Fructose-Induced SHR Rats by Lirui Zhao, Peipei Yuan, Qi Zhang, Yan Zhang, Yajuan Zheng, Saifei Li, Yi Chen, Panying Li, Yuan Ruan, Weisheng Feng and Xiaoke Zheng in Natural Product Communications
Footnotes
Acknowledgments
The author of this article wishes to thank the National Key Research and Development Program (The Major Project for Research of the Modernization of TCM): (2019YFC1708802), Major Science and Technology Projects of Henan Province (171100310500), Henan Province high-level personnel special support “ZhongYuan One Thousand People Plan” – Zhongyuan Leading Talent (ZYQR201810080), the Engineering and Technology Center for Chinese Medicine Development of Henan Province and the PhD Research Funds of Henan University of Chinese Medicine (RSBSJJ2018-04).
Author Contributions
Feng, W and Zheng, X designed the experiments. Zhao, L performed most of the experiments with the help of Yuan, P and Zhang, Q; Zhang, Y and Zheng, Y conducted immunofluorescence experiments. Li, P and Chen, Y conducted in vivo surgeries. Ruan, Y and Li, S conducted and analyzed data. All authors discussed the results and commented on the manuscript.
Funding
This work was supported by the National Key Research and Development Program (The Major Project for Research of the Modernization of TCM): (2019YFC1708802), Major Science and Technology Projects of Henan Province (171100310500), Henan Province high-level personnel special support “ZhongYuan One Thousand People Plan”-Zhongyuan Leading Talent (ZYQR201810080), the Engineering and Technology Center for Chinese Medicine Development of Henan Province and the PhD Research Funds of Henan University of Chinese Medicine (RSBSJJ2018-04).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
