Abstract
The antihypertensive mechanism was studied of the ethyl acetate fraction of the ethanol extract (EAPF) of Gardenia jasminoides var. radicans Makino (GJRM). GJRM is a fake product of Gardenia jasminoides Ellis (G. jasminoides), but in China’s Henan province, the production of GJRM is much more than G. jasminoides’s, but its traditional usage is as a dye. Gardenia jasminoides can be used to reduce blood pressure and blood glucose levels. The chemical compositions of GJRM and G. jasminoides are similar, and we previously confirmed that GJRM can also reduce blood pressure. Here, we report that the EAPF of GJRM could activate the phosphoinositide 3-kinases (PI3K) pathway in the kidneys of spontaneously hypertensive rats, thus increasing the content of nitric oxide and bradykinin in sera and decreasing endothelin-1 content. EAPF can also decrease the levels of with-no-lysine kinase 1 (WNK1) expression, WNK4 and oxidative stress-responsive kinase 1 messenger ribonucleic acid (mRNA), and Na-K-2Cl cotransporter and sodium chloride cotransporters mRNA and phosphorylation. To investigate the antihypertensive effects of the EAPF of GJRM, 5 monoterpenoids isolated from EAPF were studied for their effects on NRK52e and IMCD3 cells. These compounds inhibited the PI3K-WNK signaling pathway to varying degrees under hypotonic conditions; 4-methoxyrehmapicrogenin had the best effect.
Keywords
As living standards improve, the incidence of hypertension increase every year. Hypertension is the most common chronic disease and the most important risk factor for cardiovascular and cerebrovascular diseases. Current treatments involve drug therapies.
Gardenia jasminoides var. radicans Makino (GJRM) has been considered as a fake product of Gardenia jasminoides Ellis. Gardenia jasminoides Ellis was first reported in Shennong Herbal Medicine to reduce blood glucose levels, control blood pressure, and regulate blood lipid. 1 -3
The main active components of GJRM are similar to those of G. jasminoides. In China’s Henan province, the production of GJRM is much more than G. jasminoides’s, but its traditional usage is as a dye. A systematic study of the chemical constituents of GJRM was carried out in our laboratory and we would like to expand its application. We confirmed that GJRM had diuretic and antihypertensive effects attributed to EAPF. The purpose of this study was to investigate the antihypertensive mechanisms of ethyl acetate fraction of the ethanol extract (EAPF) in spontaneously hypertensive rats (SHR).
With-no-lysine kinase (WNK) is widely distributed in the human body and is mainly expressed in renal epithelial cells; WNK plays a regulatory role in renal ion channels. The expression of Na-K-2Cl cotransporter (NKCC2) in kidneys is specific and is one of the most important molecular bases for regulating water-saving salt balance in renal tubules; NKCC2 is also one of the main targets of bumetanide. 4,5 In recent years, WNK-oxidative stress-responsive kinase 1 (OSR1)-NKCC/sodium chloride cotransporters (NCC) has been confirmed as a pathway for the treatment of hypertension. 6
The phosphoinositide 3-kinases (PI3K)-protein kinase B (AKT) signaling pathway plays a number of roles in many diseases, 7,8 especially in diabetes. 9 In recent years, studies have shown that the activation of the PI3K-AKT pathway reduces blood pressure by producing nitric oxide (NO) and protecting cardiomyocytes in SHR. 10 Additionally, protein kinase A (PKA) inhibited the degradation of WNK to increase the activity of downstream proteins. 11 In cancer cells, PI3K-AKT regulates the WNK signaling pathway to increase the expression of NKCC. 12 Therefore, we detected the expression of related pathways in the kidney of SHR and the level of EAPF after administration of GJRM.
The WNK-OSR1-NKCC/NCC pathway is activated in hypotonic states. 13 We established a hypotonic stimulation model of renal tubular epithelial (NRK52e) cells to further study the mechanism of lowering blood pressure by GJRM at the cell level, which provided theoretical support for expanding the application of GJRM.
Materials and Methods
Materials
Dulbecco’s modified Eagle’s medium (DMEM), trypsin, fetal bovine serum (FBS), penicillin, and streptomycin were purchased from Gibco (Life Technologies, New York, USA). Bumetanide was purchased from Guilin South Pharmaceutical (Guiling, China). Bradykinin (BK), NO and endothelin-1 (ET-1). Enzyme-linked immunosorbent assay kits were purchased from Elabscience Biotechnology (Wuhan, China). Absolute ethanol was purchased from Tianjin Siyou Chemical Reagent (Tianjin, China). Chloral hydrate was purchased from Tianjin Guangfu Fine Chemical (Tianjin, China).
Extraction of GJRM and Compounds A -E
Dried powdered fruits of GJRM (9.8 kg) were extracted with 95% ethanol (8 L, extraction method by reflux extraction twice, 1 hour each time) to obtain a 95% ethanol extract (2.1 kg). The 95% ethanol extract was suspended in water (H2O, 1 L) and successively partitioned with petroleum ether (1 L × 5), ethyl acetate (1 L × 5), and n-butanol (1 L × 5) to produce petroleum ether (154.8 g), EAPF (320.1 g), and n-butanol (730.5 g) fractions. EAPF was subjected to silica gel chromatography (CC) and eluted with dichloromethane–methanol gradient mixtures (100:0-0:100) to yield 8 fractions (A1-A8).
A4 (21.0 g) was subjected to MCI gel CHP-20 CC and eluted with a methanol–water gradient solvent system (10%-100%) to give 7 subfractions (A4-1 to A4-7). A4-1 (12.6 g) was further applied to ODS CC using a gradient of methanol–water (10%-100%) to yield 18 subfractions (A4-1-1 to A4-1-18). A4-1-1 (1.3 g) was subjected to Sephadex LH-20 CC (MeOH: H2O 30:70) to give 7 subfractions (A4-1-1-1 to A4-1-1-7). Subfraction A4-1-1-4 (244.8 mg) was separated by semipreparative high-performance liquid chromatography (HPLC) (methanol [MeOH]:H2O, 30:70) to obtain crocusatin M (compound
The compounds
Animal Treatment
Eight weeks-old male SHRs and Wistar-Kyoto (WKY) rats were purchased from Vital River Laboratory Animal Technology (Beijing, China). After 1 week of acclimatization, control SHRs and WKY (untreated) received vehicle. Briefly, 5 mg/kg/day bumetanide, 360 mg/kg/day GJRM extract, and EAPF (19, 38 and 76 mg/kg/d) were administered by gavage into SHRs. After administration for 3 weeks, blood samples were collected. Kidneys were quickly isolated from the rats and frozen in liquid nitrogen. All surgery was performed under chloral hydrate. All experimental animals used in this study were treated under a protocol approved by the Animal Care and Research Committee of the Henan University of Traditional Chinese Medicine.
Western Blotting
The cytosolic and membrane proteins from the kidney tissue were obtained by a cytoplasmic membrane protein extraction kit (Applygen Technologies Institute, Beijing, China). Briefly, 60 µg of protein from each group were subjected to 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), transferred to polyvinylidene difluoride membranes and incubated with primary antibodies against WNK1 (1:500, Abcam, UK), PI3K (1:1000, Proteintech, China), p-AKT (1:500, Abcam, UK), PKA (1:1000, Abcam), and β-actin (1:5000, Proteintech, China) at 4°C overnight. The antibody–antigen complexes were detected by incubating the membranes with the respective secondary antibodies (1:30 000, LI-COR Biosciences, ME, USA) for 1 hour at room temperature. The blots were then washed 3 times with phosphate-buffered saline with Tween 20 (PBST) before Western blot. An Odyssey 2-color infrared fluorescence imaging system was used for imaging. Values were expressed after normalization to β-actin levels by Image Studio software (LI-COR Biosciences, ME, USA).
Immunohistochemistry
Rabbit anti-p-NCC antibody (1:100, PhosphoSolutions, USA) and rabbit anti-p-NKCC2 antibody 5 (1:500) were the primary antibodies used. Fluorescent Cy3-coupled or fluorescent fluorescein isothiocyanate-coupled secondary antibodies were used for detection. For both antibodies, control experiments were conducted in which the primary antibody was omitted. 18
RNA Extraction and Quantitative Reverse Transcription-PCR Analysis
RNA was extracted from the kidneys with an RNA Extraction Kit (Solarbio, China). RNA purity and concentration were determined by measuring and comparing absorbances at 260 and 280 nm. Then, 1 µg of total RNA was reverse transcribed into complementary DNA (cDNA) using a HiScript II 1st Strand cDNA Synthesis kit (Vazyme Biotech, China) and subjected to a polymerase chain reaction (PCR) using a ChamQ Universal SYBR qPCR Master Mix (Vazyme Biotech, China). Primer sequences used for WNK1, WNK4, OSR1 NKCC2, NCC, and GAPDH are listed in Table 1.
Primer Sequences

WNK1, With-no-lysine kinase 1; WNK4, With-no-lysine kinase 4; OSR1, Oxidative stress-responsive kinase 1; AKT, Protein kinase B; NKCC2, Na-K-2Cl cotransporter; NCC, Na-Cl cotransporter; GAPDH, Glyceraldehyde-3-phosphate dehydrogenase.
Cell Culture and Treatments
NRK52e cells and IMCD3 were cultured at 37°C and 5% carbon dioxide (CO2) supplemented with 10% FBS with 100 U/mL penicillin, and 100 µg/mL streptomycin.
Cells were plated on 96-well plates and incubated at 37°C in 5% CO2 overnight. After 24 hours of starvation, the medium was changed to a different osmotic pressure medium:normal (350 mOsmol/kg H2O, DMEM) and hypotonic (175 mOsmol/kg H2O, DMEM + 50% pure water). Simultaneously, the cells were stimulated with 5 µM bumetanide (Aladdin, Shanghai, China) or 5 µM compounds

The chemical structures of compounds A-E.
Statistical Analysis
All data are expressed as the mean ± standard error of mean
Results
Effect of EAPF of GJRM on BK, NO, ET-1 in Serum of SHR Rats
The EAPF of GJRM significantly increased the contents of NO and BK in the serum of SHR rats and significantly decreased the content of ET-1 (Figure 2a–c).

Effects of EAPF of GJRM on blood pressure. The effects of different concentrations of EAPF of GJRM (19, 38, and 76 mg/kg/day) on (a) NO, (b) BK, and (c) ET-1 in the serum of SHR. All data are presented as the mean ± standard deviation (n = 8). * P < 0.05 and ** P < 0.01 vs the WKY group. # P < 0.05 and ## P < 0.01 vs the SHR group. BK, bradykinin; EAPF, ethyl acetate fraction of the ethanol extract; GJRM, ET-1, endothelin-1; GJRM, Gardeniajasminoidesvar. radicans Makino; NO, nitric oxide; SHR, spontaneously hypertensive rats; WKY, Wistar-Kyoto.
Effects of EAPF of GJRM on PI3K Related Proteins and mRNA in Renal Tissue of SHR Rats
The expressions of PI3K, p-AKT, PKA protein, and AKT messenger ribonucleic acid (mRNA) in the kidney of SHR rats significantly increased due to treatment by EAPF of GJRM (Figure 3a and b).
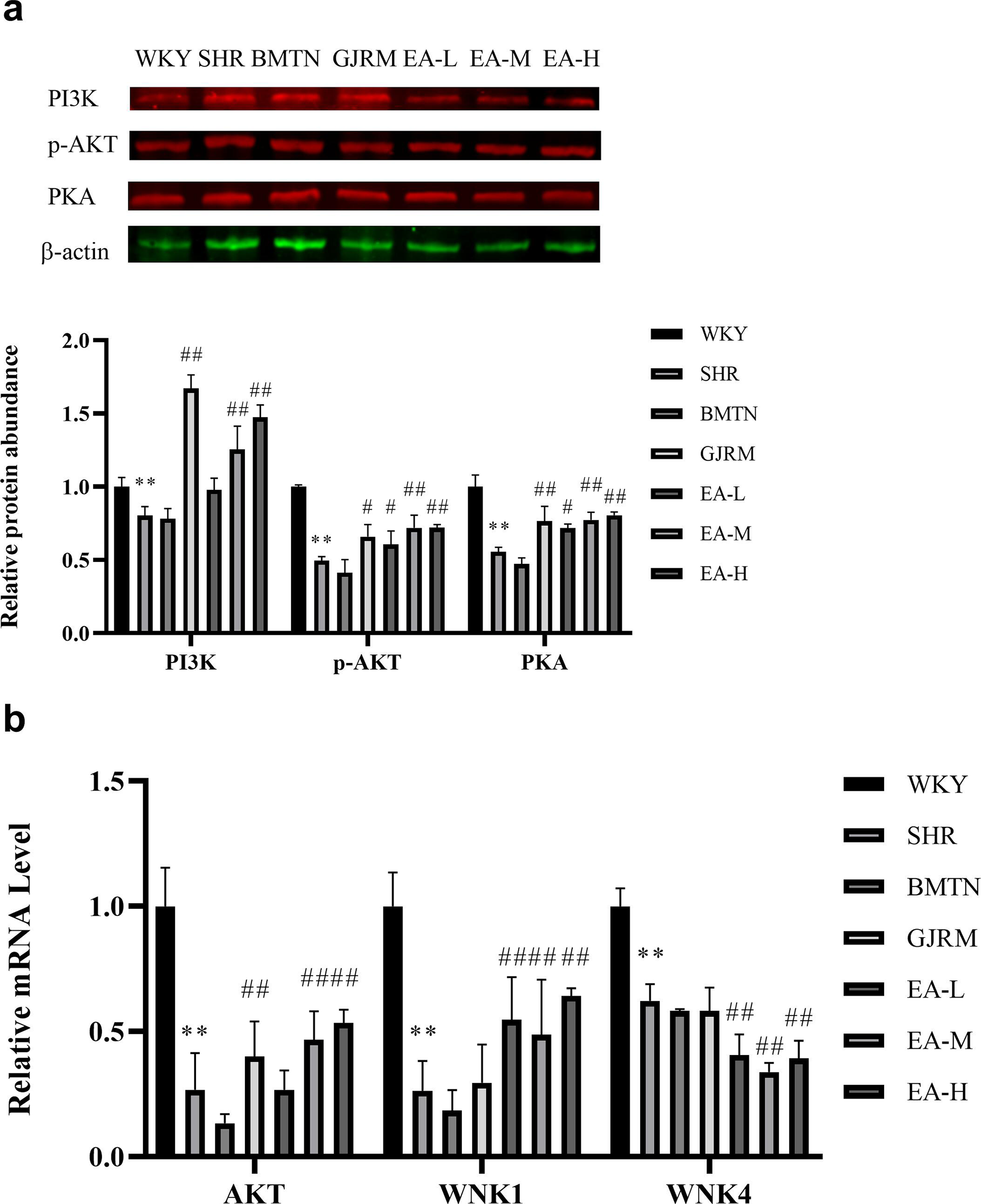
Effects of EAPF of GJRM on PI3K related proteins and mRNA in renal tissue of SHR rats. The expression of (a) PI3K, p-AKT, and PKA in the kidney tissue of SHR. (b) AKT, WNK1, and WNK4 mRNA expression. The relative level of mRNA expression was calculated as 2−ΔΔCt. All data are presented as the mean ± standard deviation (n = 8). * P < 0.05 and ** P < 0.01 vs the WKY group. # P < 0.05 and ## P < 0.01 vs the SHR group. AKT, protein kinase B; EAPF, ethyl acetate fraction of the ethanol extract; GJRM, Gardeniajasminoidesvar. radicans Makino; mRNA, messenger ribonucleic acid; PI3K, phosphoinositide3-kinase; PKA, proteinkinase A; SHR, spontaneously hypertensive rats; WNK1, with-no-lysinekinase 1; WKY, Wistar-Kyoto.
Effects of EAPF of GJRM on WNK1 and Related mRNA in Renal Tissue of SHR Rats
EAPF of GJRM decreased the expression of WNK1 protein in the kidneys of SHR rats and regulated the expression of mRNA related to the WNK pathway (Figure 4a and b).
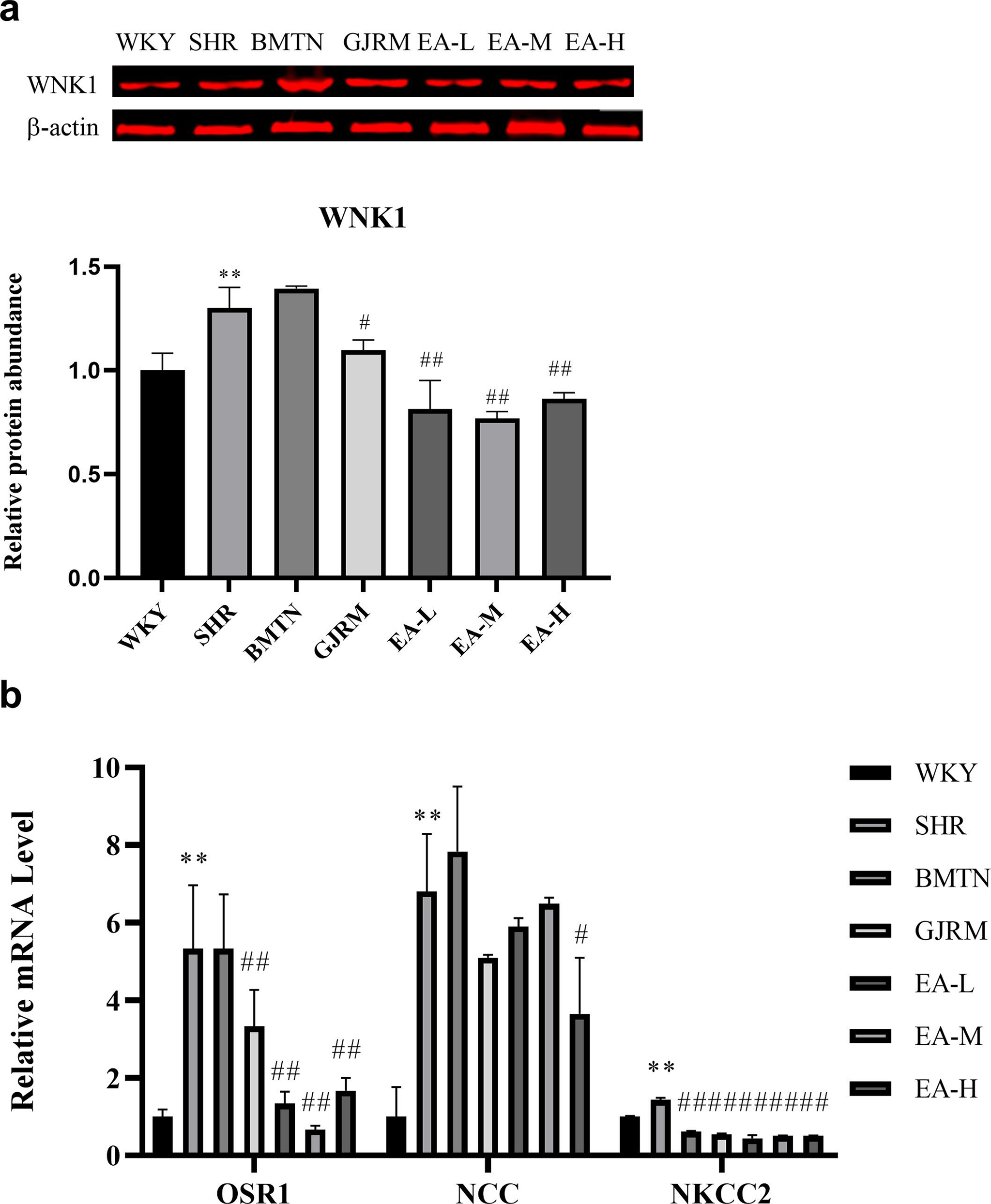
Effects of EAPF of GJRM on WNK1 and related mRNA in renal tissue of SHR rats. The expression of (a) WNK1 in the kidney tissue of SHR. (b) OSR1, NCC, and NKCC2 mRNA expression. The relative level of mRNA expression was alculated as 2−ΔΔCt. All data are presented as the mean ± standard deviation (n = 8). * P < 0.05 and ** P < 0.01 vs the WKY group. # P < 0.05 and ## P < 0.01 vs the SHR group. EAPF, ethyl acetate fraction of the ethanol extract; GJRM, Gardeniajasminoidesvar. radicans Makino; mRNA, messenger ribonucleic acid; NCC, sodiumchloride cotransporters; NKCC2, Na-K-2Clcotransporter; OSR1, oxidative stress-responsive kinase 1; SHR, spontaneously hypertensive rats; WNK1, with-no-lysinekinase 1; WKY, Wistar-Kyoto.
Effects of EAPF of GJRM on p-NCC and p-NKCC2 Protein in Kidney Tissue of SHR Rats
EAPF of GJRM decreased the phosphorylation of NCC and NKCC2 in the kidney of SHR rats (Figure 5).
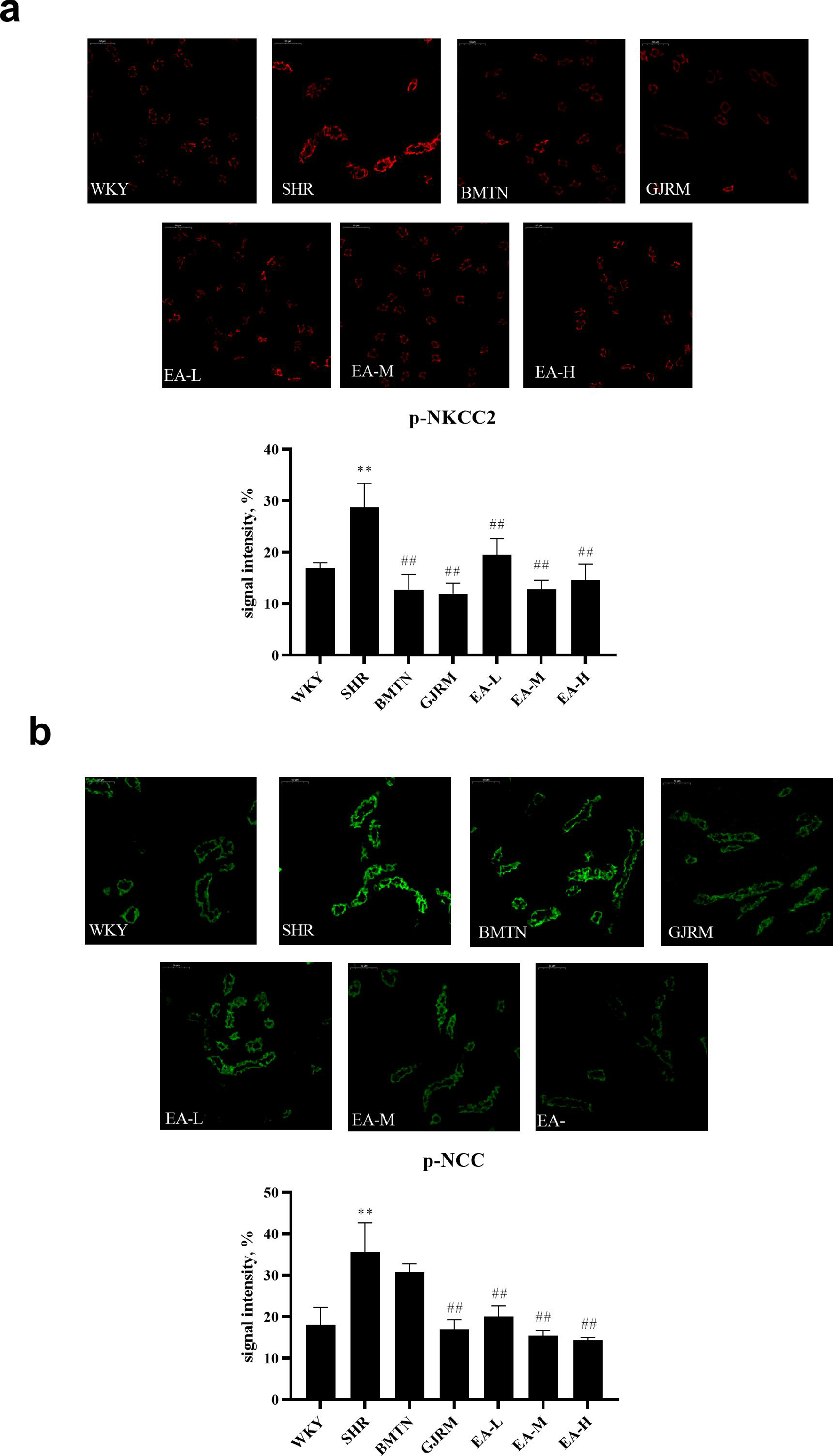
Effects of EAPF of GJRM on NCC and NKCC2 phosphorylation in kidney detected by immunofluorescence. Images of (a) NCC phosphorylation and (b) NKCC2 phosphorylation from kidneys of each groups (50 µm). Data are mean ± standard deviation from n = 6 rats/group. * P < 0.05 and ** P < 0.01 vs the WKY group. # P < 0.05 and ## P < 0.01 vs the SHR group. EAPF, ethyl acetate fraction of the ethanol extract; GJRM, Gardeniajasminoidesvar. radicans Makino; NCC, sodiumchloride cotransporters; NKCC2, Na-K-2Clcotransporter; SHR, spontaneously hypertensive rats; WKY, Wistar-Kyoto.
Effects of Monoterpenes from GJRM on PI3K-WNK Pathway in NRK52e Cells Under Hypotonic Condition
Monoterpene compounds reversed the excessive activation of PI3K-WNK pathway protein in NRK52e cells under hypotonic conditions; compound

Effects of monoterpenes from GJRM on PI3K-WNK pathway in NRK52e cells and IMCD3 cells under hypotonic condition. The expression of PI3K-WNK after incubation with monoterpenes from GJRM in (a and b) NRK52e cells under hypotonic conditions. The expression of PI3K-WNK pathway after incubation with monoterpenes from GJRM in (c and d) IMCD3 cells under hypotonic conditions. The data represent the mean ± standard deviation of 3 independent experiments. * P < 0.05 and ** P < 0.01 vs the NC group. # P < 0.05 and ## P < 0.01 vs the M group. GJRM, Gardenia jasminoides var. radicans Makino; NKCC2, Na-K-2Cl cotransporter; OSR1, oxidative stress-responsive kinase 1; PI3K, phosphoinositide 3-kinase; PKA, protein kinase A; WNK, with-no-lysinekinase.
Effects of Monoterpenes from GJRM on PI3K-WNK Pathway in IMCD3 Cells Under Hypotonic Condition
Monoterpene compounds reversed the excessive activation of PI3K-WNK pathway proteins in IMCD3 cells under hypotonic conditions; compound
Discussion
The WNK pathway has been reported to be a new pathway for the treatment of hypertension. 6,13,19 GJRM and its EAPF reduced the level of WNK1 protein and the levels of WNK4 and OSR1 mRNA in the kidneys of SHR rats, thereby reducing the mRNA and phosphorylation levels of NKCC2 and NCC to promote water and sodium excretion and lower blood pressure.
During hypertension, the PI3K-AKT signaling pathway is significantly suppressed. Activating this pathway could release NO and BK and decrease ET-1 to protect cardiovascular function and reduce blood pressure. 20 We found that the EAPF of GJRM increased NO and BK levels and decreased ET-1 levels in serum (Figure 2a–c). This could be related to the activation of the PI3K-AKT signaling pathway. We further detected the expressions of PI3K, AKT, and PKA in the kidneys of SHR rats. GJRM and its EAPF significantly upregulated the expression of 3 proteins and the mRNA level of AKT to reduce blood pressure (Figure 3).
There are plenty phenolic bioactive compounds in plant foods, such as Pomegranate, Strawberry, Onions, Strawberry tree honey, Rubus ulmifolius Schott, Ocimum basilicum var. purpurascens leaves. 21 -26 Polyphenols are widely known for their antioxidant, anti-inflammatory, anticancer, and antiaging properties. 27 In our preliminary work, a large number of phenolic compounds were extracted from the EAPF of GJRM. Several terpenoids with similar structures were selected for further study. In cell experiments, we found that these monoterpenes inhibited the PI3K-WNK signaling pathway, among which monoterpenes A and D had the greatest effects on similar structures (Figure 6). Interestingly, we found that the mechanism of action in hypoosmotic models was not exactly the same as that in hypertensive animals. The WNK pathway was significantly activated in SHR and hypotonic models. Under hypotonic injury, the PI3K-AKT pathway in NRK52e cells is activated, whereas, in SHR rats, this pathway is suppressed. We speculated that there are other upstream proteins in SHR rats that can activate the WNK pathway. However, this hypothesis requires verification.
Conclusion
Gardenia jasminoides var. radicans Makino and its EAPF may reduce blood pressure by regulating the PI3K-WNK signaling pathway.
Footnotes
Acknowledgments
We thank Dr Sebastian Bachmann and Dr Yan Xu at Department of Anatomy, Charite Universitätsmedizin in Berlin, Germany, for the gift of phosphorylated NKCC2 antibodies.
Author note
Yang Fu and Peipei Yuan are co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Key Research and Development Project (The Major Project for Research of the Modernization of TCM): Key Technology Research for the Characteristic Chinese Medicine Industry Chain of Rehmannia glutinosa (2017YFC1702800); and the Major Science and Technology Projects in Henan Province: Study on the key technology for quality control and the key characteristics of Rehmannia glutinosa, Dioscorea opposita Thunb and Achyranthes bidentata Blume from Henan Province (grant number 171100310500).
