Abstract
Objective
Although many painkillers can be used to relieve the symptoms of osteoarthritis (OA), these drugs do not address joint bone degeneration and muscle wasting, highlighting the need for alternative treatments. This study evaluated the ability of various plant extracts to inhibit joint destruction, focusing on the total glucosides of peony (TGP) obtained from the normal Chinese medication white peony.
Methods
A rabbit OA model was established by anterior cruciate ligament resection (ACLT) and TGP treatment was initiated for 3 months. The morphologies of the skeletal muscle fibers, articular cartilage, and subchondral bone of the knee joint were histologically observed via X-ray and micro-computed tomography scan.
Results
TGP significantly reduced the histopathological changes associated with skeletal muscle atrophy and inflammation, and alleviated cartilage degeneration. In addition, TGP treatment significantly improved the bone density and microstructure of subchondral bone in rabbit knee joints compared with the model group.
Conclusion
The results suggested that TGP treatment may alleviate the progression of osteoarthritis and reduce the destruction of joint cartilage and bone in ACLT.
Introduction
Osteoarthritis (OA) affects joint tissues and is characterized by bone and cartilage degradation, muscle atrophy, and synovial inflammation. It tends to manifest in middle-aged and aged people and is the most frequent kind of disabling arthritis. 1 The incidence of osteoarthritis in China is 3% to 8%, and it has become a major condition causing joint muscle pain and limb disability. 2 Osteopenia and microstructural damage to the trabecular bone are the key factors that accelerate joint cartilage degeneration in osteoarthritis. The subchondral bone is located under the articular cartilage and including a dense cortical bone plate in the top layer and an unfastened trabecular bone in the lower layer. 3 Therefore, the ordinary biomechanical residences of the subchondral bone are necessary to maintain homeostasis in articular cartilage.
Total glucosides of peony (TGP) are energetic compounds extracted from the root of the buttercup plant Peony, a mixture of physiologically effective ingredients such as peony glycosides, hydroxy-paeoniflorin, paeonin, albiflorin, benzoylpaeoniflorin, etc, collectively known as TGP. TGP is mainly composed of peony glycosides, accounting for about 95% of the active ingredients of TGP. 4 TGP has been reported to have therapeutic benefits on joint ache and is widely used to treat joint ache disorders, such as osteoarthritis and rheumatoid arthritis. 5 Previous research has proven the activate blood circulation, relieve pain, and immunomodulatory properties of TGP. 6 The anti-inflammatory effects are attributed to reducing the production of inflammatory factors such as interleukin 5 (IL-5), IL-17, and granulocyte-macrophage colony-stimulating factor in synovial cells. 7 Furthermore, TGP delays joint cartilage degeneration.8,9 Studies have shown that the chondroprotective effects of TGP may be related to the inhibition of fibroblast-like synovial cells producing matrix metalloproteinases and their inhibitors, tissue metalloproteinases.10,11 These research indicate that TGP could delay joint destruction in OA.
Therefore, TGP is hypothesized to have beneficial properties in OA-associated cartilage destruction and subchondral bone osteoporosis. The present study was undertaken to investigate the mechanism through which TGP relieves joint destruction, providing experimental evidence for the application of TGP in OA therapy.
Results and Discussion
Effect of TGP Inhibition on Cartilage Damage
None of the initial body weights of rabbits in the control, TGP, indomethacin (INDO), and normal groups had significant differences. No dead rabbits were observed in any groups, no rabbits were excluded from the study. The tibial plateau cartilage damage in every group is displayed in Figure 1. The tibial plateau articular surface of the normal group was smooth and shiny, with a normal joint capsule structure. In the control group, obvious signs of damage were visible on the cartilage surface of the medial tibial plateau, especially on the weight-bearing articular surface: the superficial cartilage layer of, the subchondral bone was exposed, severe bone hyperplasia was observed at the edge of the tibial plateau, and hypertrophic hyperplasia was seen in the surrounding joint capsule and soft tissue. Moreover, X-rays were taken after fixing the distal tibia, showing no rotation in the femurs of the normal rabbits, while rotation was observed in the femurs of the 3 other groups, which suggested knee instability after anterior cruciate ligament resection (ACLT) of the knee surgery and joint degeneration. Rabbits from the control group exhibited joint-space narrowing in the knees under radiography. However, the TGP group exhibited significant improvement in cartilage damage, with lower joint space loss compared to the control group (Figure 2). Cartilage destruction and femorotibial joint space narrowing were also observed in the INDO group.


Typical X-ray images of knee joint and articular cartilage morphology in every team of rabbits. (a) Normal group; (b) control group; . (c) TGP group and (d) INDO group. Abbreviations: INDO, indomethacin; TGP, total glucosides of peony.
TGP Reduces Inflammation of Skeletal Muscle and Cartilage Injury
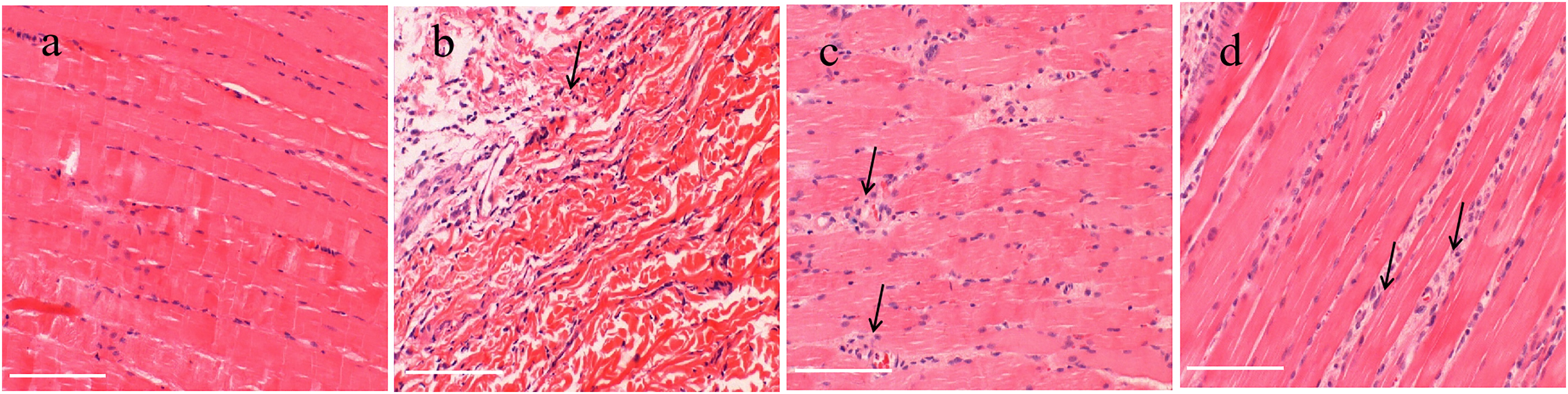
The morphological traits of the rabbit quadriceps muscle fibers and nucleus form were determined after hematoxylin and eosin (H&E) staining. Skeletal muscle from the normal group exhibited a compact structure with uniform gaps between fibers, a linear arrangement of deeply stained nuclei distributed around intact myofibrous cells, and few inflammatory cells. In contrast, the model control group showed varying degrees of inflammatory cell infiltration between fibers, and skeletal muscle fibers showed significant atrophy compared to other groups. However, the TGP group had neatly arranged myofibers, a uniform fibrous interspace, and minimal myofiber atrophy. In addition, a significantly lower number of inflammatory cells was observed around skeletal muscle fibers in the TGP-treated group. Moreover, the control group treated with INDO had a lower number of inflammatory cells than the model group (Figure 3).
Safranin O stromal staining revealed an intact articular chondrocytes layer of the normal group, with a clear structure of cartilage and subchondral bone, showing a regular chondrocyte distribution, uniformly colored matrix, and clear tide line. In the model group, superficial cartilage erosion was observed, accompanied by cracks, and a reduced number of chondrocytes in the injury area. Additionally, the safranin O stromal staining was shallow and the tidemark of cartilage was unclear. In the TGP-treated groups, the cartilage layer was barely uneven with neatly organized chondrocytes; the stromal staining was barely shallower than the regular group, and the tidemark shape was relatively clear (Figure 4). Compared with the normal group, model rabbits exhibited significantly greater Mankin rating (P < .05). Compared with the model group, the Mankin rating was notably decreased in the TGP therapy group (P < .05). However, there was no statistically significant difference in Mankin rating was found between the INDO-treated and model groups (P > .05) (Figure 4e).

TGP Inhibits Articular Bone Destruction
Micro-computed tomography (micro-CT) revealed evident ACLT-induced bone destruction and osteoporosis of subchondral bone in the distal femur (Figure 5a). The bone mineral density (BMD) value in the distal femur was significantly lower in the model group compared with the normal rabbits (P < .05). In contrast, the BMD value of the TGP-treated group was significantly higher than the model rabbits (P < .05; Figure 5b). Furthermore, ACLT treatment substantially lowered trabecular number (Tb.N), trabecular bone volume fraction (BV/TV), and trabecular thickness (Tb.Th) compared to the normal rabbits, whereas trabecular spacing (Tb.Sp) was increased as a result of ACLT-induced bone destruction (P < .05). Significant improvements in trabecular number, volume fraction, and thickness were observed in the TGP-treated group (P < .05), while Tb.Sp was markedly reduced (P < .05; Figure 5c-f). In addition, TGP notably inhibited microstructural osteoporosis of subchondral bone. Nevertheless, INDO had no significant impact on the BMD and microstructure of subchondral bone compared to the model group (P > .05).

Discussion
Through the modern pharmacological study of TGP, the mechanism of action of TGP to alleviate pain, inflammation, and immune disorders has now been clarified. Especially, TGP has been developed as a clinical formulation for the treatment of immune disorders and has been clinically used in China for decades, and its safety and efficacy have been confirmed by clinical studies.7,8 TGP capsules in combination with methotrexate in the treatment of immune disorders have the effect of increasing efficacy and reducing toxicity. 6 Knee osteoarthritis is a chronic progressive joint disorder involving the joint pain, swelling, deformity, muscle atrophy, and synovium, and tends to occur in middle-aged and elderly people; it is the most common kind of disabling arthritis. 12 Typical pathological modifications in OA include articular cartilage damage, abnormal subchondral bone remodeling and muscle loss. 13 Therefore, preventing joint destruction is essential for OA therapy. This study confirmed the inhibitory impact of TGP on cartilage degeneration and subchondral bone destruction.
OA animal models usually consist of post-traumatic and spontaneous OA animal models. Commonly used animal models of spontaneous OA, such as C57 mice, have an 80% incidence of OA at 16 months of age, but lack human OA articular cartilage fibrosis phenotype, and involve long modeling cycles and high cost. 14 ACLT is a frequently used method to establish OA models. 15 Previous studies compared the ACLT-induced OA model with the sham surgery, exhibiting typical cartilage destruction, subchondral osteosclerosis, and osteophyte formation during disease progression. Articular cartilage degeneration in the ACLT surgical model occurred within 14 days of surgery, and detectable cartilage layer degeneration and proteoglycan loss have been found as early as 1 week after surgical treatment. 16 The general morphology and pathological cartilage analysis results in this study showed a smooth tibial plateau articular surface, normal structure, and clear cell level. In contrast, the posterior part of the cartilage of the tibial plateau cartilage in the ACLT model group showed obvious cartilage damage, joint destruction, bone hyperplasia, erosion of the superficial layer of cartilage, and hypertrophic hyperplasia. In the present study, X-rays were used to confirm the narrowing of the joint space in the model rabbits. These results were consistent with previous findings. 17 Furthermore, micro-CT outcomes exhibited subchondral bone destruction in the ACLT model rabbits. The microstructural parameters of the bone were abnormal, with osteopenia and microstructural destruction of the trabecular bones. Therefore, pathological changes in the ACLT model were very similar to human OA, making this model suitable for observational studies of OA pathogenesis and drug efficacy. 18
In our study, TGP-treated group maintained a greater bone mineral density in the subchondral bone compared to model group. These findings are of interest considering that trabeculae of subchondral bone destruction is an early feature of OA. 19 Abnormal modifications in the subchondral bone are essential elements that promote the progress of OA. 20 Subchondral bone and cartilage together form osteochondral units, which are responsible for the bearing and stress dispersion of the joint load and are essential in sustaining the homeostasis of joint cartilage. In the course of OA, microstructural damage and abnormal bone remodeling occur in the subchondral bone, and its mechanical properties are affected, resulting in unusual stress on the cartilage, thereby aggravating cartilage degeneration. Subchondral bone damage might also manifest with cartilage degeneration, or even earlier than cartilage degeneration, suggesting that subchondral bone damage is intimately associated with OA. 21 In the model group, the safranin O stromal staining revealed small cysts were formed below cartilage, marking the destruction of the subchondral bone structure. These findings showed that ACLT accelerated cartilage damage and aggravated the injury to the subchondral bone structure. Subsequently, micro-CT was used to scan and analyze the subchondral bone of the distal femur in each group of samples, demonstrating notable decreases in the microstructure parameters trabecular number, volume fraction, and thickness in the ACLT model rabbits compared to the normal group. The findings indicated a damaged microstructure of the trabecular bone, which may be a key factor in cartilage degeneration.
Skeletal muscle inflammation and muscle atrophy are also important factors in the progression of osteoarthritis. 22 This study found inflammatory dysfunction in quadriceps muscle fibers and varying degrees of inflammatory cell infiltration into the space between fibers in the ACLT model group; skeletal muscle fibers were obviously atrophied compared to other groups. The muscle fibers in the TGP-treated rabbits exhibited much less atrophy, with a significantly lower number of inflammatory cells.23,24 These findings suggested the potential therapeutic value of TGP for muscle inflammation in patients with OA or other synovitis pain.25,26 INDO is a nonsteroidal anti-inflammatory drug commonly used in patients with OA pain and was used as a control drug in this study. 27 Previous research has confirmed that INDO notably inhibits the expression of pro-inflammatory cytokines in the synovial tissue of adjuvant-induced arthritis rats. 28 In this study, although the number of inflammatory cells in the control rabbits was decreased than that in the model rabbits, INDO had no significant impact on bone density and subchondral bone microstructure. In conclusion, the outcomes of the current study indicate that TGP might relieve joint destruction in rabbits with OA.
Materials and Methods
Animals and Treatments
TGP was purchased from Lihua Pharmaceutical Co., Ltd. INDO was used as a positive control medicine. Each pill contained 25 mg of INDO (provided by Meryer Biopharmaceuticals Co. Ltd, China).
A total of 24 12-month-old male New Zealand rabbits, weighing 2 to 2.5 kg, were selected from the Pudong Laboratory Animal Center. The sample size for each group was calculated based on earlier studies, which considered the overall success rate established by the model and used a power calculation with a 2-sided t-test, revealing that ∼5 rabbits were needed per group. 29 Therefore, the trial included a total of 24 rabbits. All rabbits had free access to water and food and were kept at a temperature of 22 °C and 40% humidity, with a 12-h light/dark cycle. Food intake, body weight, and animal fitness were measured daily.
Eighteen rabbits underwent ACLT under general anesthesia. The ACLT model was established as described in a previous work. 29 At the end of the sixth week, a total of 18 animals met the criteria for arthritis activity score. 30 The ACLT model rabbits were randomly divided into 3 groups: (1) control group (control, n = 6); (2) TGP group, treated with TGP by intragastric gavage at a dose of 60 mg/kg body weight per day (TGP, n = 6); (3) INDO group, receiving INDO by gavage at a dose of 3 mg/kg body weight per day (INDO, n = 6). 31 Both dosages provided the equivalent of 9 times the medical dose for a 65 kg adult. Normal rabbits did not receive any treatment and had free access to distilled water and food (normal, n = 6). The number and body weight of rabbits in each group was kept equal to minimize the experimental error.
The experiment subjects underwent treatment for 3 months. Samples were excluded if they exhibited anorexia or rapid weight loss. After 3 months, the rabbits were anesthetized and sacrificed, and knee joint and articular cartilage samples were collected for further analysis. All experiments were approved by the committee for the Animal Experiments Centre, Shanghai, China. Furthermore, all animal experimental protocols have been carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. 32
Articular Cartilage Morphology and Radiographic Analysis
Each rabbit was placed in the prone position and X-ray analysis measurements were performed to determine the bone morphology of the rabbit knee joint. A microradiography system was used for 60 s at 35 kV and 4 mA (Fujifilm, Tokyo, Japan). The collected joint capsule and tibial plateau cartilage samples were then observed.
Histological Evaluation of Cartilage Destruction and Muscle Fibers Atrophy
The quadriceps femoris muscle of the knee joint was wrapped in paraffin and sliced into 4 μm thick sections, then stained with H&E for histopathological contrast. An optical microscope with original magnification ×300 (Telmu XSP-75) was used for observation. The femoral condyles were sliced into 6 μm thick frontal lobe sections for histomorphology. These sections were stained in Safranin O-fast green. The Mankin histological scoring system was used by 2 independent pathologists to assess cartilage destruction, as previously described. 33
Microstructural Analysis of Rabbit Femoral Condyles
The distal femur was prepared for micro-CT examination with the following scan parameters: source voltage 80 kV, source current 280uA, image pixels 18 μm; a Locus SP scanner (GE Healthcare, USA) was used. The trabecular region of the subchondral interosseous compartment of the distal femur was defined as the region of interest. The microstructural features of the trabeculae, such as trabecular thickness, BV/TV (%), Tb.Sp (mm), Tb.N (1/mm), and BMD (mg/cm3), were evaluated.
Statistical Analysis
Continuous variables were presented as the mean and standard deviation. Mean variations between the control and experimental groups were analyzed using a one-way evaluation of variance (SPSS 11.0). Ridit analysis was used in order to rank data. In this study, P < .05 was considered statistically significant.
Footnotes
Authors’ Contributions
FTY performed the experiments; DYS analyzed the data; CCW and LFY performed the experiments and wrote the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this study was obtained from the Medical Ethics Committee of the Animal Care Committee of Shanghai University of Traditional Chinese Medicine (approval no. 2022007117).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Institutional Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine (approval no. 2022007117) approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
