Abstract
We examined the antiosteoarthritis effect of a mixture of powdered pomegranate concentrate, eucommiae cortex, and achyranthis radix (5:4:1 w/w) (PEA-Mix). After the injection of monosodium iodoacetate (MIA), PEA-Mixs were orally administered. To assess pain-related behaviors, a von Frey filament test and open field test were performed. We also examined the knee thickness, maximum knee extension angle, bone mineral density (BMD), and compressive strength of the knee joint and performed a histopathologic analysis. The number of COX-2, tumor necrosis factor (TNF)-α, poly (ADP-ribose) polymerase (PARP), and 5-bromo-2′-deoxyuridine immunoreactive cells, the prostaglandin E2 (PGE2) and 5-lipoxygenase (LPO) levels, matrix metalloproteinase (MMP) activity, and mRNA levels of chondrogenesis-related genes were analyzed. PEA-Mix significantly inhibited the MIA-induced decrease in the paw-withdrawal threshold and total distance moved, and the MIA-induced increases in the maximum knee extension angle and knee thickness. Also, the MIA-induced loss of the knee joint articular surface region and decrease in the BMD were significantly suppressed by PEA-Mix. The MIA-induced increases in the 5-LPO, PGE2, MMP-2, MMP-9, COX-2, and TNF-α mRNA levels were reduced by PEA-Mix. PEA-Mix increased the MIA-mediated reduction in the SOX-9 and aggrecan mRNA levels. The number of PARP-positive cells was smaller in PEA-Mix-administered rats than in MIA-administered rats, while the number of 5-bromo-2′-deoxyuridine-positive cells was larger. Therefore, PEA-Mix relieved the MIA-induced pain-related behaviors, chondrocyte proliferation, and anti-inflammatory activity.
Osteoarthritis (OA) is a disease of the joints involving the destruction of articular cartilage (AC), subchondral bone alterations, synovitis, and pain. 1 The construct capacity of AC is decided by the molecular composition of the extracellular matrix (ECM), a network of collagen and proteoglycans (PGs), aggrecan, and SRY (sex-determining region Y)-box 9 (SOX9). 2,3 Therefore, maintenance of the ECM is important in the homeostasis of AC. Changes in the ECM are triggered by mechanical damage and aging. Deficiency or loss of PGs results in tissue destruction and hypocellularity, eventually leading to joint dysfunction 4 ; also, loss of collagen contributes to cartilage deterioration.
OA is associated with the increased inflammation that occurs with aging. 5 In the pathogenesis of OA, proinflammatory cytokines stimulate cartilage resorption and inhibit the production of new ECM in AC. 6 In addition, interleukin (IL)-1β and tumor necrosis factor (TNF)-α stimulate the production of nitric oxide (NO) by chondrocytes and of prostaglandin E2 (PGE2) by inducing the expression of cyclooxygenase-2 (COX-2). 7 A high level of PGE2 and NO in cartilage influences ECM turnover. 8
OA-induced pain is related to the sensitization of knee nociceptors by peripheral proinflammatory mediators such as TNF-α. 9 OA-induced pain is a major symptom of articular disease 10 and decreases the quality of life of sufferers. Although no effective disease-modifying drug is available, nonsteroidal anti-inflammatory drugs (NSAIDs), analgesics, and hyaluronan are used to relieve the symptoms of OA. However, NSAIDs have a deleterious effect on AC; they suppress the production of PG, which plays an important role in maintaining cartilage homeostasis. 1 Therefore, natural antiarthritic agents that preserve cartilage or mixtures of medicinal foods with anti-OA activity are needed. 2,3
Pomegranates (
We reported previously that PEA-Mix (PCP:AR:EC 5:4:1 w/w) exerted an anti-inflammatory effect and protected the ECM in primary cultures of rat articular chondrocytes. 18 In a subsequent study, we used anterior cruciate ligament transection (ACLT) animals, which had a damaged knee joint. We evaluated the anti-OA effect of PEA-Mix 5:4:1 (w/w) in rats with monosodium iodoacetate (MIA)-induced OA and examined the pain-like behaviors. Unlike the surgically induced OA model, chemically induced OA models exert indirect effects on the knee joint. MIA administration into the AC of rodents decreases the cartilage proteoglycan matrix and induces functional joint impairment, features of human OA. 19 Compared with either spontaneous or surgical models in larger animals, injection of MIA into rats rapidly induces joint damage; therefore, a smaller quantity of inhibitor and a shorter time are required. Therefore, the MIA-induced OA model is suitable for the screening of anti-OA agents. 20
Materials and Methods
Animals and Husbandry
A total of 63 healthy male SPF/VAF outbred CrljOri:Wistar rats (6-week old, 150-200 g upon receipt; OrientBio, Seungnam, Korea) were housed in polycarbonate cages, 5 in each cage at 20°C to 25°C and 45°C to 55% humidity, with a 12:12 hour light:dark cycle. At 7 days, after acclimatization, the rats were divided into 6 groups of 10 rats each based on the standard deviation (SD) of body weight and knee thickness. The rats were housed according to the national regulations on the usage and welfare of laboratory animals, and the study was approved by the Institutional Animal Care and Use Committee of Daegu Haany University.
Preparation and Administration of Test Substances
PCP (ASYA Meyve Suyu ve Gıda San. A.Ş., Ankara, Turkey), EC, and AR (HL Science, Uiwang, Korea) contain 1.15 mg/g ellagic acid, 1.58 mg/g pinoresinol diglucoside, and 0.26 mg/g ecdysterone as active ingredients, respectively. PEA-Mix was prepared by dissolving PCP, EC, and AR in 5 mL of distilled water at 100, 80 and 20 mg (highest doses); 50, 40, and 10 mg (medium doses); and 25, 20, and 5 mg (lowest doses), and orally administered in a volume of 5 mL/kg (equivalent to 200, 100, and 50 mg/kg), using a stainless-steel gastric gavage attached to a 5 mL syringe, once daily for 28 days beginning 1 hour after intra-articular injection of MIA. Diclofenac was dissolved in sterile saline at 0.4 mg/mL and subcutaneously injected into the dorsal back skin at a volume of 5 mL/kg (equivalent to 2 mg/kg).
Induction of OA
The rats were anesthetized with 2%-3% isoflurane (Hana Pharm. Co., Hwasung, Korea) in a mixture of 70% nitrous oxide and 28.5% oxygen, and anesthesia was maintained with 1%-1.5% isoflurane in the same gas mixture using a rodent inhalation anesthesia apparatus (Surgivet, Waukesha, WI, USA) and ventilator (Model 687, Harvard Apparatus, Cambridge, UK). OA was induced by injection of MIA at 3 mg/rat in a volume of 50 µL of sterile saline to the intra-articular space of the left hind limb using a 26 G needle attached to a 1 mL syringe. 20
Pain Behavior Tests
Von Frey filament test
Rats were placed on an elevated wire-mesh floor and confined beneath individual overturned Perspex boxes at 1 hour after administration of the test substance. 20 Mechanical allodynia (g) was measured using a dynamic plantar aesthesiometer system (Ugo Basile SRL, Varese, Italy) and a Von Frey filament (Bioseb, Chaville, France) to apply increasing pressure to the left hind paw. The threshold was determined as the force that induced a withdrawal response, with a 50 g cut-off. Paw-withdrawal thresholds were determined before the initial administration of the test substance (day 0, baseline), and 3, 7, 14, 21, and 28 days thereafter.
Open-field test
The test was performed in a Plexiglass box (40 × 40 × 40 cm) with transparent walls and a black floor. Locomotor activity was measured in the square arena. The behavior was recorded by a video camera mounted on the ceiling, relayed to a monitor, and the total distance moved by the rat was analyzed using a rodent gait analyzer (Catwalk XT, Noldus, Wageningen, Netherlands) with tracking software (EthoVision, Noldus, Wageningen, Netherlands). Spontaneous locomotion was assessed before the initial administration of the test substance (day 0, baseline), and 3, 7, 14, 21, and 28 days thereafter. On each day, a rat was placed in the center of the arena at 1 hour after completing a Von Frey filament test and allowed to explore for 10 minutes.
Measurement of Knee Thickness
Knee thickness was measured using electronic digital calipers (Mytutoyo, Tokyo, Japan) at 1 day before and the day of MIA administration, and 1, 7, 21, 25, 27, and 28 days thereafter. Also, knee thickness after joint capsule exposure was measured after the rats were euthanized to reduce the differences in relation to surrounding tissues. 21 -24
Measurement of the Maximum Knee Extension Angle, Focal Bone Mineral Density, and Compressive Strength
Knees were dissected from the coxofemoral region to the ankle region, leaving the articular capsule intact. Next, the maximum extension angle of the knee was measured according to previous methods,
21,22,24,25
with 0° corresponding to the maximum possible extension. To minimize bias, measurements of knee extension were performed by the same veterinarian. The mean focal bone mineral density (BMD) of the total knee joint, femur, and tibia articular surface of the rats was determined after euthanasia by
Preparation of the Femur and Tibia AC with Synovial Membrane Tissue Homogenates
Portions of the femur and tibia AC with synovial membrane (SM) were separated and homogenized using a bead beater (Model Taco Pre, GeneResearch Biotechnology Corp., Taichung, Taiwan) and ultrasonic cell disruptor (Model KS-750, Madell Technology Corp., Ontario, CA, USA) in radioimmunoprecipitation assay buffer (Sigma-Aldrich, St. Louise, MO, USA). Supernatants were separated by centrifugation at 15 000 rpm at 4°C for measurement of the PGE2 level and 5-lipoxygenase (5-LPO), matrix metalloproteinase (MMP)-2, and MMP-9 activity. The tissue homogenates were stored in an ultralow-temperature freezer (Model MDF-1156, Sanyo, Tokyo, Japan) at −150°C until analysis.
Determination of the PGE2 Level and 5-LPO and MMP Activity
The PGE2 level, 5-LPO, MMP-2, and MMP-9 activity in supernatants of femur and tibia AC with SM tissue homogenates were assayed using a commercial PGE2 assay kit (ParameterTM, R&D Systems, Minneapolis, MN, USA), lipoxygenase inhibitor screening assay kit (Cayman Chemical, Ann Arbor, MI, USA), and MMP enzyme-linked immunosorbent assay kit (MBS494797 for MMP-2 and MBS722532 for MMP-9, Mybiosource, San Diego, CA, USA) with a microplate reader (Sunrise; Tecan, Männedorf, Switzerland), according to the manufacturer’s instructions.
Real-Time Reverse Transcriptase-Polymerase Chain Reaction Analysis of the mRNA Levels of ECM-Related Genes
Briefly, RNA was isolated using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). The RNA concentration and quality were determined using the CFX96 Real-Time System. RNA was reverse transcribed using the high-capacity cDNA reverse transcription kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s instructions. cDNA was amplified by polymerase chain reaction at 58°C for 30 minutes, 94°C for 2 minutes, 35 cycles of 94°C for 15 seconds, 60°C for 30 seconds, 68°C for 1 minutes, and 72°C for 5 minutes using the ABI Step One Plus Sequence Detection System (Applied Biosystems, Foster City, CA, USA). The mRNA levels were calculated relative to the vehicle control using the 2−ΔΔCq method. 26 Primer sequences for rat genes are as follows: Collagen type II, 5′-GAGTGGAAGAGCGGAGACTACTG-3′ and 5′-CTCCATGTTGCAGAAGACTTTCA-3′; SOX-9, 5′- AGAGCGTTGCTCGGAACTGT-3′ and 5′-TCCTGGACCGAAACTGGTAAA-3′; Aggrecan, 5′-GATGTCCCCTGCAATTACCA-3′, and 5′-TCTGTGCAAGTGATTCGAGG-3′; β-actin, 5′-ATCGTGGGCCGCCCTAGGCA-3′, and 5′-TGGCCTTAGGGTTCAGAGGGG-3′. The data were normalized by β-actin mRNA expression, using a comparative threshold cycle method. 27
5-Bromo-2′-Deoxyuridine Uptake Assay
To assay cell proliferation in the knee joints, proliferating cells were labeled by intraperitoneal injection of 5-bromo-2′-deoxyuridine (BrdU) (Sigma-Aldrich, St Louis, MO, USA) at 50 mg/kg in a volume of 2 mL/kg in saline, using a 26 G needle attached to a 5 mL syringe. The rats were euthanized 72 hours later. BrdU uptake in the femur and tibia AC was determined by immunohistochemistry using an anti-BrdU antibody, and in SM homogenates, as described previously. 21,22,24
Histological Analysis
Knee joints were sampled while preserving the joint capsule, fixed in 10% neutral buffered formalin, and decalcified in the decalcifying solution for 5 days. Each knee joint was longitudinally trimmed. After paraffin embedding using an automated tissue processor (Thermo Scientific, Waltham, MA, USA) and an embedding center (Thermo Scientific, Waltham, MA, USA), 3-4 µm serial sections were prepared using a microtome (Leica Biosystems, Nussloch, Germany). Representative sections were stained with Safranin O for cartilaginous tissues. 21,22,24
Immunohistochemistry
Briefly, endogenous peroxidase activity was blocked in methanol and 0.3% hydrogen peroxide for 30 minutes, and non-specific binding of immunoglobulin was blocked using normal horse serum for 1 hour in a humidity chamber after epitope retrieval by pretreatment with trypsin (Sigma-Aldrich, St Louise, MO, USA) and 2 N hydrochloric acid. The sections were treated with the primary antisera overnight at 4°C in a humidity chamber, followed by the biotinylated universal secondary antibody and ABC reagents for 1 hour at room temperature in a humidity chamber. Finally, sections were reacted with peroxidase substrate for 3 minutes at room temperature. All sections were rinsed 3 times in 0.01 M phosphate-buffered saline between the steps.
Histomorphometry
The thickness of the tibia and femur AC and the epithelial lining of the SM and the number of inflammatory cells in the SM and osteophytes in the AC were measured by histomorphometric analysis of longitudinally trimmed samples, using a computer-based image analyzer (
Statistical Analyses
Values are means ± SD of 10 rats. If there were no significant deviation from variance homogeneity, the data were subjected to one-way analysis of variance, followed by the least-significant difference multicomparison test to identify significant differences. If significant deviations from variance homogeneity were detected, the non-parametric Kruskal–Wallis H-test was used. If this indicated a significant difference, the Mann–Whitney U-test with Bonferroni’s correction was conducted to identify significant differences. Statistical analyses were conducted using SPSS for Windows software (Release 14.0K, SPSS Inc., Chicago, IL, USA); a value of
Results
PEA-Mix Inhibited the Increase in Knee Thickness
Compared with intact vehicle control rats, the MIA control rats showed significantly greater knee thickness from 1 day after intra-articular injection of MIA. In contrast, PEA-Mix 200, 100, and 50 mg/kg decreased the MIA-induced increase in knee thickness from 14 days after initial administration. Diclofenac 2 mg/kg also decreased the MIA-induced increase in knee thicknesses from 7 days after initial administration (Figure 1(a)).

Knee thickness changes (a), Von Frey filament test (b), and open-field test (c) in either intact or OA rats. Significant increases of induced knee thicknesses (a), significant decreases of paw-withdrawal threshold (b), and total distance moved (c) were demonstrated in MIA control rats after single intra-articular injection of MIA as compared with intact vehicle control (arrow). However, significant changes of knee thicknesses (a), paw-withdrawal threshold (b), and total distance moved (c) were demonstrated in diclofenac treated rats and/or in PEA-Mix administered rats, as compared with MIA control rats, respectively (dot arrows). Values are expressed mean ± standard deviation of 10 rats, mm. Day 1 means at 1 day before MIA intra-articular injection and first test article administration. Day 0 means at MIA intra-articular injection and first test article administration. Day 28 means at 24 hours after last 28th test article administration, at sacrifice. All animals were overnight fasted before first administration and sacrifice, respectively (†). a
PEA-Mix Inhibited the Decrease in the Paw-Withdrawal Threshold and Total Distance Moved
Compared with intact vehicle control rats, MIA control rats showed a decreased paw-withdrawal threshold (g) from 3 days after intra-articular injection of MIA. In contrast, PEA-Mix resulted in a dose-dependent increase in the paw-withdrawal threshold from 7 days after initial administration (Figure 1(b)). In an open-field test, MIA-induced rats showed a significant decrease in the total distance moved, while PEA-Mix 200, 100, and 50 mg/kg increased the total distance moved in a dose-dependent manner (Figure 1(c)).
PEA-Mix Inhibited the Increase in the Capsule-Exposed Knee Thicknesses and Maximum Knee-Extension Angle
MIA significantly increased the capsule-exposed knee thicknesses and maximum knee-extension angle, while PEA-Mix 200, 100, and 50 mg/kg significantly inhibited this increment in a dose-dependent manner (Figure 2(a) and (b)).

Knee thickness after capsule exposure (a), knee maximum extension angles (b), and focal CS of the femur (c) and tibia (d) AC in either intact or OA rats. Values are expressed as mean ± standard deviation of 10 rats. a
PEA-Mix Inhibited the Decreased Femur and Tibia AC Focal CS
Compared with intact vehicle control rats, MIA decreased the femur (Figure 2(c)) and tibia (Figure 2(d)) AC focal CS. PEA-Mix 200, 100, and 50 mg/kg increased the femur and tibia AC focal CS in a dose-dependent manner.
PEA-Mix Inhibited the Decrease in the BMD of the Knee Joint
MIA induced loss of the knee joint articular surface region: the femur and tibia AC erosion with osteophyte formations. In addition, MIA decreased the focal BMD of the total knee joint and femur and tibia articular surface regions. However, PEA-Mix 200, 100, and 50 mg/kg dramatically reversed the loss of the knee joint articular surface region and the decreased total knee joint and femur and tibia articular surface BMD in a dose-dependent manner (Table 1; Figure 3).

Representative dual-energy X-ray absorptiometry images of the knee joints, taken from either intact or OA rats. (a) intact vehicle control (saline intra-articular injection and vehicle-administered rats); (b) MIA control (MIA single intra-articular injection and vehicle-administered rats); (c) diclofenac (MIA treatment and diclofenac sodium 2 mg/kg subcutaneously treated rats); (d) PEA-Mix200 (MIA treatment and PEA-Mix totalized 200 mg/kg administered rats); (e) PEA-Mix100 (MIA treatment and PEA-Mix totalized 100 mg/kg administered rats); (f) PEA-Mix50 (MIA treatment and PEA-Mix totalized 50 mg/kg administered rats). AC, articular cartilage; AR, achyranthis radix; EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula.
Focal BMD on the Total Knee Joint, Femur, and Tibia Articular Regions in Intact or OA Rats.

AR, achyranthis radix; BMD, bone mineral density.EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula.
Values are expressed mean ± standard deviation of 10 rats, g/cm2.
a
b
PEA-Mix Reversed the Increased PGE2 Level and 5-LPO Activity
We evaluated the PGE2 level and 5-LPO activity as a marker of inflammation. MIA-induced rats caused increases in the PGE2 levels and 5-LPO activity in the femur and tibia AC with SM, while MIA induced rats with PEA-Mix 200, 100, and 50 mg/kg decreased the PGE2 levels in the femur and tibia AC with SM. PEA-Mix exerted a dose-dependent inhibitory effect on the MIA-induced increase in the PGE2 level in the femur and tibia AC with SM (Table 2).
Femur and Tibia AC with SM PGE2 Contents and 5-LPO Activities in Intact or OA Rats.

AC, articular cartilage; AR, achyranthis radix; EC, eucommiae cortex; LPO, lipoxygenase; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; PGE2 , prostaglandin E2; SM, synovial membrane.
Values are expressed mean ± standard deviation of 10 rats, pg/mL.
a
b
PEA-Mix Inhibited the Increased MMP-2 and MMP-9 Activities
MIA increased MMP-2 and MMP-9 activities in the femur and tibia AC with SM, while PEA-Mix 200, 100, and 50 mg/kg inhibited the increased MMP-2 and MMP-9 activity in a dose-dependent manner. The effect of PEA-Mix 50 mg/kg was comparable with that of diclofenac 2 mg/kg (Table 3).
Femur and Tibia AC with SM MMP-2 and MMP-9 Activities in Intact or OA Rats.

AC, articular cartilage; AR, achyranthis radix; EC, eucommiae cortex; MIA, mono sodium iodoacetate; MMP, matrix metalloproteinase. OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix , PCP:EC:AR 5:4:1 w/w mixed formula; SM, synovial membrane.
Values are expressed mean ± standard deviation of 10 rats.
a
b
c
PEA-Mix Reversed the Decreased Collagen II, SOX9, and Aggrecan Levels in the Femur and Tibia AC
The collagen type II, SOX9, and aggrecan mRNA levels in the femur and tibia AC were significantly lower in MIA control rats compared with intact vehicle control rats, while the SOX9 and aggrecan mRNA levels in the SM were significantly increased. PEA-Mix significantly increased the collagen type II, SOX9, and aggrecan mRNA levels, and significantly decreased those of SOX9 and aggrecan, in a dose-dependent manner (Table 4).
ECM Related Chondrogenic Gene mRNA Expressions on the Femur and Tibia AC with SM in Intact or OA Rats.

AC, articular cartilage; AR, achyranthis radix; EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; SM, synovial membrane; SOX9, SRY (sex-determining region Y)-box 9.
Values are expressed mean ± standard deviatio of 10 rats, relative to control/β-actin.
a
b
PEA-Mix Reversed the Increased Number of BrdU-Immunolabeled Cells
Compared with intact vehicle control rats, the MIA control rats showed a decreased number of BrdU-immunopositive cells in the femur and tibia AC and an increased number of SM BrdU-immunolabeled cells. PEA-Mix 200, 100, and 50 mg/kg reversed the increase in the number of BrdU-immunopositive cells and the decreased number of BrdU-immunolabeled cells in a dose-dependent manner (Table 5; Figure 4).

Representative immunohistochemistrical images of the BrdU-immunoreactive cells on the femur and tibia AC with SM, taken from either intact or OA rats. (a) intact vehicle control (saline intra-articular injection and vehicle-administered rats); (b) MIA control (MIA single intra-articular injection and vehicle-administered rats); (c) diclofenac (MIA treatment and diclofenac sodium 2 mg/kg subcutaneously treated rats); (d) PEA-Mix200 (MIA treatment and PEA-Mix totalized 200 mg/kg administered rats); (e) PEA-Mix100 (MIA treatment and PEA-Mix totalized 100 mg/kg administered rats); (f) PEA-Mix50 (MIA treatment and PEA-Mix totalized 50 mg/kg administered rats). All ABC immunostain. Scale bars = 120 µm. ABC, avidin–biotin complex; AC, articular cartilage; AR, achyranthis radix; BrdU, 5-bromo-2′-deoxyuridine; CS, compressive strength; EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; TNF, tumor necrosis factor.
Femur and Tibia AC with SM BrdU-Immunoreactive Cell Numbers in Intact or OA Rats.
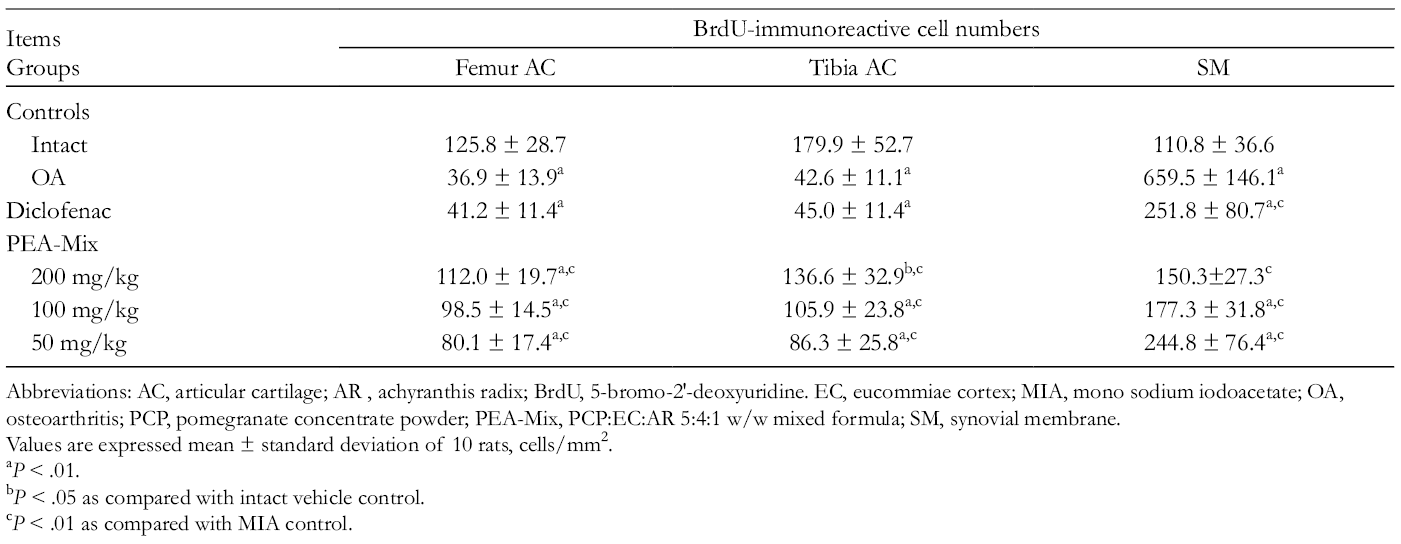
AC, articular cartilage; AR , achyranthis radix; BrdU, 5-bromo-2′-deoxyuridine. EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; SM, synovial membrane.
Values are expressed mean ± standard deviation of 10 rats, cells/mm2.
a
b
c
PEA-Mix Reversed the Increased Mankin Score in the Femur and Tibia AC
MIA increased damage to the cartilage surface and decreased the number of chondrocytes and osteophytes. The intensity of Safranin O staining of the femur and tibia AC was decreased by MIA, and so the Mankin scores of the femur and tibia AC were increased, compared with intact vehicle control rats. PEA-Mix significantly decreased the Mankin scores of the femur and tibia AC compared with MIA control rats in a dose-dependent manner (Table 6; Figure 5).

Representative general histological images of the femur and tibia AC with SM, taken from intact or OA rats. (a)Intact vehicle control (saline intra-articular injection and vehicle-administered rats); (b) MIA control (MIA single intra-articular injection and vehicle-administered rats); (c) diclofenac (MIA treatment and diclofenac sodium 2 mg/kg subcutaneously treated rats); (d) PEA-Mix200 (MIA treatment and PEA-Mix totalized 200 mg/kg administered rats); (e) PEA-Mix100 (MIA treatment and PEA-Mix totalized 100 mg/kg administered rats); (f) PEA-Mix50 (MIA treatment and PEA-Mix totalized 50 mg/kg administered rats). Arrows indicated the thicknesses of tibia or femur AC or of SM lining epithelium. All Safranin O stain. Scale bars = 160 µm. ABC, avidin–biotin complex; AC, articular cartilage; AR, achyranthis radix; CS, compressive strength; EC, eucommiae cortex; EP, epithelium; Gp, growth plate; MIA, mono sodium iodoacetate; OA, osteoarthritis; OS, osteophyte; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; TNF, tumor necrosis factor.
Femur and Tibia AC Mankin Scores in Intact or OA Rats.

AR, achyranthis radix; EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula.
Values are expressed mean ± standard deviation of 10 rats, score.
a
b
c
dMax of totalized scores = 12.
PEA-Mix Inhibited the Decreased Femur and Tibia AC Thickness
MIA significantly decreased the thicknesses of the femur and tibia AC and increased the thickness of the epithelial lining of the SM. Also, the number of infiltrating inflammatory cells in the SM and osteophytes in the AC were increased by MIA. PEA-Mix 200, 100, and 50 mg/kg inhibited the decreases in the femur and tibia AC thickness, the increase in the thickness of the epithelial lining of the SM, and the number of infiltrating inflammatory cells in the SM and of osteophytes in the AC (Table 7; Figure 5).
Femur and Tibia AC with SM General Histomorphometrical Analysis and Immunolabeled Cell Numbers in Intact or OA Rats.

AC, articular cartilage; AR, achyranthis radix; COX, cyclooxygenase; IF, inflammatory; EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; OS, osteophytes; PARP, poly(ADP-ribose) polymerase; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; SM, synovial membrane; Th, thickness; TNF, tumor necrosis factor.
Values are expressed mean ± standard deviation of 10 rats.x
a
b
c
dNot detected.
PEA-Mix Reversed the Increased Numbers of SM PARP, COX-2, and TNF-α Immunolabeled Cells in the Femur and Tibia AC
MIA significantly increased the number of PARP, COX-2 and TNF-α immunopositive cells in the femur and tibia AC. PEA-Mix 200, 100, and 50 mg/kg inhibited the increases in the number of PARP, COX-2, and TNF-α immunopositive cells in the femur and tibia AC in a dose-dependent manner (Table 7; Figures 6 -8).

Representative immunohistochemistrical images of the PARP-immunoreactive cells on the femur and tibia AC with SM, taken from either intact or OA rats. (a) Intact vehicle control (saline intra-articular injection and vehicle-administered rats); (b) MIA control (MIA single intra-articular injection and vehicle-administered rats); (c) diclofenac (MIA treatment and diclofenac sodium 2 mg/kg subcutaneously treated rats); (d) PEA-Mix200 (MIA treatment and PEA-Mix totalized 200 mg/kg administered rats); (E) PEA-Mix100 (MIA treatment and PEA-Mix totalized 100 mg/kg administered rats); (f) PEA-Mix50 (MIA treatment and PEA-Mix totalized 50 mg/kg administered rats). All ABC immunostain. Scale bars = 120 µm.ABC, avidin–biotin complex; AC, articular cartilage; AR, achyranthis radix; CS, compressive strength; EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PARP, poly(ADP-ribose) polymerase; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula.
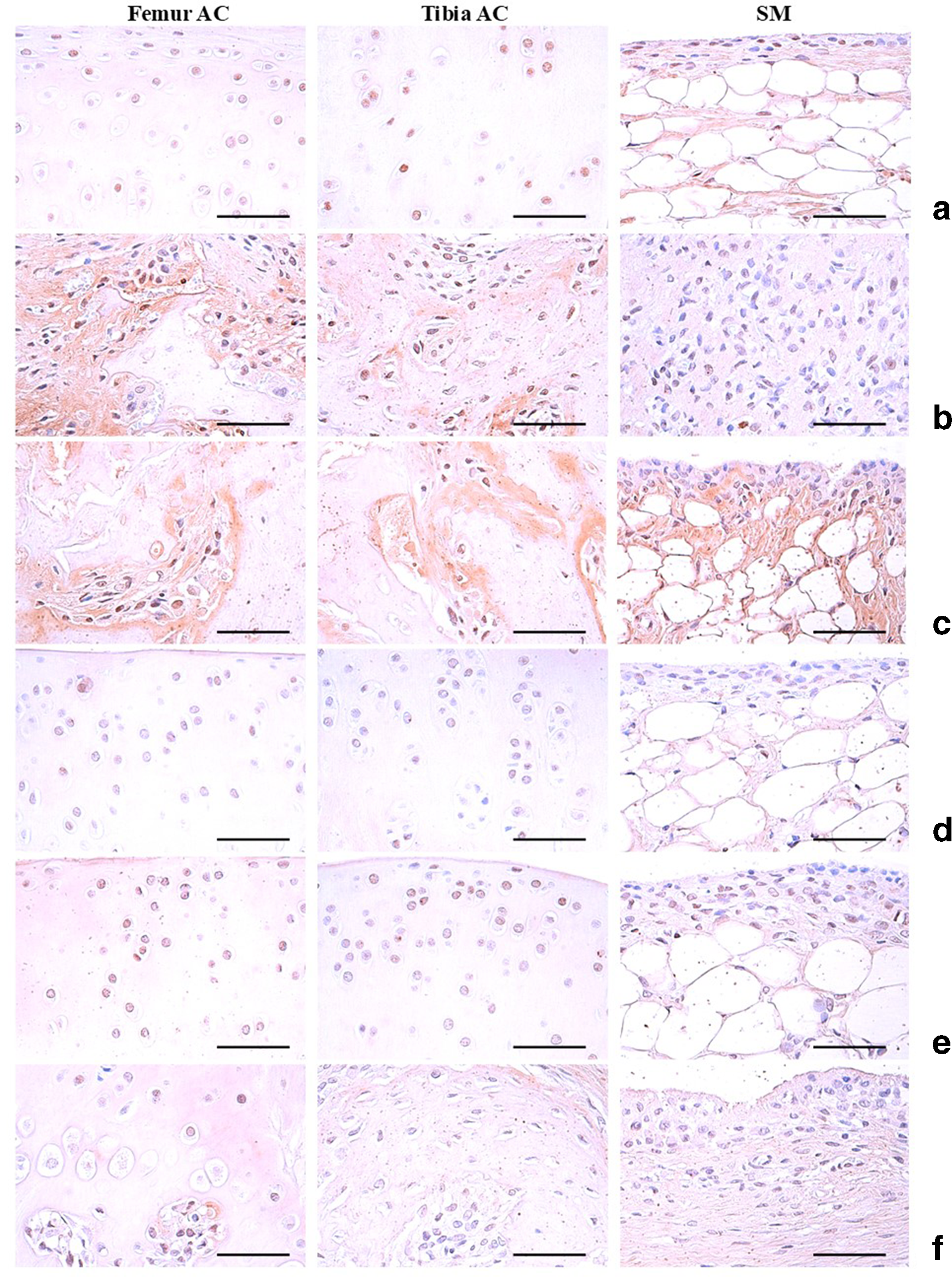
Representative immunohistochemistrical images of the COX-2-immunoreactive cells on the femur and tibia AC with SM, taken from either intact or OA rats. (a) Intact vehicle control (saline intra-articular injection and vehicle-administered rats); (b) MIA control (MIA single intra-articular injection and vehicle-administered rats); (c) diclofenac (MIA treatment and diclofenac sodium 2 mg/kg subcutaneously treated rats); (d) PEA-Mix200 (MIA treatment and PEA-Mix totalized 200 mg/kg administered rats); (e) PEA-Mix100 (MIA treatment and PEA-Mix totalized 100 mg/kg administered rats); (f) PEA-Mix50 (MIA treatment and PEA-Mix totalized 50 mg/kg administered rats). All ABC immunostain. Scale bars = 120 µm. ABC, avidin–biotin complex; AC, articular cartilage; AR, achyranthis radix; CS, compressive strength; COX, cyclooxygenase; EC, eucommiae cortex; MIA, mono sodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; SM, synovial membrane.
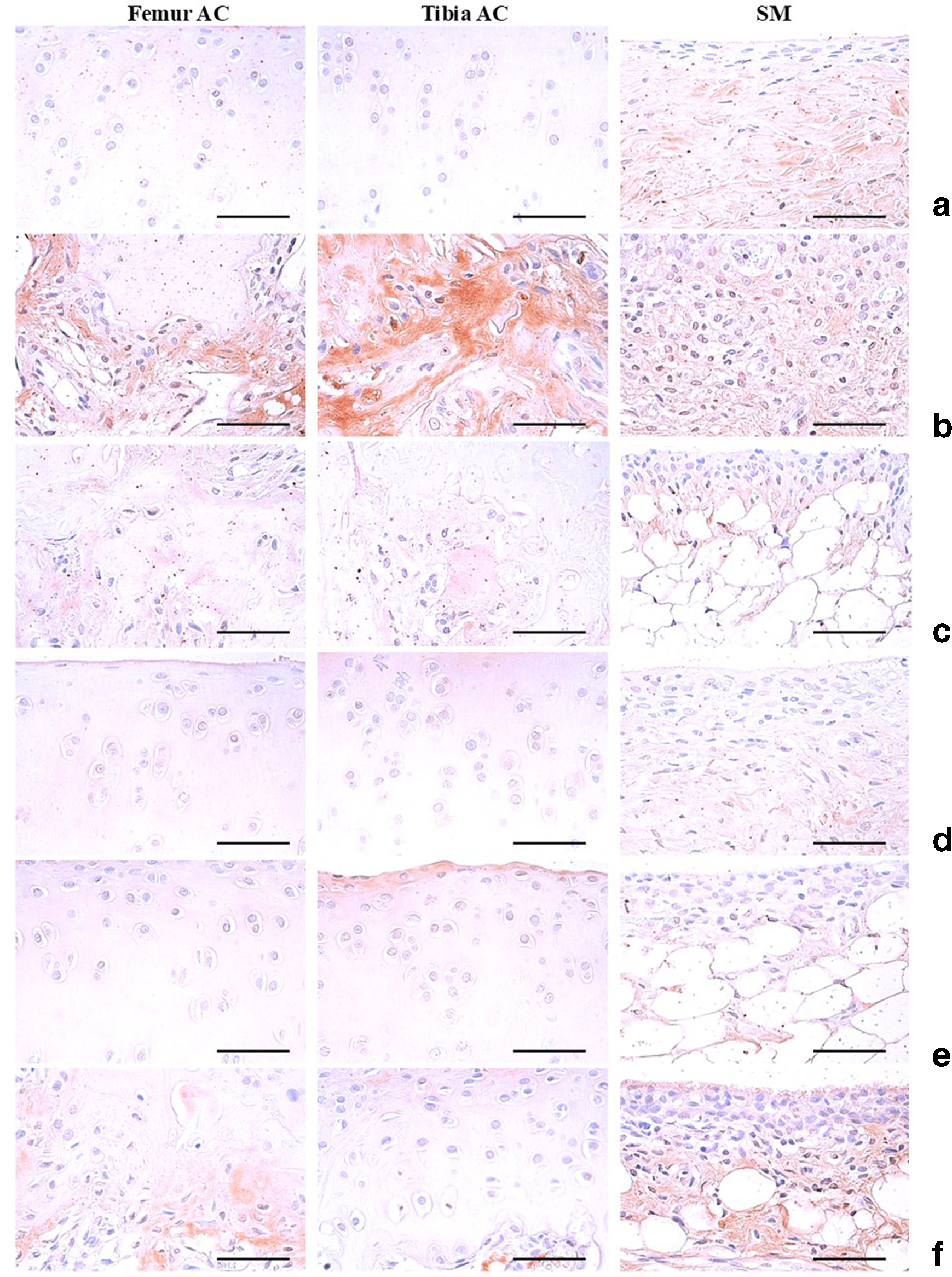
Representative immunohistochemistrical images of the TNF-α immunoreactive cells on the femur and tibia AC with SM, taken from intact or OA rats. (a) Intact vehicle control (saline intra-articular injection and vehicle-administered rats); (b) MIA control (MIA single intra-articular injection and vehicle administered rats); (c) diclofenac (MIA treatment and diclofenac sodium 2 mg/kg subcutaneously treated rats); (d) PEA-Mix200 (MIA treatment and PEA-Mix totalized 200 mg/kg administered rats); (e) PEA-Mix100 (MIA treatment and PEA-Mix totalized 100 mg/kg administered rats); (f) PEA-Mix50 (MIA treatment and PEA-Mix totalized 50 mg/kg administered rats). All ABC immunostain. Scale bars = 120 µm. ABC, avidin–biotin complex; AC, articular cartilage; AR, achyranthis radix; CS, compressive strength; EC, eucommiae cortex; MIA, monosodium iodoacetate; OA, osteoarthritis; PCP, pomegranate concentrate powder; PEA-Mix, PCP:EC:AR 5:4:1 w/w mixed formula; SM, synovial membrane; TNF, tumor necrosis factor.
Discussion
PEA-Mix 200, 100, and 50 mg/kg ameliorated MIA induced OA, and the effect was comparable with that of PCP, EC, and AR and diclofenac sodium 2 mg/kg. In rats with ACLT OA, the anti-inflammatory and chondrogenic activities of PEA-Mix synergistically enhanced the anti-OA effect of PCP, EC, and AR. 28 However, ACLT OA does not accurately reflect the pathogenesis of OA. MIA causes inflammation and joint destruction, while ACLT directly induces joint destruction and then synovial inflammation; both mimic OA. However, the pain and behavioral changes are induced by different mechanisms. 29 -31
MIA resulted in a significant decrease in the paw-withdrawal threshold and the total distance moved, and these effects were significantly reversed by PEA-Mix. Therefore, PEA-Mix relieved MIA-induced pain-related behaviors. Chemical-OA and surgery-OA models may induce different degrees of pain over time. Multiple methods have been used to analyze pain behaviors 32 ; the Von Frey filament test and open field test are generally used to assess these. At the later stages of MIA-induced OA, the damage to exposed subchondral bone and synovia resembles human OA and is related to joint pain. 20,33 A decreased paw-withdrawal threshold indicates increased mechanical allodynia, 20 and a decreased total distance moved in an open field test indicates pain-related loss of spontaneous locomotor activity. 20
MIA injection caused an increase in knee thicknesses, a decrease in the femur and tibia AC thickness, and significant increases in the number of COX-2 and TNF-α immunolabeled cells. The MIA control rats showed markedly elevated PGE2 level and 5-LPO activity in the femur and tibia AC with SM, respectively. However, these effects were significantly reversed by PEA-Mix. Therefore, PEA-Mix relieved the symptoms of OA by exerting an anti-inflammatory effect. Inflammation is an important factor in the pathogenesis of OA. Nam et al reported that the expression of proinflammatory genes is activated by MIA-induced cartilage damage. 34 Mechanical stress and age-related wear/tear stimulate the production of inflammatory mediators such as PGE2 and leukotriene by increasing the expression of COX-2 and 5-LPO. The resulting acute inflammation induces edema in surrounding tissues, increases the joint thickness, and triggers the formation of abnormal osteophytes. 34 Additionally, in patients with OA, the proinflammatory cytokines IL-1β and TNF-α promote ECM degradation by triggering the release of proteases such as MMPs. 35
The mRNA levels of collagen type II, aggrecan, and SOX9 in the femur and tibia of MIA control rats were significantly lower than those of intact vehicle control rats, and the mRNA levels of aggrecan and SOX9 were increased in SMs. The mRNA level of collagen type II in the SM was significantly increased, suggesting focal fibrosis and stiffness, classic signs of OA. 21 -25 PEA-Mix significantly inhibited these effects. The ECM of AC is principally composed of collagen and GAGs, PG, aggrecan, and SOX9. 2,3,36 Aggrecan and collagen are abundant in healthy cartilage. 36 The transcription factor SOX9 plays a pivotal role in chondrogenic differentiation. 37 Haag et al reported that SOX9 expression is downregulated in terminally damaged cartilage from patients with OA. 38 ECM preservation is important for the maintenance of AC. If ECM homeostasis breaks down, the synthesis of ECM components is inhibited, resulting in tissue destruction and hypocellularity, leading to functional joint impairments. 4 The degradation of collagen, aggrecan, and SOX9, due to the inflammation-mediated death of chondrocytes and damage, is involved in the deterioration of the homeostasis and functioning of AC in OA. 39 Therefore, preservation of ECM components should be a goal of OA treatment. 36
The number of BrdU-positive cells was decreased and increased in the femur and tibia AC and SM, respectively, of MIA control rats, in comparison with intact vehicle control rats. These effects were reversed by PEA-Mix. Therefore, PEA-Mix enhanced OA relief activities through AC proliferating activities. BrdU staining has been used to assay cell proliferation in models of OA. 21 -24 Chondrocytes proliferate in response to mechanical trauma or pathological damage, and their regenerative capacity decreases with age. 40 In patients with OA, loss of chondrocytes is a feature of cartilage degeneration, as are hyperplasia and fibrotic changes in the SM. Thus, increased chondrocyte proliferation in the femur and tibia AC indicates amelioration of OA. Some anti-OA agents, for example, lubricin, inhibit the overgrowth of synovial cells. 41
The number of PARP-immunoreactive cells in the femur and tibia AC with SM was increased in the MIA control rats and was significantly reversed by PEA-Mix 200, 100, and 50 mg/kg. Therefore, PEA-Mix ameliorated OA by inhibiting apoptosis. During apoptosis, cytochrome
The MIA-induced focal loss of AC and osteophytes and the decreased BMD and CS in knee joints were significantly inhibited by PEA-Mix in a dose-dependent manner. Therefore, PEA-Mix ameliorated OA by preserving the AC. During the development of OA, physical injury can damage the joint surface, leading to cartilage erosion and osteophyte formation. 48 Suppression of cartilage damage, detected on the roentgenographic survey, is a marker of the effectiveness of anti-OA agents. 48 BMD is used to assess bone quality in clinical research. 49,50 In addition, the focal BMD of the joint surface is used to assess the progression of OA and the efficacy of drugs targeted at bone tissue. 51 We found that the AC CSs decreased markedly in rats with OA.
MIA also increased the maximum knee-extension angle and can induce hypertrophic and hyperplasia in the SM. These changes were significantly relieved by PEA-Mix. The effect of PEA-Mix 50 mg/kg was comparable with that of diclofenac 2 mg/kg. Therefore, PEA-Mix alleviated OA-related joint stiffness. In patients with OA, chronic inflammation leads to fibrosis, which limits joint motion and decreases the quality of life. Thus, the evaluation of joint stiffness is useful for assessing the severity of OA. Maximum extension is 0°; the larger the value, the worse the knee function. 21 -25
MIA increased the femur and tibia AC Mankin scores, while PEA-Mix significantly reduced the AC thickness. Therefore, PEA-Mix ameliorated OA by preserving the AC. The AC thickness is decreased in animal models of OA. 21,22,24 MIA disrupts glycolysis in chondrocytes, leading to cartilage degeneration. The histological changes in the degenerating joint resemble those in human OA. 52 The Mankin score encompasses cartilage surface damage, chondrocyte number, clone/osteophyte formation, and the intensity of Safranin O staining of PG in cartilage. The lower the Mankin score, the less the severity of OA. 21 -25
In conclusion, the features of MIA-induced OA (disruption of the femur and tibia AC, inflammation, fibrosis of the SM, knee stiffness, and pain) were significantly ameliorated by PEA-Mix. The effect of PEA-Mix 50 mg/kg was similar to that of diclofenac sodium 2 mg/kg. Unlike diclofenac sodium 2 mg/kg, PEA-Mix dose-dependently inhibited the proliferation of chondrocytes in the femur and tibia AC. Therefore, PEA-Mix ameliorated OA not only by exerting an anti-inflammatory effect and preserving the AC but also through AC proliferating and related ECM production and secretion activities. Thus, PEA-Mix shows promise for the treatment of OA, and its efficacy should be evaluated in preclinical and clinical trials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea grant funded by the Korea government (MSIT) (No.2018R1A5A2025272).
