Abstract
Proteoglycan (PG) is a type of glycoprotein which forms an extracellular matrix with collagen and hyaluronic acid to maintain articular cartilage, synovial membrane, and synovial fluid. This study aimed to evaluate the antiosteoarthritis effects of salmon nasal cartilage-derived PG in alleviating knee osteoarthritis in an osteoarthritis rat model. Knee osteoarthritis was induced in rats by intra-articular injection of monosodium iodoacetate (MIA), 3 mg/knee, to the right knee. Animals were then administered either diclofenac (3 mg/kg body weight [b.w]/day) or proteoglycan F (PGF; 40 mg/kg and 120 mg/kg b.w/day) by oral gavage for 6 consecutive weeks. Knee diameters were measured throughout the experimental period; serum interleukin-1β and tumor necrosis factor-alpha (TNF-α) levels, and histological analysis of the ligament were carried out at the end of the experiment. Salmon cartilage PG considerably alleviated the osteoarthritis symptoms in the model and lowered the serum concentrations of interleukin-1β and TNF-α. Diclofenac 3 mg/kg/day and PGF at doses of 40 mg/kg/day and 120 mg/kg/day also improved articular cartilage structure on further histological studies. This study demonstrated the in vivo effect of salmon cartilage PG in attenuating symptoms in an MIA-induced rat model, including reduction of inflammatory markers and histological improvement of cartilage tissue.
Osteoarthritis (OA) is a multifactorial, chronic disease of the joint that damages and ultimately lead to deformation and destruction of the affected joints. 1 Though the pathogenesis of OA remains unclear, it is often linked to aging as degeneration of cartilage protein components and bone remodeling occurs in response to physical wear and tear on the affected joints. 2 The degeneration of cartilage structure can be primary, due to a combination of genetic makeup and lifestyle, or secondary due to physical trauma and metabolic/endocrine disorders. 1 OA is one of the main causes of loss of ambulation, disability, and lower quality of life, especially in the elderly population. 3 There are different approaches to the management of OA, from physiotherapeutic measures and drugs, such as nonsteroidal anti-inflammatory drugs to alleviate symptoms, to joint replacement surgery for unmanageable cases. 4 These options, however, often incur side effects, and surgery is not always an option for the patient. Therefore, alternative therapy and complementary supplements to help restructure and modulate the immune system to respond to OA are being investigated in depth.
Proteoglycans (PGs) are important structural components that are largely present in connective tissues, especially in the cartilage cellular matrix where they form complexes with collagen, fibronectin, hyaluronic acid, and other glycoproteins to maintain cartilage integrity. Studies have demonstrated that the immunomodulatory and antirheumatoarthritis effects of salmon nasal cartilage in vitro and in vivo. 5 Most notably, a clinical study was conducted to investigate the safety and efficiency of inflammatory markers of individuals with knee-joint discomfort, the results of which suggested an improvement of cartilage metabolism with minimal adverse effect 6 ; however, the study excluded individuals diagnosed with OA. Thus, we conducted this study to investigate the effectiveness of PG in attenuating OA in an animal model of monosodium iodoacetate (MIA)-induced Wistar rats.
Materials and Methods
Salmon Cartilage PG
Salmon cartilage PG (Proteoglycan F), processed into powder and containing 26.1% PG, was extracted from salmon cartilage. A specimen was deposited at ICHIMARU PHARCOS Ltd., Co. at 318‐1 Asagi, Motosu-shi, Gifu 501‐0475 Japan, Japan.
Experimental Animals
Normal healthy Wistar albino (aged 20 weeks) rats, with weight ranging from 180 g to 200 g, were purchased from a certified breeding center in Hanoi and housed under controlled conditions with a 12-hour light/dark cycle at 22 ± 2 °C and 55 ± 15% humidity. Rats were provided with a laboratory diet and water ad libitum. All experimental procedures were performed in compliance with the National Institutes of Health guidelines.
Experimental Design
Fifty male and female Wistar albino rats were fed in the laboratory according to standard criteria for 10 days and then divided into 5 groups (10 animals/group).
Group I : normal control
Group II (MIA + physiological saline gavage): MIA control group
Group III (MIA + diclofenac 3 mg/kg/day)
Group IV (MIA + PGF 40 mg/kg/day)
Group V (MIA + PGF 120 mg/kg/day)
At the start of the experiment, the group I animals (control) were given an intra-articular injection of physiological saline; groups II-V were injected intra-articularly with a single injection of MIA (25 g from Sigma-Aldrich, Singapore) at a dose of 3 mg/knee. The volume of injection was the same for every rat at 50 µL. Groups III, IV, and V were administered orally with diclofenac and PGF powder at doses of 40 mg/kg/day and 120 mg/kg/day, respectively, over a 6-week period (the PG level was based on previous studies). 5 The antiosteoarthritic effect of PGF was evaluated through the change in diameter of the right knee joints, serum interleukin (IL)-1β, tumor necrosis factor-alpha (TNF-α) assay, and histological assessment of the extracted tissue by the end of the experiment.
Knee Diameter
The right knee diameter was recorded by 1 researcher at all times to ensure consistency using an electric measurement (MC 555, HTMT Co., Ltd., China) at days 0, 3, 5, 7, 14, 21, 28, 35, and 42 of the treatment period. The knee was measured twice each time, and the higher result was recorded. The evaluation index was the increase in knee diameter compared with the previous measurement. 6
Serum Cytokine Levels
The blood samples were centrifuged at 1500
Histopathology of Knee Joints
Knee joint histology was randomly assessed from 3/10 rats in each group at the end of the study. Tissue specimens from the knee joint of rats were removed, fixed in 10% formalin, embedded in paraffin, and serially sectioned at 7 µm. Tissue sections were then stained with hematoxylin and eosin (H&E). Histological changes were examined by light microscopy (Olympus CX31/BX51, Olympus Optical Co., Tokyo, Japan) and photographed (Olympus DP70); the histological images were assessed by the Department of Pathological Anatomy.
Statistical Analysis
Statistical analysis and data plotting were carried out using SPSS version 20.0. Results are expressed as mean ± SD. The Shapiro-Wilk test was used for testing normal distribution. One-way analysis of variance (ANOVA) was utilized to assess the significance of serum cytokine level; two-way ANOVA was used for statistical testing of joint diameter changes. Student’s
Results
Changes in Knee Diameter
The increase in knee-joint diameter of MIA injected rats was significantly higher compared to with non-MIA rats (

Increase in knee diameter (mm) compared with baseline pretreatment. MIA, monosodium iodoacetate.
Serum Cytokine Levels
IL-1β serum levels were quantified at the end of the treatment period; the results followed normal distribution based on the Shapiro-Wilk test (Figure 2). One-way ANOVA returned an f-ratio value of 3.22; the

Concerntration of serum interleukin-1β. **
One-way ANOVA analysis of TNF-α serum levels returned the f-ratio value at 12.18 and a
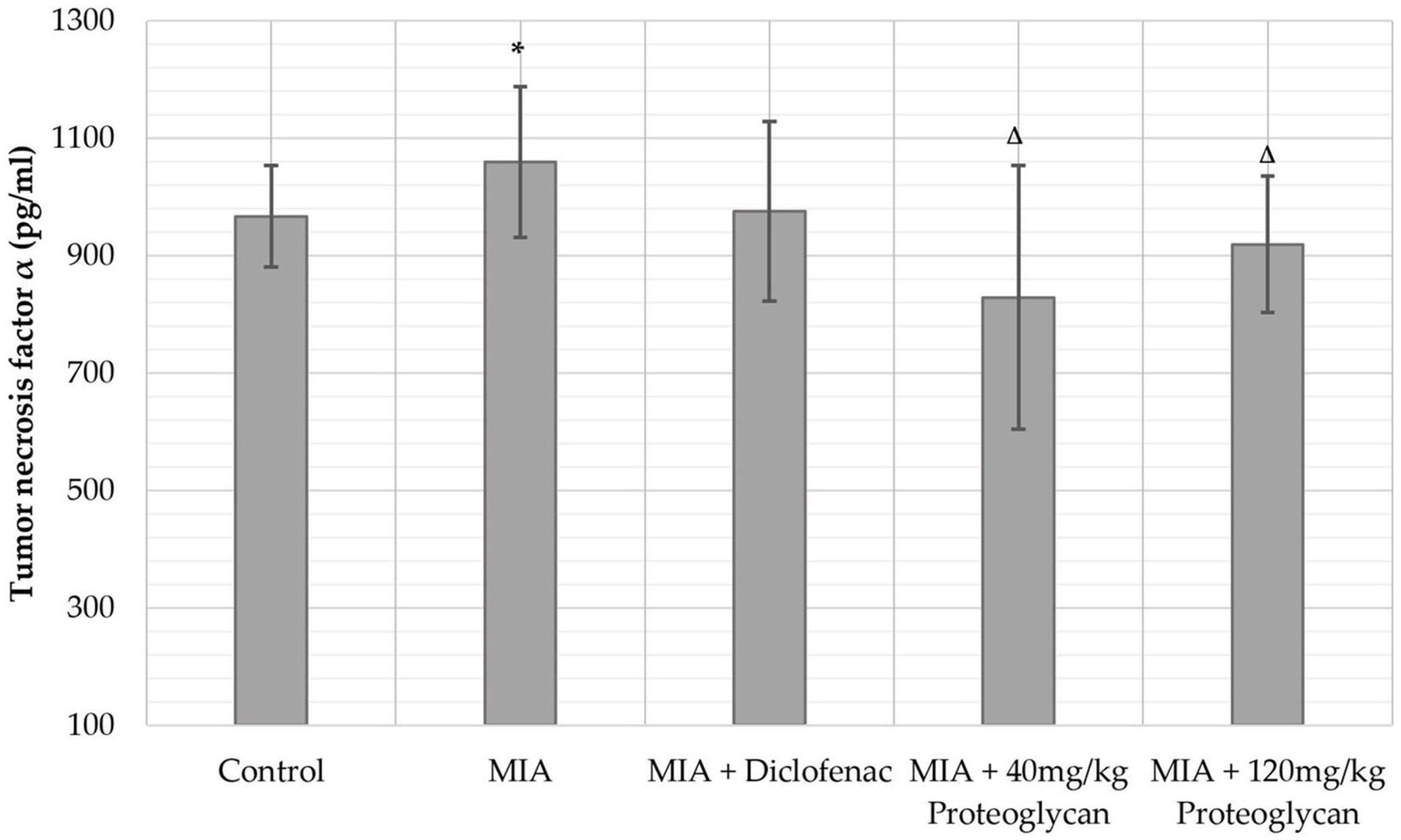
Concentration of serum tumor necrosis factor-alpha. *
Histopathological Assessment
Histopathological changes in the knee joints of rats are shown in Figure 4.

Histological assessment of the knee joint by the end of the experiment. MIA, monosodium iodoacetate; OA, osteoarthritis; PF, patellofemoral.
Group I (normal control group): there was no subchondral bone and cartilage damage and no loss in the PG dye layer. There was no degeneration of synovial cells and no necrotic chondrocytes.
Group II (MIA control group): moderate-to-severe subchondral bone and cartilage damage, loss of PG staining, and degenerative synovial cells were observed. Ghost chondrocytes (no nuclei) and necrotic chondrocytes appeared, and there was a marked irregularity of tidemark. Irregular thinning, fragmentation, and fibrillation of thinned cartilage. Joint space was narrower compared with the normal joint. Subchondral cysts with mucoid fluid were surrounded by sclerotic bone. Many osteophytes appeared together with the synovial area with granulation tissue and chronic inflammation.
Group III, IV, and V: moderate subchondral bone and cartilage damage, mild to moderate loss of PG staining, and moderate synovial cell degeneration were found upon histological assessment. Milder narrowing of the joint space and fewer osteophytes were observed. There was fibrous and chondrocyte proliferation. The degree of damage to the cartilage structure was decreased compared with the MIA control group (detailed assessment is provided in the Supplemental Information).
Discussion
In order to study the pathophysiology and research into novel therapeutic agents, a number of animal models for OA have been developed. 7 They generally fall into 3 categories: (1) a naturally occurring or genetically engineered OA animal model; (2) physically induced OA animal model either by trauma or surgery; (3) intra-articular injection-induced OA animal model. The intra-articular injection model is often preferred due to the pathophysiological similarity to primary OA and has no association with post-traumatic OA. 8 In this study, an MIA-induced rat model was chosen because it has been well established and bears resemblance to human OA regarding the histological, morphological, and metabolic changes. MIA inhibits glyceraldehyde-3-phosphate dehydrogenase enzymatic activity in chondrocytes, which disrupts the cell metabolism, consequently leading to chondrocyte apoptosis. Furthermore, the model has been shown to affect both the tibiofemoral and patellofemoral compartments of the knee joints, 9 which prove useful for studying new therapeutic agents.
Even though OA has less immunological involvement than rheumatoid arthritis, cytokine changes do occur because of chondrocyte apoptosis and subsequent immune cell migration into the synovial tissue. IL-1β and TNF-α have been implicated in the pathogenesis of OA, and there have been hypotheses that by interacting with innervating joint nociceptors, these cytokines also generate pain in affected individuals. Therefore, in the assessment of these cytokines, we can investigate changes in both pathophysiology and inflammatory pain states upon treatment with salmon nasal cartilage PG. In this study, compared with the MIA control and diclofenac-treatment groups, the PG-treated groups at both doses exhibited lower serum levels of IL-1β and TNF-α, which suggest better immune modulation and lesser pain generation in these groups by the end of the experiment. Future clinical trials would have to include pain assessment to confirm the efficacy of PG in the control of pain in OA patients.
The changes in knee diameter were monitored closely to assess treatment effectiveness; the PG-treated group saw a delayed improvement in knee diameter compared with the diclofenac group. However, by the end of the experimental period, the effectiveness of PG was comparable to that of diclofenac at both doses. Upon histological assessment, both diclofenac and salmon nasal cartilage PG showed an effective reduction of inflammation and necrosis. The treated group showed relatively normal synovial structure, similar to that of the normal control group, which proved the protective effect of salmon nasal cartilage PG against chondrotoxic and proinflammatory agents.
Till now, the exact mechanism by which salmon nasal cartilage provides a protective action to the cartilage structure remains to be elucidated. Previous in vitro studies showed that salmon nasal PG promotes chondrocyte proliferation and innate PG production. PG or its metabolites may exert anti-inflammatory actions via the suppression of inflammatory cytokine production, as well as a chondroprotective effect at the joint tissue via the proliferation of chondrocytes and the production of PGs. 10 In an in vivo study by Yo Tsuchiya et al, PG was shown to be absorbed through the small intestine via clathrin-mediated endocytosis. 11 There are also many instances, such as genetic disorders, where changes in PG production directly affect cartilage tissue. Camptodactyly-arthropathy-coxa vara-pericarditis syndrome (Jacob’s syndrome, OMIM # 208250), which is caused by decreased PG 4 (Lubricin) production, exhibits clear joint involvement, synovial hypertrophy, polyarticular large joint arthritis, and arthropathy. Induced expression of PG 4 in mice showed a protective effect against age-related and post-traumatic OA, which suggests a direct link between PG level and OA. 12 Even with this evidence, the detailed mechanism for the chondroprotective action of salmon nasal cartilage PG remains to be elucidated and requires further clinical assessment.
To our knowledge, this is the first study to investigate the effect of oral administration of salmon nasal PG on MIA-induced OA in a rat model. The efficacy of PGF observed in the present study suggests that a dietary supplement containing salmon nasal PG may exert anti-inflammatory and chondroprotective actions in rats with knee joint discomfort by alleviating the swelling of the knee joint, decreasing the serum concentrations of IL-1β and TNF-α, and partly improving the articular structure. Thus, salmon nasal PG-containing supplement is a potential candidate for treating OA of the knee. Future clinical studies should have an extended observation period to assess long-term changes in articular structure and pain status of the patients.
Conclusion
This study demonstrated that salmon cartilage PG at the doses of 40 mg/kg/day and 120 mg/kg/day can attenuate OA progression in MIA-induced OA in rats. The result from this study would suggest the further clinical application of salmon cartilage PG for management and prophylaxis of OA.
Supplemental Material
online supplementary file 1 - Supplemental material for Oral Administration of Salmon Cartilage Proteoglycan Attenuates Osteoarthritis in a Monosodium Iodoacetate-Induced Rat Model
Supplemental material, online supplementary file 1, for Oral Administration of Salmon Cartilage Proteoglycan Attenuates Osteoarthritis in a Monosodium Iodoacetate-Induced Rat Model by Tuyen Danh Le, Hien Thi Thu Vu, Iddamalgoda Arunasiri, Kenichi Ito, Tadahiro Makise, Hang Thi Le, Truong Hong Nguyen, Lai Thi Hong Nguyen, An Thai Nguyen, Binh Quoc Pham, Phuong Thuy Pham, Ha Thi Thanh Nguyen, Minh Hong Phan, Long Hoang Luong and Van Anh Pham Thi in Natural Product Communications
Footnotes
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Hanoi Medical University Institutional Ethical Review Board approved protocols.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was self-funded by the Vietnam University of Traditional Medicine and Ichimaru Pharcos Co. Ltd, Japan.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
