Abstract
Objective
Fuyuan Huoxue decoction (FHD) is a traditional Chinese medicine that can promote blood circulation, inhibit inflammatory reactions and effectively relieve pain. This study investigated the effect of FHD on rat skin flap survival and the p38 MAPK/IKK/NF-κB pathway.
Methods
The pathological condition of the flap tissue was examined by H&E staining. The expression levels of CD34 and VEGF in skin flap tissues were examined by immunohistochemistry. The levels of TNF-α, IL-6, CRP, and HO-1, superoxide dismutase (SOD) activity, and MDA content were detected by ELISA. The protein expression levels of Bax, Bcl-2, cleaved-caspase 3, p-IKKαβ, p-p38, p-p65, α-SMA, CK14, CK15, and SOX2 were detected by western blotting.
Results
Fuyuan Huoxue decoction upregulated the levels of CD34, VEGF, and Bcl-2, downregulated the expression of TNF-α, IL-6, p-IKKαβ, P-P38, P-P65, Bax, and cleaved-caspase 3, increased the activity of SOD, and inhibited the level of MDA. In addition, FHD promoted the proliferation and differentiation of skin flap.
Conclusions
Fuyuan Huoxue decoction improved the survival of the rat skin flap model by regulating the p38MAPK/IKK/NF-κB signaling pathway, and the mechanism may be related to the promotion of skin flap proliferation, differentiation, and revascularization and the inhibition of inflammation and oxidative stress.
Introduction
The skin flap is a significant and universal surgical material. It is widely used in plastic surgery to repair skin defects caused by tumor resection, 1 trauma or congenital defects, 2 such as nasal rhinoplasty and breast reconstruction. 3 During skin flap transplantation, there are different degrees of oxidative stress damage and various inflammatory reactions, which affect skin flap angiogenesis and cause some tissue cell necrosis, reducing the survival rate of the skin flap.4,5 Currently, several drugs have been used to increase skin flap survival, such as calcitriol, aspirin, and vitamin C.6–8 However, the therapeutic outcomes are unsatisfactory. Traditional Chinese medicine (TCM) has a unique advantage in the treatment of skin flap injury due to its multicenter and multitarget effects.9,10 Fuyuan Huoxue decoction (FHD) is a classic prescription to promote blood circulation and remove blood stasis, which has achieved remarkable results in clinical treatments. 11 It has the effect of reducing blood viscosity and significantly improving microcirculation. 12 Although it is recognized for its therapeutic effects in clinical applications, its mechanism of action has been less well studied. Recent studies have shown that NF-κB (nuclear factor κB) plays an important role in chronic inflammation and regulates large numbers of genes expressed at the transcriptional level, including chemokines, inflammatory mediators, and immune-related receptor genes. 13 It has been reported that NF-κB could be a target of MAPK kinases, which could inhibit p38MAPK and JNK activation and decrease IL-1, IL-6, and TNF-α expression by preventing p65 nuclear translocation. 14 Research has shown that NF-κB plays a critical role in the response to ischemia/reperfusion (I/R) injury in skin flap surgery. 15 Previous studies have indicated that the p38/NF-κB pathway could improve skin flap viability through the anti-inflammatory effect of natural hirudin. 14 In addition, hyperbaric oxygen preconditioning could promote abdominal skin flap survival via the ICAM-1/VCAM-1/NF-κB pathway during ischemia‒reperfusion injury. 15 Fuyuan Huoxue Tang may be used to treat type III prostatitis by affecting NF-κB signaling pathways through multiple herbal components and multiple targets. 16 However, the protective effect of FHD on skin flap necrosis and its related mechanism have not been reported.
In this study, we investigated the effect of FHD on the P38 MAPK/IKK/NF-κB signaling pathway by establishing a rat skin flap injury model with narrow tips and exploring the mechanism by which FHD promotes skin flap survival.
Results and Discussion
Results
LC-MS/MS analysis of FHD
LC-MS/MS was used to detect the chemical constituents of FHD (Figure 1). The representative chemical composition was gaultherin, aleuretic acid, pristimerin, baimaside, saikosaponin A, glycyrrhizic acid, and notoginsenoside Fe.
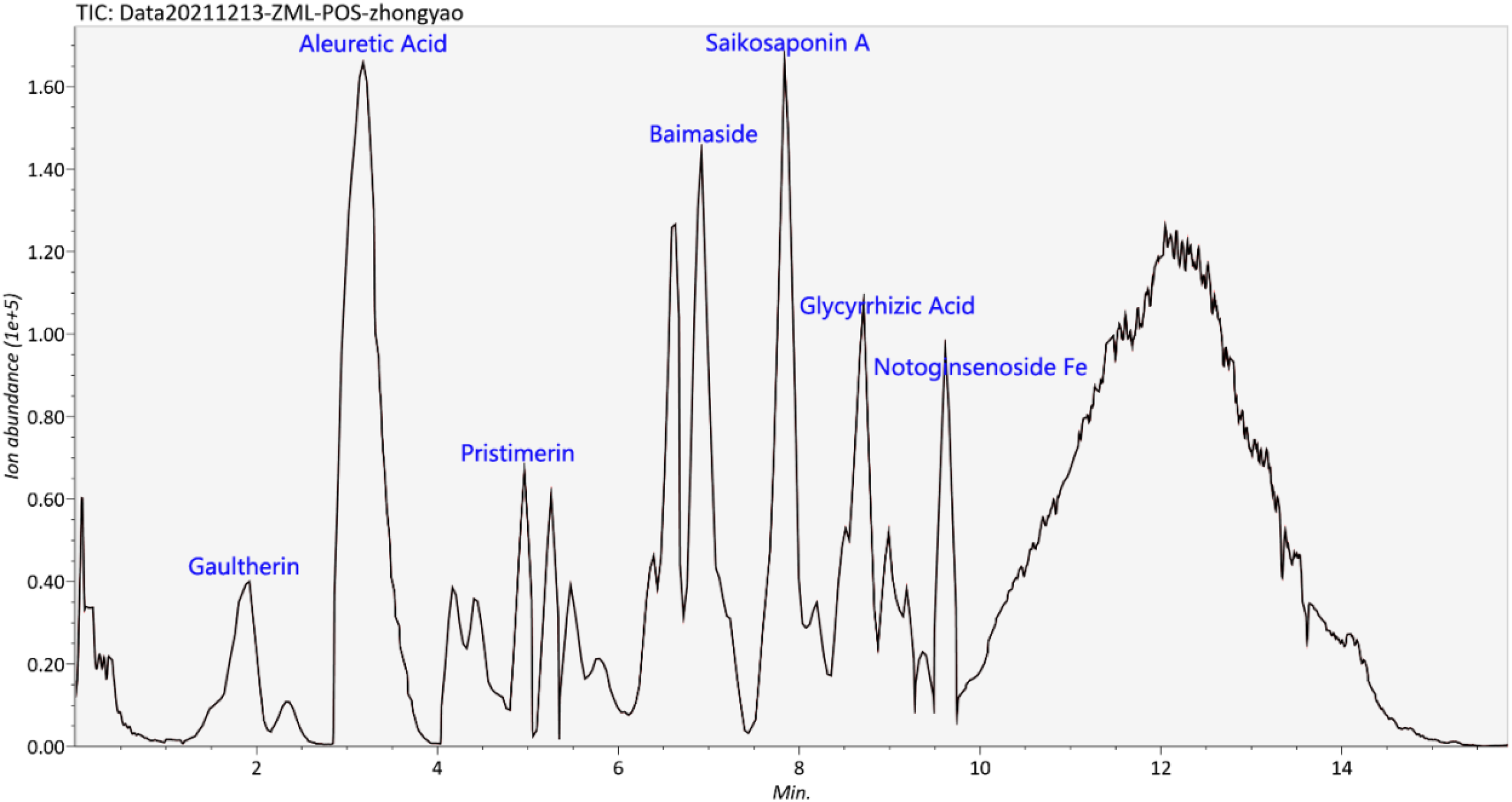
LC-MS/MS analysis of Fuyuan Huoxue decoction.
Fuyuan Huoxue decoction improved skin flap survival
To investigate the effect of FHD on skin flap survival, a 2 × 8 cm skin flap model was established (Figure 2A). The survival of the skin flap was recorded on the seventh postoperative day. A clear border between the surviving and necrotic areas of the skin flap was visible to the naked eye. In the survival area, the new skin was soft and warm, while in the necrotic area, the skin flap was dark, stiff, cold and lacked hair, and bleeding occurred at the incision. Fuyuan Huoxue decoction significantly increased the survival rate of the skin flap in a concentration-dependent manner (Figure 2B). The effect of FHD on edema and neutrophil infiltration was evaluated by H&E staining. The results showed that the distal areas of the skin flap were morphologically similar. On the seventh day, structural damage, edema, and inflammatory cell infiltration were observed in the model group. Fuyuan Huoxue decoction treatment significantly improved inflammatory damage (Figure 2C). The histological score of the model group was significantly higher than the control group; FHD treatment markedly reduced the histological score (Figure 2D).

Fuyuan Huoxue decoction improved skin flap survival. (A) A random 2 × 8 cm flap was established in the middle of the back of each rat. (B) Skin flap survival. (C) H&E staining showing histopathological changes in the skin flap (original magnification, 100×). (D) Histological score of H&E staining. Values are expressed as the means ± SDs, n = 6 per group. *P < .05, ***P < .001 versus control group; #P < .05, ###P < .001 versus model group; $P < .05 versus FHD-L group. FHD, Fuyuan Huoxue decoction; SD, standard deviation.
Fuyuan Huoxue decoction promoted the proliferation and differentiation of skin flap
To investigate the molecular mechanisms underlying the promotion of proliferation and differentiation in skin flap by FHD, we measured the expression levels of α-SMA, CK14, CK15, and SOX2. Western blotting showed that the protein levels of α-SMA, CK14, CK15, and SOX2 were significantly decreased in the model group. However, FHD treatment reversed this change in a concentration-dependent manner (Figure 3). Taken together, these results suggested that FHD promoted the proliferation and differentiation of skin flap.

Fuyuan Huoxue decoction promoted the proliferation and differentiation of skin flap. Western blot analysis of α-SMA, CK14, CK15, and SOX2. Values are expressed as the means ± SDs, n = 6 per group. *P < .05, ***P < .001 versus control group; #P < .05 versus model group; $P < .05 versus FHD-L group. FHD, Fuyuan Huoxue decoction; SD, standard deviation.
Fuyuan Huoxue decoction promoted angiogenesis in skin flap
Blood supply and angiogenesis are the key factors determining the survival of the flap. 17 Immunohistochemistry was used to study the positive effects of FHD on microvessels during skin flap survival. The results showed that the expression levels of VEGF and CD34 in the model group were significantly decreased. However, FHD reversed this effect in a concentration-dependent manner (Figure 4). Therefore, FHD could promote the formation of new blood vessels.
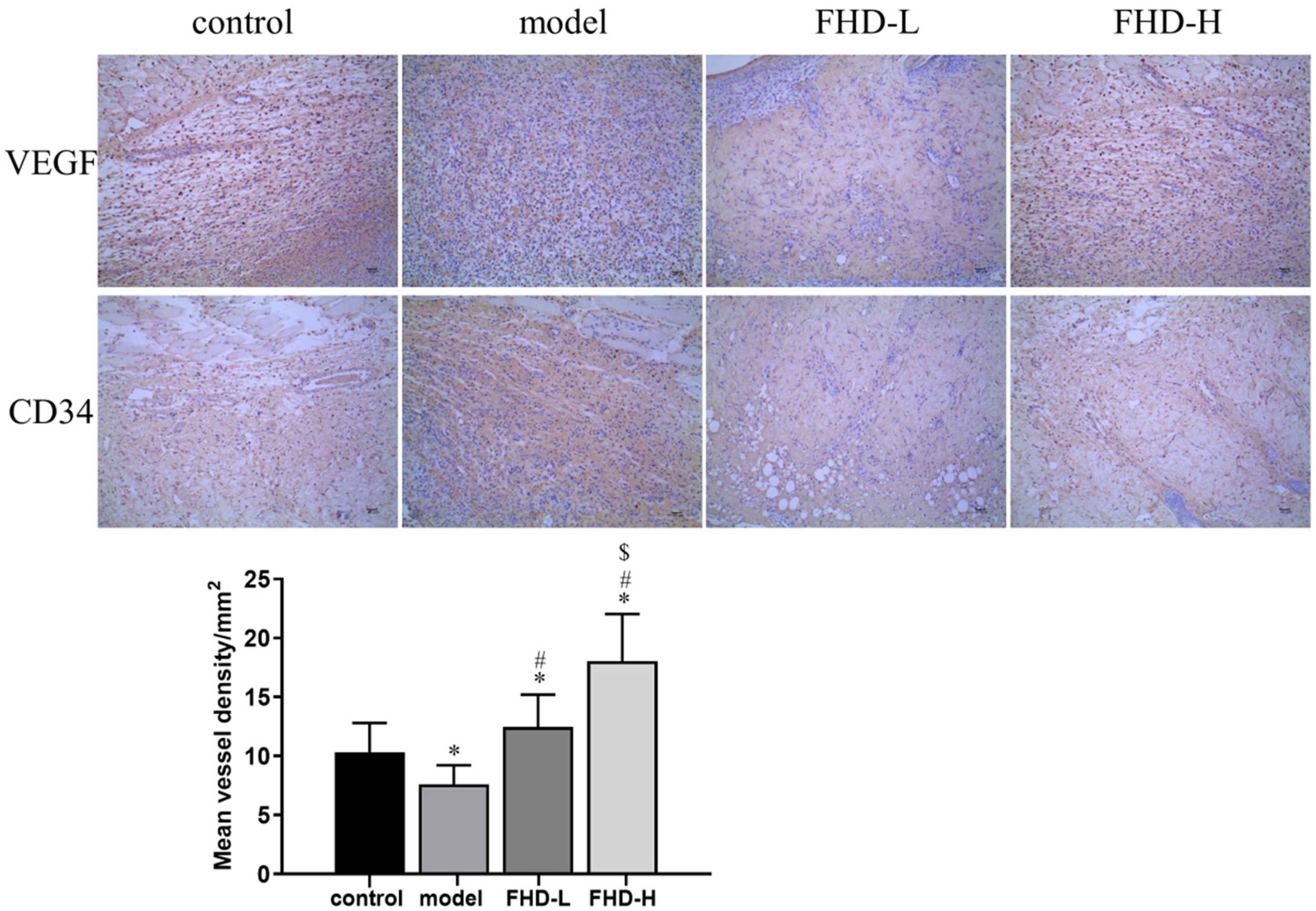
Fuyuan Huoxue decoction promoted angiogenesis in skin flap. IHC was used to detect the protein expression of CD34 and VEGF (original magnification, 100×). Mean vessel density/mm2. Values are expressed as the means ± SDs, n = 6 per group. *P < .05 versus control group; #P < .05 versus model group; $P < .05 versus FHD-L group. FHD, Fuyuan Huoxue decoction; SD, standard deviation.
Fuyuan Huoxue decoction inhibited oxidative stress and apoptosis in skin flap
The content of MDA, the activity of superoxide dismutase (SOD), and the level of HO-1 were detected by ELISA to explore the effect of FHD on oxidative stress in skin flap. The activity of SOD and the level of HO-1 were markedly increased, and the level of MDA was significantly reduced in the model group (Figure 5A). However, treatment with FHD reversed those changes in a concentration-dependent manner, suggesting that FHD protected the skin flap from oxidative stress. The effect of FHD on the expression of apoptotic proteins in skin flap was studied. The levels of Bax, Bcl-2, and cleaved-caspase 3 in skin flap were measured by Western blotting. The results showed that the expression levels of Bax and cleaved-caspase 3 were upregulated, and Bcl-2 was downregulated in the model group. After FHD treatment, the expression levels of Bax and cleaved-caspase 3 were downregulated, and Bcl-2 was upregulated. This finding suggested that FHD inhibited the expression of apoptotic proteins in a concentration-dependent manner (Figure 5B).

Fuyuan Huoxue decoction inhibited oxidative stress and apoptosis in skin flap. (A) ELISA analysis of SOD, MDA, and HO-1. (B) Western blot analysis of Bax, Bcl-2, and Cleaved-caspase 3. Values are expressed as means ± SDs, n = 6 per group. *P < .05, ***P < .001 versus control group; #P < .05 versus model group; $P < .05 versus FHD-L group. FHD, Fuyuan Huoxue decoction; SOD, superoxide dismutase; SD, standard deviation.
Fuyuan Huoxue decoction inhibited inflammation in skin flap
The levels of the pro-inflammatory cytokines TNF-α, CRP, and IL-6 were measured by ELISA to evaluate the effect of FHD on the inflammatory response. The levels of TNF-α, CRP, and IL-6 were increased in the model group. This finding indicated that an inflammatory reaction occurred in model rats. Fuyuan Huoxue decoction treatment decreased the levels of TNF-α, CRP, and IL-6 in a concentration-dependent manner (Figure 6). Thus, these data indicated that FHD inhibited the inflammatory reaction.
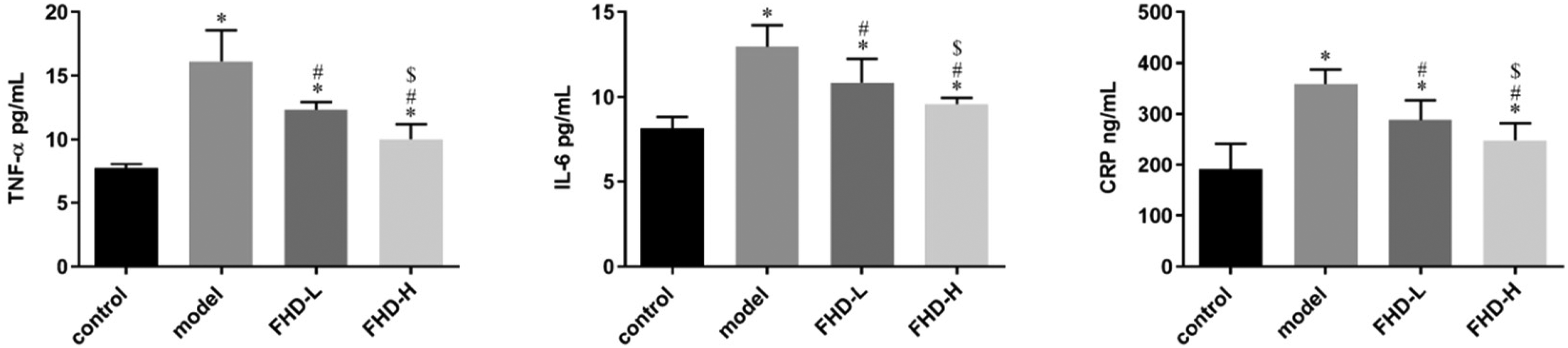
Fuyuan Huoxue decoction inhibited inflammation in skin flap. ELISA analysis of TNF-α, IL-6, and CRP. Values are expressed as the means ± SDs, n = 6 per group. *P < .05 versus control group; #P < .05 versus model group; $P < .05 versus FHD-L group. FHD, Fuyuan Huoxue decoction; SD, standard deviation.
Fuyuan Huoxue decoction promoted skin flap survival by inhibiting the p38MAPK/IKK/NF-κB signaling pathway
NF-κB can regulate the expression of large numbers of genes at the transcriptional level, including chemokines and inflammatory mediators. Western blotting was used to detect the protein levels of p-IKK αβ, p-p38, and p-p65 to verify that activation of the p38MAPK/IKK/NF-κB signaling pathway is the main mechanism by which FHD promotes skin flap survival. The results showed that the protein levels of p-IKKαβ, p-p38, and p-p65 were significantly increased in the model group compared with the control group. The levels of p-IKKαβ, p-p38, and p-p65 were significantly decreased after FHD treatment (Figure 7). These data suggest that FHD regulates skin flap survival by inhibiting the p38MAPK/IKK/NF-κB signaling pathway.

Fuyuan Huoxue decoction promoted skin flap survival by inhibiting the p38MAPK/IKK/NF-κB signaling pathway. Western analysis of p-IΚΚαβ, p-p38, and p-p65. Values are expressed as means ± SDs, n = 6 per group. *P < .05, ***P < .001 versus control group; #P < .05 versus model group; $P < .05 versus FHD-L group. p-IΚΚαβ: Phospho-IKK alpha + IKK beta. FHD, Fuyuan Huoxue decoction; SD, standard deviation.
Discussion
Due to the complexity of the mechanism of skin flap injury, curing or preventing skin flap injury is still a major problem for clinicians. 18 In recent years, TCM has made significant breakthroughs in the clinical treatment of ischemia‒reperfusion injury in the flap. 19 Some TCM preparations (eg, Dilong injection) with blood-activating and blood-stasis-transforming effects have been shown to increase the survival area of random flap in rats. 20 Although the therapeutic effects of TCM on ischemia‒reperfusion injury in flap have been clinically recognized, the specific mechanisms of action are still unclear. Therefore, it is important to study and analyze the mechanism of TCM in the treatment of flap injury. Our results showed that FHD regulated the survival of random skin flap by inhibiting the p38MAPK/IKK/NF-κB signaling pathway. Mechanistically, FHD inhibited the activation of NF-κB by downregulating the expression of P65 and p-IKKαβ; furthermore, the activation of p38MAPK was suppressed.
In pathological conditions, fibroblasts convert to myofibroblasts, which characteristically express α-SMA and produce contractile forces. 21 The higher the α-SMA-positive expression rate, the more severe the flap contracture. SOX2 is involved in all stages of organism development, as well as in the development of various diseases, and is essential for the maintenance of many adult stem cells. Cytokeratin (CK) is mainly expressed in human epithelial tissues. 22 This protein consists of approximately 20 isoforms and is involved in signal transduction, the regulation of cell proliferation, apoptosis, malignant transformation, and various stress responses in epithelial cells, in addition to maintaining the structural stability of epithelial cells or tissues. In this study, the results showed that FHD promoted the protein levels of α-SMA, CK14, CK15, and SOX2. Accordingly, FHD promoted the proliferation and differentiation of skin flap.
The survival rates of random skin flap are closely related to angiogenesis. Previous studies have shown that neovascularization can increase flap survival.23,24 Angiogenesis is a complex process involving the proliferation and migration of endothelial cells.25,26 VEGF is a vascular endothelial cell-specific heparin-binding growth factor that increases microvascular permeability and induces angiogenesis in vivo. It has been reported that VEGF promotes skin flap survival by stimulating endothelial cell proliferation and vascular angiogenesis. CD34+ cells promote proliferation in the vascular endothelium and the sprouting and growth of capillaries and induce the formation of proteolytic enzymes, which is conducive to endothelial cells passing through the matrix and promoting neovascularization. 27 Paeoniflorin effectively enhances the survival of rat random skin flap by promoting vascular hyperplasia and decreasing inflammation. 28 The results showed that the levels of VEGF and CD34 were significantly increased after the administration of FHD, which proved that FHD could promote the formation of new blood vessels and increase vascular density.
Skin flap necrosis is a common postoperative complication that may be caused by reactive oxygen species.29,30 This is because the accumulation of ROS can destroy the structure of cell membranes, nucleic acids, and chromosomes, leading to oxidative stress and apoptosis. 31 Superoxide dismutase, an antioxidant enzyme, is considered to be an important antioxidant defense that prevents cells from being damaged by oxygen free radicals by scavenging superoxide (O2−). MDA is one of the products of lipid peroxidation and is often used to reflect the degree of oxidative stress. HO-1 is an enzyme that disassembles heme into ferrous iron, biliverdin, and carbon monoxide (CO). In our study, the ELISA results demonstrated that FHD increased SOD activity and HO-1 level and decreased MDA content in random skin flap, which confirmed that it increased free radical scavenging, inhibited lipid peroxidation, and further inhibited oxidative stress. Bax is an apoptotic factor. The Bcl-2/Bax ratio determines whether cells survive after receiving apoptotic signals. When the ratio is increased, the antiapoptotic effect is stronger, and the cell survival rate is higher. As the upstream regulatory mechanism of caspase, overexpression of Bcl-2 can inhibit caspase activation and apoptosis induced by various factors. In the high-dose FHD group, Bax and cleaved-caspase 3 were decreased, and Bcl-2 was increased. In conclusion, FHD may promote the survival of skin flap by inhibiting oxidative stress and apoptosis.
Recent studies have shown that inflammation is the main cause of skin flap necrosis. The P38MAPK signaling pathway mediates the inflammatory response, and NF-κB is the downstream transcription factor of this signaling pathway. The activation of this signaling pathway could increase the expression of NF-κB, promote its translocation to the nucleus, and then activate the expression of various inflammatory mediators such as IL-6 and TNF-α. Natural hirudin increases rat flap viability by inhibiting inflammation via the PARs/p38/NF-κB pathway. 14 CRP is a sensitive index of acute and chronic inflammation and is a closely related indicator of inflammation and thrombosis. Ultrasound debridement can downregulate hs-CRP level, effectively reduce postoperative inflammatory reactions, reduce postoperative wound infection, and relieve pain in patients. 32 The results showed that the expression levels of CRP, IL-6, and TNF-α in the skin flap in the FHD group were significantly lower than those in the model group. These results suggested that FHD could inhibit the inflammatory reaction by inhibiting the expression of inflammatory factors. NF-κB is an inducible transcription factor formed by the 5 distinct subunits RELA (p65), RELB, c-REL, NFΚB1, and NFΚB2. It is sequestered in the cytoplasm by IκB proteins containing anκyrin repeats. The inactivation of NF-κB and its related pathway is associated with many diseases such as inflammation, cancer, and viral infection. When this pathway is not activated, NF-κB is located in the cytoplasm and bound to IκB isomers. When the pathway is activated, IκB proteins are phosphorylated on critical serine residues to facilitate their ubiquitination. NF-κB is freed and can then translocate to the nucleus and bind to the promoter region of its target genes. Then, the phosphorylation of IκB regulates the activation of the NF-κB pathway. The results showed that the expression of IΚΚαβ, p38, and p65 in the FHD group was significantly lower than that in model group, which inhibited IΚB phosphorylation and P65 nuclear translocation and then inhibited the activation of NF-κB, further suppressing the activation of p38MAPΚ, which promoted the survival of the skin flap.
In summary, this study demonstrated that FHD is a new therapeutic drug for flap necrosis. Fuyuan Huoxue decoction could promote random skin flap survival by inhibiting the P38MAPΚ/IΚΚ/NF-κB pathway.
Materials and Methods
Reagents
Based on the literature, 12 FHD is composed of 7 traditional medicines: 15 g of Radix Bupleuri, 9 g of Radix Trichosanthis, 10 g of Angelicae Sinensis Radix, 6 g of Carthami Flos, 6 g of Radix Rhizoma Glycyrrhizae, 9 g of Persicae Semen, and 10 g of Rheum officinale Baill. These 7 herbs were soaked in 10 times the amount of pure water for 60 min, boiled at high heat, simmered at low heat for 30 min, decocted, and extracted twice. The solution was mixed with the dregs, filtered, heated, and concentrated until the concentration of the raw drug was as indicated. The herbal medicines were purchased from the pharmacy of the Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University in Luzhou, Sichuan Province, China.
Liquid Chromatography‐Mass Spectrometry Analysis (LC-MS/MS)
The chemical composition of FHD was tested by a hybrid quadrupole-TOF LC/MS/MS mass spectrometer. The chromatographic conditions were as follows: the chromatographic column was a Waters BEH C18 (150 × 2.1 mm, 2.5 µm). The mobile phase ratio is shown in Table 1. The mass spectrometry conditions were as follows: the positive and negative ionization modes of electrospray ionization (ESI) were used for detection. The ESI source conditions were as follows: Ion Source Gas1 (Gas 1): 50; Ion Source Gas 2 (Gas 2): 50; Curtain Gas (CUR): 25; Source Temperature: 500 °C (positive ion) and 450 °C (negative ion); Ion Spray Voltage Floating 5500 V (positive ion) and 4400 V (negative ion); TOF MS scan range: 100 to 1200 Da; product ion scan range: 50 to 1200 Da; TOF MS scan accumulation time 0.2 s: and product ion scan accumulation time 0.01 s. Second-order mass spectrometry was obtained by information-dependent acquisition and had a high-sensitivity mode, declustering potential of ±60 V, and a Collision Energy of 35 ± 15 eV.
Gradient Elution Conditions of Acetonitrile-Formic Acid.

Flow rate: 0.3 mL/min, column temperature: 40°C, mobile phase: A. Equate = “0.1% HCOOH-H2O,” B. Equate = “ acetonitrile,” detection wavelength: 254 nm, injection volume: 10 μL.
Animals
Twenty-four adult male Sprague-Dawley rats, weighing 230 to 250 g, were supplied by Chengdu Dashuo Biological Co., Ltd. The rats were placed in separate cages with an ambient temperature of 22 °C to 24 °C, humidity of 40% to 60%, and a 12 h light/dark cycle with enough drinking water and chow and were used after one week of acclimatization.
The rats were randomly divided into 4 groups: control, model, FHD low-dose (FHD-L), and FHD high-dose (FHD-H) groups. Each group contained 6 rats.
Skin Flap Model
The rats were preoperatively anesthetized with sodium pentobarbital (100 mg/kg, intraperitoneal injection). When the rats were unconscious, there was no resistance and no response to a painful stimulus, and then the rats were fixed in a prone position. An electric shaver was used to shave the dorsal hair. The size of skin flap was designed to be 2 cm × 8 cm with the bilateral iliac ridge as the bottom line on the dorsum of each rat. 15 Under aseptic conditions, the skin and subcutaneous tissue were cut, the flap was separated from the deep fascia, and the sacral arteries were cutoff. The skin flap was immediately sutured back onto the rats using nylon and a wedged-on cutting needle.
Postoperatively, the FHD-L group was given 7.14 g/kg FHD, and the FHD-H group was given 3.57 g/kg FHD. The rats were given the treatments by intragastric administration once a day for 7 days. The control and model groups were given equal 0.9% saline solution. During the study, the rats were kept separately in standard laboratory cages and fed standard food and water at standard room temperature.
Skin Flap Survival
Skin flap appearance, tissue elasticity, and hair condition were evaluated to observe survival after daily gavage administration. At 7 days postoperatively, all skin flap was photographed, and the total area was measured. Images were analyzed using Image J software, and the percentage of the survival area was calculated (survival rate % = survival area/skin flap area).
H&E Staining
At 7 days postoperatively, the rats were sacrificed by dislocation of cervical vertebra. The flap tissue 3 to 4 cm away from the tail end was collected. Each specimen was fixed in 10% paraformaldehyde (10 g paraformaldehyde/0.1 M 100 mL PBS) for 24 h, embedded in paraffin, sliced into 4 mm slices, and prepared for hematoxylin and eosin (H&E) staining.
Immunohistochemistry
Skin flap tissue sections of paraffin-embedded blocks were dewaxed and rehydrated in gradient alcohol and then rinsed with pH 7.4 PBS (phosphate buffer). The slices were placed in 10 mM sodium citrate and incubated at 36 °C for 30 min to repair the antigen. The sections were treated with 3% hydrogen peroxide solution to block endogenous peroxidase activity and then washed with PBS. The slides were then incubated with normal goat serum at room temperature for 1 h to prevent nonspecific binding. The sections were incubated overnight at 4 °C with an anti-VEGF antibody (Abcam, ab232858, 1:1000) and anti-CD34 antibody (Abcam, ab81289, 1:2000). The slides were rinsed with the PBS and then incubated with a goat antirabbit IgG (Abcam, ab205718, 1:2000) for 45 min at room temperature. After being washed, the slides were treated with diaminobenzidine for 10 min at room temperature. Finally, the sections were counterstained with hematoxylin. The sections were photographed under a light microscope, and the number of microvessels was counted using the Weidner method.
ELISA
Skin flap samples were taken from each group, weighed, homogenized, and diluted. The supernatant was used to calculate the linear regression equation of the standard curve according to the concentration of the standard in the ELISA kits and the corresponding OD value, and then the concentrations of the corresponding cytokines (including IL-6, TNF-α, SOD, MDA, CRP, and HO-1) were calculated based on the regression equation according to the OD value of each group.
Western Blotting
The skin flap was cut into small pieces, 1 mL RIPA lysis buffer (Abcam, ab156034) was added, and the sample was ground, homogenated, and centrifuged for 15 min at 3000 g and 4 °C. The protein concentration of the supernatant was determined by a Pierce™ BCA Protein Assay Kit (Thermo Scientific, 23227). After SDS-PAGE, the protein was transferred onto a polyvinylidene fluoride membrane. After being blocked with 5% skim milk powder, primary antibodies, including rabbit anti-Bax antibody (ab32503, 1:1000, Abcam), rabbit anti-Bcl-2 antibody (ab32124, 1:1000, Abcam), rabbit anti-Caspase-3 antibody (ab13847, 1:500, Abcam), phospho-IΚΚαβ (ybs-3237R, 1:1000, Shanghai Yanjin Biological), phospho-p38 (4511T, 1:1000, Cell Signaling Technology), phospho-p65 (ab76302, 1:1000, Abcam), α-SMA monoclonal antibody (ABM0052, 1:10,000, Abbkine), anti-CK 14 antibody (ab119695, 1:25, Abcam), anti-CK 15 antibody (ab52816, 1:10000, Abcam), and anti-SOX2 antibody (ab92494, 1:1500, Abcam) were added to the membrane and incubated overnight at 4 °C. Subsequently, goat antirabbit IgG (HRP) secondary antibodies (Abcam, ab205718, 1:2000) were applied to the membranes and incubated at room temperature for 1 h. The antibodies were diluted with PBS (vol/vol). The blots were assayed by using the Novex™ ECL Chemiluminescent Substrate Reagent Kit (Thermo Fisher Scientific™, WP20005). The β-actin was used as an internal reference protein. The results were analyzed with ImageJ software.
Statistical Analysis
All data were analyzed with SPSS software 22.0 and GraphPad Prism. The results are expressed as the mean ± standard deviation. A P value less than .05 was considered statistically significant. Student t test was used for comparisons between 2 groups. One-way analysis of variance was used for comparisons between more than 2 groups. The least significant difference test was used to determine homogeneity of variance, and Dunnett's t test was used to determine heterogeneity of variance.
Footnotes
Authors’ Note
This study was approved by the Administration Committee of Experimental Animals, Sichuan Province, China. This study was approved by the Ethics Committee of Southwest Medical University (ID: swmu20220105). All procedures in this study were conducted in accordance with the *Ethics Committee of Southwest Medical University (ID: swmu20220105)* approved protocols.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The research project of Luzhou City-Southwest Medical University (grant number 2019LZXNYDJ37).
