Abstract
Objective
Papillary thyroid cancer (PTC) is one of the most common thyroid cancers and its prevalence has increased in recent years. Studies have shown that autophagy, angiogenesis, inflammation, oxidative damage, apoptosis, and some signaling pathways such as PI3K/AKT/mTOR play an important role in cancer progression. Moreover, most of their signaling molecules are the clients of heat shock protein 90 (HSP90), and its inhibition attenuates tumorigenesis. Diphenyl difluoroketone (EF-24), a curcumin analog, has antioxidative, anti-inflammatory, and anticancer properties and, unlike curcumin, has high efficacy, and may treat PTC. Therefore, the antioxidant, anti-inflammatory, angiogenic, apoptotic, and anticancer effects of EF-24 on PTC cell lines (BCPAP and TPC-1) were investigated in this study.
Methods
In this study, the effect of different concentrations of EF-24 and cisplatin on cell viability, apoptosis, inflammation, autophagy, oxidative status, and angiogenesis, along with HSP90 expression was evaluated in BCPAP and TPC-1 cell lines.
Results
The results showed that treatment with different EF-24 concentrations could significantly decrease cell viability by inducing apoptosis. EF-24 (40 µg/mL) also significantly downregulated HSP90 and the gene and protein involved in oxidative damage, angiogenesis, inflammation, and autophagy.
Conclusions
In conclusion, EF-24 could decrease cell viability by inducing apoptosis mediated by autophagy, oxidative stress, angiogenesis, inflammation, and the PI3K/AKT/mTOR pathway. It is possible that a decrease in HSP90 may mediate these effects. These results indicate that EF-24 is a viable option for the prevention and treatment of PTC.
Keywords
Introduction
Papillary thyroid carcinoma (PTC) occurs as an irregular solid or cystic mass or nodule in normal thyroid parenchyma. 1 PTC is accounting for 80% to 85% of all thyroid cancer cases and its incidence increased between 1975 and 2012 from 4.8 to 14.9 per 100 000. 2 It is most often diagnosed in people aged 45 to 54 years. However, its prevalence has been increasing in children and women in recent decades.3,4 Obesity, overweight, age, sex, race, exposure to ionizing and nonionizing waves, in some cases iodine deficiency, and mutations are the most important risk factors for thyroid cancers.3,5–8
Studies have also shown that angiogenesis, inflammation, autophagy, oxidative damage, and apoptosis play an important role in diseases such as cancer progression.9–14 Mediators of these (patho)physiological conditions are clients of heat shock protein 90 (HSP90).15,16 Inhibition of HSP90 decreases the activity of the phosphoinositide 3-kinases (PI3K)/protein kinase B (AKT) pathway and induces apoptosis in cancer cells by inhibiting the activity of antiapoptotic proteins. 17 On the other hand, the activation of HSP90 inhibits tumor cell apoptosis through the AKT pathway, tumor necrosis factor receptors, and nuclear factor-κB (NF-κB). 18 These results suggested that the inhibition of this protein through these pathways could be one of the therapeutic goals for cancer. Moreover, HSP90 enhances angiogenesis by upregulating vascular endothelial growth factor (VEGF) and endothelial nitric oxide synthase (eNOS), which are required for tumor progression. 19 Therefore, modulating the activity of HSP90 by medicine and herbal compounds may be a promising therapeutic mechanism for cancer. Interestingly, recently the use of herbs and their derivatives has been able to play a significant role in the improvement of some diseases such as cancer.14,20–23
Curcumin, a constituent of Curcuma longa, has recently been considered by researchers due to its extensive pharmacological properties such as anti-inflammatory, antioxidant, and anticancer. However, due to low stability and low bioavailability, it has very low efficacy. 21 Diphenyl difluoroketone (EF-24) is a bioactive synthetic analog of curcumin. 24 EF-24 has also been shown to have much higher systemic bioavailability than curcumin. 25 In addition, its biological properties, including anticancer, anti-inflammatory, and antibacterial activities, are about 10 times stronger than those of curcumin. 25 So, it seems that this curcumin analog is a good treatment option to inhibit the progression of cancers such as PTC. Therefore, this study was designed to evaluate the antioxidant, anti-inflammatory, antiapoptotic, and anticancer effects of EF-24 on PTC cell lines (BCPAP and TPC-1). First, the effect of different concentrations of EF-24 on cell viability was evaluated in BCPAP and TPC-1. After obtaining the appropriate dose, the status of apoptosis, inflammation, oxidative damage, and angiogenesis was evaluated along with the PI3K/AKT/mTORc1 pathway.
Materials and Methods
Cell Lines and Cell Culture
Human papillary thyroid carcinoma (BCPAP and TPC1) cell lines were used to evaluate the cellular responses (cell viability, inflammation, oxidative damage, and apoptosis) to EF-24 and cisplatin in this study. In addition, a primary human thyroid follicular epithelial cell line (Nthy-ori 3-1) was used as a control to evaluate EF-24-induced toxicity. Nthy-ori 3-1 along with BCPAP and TPC-I were cultured in RPMI-1640 (Biosera) medium in a humidified atmosphere containing 5% CO2 at 37 °C. The culture medium was supplemented with FBS (10% vol/vol), penicillin (100 U/mL), streptomycin (100 U/mL), glutamax (1%), and 5% RPMI. Cells were washed with culture media every 1 to 3 days, and the third passages of cells (105 cells/well) were typically grown to 75% confluence. The plates were treated with different concentrations of EF-24 and cisplatin (0, 2.5, 5, 10, 20, 40, and 80 μg/mL) (Sigma-Alderich). To prepare EF-24 and cisplatin, first, stock solutions with a concentration of 0.01 mM were made in dimethyl sulfoxide (DMSO), and then the second solution was diluted by the cell culture medium and used as previously described. 26 Cell samples (2 × 105 cells per well) were then incubated in a humidified atmosphere with 5% CO2 at 37 °C for 6 h post-treatment. Finally, they were collected at 24 h after treatment for further analysis.
MTT Assay
The viability of BCPAP and TPC1 cells was determined using the 2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. Briefly, 24 h after treatment, 20 μL of MTT reagent was added to each well. It was then incubated at 37 °C for 4 h. The medium was then washed, added to DMSO (Sigma Aldrich), and gently shaken to dissolve the crystals, which were evaluated at a wavelength of 590 nm by a microplate reader (Thermo Scientific).
Measurement of Oxidative and Inflammatory Parameters
The activities of superoxide dismutase (SOD) and catalase (CAT) were measured by the method described by Misra and Fridovich 27 and Aebi, 28 respectively. The malondialdehyde (MDA) level was measured based on the method described by Ohkawa et al. 29 The levels of TNF-α and IL-1β were determined using enzyme-linked immunosorbent assay kits.
Real-Time PCR
The expression of B-cell lymphoma 2 (Bcl-2), bcl-2-like protein 4 (BAX), caspase-3, AKT, mammalian target of rapamycin (mTOR), HSP90, eNOS, VEGF, NF-κB, and nuclear factor erythroid 2-related factor 2 (Nrf2) genes were evaluated. The sequence of primers used in this study is presented in Table 1. Total RNA was extracted with TRIzol reagent according to the manufacturer's instructions from cell lines (Invitrogen). Complementary DNA was synthesized by using random hexamer priming and a Prime ScriptTM-RT reagent kit (TaKaRa). The abundance of target transcripts was assessed using SYBR TM Green PCR Master Mix (Thermo Fisher Scientific) by a real-time PCR system (RotorGene 6000, Corbett Research). The expression of target genes was normalized against the expression of β-actin as an internal control and the relative expression level was calculated using the comparative cycle of threshold (CT) method 2−ΔΔCT.
The Primer Sequences Used in This Study.
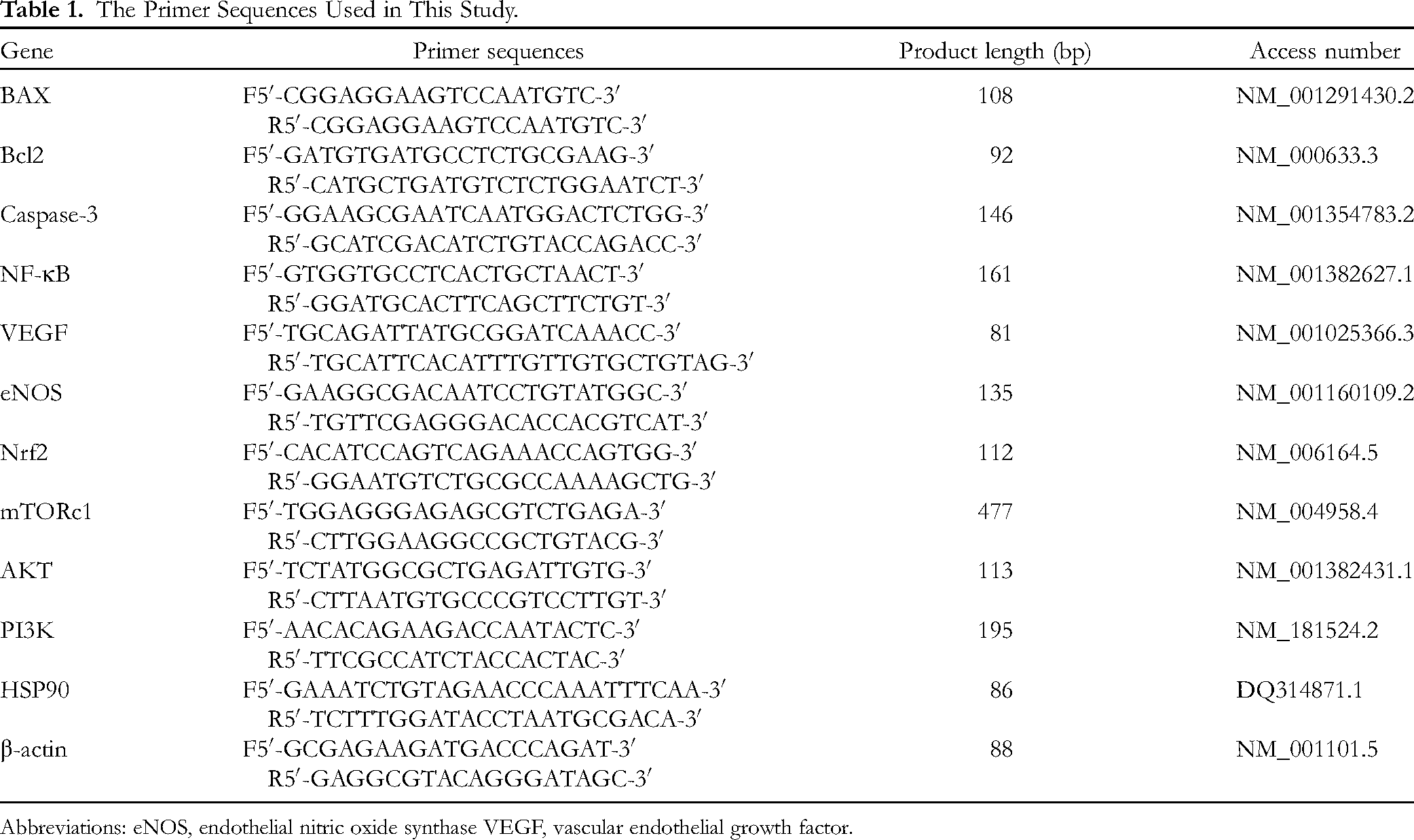
Abbreviations: eNOS, endothelial nitric oxide synthase VEGF, vascular endothelial growth factor.
Western Blot
After treatment for 24 h of BCPAP and TPC1 cell lines with EF-24, they were lysed with lysis buffer (Roche). Protein samples were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred to nitrocellulose membranes (Maibio). They were then blocked by immersion in 5% skim milk for 2 h at room temperature. The membrane was incubated overnight with specific primary antibodies (Casp3 (ab2171), Bcl-2 (ab32124), tumor necrosis factor (TNF)-α (ab155256), NF-κB (ab16502), HSP90 (ab32568), p-AMP-activated protein kinase (AMPK) (ab32047), total AMPK (ab222491), Nrf2 (ab76026), p-AKT (ab38449), AKT (ab8805), p-mTOR (ab109265), and mTOR (ab32028) with β-actin (ab8227)) as an internal control (Abcam) at 4 °C. The membranes were incubated for 2 h at room temperature after washing with phosphate-buffered saline in horseradish peroxidase conjugated secondary antibodies. Positive signals were developed by ECL Plus Western Blotting Substrate (Thermo Scientific) and analyzed by ImageJ 1.49 (National Institutes of Health).
Statistical Analysis
In this study, an independent t test was used for the statistical analysis of the dose-dependent and time-dependent study to determine the appropriate dose and the appropriate time of treatment. The obtained results from biochemical and molecular data in controlled and treated groups were analyzed by one-way analysis of variance and compared with the Tukey post hoc test using GraphPad Prism 5.0 software (GraphPad Software). P < .05 was set as the minimum statistically significant difference between the groups. Results are shown as mean ± standard error of the mean (SEM).
Results
Assessment of EF-24 Toxicity
Figure 1 shows the results of the MTT test related to the effect of different concentrations of EF-24 and times on the cell viability of primary human thyroid follicular epithelial cell line (Nthy-ori 3-1). The results showed that exposure to different concentrations of EF-24 for different times did not significantly change the cell viability. In other words, at these concentrations and for this duration, Ef-24 has no toxic effects.

The results of MTT test analysis to evaluate the toxicity of EF-24 concentrations at different times on the cell viability of Nthy-ori 3-1 cell line.
EF-24 Could Reduce Cell Viability in BCPAP and TPC1 Cell Lines
The MTT assay was used to determine the effects of different concentrations of EF-24 and cisplatin on the viability of BCPAP and TPC1 cells. The results showed that cell viability significantly decreased by increasing EF-24 and cisplatin in a concentration-dependent manner compared to the control group (Figure 2A, P < .05). Figure 2B shows a comparison of cell viability (%) in response to different concentrations of EF-24 over 24 h (Figure 2B, P < .05) and the highest rate of cytotoxicity was observed after 24. The results also showed that the highest concentration had a significant effect on cell survival after at least 12 h (Figure 2C, P < .05). In addition, the results showed that the onset of the effect of EF-24 depended on the concentration, and the higher the concentration, the faster the onset of the effect. Comparing the results of EF-24 with those of cisplatin showed that although EF-24 was not as effective as cisplatin, it was able to significantly reduce the viability of BCPAP and TPC1 cell lines in an almost similar pattern (Figure 2D, P < .05). Considering that in a concentration-dependent behavior, EF-24 could have a significant effect on cell viability in both cell lines (BCPAP and TPC1), we selected a concentration of 40 µg/mL to study cellular and molecular responses.
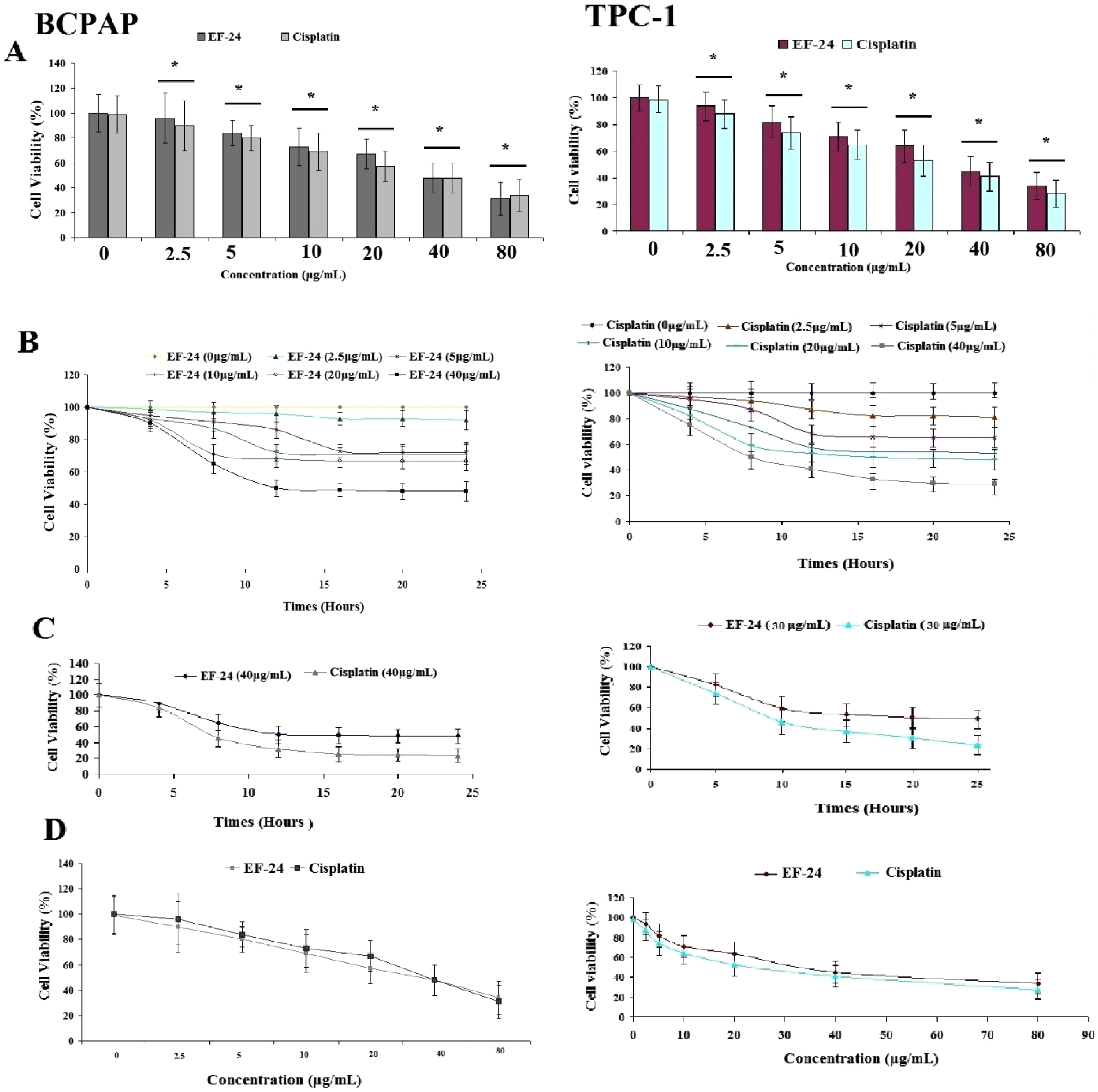
Comparison of the effects of EF-24 and cisplatin on the viability of BCPAP and TPC1 cells. Panels A and B show the effect of different concentrations of EF24 and cisplatin on the viability of BCPAP and TPC1 cells (n = 25). Panel C shows the effect of EF-24 and cisplatin on the viability of BCPAP and TPC1 cells 24 h after treatment (n = 16). Panel D shows the effect of different concentrations of EF-24 and cisplatin on the viability of BCPAP and TPC1 cells 24 h after treatment (n = 25). Asterisk shows a significant difference at the level of P < .05.
EF-24 Could Modulate Oxidative Damage and Inflammation in BCPAP and TPC1 Cell Lines
In this study, we measured the activity of SOD and CAT and the MDA level as a byproduct of lipid peroxidation to evaluate the status of oxidative stress (Figure 3A, P < .05). EF-24 treatment could significantly reverse the levels of these parameters compared to the control group. The level of antioxidant enzymes (SOD and CAT) significantly decreased and the level of MDA increased significantly 24 h after EF-24 treatment. In this study, we also measured the gene and protein expression of Nrf-2 to identify the molecular mechanisms involved in these cellular responses. Although its gene expression was significantly decreased by EF-24, its protein expression was not significantly decreased. The results also showed that the expression of NF-κB and the levels of TNF-α and IL-1β significantly decreased with EF-24 treatment. The results also showed that the expression of AMPK significantly increased with EF-24 treatment (Figure 4).
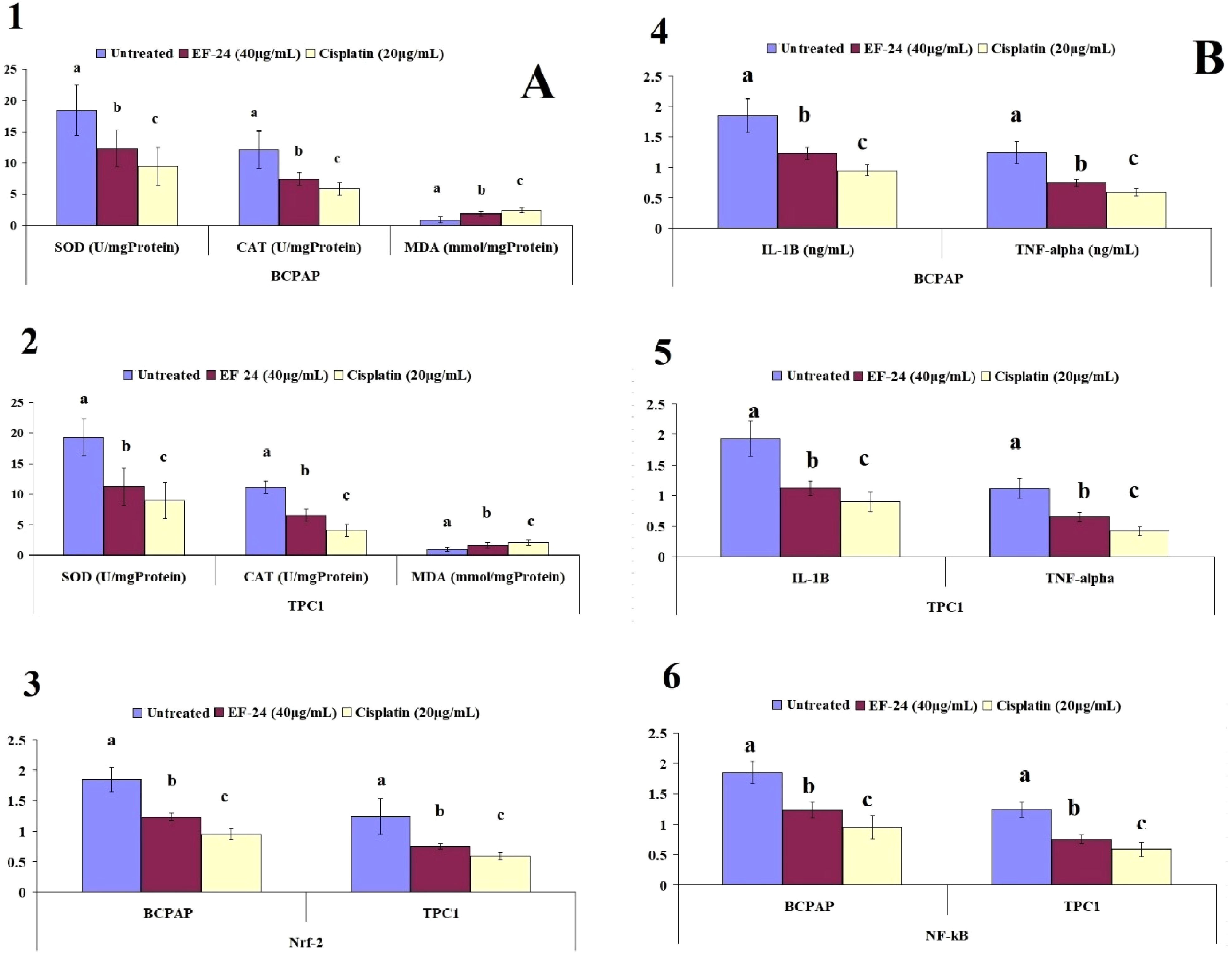
Comparison of the effects of EF24 on the parameters of oxidative damage (A panel) and inflammation (B panel), and gene and protein expression of Nrf2 and NF-ĸB in BCPAP and TPC1 cell lines. Dissimilar letters indicate a significant difference at the P < .05 level (n = 8).

Results of Western blot measurements in treated and control groups. The asterisk indicates a significant difference at the level of P < .05 with the untreated group.
EF-24 Could Inhibit Angiogenesis by Downregulating VEGF and eNOS Expression in BCPAP and TPC1 Cell Lines
The results showed that EF-24 could inhibit angiogenesis by reducing the expression of VEGF and eNOS in BCPAP and TPC1 cell lines compared with the control group. In addition, the expression of VEGF and eNOS was significantly reduced by cisplatin treatment compared with EF-24 in BCPAP and TPC1 cell lines (Figure 5, P < .05).

Comparison of the EF-24 effects on vascular endothelial growth factor (VEGF) and endothelial nitric oxide synthase (eNOS) expression in BCPAP and TPC1 cell lines. Dissimilar letters show a significant difference at the level of P < .05 (n = 8).
EF-24 Could Induce Apoptosis by Downregulating Antiapoptotic and Upregulating Proapoptotic Gene Expression in BCPAP and TPC1 Cell Lines
The results showed that EF-24 treatment could reduce the expression of gene Bcl2 and increase the expression of BAX and Caspase-3 genes in the BCPAP and TPC-1 cell lines. A comparison of the results showed that there was no significant difference between them in BCPAP and TPC-1 cell lines (Figure 6, P < .05).

Comparison of EF24 effects on Bcl2, BAX, and caspase-3 expression gene (down panel) and protein (up panel). Dissimilar letters show a significant difference at the level of P < .05. Asterisk indicates a significant difference at the level of P < .05 with the untreated group (n = 8).
EF-24 Could Inhibit Autophagy in BCPAP and TPC1 Cell Lines
EF-24 could decrease the expression of mTORC1, AKT, and PI3K in a similar pattern to that of cisplatin (Figure 7, P < .05).

Comparison of the effects of EF24 on PI3K, Akt, and mTORc1 expression gene (down panel) and protein (up panel) in BCPAP and TPC1. Dissimilar letters show a significant difference at the level of P < .05. Asterisk indicates a significant difference at the level of P < .05 with the untreated group (n = 8).
EF-24 Could Inhibit HSP90 Expression in BCPAP and TPC1 Cell Lines
HSP90 expression was significantly increased in untreated cancer cell lines. However, 24 h after EF-24 treatment, its expression was significantly decreased in the BCPAP and TPC1 cell lines (Figure 8, P < .05).

Comparison of EF-24 effects on HSP90 expression gene (right panel) and protein (left panel) in BCPAP and TPC1 cell lines. Dissimilar letters show a significant difference at the level of P < .05 (n = 8). Asterisk indicates a significant difference at the level of P < .05 with the untreated group.
Discussion
In order to address the regulatory role of HSP90 in the signaling mechanisms of cancer cells, the present study focused on signaling pathways of PI3K/Akt/mTORc1, inflammation, oxidative damage, autophagy, apoptosis, and angiogenesis. It was well documented that many of the processes that lead to the induction of either tumorigenesis or inhibition of tumorigenesis can be controlled by HSP90. In this study, we hypothesized that inflammation, cellular oxidative status, autophagy, apoptosis, and angiogenesis, along with the PI3K/Akt/mTORc1 pathway, can be clients of HSP90. So, by targeting this protein, these processes could be controlled to stop tumorigenesis. The molecular findings of this study are in line with previous studies30–33 showing that treatment with EF-24 for 24 h significantly decreased the expression of HSP90 in BCPAP and TPC1 cell lines. It is well documented that the inhibition of HSP90 is a therapeutic target for the treatment of various cancers.33–35 Clarke et al reported that gene expression profiles of human colon cancer cells following exposure to 17-allylamino-17-dimethoxy goldamycin, a molecular inhibitor of HSP90, showed inhibition of signal transduction and tumorigenesis. 35 Moreover, Mshaik et al reported that the inhibition of HSP90 decreased the growth of acute lymphoblastic leukemia T and B cells by affecting the stability of SRC kinases. 34 In line with this evidence, treatment with different doses of EF-24 could significantly reduce the viability of BCPAP and TPC1 cell lines. So, it may be an inhibitor of HSP90. Our results are in line with previous studies that showed that EF-24 could reduce the viability of different cancer cell lines.25,31,36,37 He et al showed that EF-24 inhibited tumor growth by inducing cell cycle arrest and apoptosis, mainly through its inhibitory effect on the NF-κB pathway and by regulating key genes through microRNA or the proteosomal pathway. 25 Yin et al showed that EF-24 inhibited tumor growth and metastasis via suppression of the NF-κB-dependent pathways in human cholangiocarcinoma. 36 Khan et al indicated that curcumin synergistically enhances the anticancer activity of cisplatin in PTC cells by targeting signal transducer and activator of transcription 3. 31 Li et al showed that curcumin suppresses proliferation and the Warburg effect by inhibiting LINC00691 and induces apoptosis through the Akt signaling pathway in B-CPAP cells. 38 Our findings also showed that EF-24 induced apoptosis by increasing the expression of BAX and capase-3 and reducing the expression of Bcl2. There was also a positive correlation between the inhibition of gene and protein expression of HSP90 with Bcl2 and a negative correlation between HSP90 with BAX and Cas-3 in EF-24-treated BCPAP and TPC1 cell lines. Adams et al who first designed and synthesized EF-24 stated that “EF-24 induced cell cycle arrest and apoptosis by means of a redox-dependent mechanism in MDA-MB-231 human breast cancer cells and DU-145 human prostate cancer cells.” 24 Zou et al also reported that “EF-24 may interact with thioredoxin reductase 1 to induce reactive oxygen species (ROS)-mediated apoptosis in human gastric cancer cells.” 39 In addition the combined treatment of EF-24 with mTOR inhibitor (rapamycin) 40 and Akt inhibitor 41 could induce apoptosis in gastric cancer cells by increasing the production of ROS. He et al reported that “EF-24 treatment dose-dependently decreased cell viability and caused cell cycle arrest in colon cancer cell lines.” 42 They also reported that “EF-24 treatment induced apoptosis via enhancing intracellular accumulation of ROS.” 42 However, some other researchers showed that EF-24 inhibited oxidative stress by decreasing ROS production, 43 and EF-24-induced apoptosis in human leukemia cells is not mediated by oxidative stress-related mechanisms. 44 Tan et al showed that EF-24 inhibited ROS generation and activated antioxidant response element–dependent gene transcription in human ovarian cancer cells. 43 Our results showed that the expression of Nrf2 and the activity of SOD and CAT decreased after treatment with EF-24. However, the results for MDA were different from what was predicted. It seems that EF-24 has only been able to affect the factors that control the antioxidant status, SOD and CAT, and gene Nrf2 and has not been able to change the consequence of oxidative damage, the MDA level. However, it should be noted that in the case of cancer, the cancer cells are very dynamic and their environment is very variable. Therefore, it seems that the role of EF-24 in inducing oxidative damage by altering ROS levels may be cell-dependent and the effects of EF-24 on ROS-mediated and ROS-independent apoptosis in cancer cells should be further studied. However, our results well showed that EF-24 clearly reduced the viability of BCPAP and TPC1 cell lines by altering the expression of genes involved in apoptosis and oxidative stress.
Our results showed that IL-1β and TNF-α levels, along with NF-ĸB expression, were significantly decreased in BCPAP and TPC1 cell lines by EF-24. It is well established that these genes interact and control each other's status. 45 It was reported that EF-24 inhibits the expression of NF-κB and levels of IL-1β and TNF-α and the signaling pathways associated with toll-like receptor (TLR)-4 and IL-1 receptor. 46 The signaling pathways associated with these cases control cell viability by regulating apoptosis.47,48 The NF-κB signaling pathway is activated by TLR-4 48 and plays a key role in the production of TNF-α and IL-1β.46,49 Vilekar et al reported that EF24 could inhibit the IL-1R and TLRs signaling pathways. 46 Moreover, the expression of NF-κB and the levels of TNF-α and IL-1β in BCPAP and TPC1 cell lines were decreased by EF-24. This condition leads to an inflammatory state via activation of the redox-sensitive transcription factor NF-κB by the production of pro-inflammatory cytokines such as TNF-α and IL-1. In addition, consistent with our results, some studies have shown that EF-24 causes oxidative damage by increasing ROS production. 24
The results of this study are in line with a previous one 43 that showed that the expression of VEGF and eNOS in these cell lines was significantly reduced by EF-24. Inhibition of these 2 genes plays an important role in inhibiting angiogenesis and tumorigenesis.50,51 Tan et al showed that EF-24 could significantly inhibit H2O2-induced VEGF expression. 43 VEGF is not only a mitogen but also a potent factor for endothelial cell development in newly formed immature vessels. 51 It suppresses the proapoptotic pathway and activates cell survival and antiapoptotic pathways such as PI3k/Akt, p42/44 MAP kinases and Bcl-2, respectively.52,53 In addition, the receptors of these angiogenesis-promoting growth factors are the client of HSP90, and their stability and performance depend on HSP90-associated machines. 54 Therefore, it seems that EF-24 can inhibit angiogenesis by inhibiting the expression of angiogenesis-promoting growth factors such as VEGF and eNOS and through HSP90-associated pathways.
Our results also showed that the PI3K/AKT/mTOR pathway changed dramatically after EF-24 treatment, which leads to impairment of autophagy and metabolism of BCPAP and TPC-1 cell lines. Moreover, a positive correlation was observed between HSP90 and the PI3K/Akt/mTOR pathway in BCPAP and TPC-1 cell lines. The PI3K/AKT/mTOR pathway is activated in tumors to regulate gene transcription involved in tumor metabolism, autophagy, and protein synthesis to increase cell proliferation. 55 The PI3K/AKT/mTOR pathway is stimulated by growth factors, insulin, and other signaling molecules that are essential for the survival of cancer cells. It is well known that the receptors of these growth factors and most mediators involved in their signaling pathways are HSP90 clients. Wang et al showed that “HSP90 regulates autophagy and plays a role in cancer therapy.” They stated that “HSP90 plays a role in autophagy via regulating the stability and activity of signaling proteins, and some HSP90 inhibitors can induce autophagy.” 56 Moreover, the PI3K/AKT/mTOR pathway, which is a major regulator of nutrient sensitivity, is an HSP90 client. 57 Moreover, EF-24 treatment could induce autophagy by changing the PI3K/AKT/mTOR pathway. Aryal et al showed that the mTORC1/ULK1 pathway can regulate autophagy. 58 mTORc1 also plays an important role in energy homeostasis in cancer cells. It was reported that EF-24 inhibited glucose uptake and glycolysis and reduced cell migration. 59 EF24 could also inhibit the activity of HIF-1α, as an oncogenic factor that regulates aerobic glycolysis and the expression of GLUT1, thereby regulating cancer cell survival. 60 So, inhibition of PI3K/AKT/mTOR gene expression and reduction of their protein content in the BCPAP and TPC-1 cell lines by EF-24 play an important role in reducing the energy available to the cell and can be one of the factors in reducing the viability of the BCPAP and TPC-1 cell lines. These results suggested that EF-24 has broad molecular targets and that its contribution to the control of energy metabolism and homeostasis in cancer cells seems to be another therapeutic feature that needs further study. It seems that EF-24, in addition to having a direct effect on the expression of genes and proteins of the PI3K/AKT/mTOR pathway through HSP90, also regulates the activity of this pathway. In fact, a review of the results of Western analysis showed that EF-24 changed the activity of the PI3K/AKT/mTOR pathway. This effect can be due to either the direct or HSP90-mediated effect. In addition, these findings are supported by the evidence that mTORc1 can be a therapeutic target for cancer treatment by regulating mitochondrial function 61 and the MAPK pathway. 62 Moreover, mTOR inhibitors alone or in combination with a variety of antitumor drugs are highly effective in treating cancer.57,63 Summing up, the findings of these studies suggest that EF-24 either alone or in combination with curcumin as adjunctive therapy, along with chemotherapy and angiogenic inhibitors can be recommended for preclinical and clinical studies.
Conclusion
The findings of this study showed that EF-24 was able to inhibit tumorigenesis in BCPAP and TPC1 cell lines by modulating the genes involved in inflammation, oxidative stress, angiogenesis, and autophagy. These effects may be mediated through HSP90-clients associated with signaling pathways controlled by EF-24. Given that the use of dietary phytochemicals or their analogs such as EF-24 can be an ideal strategy in preventing the development and treatment of cancer, it can be suggested that EF-24 could be used in combination with chemotherapeutic agents as an adjunctive therapy.
Footnotes
Acknowledgments
The authors thank Department of Endocrinology, Xi’an Gaoxin Hospital, Xi'an, China for financial support to carry out the study. The authors state that the intellectual property of this research belongs to the scientific and research group affiliated with Dr Abolfazl Akbari.
Author Contributions
GZ: Methodology, investigation, writing original draft. YZ and XW: Methodology, writing original draft and resources. AA: Conceptualization, supervision, project administration, writing review, and editing, JW: Conceptualization, supervision, analysis, project administration, writing review, editing, and resources.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
