Abstract
Objective
In this study, an online CMC–LC/MS method was established in order to screen the active components of Yiqi Fumai lyophilization injection (YQFM) related to angiogenesis. The pro-angiogenic active components of YQFM were screened using this system and were then further verified.
Methods
The ECV304/CMC model coupled online with an HPLC–MS system was initially developed to screen, analyze, and identify the allergenic components of YQFM. A fraction was retained on the ECV304/CMC column, which was identified as ginsenoside Rb2. A cell proliferation assay and zebrafish intestinal microvascular model labeled with green fluorescence were used to detect the proliferation effect of ginsenoside Rb2.
Results
(1) The active component in YQFM, which was retained on the column, was ginsenoside Rb2. (2) Within a concentration range of 2.5–20 μg/mL, ginsenoside Rb2 was shown to promote the proliferation of ECV304 and EPCs cells and increase the area of sub-intestinal vessels in zebrafish.
Conclusion
Ginsenoside Rb2, the active component in YQFM, can promote angiogenesis. The ECV304/CMC-online-HPLC system may serve as an effective screening tool for the vasoactive components from the traditional Chinese medicine injection.
Introduction
Yiqi Fumai lyophilization injection (YQFM) is a traditional Chinese medicine injection used in the treatment of cardiovascular disease, 1 as well as the diagnosis and treatment of chronic heart failure and acute myocardial infarction. 2 It is an improved Shengmai powder extracted from ginseng, Ophiopogon japonicus and Schisandra. According to traditional Chinese medicine, its prescription can supplement qi, nourish yin, prevent fatigue and tonify fluid. 3 A large number of studies and clinical trials have shown that YQFM can dilate coronary arteries and improve cardiac dysfunction in patients with heart failure. 4 Endothelial dysfunction is a key event in cardiovascular disease. Endothelial cells can regulate cardiac contractility, myocardial metabolism and the heart rate. It is necessary to screen the potential angiogenesis promoting effect of YQFM in order to ensure the effectiveness of the injection, and ultimately to provide a guideline for the production control of the injection. Therefore, chromatography can be taken into consideration.
Cell membrane chromatography (CMC) has been shown to serve as an effective method for screening active compounds that interact with specific receptors in traditional Chinese medicine. 5 It is a biomimetic affinity chromatographic technology with the cell membrane as the stationary phase, which is based on the selective interaction between cell membrane receptors and their ligands in vivo, helping to determine the binding characteristics between ligands and cell membrane receptors. 6 It has both biological and chromatographic separation characteristics. 7 CMC is mainly used to study the interaction between drugs and cell membrane receptors in vitro and can effectively identify specific target components that produce the biological effects of ligand cells in complex samples. The separation and identification of the bioactive components can be accomplished by liquid chromatography–mass spectrometry (LC–MS). 8 CMC technology is faster and more efficient than traditional high-throughput drug screening methods. Thus, a CMC coupled with LC–MS method was chosen for this work.
Angiogenesis is the formation of new blood vessels from existing blood ones, which takes place in a variety of physiological processes and is one of the main mechanisms of the formation of organ systems and tissues. 9 Angiogenesis plays a vital role in repair, remodeling and regeneration of the body.10,11 Vascular endothelial growth factor (VEGF) and its receptor (VEGFR) play an important role in physiological and pathological angiogenesis as well as in the establishment of the circulatory system under physiological conditions. A large number of cardiovascular diseases eventually lead to heart failure, which can be prevented by promoting angiogenesis during the formation of this disease. Vascular growth factor can regulate angiogenesis, which is mainly regulated by VEGF. 12 The unique role of VEGF depends on its binding to different receptors, binding to VEGFR-2 and activation of downstream signaling pathways, which can induce cell proliferation and angiogenesis. 13 Cell lines with high expression of VEGFR receptor are considered as a good model for studying angiogenesis.
ECV304 was reported first in 1990 as a spontaneously-transformed and immortalized cell line derived from a Japanese HUVEC. Subsequently, many studies have validated that ECV304 is a permanent endothelial cell line. It represents a potentially significant advance in this area of research. ECV304 maintains an apparently typical endothelial morphology in the absence of specific growth factors. We found that the ECV304 cell line highly expressed VEGFR, as shown in supplementary Figure 1. The endothelial progenitor cells (EPCs) are precursor cells of vascular endothelial cells, which are also known as angioblasts for angiogenesis and vascular function experiments. In this manuscript, EPCs were used to detect the effect of the active substances on the proliferation of vascular endothelial cells. Therefore, ECV304 and EPCs were used to study angiogenesis.

Summary of the study.
This study hypothesizes that YQFM contains pro-angiogenic active components, hence an ECV304/CMC-online-HPLC/MS system was constructed in order to screen the active ingredients of YQFM. The pro-angiogenic effects of the active ingredients with EPCs cells and zebrafish model fluorescent labeling of intestinal microvessels were then tested, as shown in Figure 1.
Materials and Methods
Reagents and Materials
DMEM high glucose medium was purchased from Thermo Fisher Scientific (Massachusetts, USA), fetal bovine serum from HYCLONE (Utah, USA), trypsin and the CCK-8 kit from Beyotime (Shanghai, China), penicillin–streptomycin and phosphate buffer from Gibco (New York, USA), acetonitrile, methanol and formic acid from Merck (Darmstadt, Germany), ginsenoside Rb2 (Batch No. HR27160W5) (mass fraction > 98%) from Baoji Chenguang Biotechnology Co., Ltd (Shan xi, China), Sorafenib from Beijing Beina Chuanglian Institute of Biotechnology (Beijing, China), methylcellulose (Batch No. B2006074) from Shanghai Aladdin Biochemical Technology Co., Ltd, simvastatin (Batch No. U2DBF-1K) from Tihi Ai (Shanghai, China) Chemical Industry Development Co., Ltd, astragaloside IV (Batch No. L2007265) from Shanghai Aladdin Biochemical Technology Co., Ltd, and YQFM from Tianjin Tasly Pride Pharmaceutical Co. Ltd (Tianjin, China). Ultra-pure water, produced using a MK-459 Millipore Milli-Q Plus ultra-pure water system, was used throughout the experiment.
Instruments
A one-dimensional chromatographic column (10.0 mm×2 mm) and column loader (Dalian Ripuli Technology Instrument Co., Ltd) were used in this study. The second column was a Waters Symmetry column (4.6 × 150 mm). In addition, High Performance Liquid Chromatography [MSIC (Hainan, China) Technology Co., Ltd], a SZX7 anatomy microscope (OLYMPUS,Japan), a VertA1 CCD camera (Shang hai, China), an IM300 microinjector (Narishige, Japan), and an AZ100 electric focusing continuous zoom fluorescence microscope (Nikon,Japan) were used in the second dimensional analytical system.
Operating Conditions
The ECV304/CMC-online-LC–MS system was first constructed, which used ECV304/CMC as the first dimension and LC–MS as the second. 14
One-dimensional conditions: the mobile phase was ultra-pure water, with a flow rate of 0.2 mL/min, column temperature of 37°C, and detection wavelength of 203 nm.
Two-dimensional conditions: a VP-ODS chromatographic column (4.6 × 150 mm, Waters) was used. The mobile phase A was 0.1% formic acid water, while B was 0.1% formic acid acetonitrile. The flow rate was 0.4 mL/min, and the column temperature 37°C, with DAD detection.
Mass spectrometry conditions: Flow rate: 0.12 mL/min; interface temperature: 300°C; CDL temperature: 250°C; detection voltage: 2.22 kV.
Preparation of Samples and Standard Solutions
Injection of ECV304/CMC-Online-LC/MS System Experiment
Sorafenib and ginsenoside Rb2 were dissolved in methanol to prepare a 1-mg/mL solution. About 100 mg of the YQFM sample was weighed, dissolved in 1 mL ultra-pure water, and filtered through a 0.45-μm filter.
Fluorescence Labeling Experiment of Zebrafish Promoting Angiogenesis
YQFM was dissolved in ultra-pure water to obtain individual stock solutions with concentrations of 12.5, 25.0, 50.0, 100.0 and 200.0 mg/mL. Ginsenoside Rb2 was prepared into a 50.0-mg/mL mother liquor with 10% DMSO and stored at −20°C. The mother liquor was diluted to 0.195, 0.391, 0.781 and 1.560 mg/mL during administration. Astragaloside IV was prepared as a 23.5-mg/mL mother solution in DMSO and stored in the dark at −20°C. Finally, astragaloside IV was diluted to 23.5 μg/mL. The low permeability solution(1.22 mg/mL Tris, 0.74 mg/mL KCl, and 0.5 mg/mL MgSO4) was adjusted to pH 7.4 with HCl.
Cell Culture
ECV304 was derived from the Cell Bank of the Academy of Life Sciences, Chinese Academy of Sciences. The cells were cultured in DMEM complete medium supplemented with 10% fetal bovine serum and 100 μg/mL penicillin–streptomycin. EPCs (ATCC) were cultured in DMEM supplemented with 10% fetal bovine serum, 100 µg/mL penicillin–streptomycin, and 100 ng/mL recombinant human ECGS. Both cell lines were grown in a flask and incubated at 37 °C in a 5% CO2 atmosphere. 15
Preparation of Cell Membrane Chromatographic Column
The ECV304 cells (2 × 107 cells/mL) were collected when the cells were in the exponential phase. The cells were washed three times with physiological saline by centrifugation at 1000 g at 4 °C for 5 minutes, and then re-suspended by sonic disruption in an ice-bath for 30 minutes. After that, the suspension was separated by centrifugation at 1000 g for 10 minutes. The supernatant was then collected and centrifuged at 12 000 g at 4 °C for 20 minutes. The precipitate was re-suspended in 5 mL physiological saline. 16 The cell membrane suspension was slowly added to the silica (50 mg, activated at 105 °C for 30 minutes) under vacuum with continuous vortex. The mixture was mixed with a cell membrane suspension to prepare the cell membrane chromatographic stationary phase. The stationary phase was stirred in a magnetic stirrer for 30 minutes and placed in a refrigerator overnight at 4 °C to produce the ECV304 membrane stationary phase, which was finally packed into a CMC column (10 mm × 2.0 mm) on a column-loading machine following a wet packing method. 17 The next day, wet packing was performed. 18
Method Validation
In order to screen for angiogenesis promoting components of YQFM, we developed a method of two-dimensional ECV304/CMC coupled online with HPLC–MS, via a 10-port two position switching valve and two C18 enrichment columns. ECV304/CMC was used to screen and retain the angiogenesis promoting components, which were then separated and identified by HPLC–MS, as shown in Figure 2. Firstly, various drugs (sorafenib, gefitinib and ciprofloxacin) acting on specific receptors were used to verify the selectivity of the ECV304 column and sorafenib was selected as the positive control drug to investigate the suitability of the ECV304/CMC-online-LC/MS. The positive drug sorafenib, as well as the negative control drugs, gefitinib and ciprofloxacin, were injected on the same ECV304/CMC column under the same conditions so as to observe the retention behavior of the three drugs on the ECV304/CMC column.19,20

ECV304/CMC-online-HPLC system diagram.
The repeatability of ECV304/CMC was then investigated, and the binding time was observed by performing 10 injections of sorafenib on the same ECV304/CMC column.
Screening of Active Components of YQFM
The active components of YQFM were screened by the ECV304/CMC-online-LC/MS system. For further identification, the corresponding standard compound of the retained constituent identified by the online system was also analyzed by the online-coupled system under the same experimental conditions.
Cell Growth Assay
1.5 × 105 cells/mL cell suspensions were inoculated on a 96-well plate, which was cultured for 24 hours. After the drug effect ended, CCK-8 was added to each well and incubated for 40 minutes. Ginsenoside Rb2, at different concentrations, was then added for 6 hours, and CCK-8 was added for incubation for 40 minutes. Then, the absorbance (OD) value was detected by a microplate reader at 450 nm in order to calculate the proliferation rate.
Validation of Angiogenesis in the Fluorescence Labeled Zebrafish Model
The normal group was treated with fish circulating water, and the model group was treated with simvastatin solution (1 mg/mL) to create the zebrafish intestinal vascular injury model. The administration groups were given 12.5, 25, 50, 100 and 200.0 mg/mL YQFM, as well as 0.195, 0.391, 0.781 and 1.560 mg/mL ginsenoside Rb2, respectively, to observe the angiogenesis of the zebrafish intestinal vascular area.
Statistical Analysis
All values were expressed as
Results
Method Validation
Specificity
Sorafenib, gefitinib and ciprofloxacin were first analyzed by the first-dimensional column and the chromatograms obtained using ECV304/CMC are shown in Figure 3B to D. Sorafenib is a VEGF receptor inhibitor; gefitinib acts on the EGF receptor. Gefitinib and ciprofloxacin were the negative controls. The chromatograms of non-retained and retained fractions suggest that only sorafenib was retained on the column, which demonstrated that sorafenib had better retaining characteristics. The chromatograms obtained using ECV304/CMC are shown in Figure 3A. No binding peak was observed on the empty silica gel column; however, binding behavior was evident on the ECV304/CMC, indicating that sorafenib was kept on the column. The results of the sorafenib injection on ECV304/CMC demonstrated the presence of a binding peak, for which the binding time was 50 minutes. Meanwhile, when gefitinib and ciprofloxacin were injected onto the ECV304/CMC, no binding was demonstrated, indicating that the chromatographic column of the cell membrane had normal column efficiency and specificity. The experimental results are shown in Figure 3.

Specificity of the ECV304/CMC column: (A) sorafenib empty silica gel column; (B) sorafenib; (C) gefitinib; (D) ciprofloxacin; gefitinib and ciprofloxacin are negative controls.
Repeatability
Using ECV304/CMC, sorafenib was continuously tested 10 times under the same conditions, and the sample volume was 10 µL each time. No significant difference was observed between the 10 chromatograms; the binding time was 50 minutes, indicating that the repeatability of ECV304/CMC was stable, and the RSD was 2.82%. The results are shown in Figure 4.

Repeatability of the chromatogram of sorafenib retained on ECV304/CMC.
System Adaptability
The ECV304/CMC-online-HPLC/MS system was verified with Sorafenib, a control drug acting with the VEGFR in research of angiogenesis, as shown in Figure 5. According to Figure 5A, sorafenib had good retention characteristics (R1, with a retention time of about 50 minutes) on ECV304/CMC. The retention component was enriched on the enrichment column by converting a six-way valve, which was then injected into the HPLC–MS system for detection, as shown in Figure 5B. Subsequently, it was compared with the chromatogram (Figure 5C) obtained by direct injection of sorafenib, for which the retained component was sorafenib. The chromatogram of the retained fraction (R1) in Figure 5B was the same as that obtained from a direct injection with sorafenib (Figure 2C). Therefore, the results obtained with the ECV304/CMC-online-HPLC/MS system were accurate and could be used to determine components that act on the cell membrane.
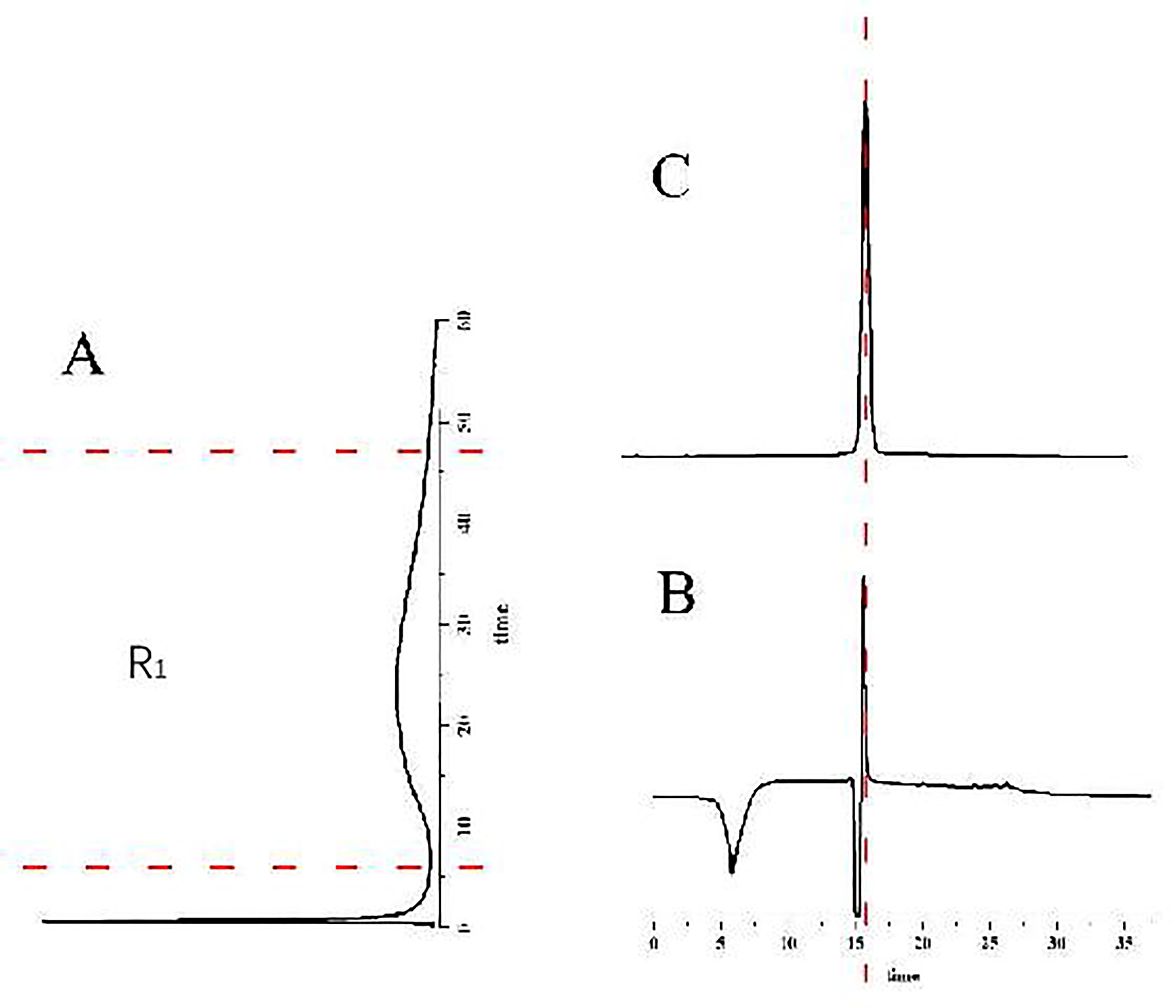
Analysis of sorafenib by ECV304/CMC-online-HPLC/MS system: (A) chromatogram of sorafenib retained on ECV304/CMC, (B) HPLC–MS chromatogram of retained fraction (R1) and (C) direct HPLC–MS chromatogram of sorafenib.
Screening of Active Components of YQFM
The established method was applied to screen potential angiogenic components of YQFM (Figure 6). One hundred mg/mL YQFM was injected into ECV304/CMC, after which the active components were screened. As shown in Figure 6A, R0 represents the unretained part, and R1 the retained part (retention time is 3 minutes). The retained fraction, R1, was enriched and switched to the HPLC–MS system (shown in Figure 6B). The chromatogram of YQFM obtained by enriching the retained components (between the two dotted lines) on the enrichment column showed the same characteristics, both in retention behavior and their precursor ions. The HPLC system is shown in Figure 6B, which was compared with the chromatogram obtained by direct injection of YQFM for HPLC analysis. As shown in Figure 6C, the results were the same.

Chromatogram of YQFM analyzed by ECV304/CMC-online-HPLC/MS system: (A) chromatogram of YQFM retained on ECV304/CMC, (B) HPLC–MS chromatogram of the retained fraction (R1) and (C) direct HPLC–MS chromatogram of YQFM.
In order to confirm the screened component, standard ginsenoside Rb2 was also analyzed through the whole system with a similar “screen-enrich-switch-identify” mode (Figure 7). As shown in Figure 7A, R0 represents the unretained fraction and R1 the retained fraction (with a retention time of about 8 minutes). Enriched components were then injected into the HPLC system, as shown in Figure 7B. The chromatogram obtained by directly injecting ginsenoside Rb2 standard solution into the HPLC system is shown in Figure 7C. The results showed a similar retention time of Rb2 retained on the ECV304/CMC column and displayed the same retention behavior and MS precursor on the second dimension (Figure 8).

Chromatogram of Rb2 analyzed by ECV304/CMC-online-HPLC/MS system: (A) chromatogram of Rb2 retained on ECV304/CMC; (B) HPLC–MS chromatogram of the retained fraction (R1); (C) direct HPLC–MS chromatogram of Rb2.

Mass spectrometry detection chart: (A) mass spectrum of YQFM; (B) mass spectrum of ginsenoside Rb2; (C) ginsenoside Rb2 structural formula.
By comparing the two-dimensional chromatograms of YQFM and ginsenoside Rb2, the peak time of the two was found to be relatively close. Therefore, mass spectrometry was used for further verification in order to determine whether the fragment in YQFM was ginsenoside Rb2. The corresponding experimental results are shown in Figure 8. Hence, we concluded that the screened component of YQFM was indeed Rb2.
Cell Proliferative Assay
CCK-8 was used to detect the proliferation effect of ginsenoside Rb2 on EPC cells, as shown in Figure 9. Ginsenoside Rb2 was shown to promote the proliferation of EPC cells in a dose dependent manner in the range of 5–20 μg/mL.

Effect of ginsenoside Rb2 on proliferation of EPCs cells by CCK-8 assay. Notes: n = 6, *P < 0.05.
Validation of Angiogenesis in the Fluorescence Labeled Zebrafish Model
Through the detection and analysis of the sub-intestinal vascular area of zebrafish in each group and comparison with the normal control group, the sub-intestinal vascular area of zebrafish in the model group was noted to be significantly reduced (P < 0.01), indicating that the simvastatin-induced sub-intestinal vascular promotion model of zebrafish was successfully constructed. Compared with the model group, 1.560 mg/mL ginsenoside Rb2 and 200.0 mg/mL YQFM were found to have significant effects in promoting the angiogenesis of zebrafish (P < 0.05). However, 200.0 mg/mL YQFM was shown to produce more significant effects in promoting angiogenesis (P < 0.01). The experimental results are shown in Figure 10.

Typical map of sub-intestinal vascular area of zebrafish treated with YQFM and ginsenoside Rb2. *P < 0.05, **P < 0.01, ***P < 0.001.
Discussion
In the present study, an ECV304/CMC-online-HPLC system was successfully constructed, in which the specificity, repeatability and system adaptability were investigated. Accordingly, its repeatability can only ensure the stability of intra-column repeatability; however, inter-column repeatability was poor, which should be further improved.
ECV304 cells have been widely used in experimental research. Now they were used to make cell membrane chromatographic columns because of the high expression of VEGFR, as shown in Supplementary Figure 1.
Angiogenesis refers to the process of sprouting or non-germinating new capillaries from pre-existing blood vessels through the proliferation and migration of endothelial cells, and then developing from new microvessels into a blood supply system. Angiogenesis can promote or inhibit angiogenesis by regulating VEGF and its receptors.
YQFM is mainly used for the treatment of cardiovascular diseases. This study attempted to offer insight into improving cardiovascular disease treatment pertaining to angiogenesis, for which VEGF is a key factor. The VEGF family includes VEGF A, VEGF B, VEGF C, VEGF D and placental growth factor. 21 These peptides have shown different affinities for VEGFR subtypes, where VEGFR has been known to exist as three subtypes of VEGFR-1, VEGFR-2 and VEGFR-3. VEGFR-2 is mainly responsible for the response of vascular endothelial cells to VEGF. 22 Moreover, it is the main kinase in multiple angiogenesis processes, including vascular permeability, endothelial cell proliferation, migration and survival. 23 The expression of VEGFR has previously been verified in wild-type cells. 24 However, our previous study found that ECV304 cells contained a large amount of VEGFR; thus, ECV304 cells were selected for the preparation of the cell membrane.
Traditional Chinese medicine and its injections have a bidirectional regulation on angiogenesis, which can be promoted within a certain dose range. The results showed that 2.5–20 μg/mL ginsenoside Rb2 promoted the proliferation of EPCs cells. Zebrafish experiments in which 50, 100, and 200 mg/mL YQFM, as well as 0.391, 0.781, and 1.56 mg/mL ginsenoside Rb2, were administered, demonstrated an increased promotion of intestinal vascular area, indicating the promotion of angiogenesis within this range. In addition, the effect of YQFM was noted to be significantly higher than that of ginsenoside Rb2, indicating that YQFM compound contains other angiogenesis promoting components besides Rb2, which were not retained on the column.
CMC has certain advantages, such as specific binding and high-throughput screening. By constructing the corresponding CMC combined with an HPLC–MS system, a single active ingredient in drugs can be quickly, efficiently and conveniently screened, which may address issues of the many components and complex compositions of traditional Chinese drugs and injections, providing an effective tool for the study of TCM modernization.
Conclusions
Ginsenoside Rb2, the active ingredient in YQFM, may promote angiogenesis. The ECV304/CMC-online-LC/MS system is stable and reliable, which may be used for the screening of active components related to angiogenesis in YQFM.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231182209 - Supplemental material for An Online CMC–LC/MS Method for Screening Angiogenesis Promoting Components in Yiqi Fumai Lyophilization Injection (YQFM)
Supplemental material, sj-docx-1-npx-10.1177_1934578X231182209 for An Online CMC–LC/MS Method for Screening Angiogenesis Promoting Components in Yiqi Fumai Lyophilization Injection (YQFM) by Qing Xu, Dekun Li, Meixu Wan, Yanxin Zhang, Zhi Li, and Aichun Ju in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Administration Committee of Experimental Animals, Tianjin, China.
Statement of Human and Animal Rights
All of the Experimental procedures involving animals were conducted in accordance with the Institutional Animal Care Guidelines of Tianjin University, China and approved by the Administration Committee of Experimental Animals, Tianjin, China.
Informed Consent
There are no human subjects in this articale and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
