Abstract
Objectives
Here we sought to determine the specific mechanism by which resveratrol targets microRNA-4325p and PDGFB in an osteosarcoma (OS) cell line and investigate its effect on the OS cell line.
Methods
As a result of the study related to the effect of resveratrol on the conditioned media of microRNA-4325p mimic transfected MG-63 and HOS (a fibroblast and epithelial-like cell line) cells, we determined that resveratrol decreased survival, and growth, migration, and aortic ring formation of endothelial cells. A combination of miRNA mimics and HOS and MG-63 cell lines were transfected with inhibitors to explore the mechanisms behind angiogenesis formation induced by resveratrol.
Results
We found that resveratrol improved the activity of microRNA-4325p mimic transfection in OS, thus promoting angiogenesis, as demonstrated by angiogenic parameters and gene expression in OS. Following these results, it is evident that resveratrol can modulate angiogenesis by regulating microRNA-4325p expression by targeting PDGFB genes in OS cells.
Conclusion
The results demonstrate that resveratrol promotes tumor angiogenesis of OS cells through microRNA-4325p/PDGBB-mediated pathways and provides theoretical and experimental support for the discovery of new bioactive phytochemicals that are effective therapeutic agents in the prevention and treatment of OS diseases.
Introduction
A malignant bone tumor, osteosarcoma (OS), frequently occurs in malignancies in children and adolescents.1,2,3 The mortality rate for OSs has remained unchanged, despite technological advances in surgery and chemotherapy. The presence of metastases in lung and the fact that these metastases resist conventional chemotherapy are the reasons why tumor recurrence occurs.4,5 Therefore, it is of the utmost importance to determine the molecular mechanism underlying OS growth of OS so that therapeutic approaches can be developed effectively. Two distinct processes are involved in the formation of newly formed blood during angiogenesis. Dysfunctional angiogenesis is among the leading causes of tumor development and growth. Tumor angiogenesis blockade is currently the second most popular method of treating cancer. Currently, clinically approved antiangiogenic drugs do not show any clinical benefit. Various compensatory mechanisms are activated and involved in the cascade of macromolecules required for tumor angiogenesis. Therefore, it is essential to investigate these compensatory pathways to develop broad antiangiogenic treatments in addition to chemotherapy. The development and advancement of OS are caused by the interference of cancer cells with the endothelium, macrophages involved with tumors, and smooth muscle cells. 3 The interactions between these also result in the release of transcription factors and cytokines, resulting in tumor cell growth, such as endothelin-1, TGF-β, angiotensin II and PDGF. 6 The PDGFB transcription factor is involved in the regulation of angiogenesis in tumors through the activation of transcription factors and molecular mechanisms.6,3
PDGFB may participate in the regulation of vascular remodeling, maturation, and stability through its participation in the recruitment of pericytes and vascular smooth muscle cells that produce new vessels. In the absence of the PDGFB gene, early embryonic defects result from the loss of pericytes and an excessive degree of vascular leakiness. 7 There was also evidence that overexpression of PDGFB causes an increase in high-density primary vascular plexus formation in murine fibrosarcomas, leading to neovascularization and the development of tumor metastases. 3 In addition, there is a relationship between the PDGFB levels that a tumor cell produces and the effects of drugs that inhibit PDGF on the remodeling of vascular tissue and the growth of metastases. 6
There is substantial evidence in the literature to indicate that microRNAs (miRNAs), a kind of noncoding RNA molecule naturally occurring within the body, regulate tumor growth and progression. MiRNAs have been shown to modulate gene expression by interacting with the specific mRNA 3′UTR. 8 MiRNAs control various vascular remodeling diseases. Therefore, targeting these miRNAs would have a beneficial effect on the severity of these diseases. During the progression of PDGF signaling in human pulmonary artery smooth muscle cells, miR-4632 has been found to target c-JUN and regulate apoptosis and proliferation. 9 MiR-221 expression induced by PDGF is significant in primary VSMCs because it decreases the expression of c-Kit and p27-Kip1. 10 Smad1 is an important target for miR-26a, as it plays an important role in the phenotypic changes induced by PDGFB in VSMCs. 11 It is significant for both the scientific and therapeutic communities to identify new regulatory molecules involved in the PGDF signaling pathway. Numerous research reports have shown that microRNA is essential for its function in regulating PDGF-induced cellular changes.
A polyphenolic compound, resveratrol (3, 5, 4′-trihydroxy-trans-stilbene), is a constituent of grapes, raspberries, mulberries, and cranberries and is thought to help increase health. Recently, it has been discovered that resveratrol has multiple pharmacological functions that are of interest to the scientific community. Researchers have found that resveratrol contains properties that prevent oxidation, inflammation, viral infections, and cancers. Initially, resveratrol was found to have anticancer properties by inhibiting induction and promoting tumorigenesis. However, several scientific studies published since 1997 have established that resveratrol exerts anti-tumor effects against various cancers, such as breast, cervical, colorectal, liver, prostate, and ovarian.12,13,14
According to research, resveratrol reverses chemotherapy resistance, which is present in gastric cancer due to inhibition of the epithelial-mesenchymal transition through modulation of the PTEN/Akt signaling pathway. 15 Furthermore, the study demonstrated that resveratrol inhibits autophagy in prostate cancer cells by reducing STIM1, which down-regulates the transcription factor mTOR. 16 Furthermore, increasing evidence suggests that resveratrol plays a significant role in reducing the risk of OS.17,18 However, the precise mechanism of resveratrol on OS is not yet completely understood.
Our aim in this research was to examine the role that resveratrol plays in the PGDF signaling pathway and its downstream targets that are involved in compensatory mechanisms as a result of tumor angiogenesis and to clarify the significance of angiogenesis in the microenvironment of OS. Furthermore, we investigated the level of expression of microRNA-4325p and PGDFB in OS cells. This study indicates that resveratrol exhibits a potent inhibitory effect on OS cells in conjunction with microRNA-4325p/PGDFB signaling. Hence, this research established a causal relationship between microRNA-4325p modulation by resveratrol and anti-tumor effects and offered evidence for a possible relationship between these molecular mechanisms and anti-cancer therapy regimens that effectively inhibit angiogenesis.
Materials and Methods
Culture of Cells
The resveratrol used in this study was purchased from Sigma-Aldrich, USA, then solubilized in DMSO to a level of 5 mg/mL and diluted with DMEM to the appropriate concentrations. DMEM medium containing fetal bovine serum (10% FBS), 100 units/mL of penicillin, and 100 units/mL of streptomycin were used in an incubator containing 5% carbon dioxide at 37 °C to cultivate human osteoblastic cells and human OS cell lines, HOS, and MG-63 cells (Chinese Academy of Sciences in Shanghai, China).
Cell Transfection
The 4 × 105 OS cells were plated on an 18 mm cover-slip under standard conditions at 37 °C in 5% CO2, supplemented with 400 µL of medium until the cells reached 80% confluence. We carried out transient transfection of miRNAs as previously reported. 18 Either a control miRNA (40 nM) microRNA-4325p mimic (40 nM), or a microRNA-4325p inhibitor (40 nM) was added to the transfecting reagent and the process was completed as specified in the supplier's guidelines. An inhibitor of microRNA-4325p binds to and inhibits endogenous microRNA-4325p, while a mimic of microRNA-4325p is a similar sequence to the endogenous microRNA-4325p. We examined the effects of either mimicking or inhibiting microRNA-4325p with a control miRNA. The cells were then incubated at 37 °C in 5% CO2 under standard conditions after transfection to achieve 100% confluence.
Analysis of Cell Apoptosis by Flow Cytometry
In the present study, HOS cells and MG-63 cells were divided into different groups so that they received resveratrol (5, 10, or 20 µM), a control miRNA, or a miRNA mimic or inhibitor. The test was conducted utilizing an Annexin-V/PI double staining kit, following the directions provided by the manufacturer. We then analyzed the cells with a FACS-Caliber system.
MTT Testing
Osteoblastic cells (human), the MG-63 cell line, and HOS cells were treated with either resveratrol (5, 10 or 20 µM) or transfected with a control (miRNA), miRNA mimic or inhibitor, seeded in 96-well plates (1 × 104 cells per well), and cultured overnight. Each well was incubated for 4 h with 10 µL of MTT solution (5 mg/mL in PBS, pH 7.4). Each well was carefully aspirated and DMSO was added to dissolve the crystals for 10 min. Absorbance measurements were made at 490 nm. Inhibition rate of proliferation (%) = (1−OD value of the treated group/OD value in the control group) × 100%.
A Real-Time RT-PCR Assay
Trizol reagent was used to isolate total RNA from the sample according to the manufacturer's guidelines. Cyclic DNA was formed with the reverse transcriptase kit. We performed a PCR assay utilizing SYBR-premix Ex-Taq II. For normalizing mRNA expression and miRNA for cell line studies, the GAPDH primer and U6 primer were used, respectively, and CAM (chick embryo chorioallantois membrane) assay-based mRNA expression was the normalized β-actin primer. For reverse transcription, a MicroScript II RT kit was used to detect mature miRNAs. In summary, 1 µg of RNA template was added along with buffer and nucleic mixture, and the volume of the reverse transcription reaction mix was increased to 20 µL using RNase-free water. We followed the following thermal cycling profile for the PCR: 90 °C for 6 min to denature, followed by 30 cycles of 90 °C for 40 s, 60 °C for 40 s, 75 °C for 50 s, and then 75 °C for 7 min. The relative levels of miRNA to mRNA expression were calculated using the ΔΔCt method. 19
Western Blot Analysis
Western blot assay was carried out following the procedure described earlier. 20 The cells were lysed in ice water for 30 min, and then the protein concentration in each sample was determined using a BCA protein quantification kit. After that, the protein extract (30 µg) was separated using SDS-polyacrylamide gel (10%) and applied to a PVDF membrane. As part of the preparation of whole cell lysates, RIPA buffer was combined with a protease inhibitor cocktail. Twenty microgram of isolated protein was then utilized or Western blot analyzed utilizing antibodies against PDGFB (1:1200, MAB2201, Biotechne, R&D systems, USA), Tie-2 (1:800; Biotechne R&D Systems, USA), Cycline A (1: 1000; Biotechne R&D Systems), Cdk1 (1:1000, Sigma Aldrich, USA), Cald1 (1:1000; Sigma Aldrich, USA), caspase-3 (Cell Signaling Technology, USA), Ephrin-B2 (1:800; Biotechne R&D systems, USA), CXCR4 (1:800; FAB170P, Biotechne R&D systems, USA), and α-tubulin (1:1200; Sigma Aldrich, USA) and CDK-2 (1:1200, Santa Cruz Biotechnology, USA). Visualization and data analysis were performed using a chemiluminescence imaging system. β-actin was used as a loading control for visual analysis.
Luciferase Reporter Assay
Transient transfection with control miRNA or microRNA-4325p mimic or inhibitor (50 nM) or resveratrol (20 mM) was performed in the presence of either mutant or wild-type PDGF-B 3′ UTR constructs (300 ng) with transfection reagent (Lipofectamine-2000). During 24 h, luciferase activity was evaluated with the help of a dual-luciferase reporter assay system. The luciferase function of Firefly luciferase was standardized with Renilla luciferase activity.19,8,21
Angiogenesis Test for Egg Yolks
The poultry station in Beijing, China, gave us eggs of white Leghorn chicks of Gallus gallus that had already been incubated. The incubation was carried out on eggs at 37 °C and 60% humidity under standard conditions. The assays were performed on Hamburger Hamilton embryos (HH). The eggs were broken open and inserted into foam cups. The embryos were placed upward and a breathable polyethylene cover was applied to cover them. An incubator was used to incubate the embryos without shells at room temperature and 70% humidity. 20 During the experiment, cover-slips (18 mm) were plated with HOS, and MG-63 cells in the presence and absence of miRNAs and resveratrol (20 µM) were applied to the bed of the vascular system so that the cells could easily contact the embryos. In this study, stereomicroscope images were obtained and analyzed using AngioQuant software after 6 h of treatment with a Magnus digital camera. 22
Analysis of Sprouting of the Chick Aortic Ring
The aortic ring assay of chicks was performed as previously reported. 23 The first step was to coat cover-slips with Matrigel (50 µL), then incubate them in CO2 for 30 min at 37 °C to solidify them. This study consisted of removing the aortic arch of the chick that evolved out of the heart of 12-day-old chick embryos and from pedicled arches of the aorta as ring-shaped sections, slicing, and mounting on covered cover slips coated with Matrigel. Then more Matrigel (20 µL) was added above the pedicled aortic ring. The aortic rings were incubated for 36 h with conditioned culture medium of MG-63, and HOS cells with and without either miRNAs or resveratrol (20 µM) prior to treatment. They were then rinsed with 1×PBS and imaged using an inverted microscope after the arches were treated. The captured images were then processed using AngioQuant software to determine the vessels’ ring junction length and size.
Proliferation of Cells and fluorescein diacetate
Three ×104 EA-hy926 cells were seeded in a 24-well plate and MTT assay was carried out in compliance with a previous report. 24 The EA-hy926 cells were cultivated in media supplemented with MG-63 and HOS, with and without miRNA transfection or with and without resveratrol (5, 10, or 20 µM). Cells were rinsed with phosphate buffer saline 48 h after treatment and subsequently added to a solution of MTT (5 mg/mL) and allowed to incubate at room temperature for 3 h. The MTT solution was replaced with DMSO to dissolve any crystals formed after incubation. A spectrophotometer carried out an optical density measurement at 565 nm at the end of the process. After treating the cells, a solution of fluorescein diacetate (FDA) was added (40 µg/mL). A fluorescent microscope was used to obtain the images.
Wound Scratch Test
A total of 1 × 106 EA-hy926 cells were seeded in 24-well plates and incubated for up to 24 h until they reached confluence. To produce a linear wound in monolayer cells, a 20 µL enzyme tip was used and washed with phosphate buffer saline, followed by incubation in a conditioned medium of HOS and MG-63 cells in the presence and absence of miRNA and resveratrol (20 µM). After 24 h, an inverted microscope was used to collect bright-field images. The wound closure rate was determined using Image J software based on the images.
Quantification of Human PDGFB by ELISA kit
Cell lines from OS, MG-63, and HOS were transfected with microRNA-4325p mimics or inhibitors or with resveratrol (20 µM) and cultured for 24 h. The conditioned medium was supplemented following the instructions provided by the manufacturer, and the level of PDGFB was determined with the assistance of a human PDGFB ELISA kit.
Statistical Analyses
Findings were displayed as mean ± SD. Student's t-test was employed to ascertain whether a difference was statistically significant (P < .05) between the groups. A one-way analysis of variance was performed to determine the statistical significance of the differences between the groups, and then Tukey's multiple comparison test (involving multiple groups). A value of P less than .05 was considered statistically significant.
Results
Inhibition of OS Cell Proliferation
To determine the rate of osteoblast proliferation, either MG-63 or HOS cells were subjected to MTT assay. Based on Figure 1a, it can be concluded that the optical density values of human osteoblast cells do not differ significantly after being treated with resveratrol (5, 10, and 20 µM) at each time point, indicating that resveratrol does not inhibit osteoblast growth. However, when either HOS or MG-63 cells were treated with resveratrol (5, 10, and 20 µM) for 48 h, there was a notable reduction in the proliferation of this compound in a dose-dependent manner (Figure 1a and b).

(Top) Proliferation of OS cells is inhibited by resveratrol (RST) in HOS cells. (A) MTT assays were performed to determine the survival rate of human osteoblast cells treated with resveratrol at different concentrations; (B) MTT assay was used to detect the survival rate of HOS cells treated with resveratrol at various concentrations; (C) Western blotting was used to determine the levels of cell cycle proteins (Cyclin A and Cdk1) in HOS cells after treatment with different concentrations of resveratrol. The results are expressed as the mean ± standard deviation. *P < 0.05 and **P < 0.01 indicate a significant decrease when compared to untreated (control) and treated HOS cells with RST at various concentrations (5, 10 and 20 µM) using one-way ANOVA followed by Tukey's multiple comparisons. (Bottom) Proliferation of OS cells is inhibited by resveratrol (RST) in MG-63 cells. (A) MTT assay was used to detect the survival rate of MG-63 cells treated with resveratrol at various concentrations; (B) Western blotting was used to determine the levels of cell cycle proteins (Cyclin A and Cdk1) in MG-63 cells after treatment with different concentrations of resveratrol. The results are expressed as the mean ± standard deviation. *P < 0.05 and **P < 0.01 indicate a significant decrease when compared to untreated (control) and treated MG-63 cells with RST at various concentrations (5, 10 and 20 µM) using one-way ANOVA followed by Tukey's multiple comparisons.
Effects of Resveratrol on Apoptosis of OS Cells
The reason for conducting this investigation was to determine whether resveratrol affects OS. In this experiment, HOS and MG-63 cells were co-cultured with resveratrol at varying concentrations (5, 10, and 20 µM) for 48 h. The results shown in Figure 2 demonstrate that resveratrol affects apoptosis of HOS cells (Figure 2A) and MG-63 cells (Figure 2B), and also caspase-3 activity (Figure 2C and D) in a dose-related manner.

Apoptosis is induced by resveratrol both in HOS and MG-63 cells. (A) and (B) Flow cytometry was performed to determine the apoptosis rate of HOS and MG-63 cells treated with resveratrol; (C) and (D) Western blotting was used in order to identify the levels of cleaved-caspase-3 and caspase-3, and β-actin served as a loading control. The results are expressed as the mean ± standard deviation. *P < 0.05, **P < 0.01 and ***P < 0.001 indicate a significant decrease when compared to control and treated HOS and MG-63 cells with RST at various concentrations (5, 10 and 20 µM) using one-way ANOVA followed by Tukey's multiple comparisons.
Resveratrol Stimulates the Reduction of microRNA-4325p Expression, Which is Implicated in the Apoptosis Induction and Proliferation Suppression in the MG-63 and HOS Cell Lines
MicroRNA-4325p, which has been identified as a substance that inhibits the growth of tumors in OS, was determined by measuring the expression of microRNA-4325p in MG-63 and HOS cells, and the results indicate that the expression levels of microRNA-4325p in MG-63 and HOS cells are significantly lower than those of normal osteoblasts (Figure 3A). The expression levels of microRNA-4325p in MG-63 and HOS cells were revealed to have increased in a dose-dependent manner after being exposed to resveratrol (5, 10, and 20 µM) for 48 h (Figure 3B and C). To determine whether resveratrol plays a role in suppressing microRNA-4325p involved in apoptosis and cell proliferation in MG-63 and HOS cells, either a microRNA-4325p mimic or inhibitor was utilized to either overexpress or suppress microRNA-4325p expression (Figure 3D to G). Figure 4 illustrates that up-regulation of microRNA-4325p increased the rate of apoptosis in cell lines compared to the control, while microRNA-4325p mimics accelerated the rate of resveratrol-induced apoptosis (20 µM).
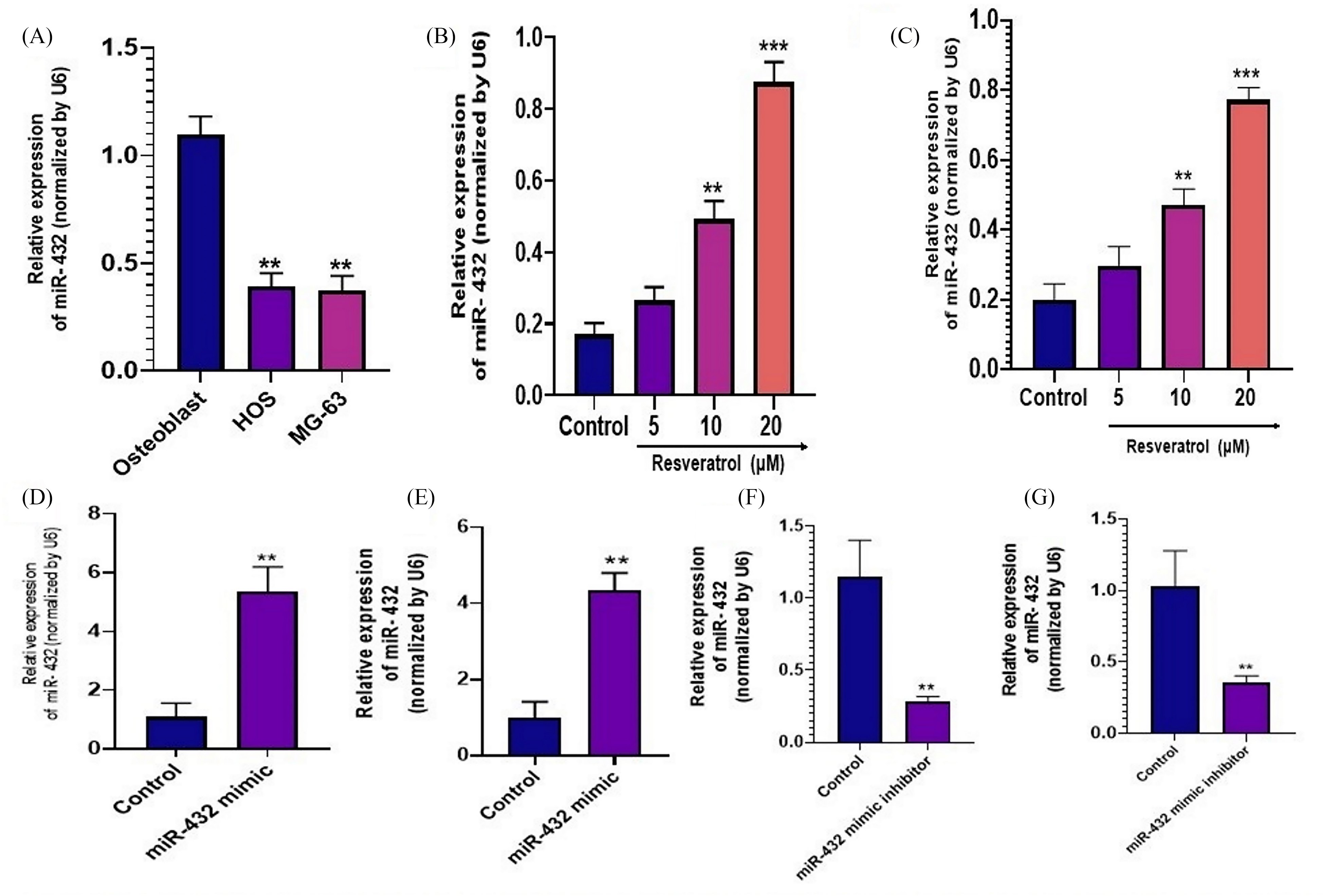
Resveratrol induces miR-432-5p expression. (A) qRT-PCR was used to analyze the expression levels of miR-432-5p in HOS, MG-63, and osteoblast cells; (B) and (C) Induction of miR-432-5p expression by resveratrol was dose dependent in HOS and MG-63 cells. D and E) miR-432 expression was examined in HOS and MG-63 cells treated with miR-432 mimic or miR-432 inhibitor by qRT-PCR. The results are expressed as the mean ± standard deviation. *P < 0.05, **P < 0.01 and ***P < 0.001 indicate a significant decrease when compared to control and treated HOS and MG-63 cells with RST at various concentrations (5, 10 and 20 µM) using one-way ANOVA followed by Tukey's multiple comparisons. **P < 0.01 indicates a significant decrease when compared to control and cells transfected with miR-432-5p mimics or miR-432-5p inhibitors respectively.

MicroRNA-432-5p induces apoptosis and suppresses MG-63 and HOS cell proliferation. (A–D) Flow cytometry was used to measure apoptosis rates in HOS and MG-63 cells transfected with miR-432-5P mimics or miR-432-5p inhibitors with or without resveratrol; (E–F) Western blots were used to measure expressions of cleaved-caspase-3 and caspase-3, and β-actin was used as a loading control; (G–H) Apoptosis rates in HOS and MG-63 cells transfected with miR-432-5P mimics or miR-432-5p inhibitors in the presence of resveratrol. The results are expressed as the mean ± standard deviation. * and # P < 0.05 indicate a significant decrease when compared to scramble and HOS and MG-63 cells transfected with miR-432-5p mimics or miR-432-5p inhibitors with or without resveratrol at 20 μM using one-way ANOVA followed by Tukey's multiple comparisons.
On the other hand, microRNA-4325p inhibitors were able to suppress resveratrol-induced apoptosis in a dose-dependent approach (Figure 4A to D). Therefore, we analyzed the level of apoptosis-related protein expression to gain further insight into the mechanism by which miR-432-5p induces apoptosis. Based on the results shown in Figure 4E to H, it is evident that the protein level of cleaved caspase-3 observed in MG-63 and HOS cells treated with the microRNA-4325p mimic is elevated in response to resveratrol treatment, demonstrated by the determining apoptosis. On the other hand, decreased caspase-3 cleaved levels were reversed by resveratrol. Furthermore, microRNA-4325p mimics inhibited the proliferation of cells concurrently with the induction of apoptosis, which was increased by the microRNA-4325p inhibitor (Figure 5A and B), and the effect stimulated by the microRNA-4325p inhibitor was reversed by resveratrol. Based on these findings, resveratrol suppresses the cellular proliferation of HOS and MG-63 and promotes apoptosis by including microRNA-4325p expression.
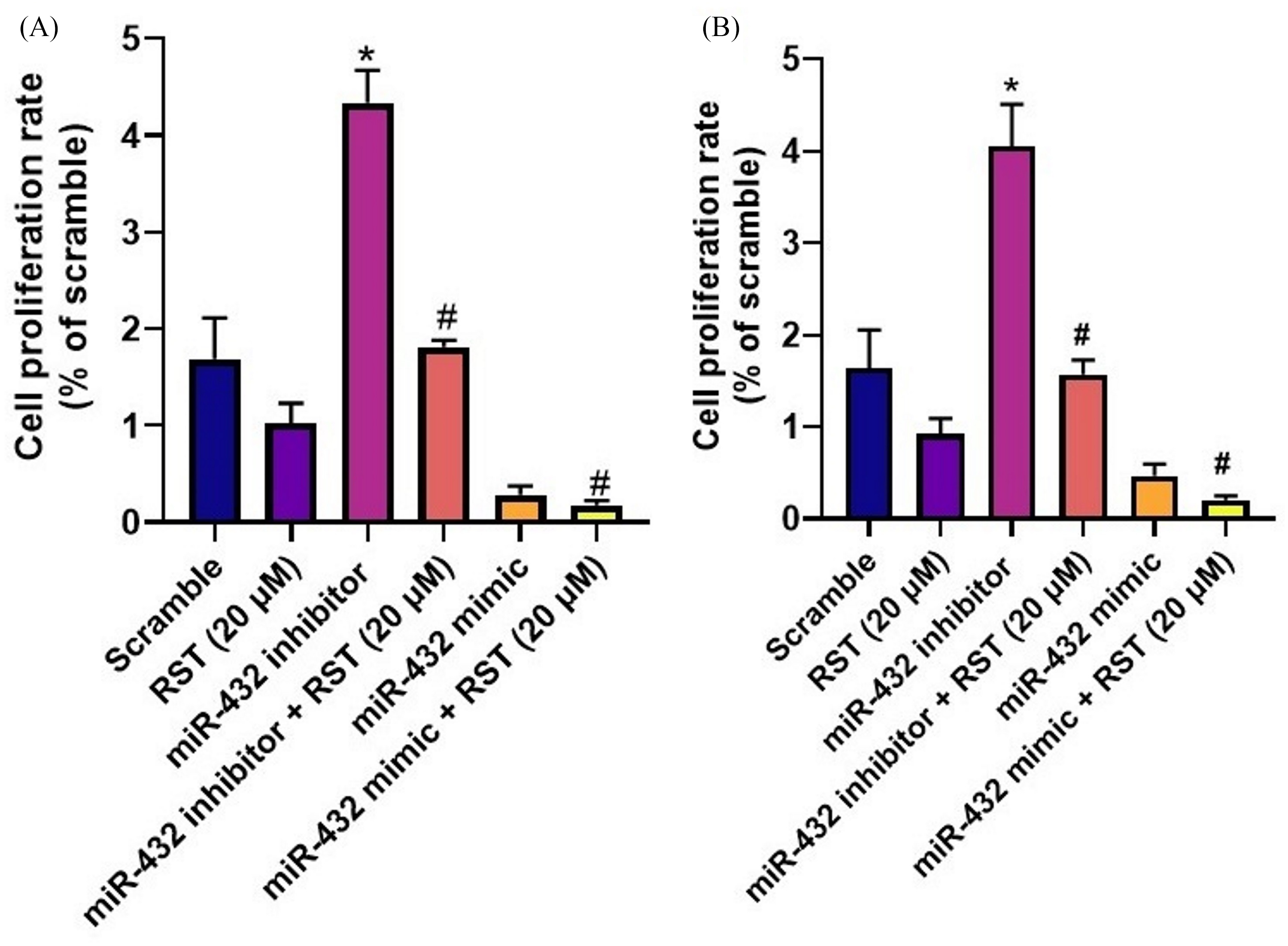
MicroRNA-432-5p induces apoptosis and suppresses HOS (A) and MG-63 (B) cell proliferation in the presence and absence of resveratrol. The MTT assay was used to measure survival rate of HOS and MG-63 cells transfected with miR-139-5p mimics or inhibitors with and without resveratrol. The results are expressed as the mean ± standard deviation. * and # P < 0.01 indicate a significant decrease when compared to scramble and HOS and MG-63 cells transfected with miR-432-5p mimics or miR-432-5p inhibitors with or without resveratrol at 20 µM using one-way ANOVA followed by Tukey's multiple comparisons.
MicroRNA-4325p Leads to Apoptosis of HOS Cells by Negatively Regulating the Expression Levels of PDGFB mRNA Expression
It is critical to understand the signaling mechanisms of the OS microenvironment to identify possible markers involved in the delay or progression of this disease. Therefore, it was of great interest to explore the part that signaling molecules play in how OS regulates tumor angiogenesis. As a first step, we evaluated the expression pattern in tumor samples and compared it with normal tissues. The results indicated that PDGFB is expressed in both cancerous and normal cells. In addition, several publications have revealed an elevated level of PDGF expression in clinical OS specimens.18,25,26
As seen in Figure 6, the expression of microRNA-4325p is associated with the angiogenesis induced by resveratrol in OS cells, which was thought to be involved in its regulation. Therefore, in this investigation, the effects were investigated of resveratrol on the putative target genes of microRNA-4325p by transiently transfecting MG-63 cells with the mimic or inhibitor, control miRNA or resveratrol (20 µM), followed by total RNA and real-time transcription-PCR analysis of the resulting RNA. Based on Figure 6, up-regulation of microRNA-4325p significantly decreased the mRNA expression level of PDGFB in MG-63 and HOS cells compared with the control, while microRNA-4325p mimics significantly reduced the level of PDGFB induced by resveratrol (20 μM).

PDGFB expression is downregulated by microRNA-432-5p in OS. miRNA-432-5p was chosen for further elucidation. HOS and MG63 cells were transfected with miRNA-432-5p mimics and inhibitors to study PDGFB expression. (A) Total RNA was isolated and real-time RT-PCR on PDGFB-based primers was carried out using the cDNA for the gene. The fold change in mRNAs was calculated by normalizing them with GAPDH; (B) The whole cell lysate was prepared after transfection and subjected to Western blot analysis with CDK-2 as a loading control; (C–D) A quantitative analysis of PDGFB secreted in the medium by MG63 and HOS cells, expressed in terms of ELISA-based relative fold changes. The results are expressed as the mean ± standard deviation. *P < 0.05 and **P < 0.01 indicate a significant decrease when compared to scramble and HOS and MG-63 cells transfected with miR-432-5p mimics or miR-432-5p inhibitors with or without resveratrol at 20 μM using one-way ANOVA followed by Tukey's multiple comparisons.
Furthermore, a conditioned medium obtained after transfection of MG-63 and HOS cells with miRNA-4325p mimic or inhibitor or control miRNA or resveratrol (20 µM) was analyzed for PDGFB using an ELISA kit (Figure 6C and D). PDGFB levels showed a similar pattern of considerable decline after resveratrol treatment, which is consistent with their levels of mRNA and protein.
Furthermore, in this study, a dual luciferase reporter system was used to confirm experimentally that PDGFB affects miRNA-4325p directly. Using a pair-GLO construct, which includes the Firefly and Renilla luciferase genes, the 3 ‘UTR target region of PDGFB was cloned. The 3 ‘UTR sequences of PDGFB from mutant and wild PDGFB were chemically produced, fused, and cloned into pmir-GLO vectors. Since Renilla luciferase is always active, it was used to establish a baseline for the amount of firefly luciferase activity included within the pmirGLO vector. The pmirGLO vector, which included either the wild type or the mutant PDGFB 3′ UTR downstream from the firefly luciferase reporter gene, was co-transfected effectively with miRNA-4325p mimic or inhibitor or control miRNA, or resveratrol (20 µM), in MG63 (Figure 7A) and HOS cells (Figure 7B). Luciferase activity was determined 24 h after transfection.

miR-432-5p binds to the 3'UTR of PDGFB. There was a putative prediction that miR-432-5p is capable of binding to a portion of the 3′UTR region of PDGFB. The putative target sequence of PDGFB and its mutated sequence have been cloned and inserted into pmirGLO. In (A) MG63 and (B) HOS cells, the pmirGLO construct (containing either wild-type or mutant PDGFB 3’ UTR) as well as miR-432-5p mimic/inhibitor, or control miRNA, were transiently co-transfected. The results are expressed as the mean ± standard deviation. *P < 0.01 implies a significant decrease in relative luciferase activity. Firefly luciferase activity was normalized with Renilla luciferase activity after 24 hours when compared to control and cells transfected with miR-432-5p mimics or miR-432-5p inhibitors with or without resveratrol at 20 µM using one-way ANOVA followed by Tukey's multiple comparisons.
According to the experimental results, the luciferase activity was significantly decreased by resveratrol (20 µM) in the cells that were co-transfected with a construct comprising the PDGFB wild 3′ UTR and the pmirGLO gene along with microRNA-4325p mimic. On the contrary, the control miRNA and the microRNA-4325p inhibitor did not show a discernible change in the amount of luciferase action (Figure 7A and B). Based on the results, it can be concluded that the 3′ UTR of PDGFB is specific for microRNA-4325p in OS cells. Therefore, the results of this experiment suggest that microRNA-4325p activates PDGFB 3′-UTR in OS cell lines. However, other potential targets of microRNA-4325p cannot be ruled out.
Proliferation, Migration, and Sprouting of Endothelial Cells in OS are Controlled by microRNA-4325p/PDGFB Signaling Regulation
We found that microRNA-4325p suppressed PDGFB expression (Figure 8A). As a result, to establish if microRNA-4325p/PDGFB signaling influences tumor angiogenesis in OS caused by resveratrol, a proliferation of endothelial cells, recruitment and sprouting were investigated. Transient transfection with control miRNA or microRNA-4325p mimic/inhibitor or resveratrol (20 µM) subsequently 24 or 48 h, endothelial cells were incubated in conditioned medium (EA-hy926 cells) to test their viability, proliferation and sprouting, evaluated by the MTT assay (Figure 8A) and FDA staining (Figure 8B). According to our results, the conditioned medium from transfected OS cells transfected with the microRNA-4325p mimic significantly reduced the proliferation of endothelial cells caused by resveratrol without affecting viability relative to the conditioned medium from transfected OS cells transfected with control miRNAs or microRNA-4325p mimics or resveratrol (Figure 8A). Similarly, the other characteristic of angiogenesis, known as endothelial cell migration, was evaluated using wound scratch assays (Figure 8B). Upon treatment with conditioned medium from either a control microRNA transfection or a microRNA-4325p inhibitor transfection, the findings indicated a discernible slowdown in endothelial cell migration and wound healing due to resveratrol compared to the supplemented medium from a control miRNA transfection or microRNA-4325p inhibitor transfection.

(Top) Endothelial cell (EA. hy926 cells) proliferation is influenced by miR-432-5p/PDGFB signaling in OS. Cell proliferation (A) and viability (B) were evaluated using condition medium prepared from miR-432-5p mimic/inhibitor transfected MG-63 and HOS cells and treated with RST (20 µM). The migration of EA. hy926 cells was assessed. The EA. hy926 cell migration was calculated using scratch wound assays. The results are expressed as the mean and standard deviation. *P < 0.05 and **P < 0.01 indicate a significant decrease in wound closure compared to either the control or miRNA mimic and indicate a significant increase in wound healing compared to the control miRNA. The data were analyzed using one-way ANOVA followed by Tukey's multiple comparison test. (Bottom) Fluorescein Diacetate (FDA) staining was performed to analyze the morphology of the endothelial cells (EA-hy926 cells) to determine the effects of RST. The HOS and MG-63 treated cells were examined for the presence of live cells using FDA staining. Green fluorescence images were taken (20×) as an index of strong enzyme activity in live cells suggesting MiR-432-5p/PDGFB-mediated endothelial cell migration in HOS and MG-63 cells is regulated by miR-432-5p/PDGFB signaling. The scale bar represents 50.0 µm.
Further, an analysis of the sprouting of chick aortic rings was carried out to understand better the endothelial sprouting. In the present study, an aortic ring was made, and resveratrol was added to it in the medium of cells (MG-63 or HOS) transfected with control miRNA or microRNA-4325p mimic or inhibitor. In the quantitative analysis of angiogenesis parameters, such as ring number, size, length, and junction count, we discovered that resveratrol caused a significant reduction in angiogenesis after miRNA mimic transfection compared to control miRNA or inhibitor transfection (Figure 9A to D). It is suggested that the expression of these factors revealed that PDGFB in OS cells controls tumor angiogenesis.

Analyses of aortic ring sprouting in chicks. Twelve-day-old chick embryo aortas were cut into small pieces. Matrigel was placed between the aortas, and conditioned media prepared from MG-63 and HOS cells transfected with miR-432-5p mimic/inhibitor or control miRNA was applied. The number of rings, the length, the size of the tubule, and the number of junctions were quantified and plotted. (A) The number of rings and junctions under treatment was compared to the number of rings and junctions under control transfection; (B) The length and size were also measured and plotted on the graph; (C–D) Representative images of a spheroid sprouting assay (bright-field, magnification 400x) of chick aortic ring incubated for 36 hours with conditioned culture medium of HOS and MG-63 cells with and without miRNAs or resveratrol (20 µM). Scale bar = 500 µM. The results are expressed as the mean ± standard deviation. *P < 0.05 & **P < 0.01 indicate a significant decrease when compared to control and/or inhibitor. Using one-way ANOVA followed by Tukey's multiple comparisons, the data was analyzed.
Regulation of microRNA-4325p/PDGFB Signaling in OS Increases Angiogenesis
In the recent past, a unique model was developed to test the angiogenic potential of tumor cells with chick embryos. 20 Observation of the findings demonstrated that the same pattern of angiogenesis occurred in cells from the OS with similar characteristics. In addition, the length, size, and junction count of blood vessels in OS, MG-63, and HOS cell lines were significantly increased by resveratrol treatment relative to primary osteoblasts.
The CAM model is used to understand how microRNA-4325p/PDGFB signaling regulation contributes to tumor angiogenesis in OS-regulated tumors. First, the vasculature of the CAM was photographed and then Angioquant software was used to perform a study of the angiogenic parameters. 27 Finally, angiogenic parameters were analyzed on the 10th day of angiogenesis, and it was found that the result was ideal. Therefore, in the present study, we determined that microRNA-4325p/PDGFB signaling in OS is regulated by resveratrol using 10 days in the CAM development stage.
After 24 h, a cover slip comprising OS cells was inverted and placed on the CAM vasculature for 10 days. After that, the cover-slip containing either HOS or MG-63 cells was transfected transiently with control miRNA, miRNA mimic, and resveratrol. Angioquant software was used to quantify the angiogenic parameters, and these results were relative to each other. As a result, transfected with microRNA-4325p mimic MG-63 and HOS cells showed increased blood vessel size, length, and junctions count in response to resveratrol treatment (Figure 9), compared with control miRNA (control miRNA and microRNA-4325p transfected cells).
Furthermore, the expression of markers was examined by real-time RT-PCR to understand the role of microRNA-4325p/PDGFB signaling in OS regulation. MG-63 and HOS cells were grown on cover-slips and transfected with control mRNAs or microRNA-4325p mimics or inhibitors and subsequently resveratrol and deposited in the CAM for 10 days. A complete RNA sample was extracted from the CAM vasculature and was subjected to real-time reverse transcription-PCR analysis to search for mRNAs encoding Tie-2, Ephrin-B2, CALD1, TEM8, and CXCR4 (Figure 10). In the present study, we confirm that Tie-2, Ephrin-B2, CALD1, TEM8, and CXCR4 are associated with angiogenesis. 20 According to the expression analysis of markers, the expression of angiogenesis proteins, TEM8, CALD1, and CXCR4 mRNA at the CAM phase was decreased in HOS and MG-63 cells transfected with microRNA-4325p mimic induced by resveratrol relative to miR-4325p inhibitor or control miRNA. The other angiogenic indicators (Tie-2 and Ephrin-B2 mRNAs) were not noticeably changed by resveratrol therapy at the various stages of the CAM procedure. According to these results, resveratrol appears to regulate angiogenesis through microRNA-4325p and PDGFB signaling during the development of OS.

Angiogenesis is promoted in the sprouting phase of CAM by miR-432-5p/PDGFB signaling. After exposure to MG-63 cells or HOS cells containing coverslip, 6-day-old embryos' CAM was excised and subjected to RT-PCR analysis. TEM8, CALD1, CXCR4, Tie-2 and Ephrin-B2 have been identified as angiogenesis-associated genes. The expression of β-actin mRNA was used to normalize the expression of these mRNAs. The results are expressed as the mean ± SD. *P < 0.05 & **P < 0.01 represent a significant decrease when compared to the control or inhibitor. One-way ANOVA followed by Tukey's multiple comparison was used to analyze the data.
Discussion
Resveratrol is known to possess anticancer properties, and these benefits have been seen in both in vitro and in vivo models that simulate various cancers. In addition, it has previously been found that this substance induces the apoptosis of cancer cells in breast, lung, and colorectal tissues.18,25 However, a limited number of studies characterize its anticancer activity on OS, even though a wide variety of herbal supplements containing it are widely available without any regulation. The present study subjected OS cell lines (MG-63 and HOS) to various concentrations (0, 5, 10, 20 µM) of resveratrol for 48 h. The results showed that resveratrol induced apoptosis in HOS cells, as well as inhibiting the proliferation of OS cells in a dose- and time-dependent manner, which is in line with the findings of research carried out by Liu et al 9 and shows that resveratrol can cause apoptosis in OS cells and inhibit their proliferation. In addition, resveratrol increased the expression of miR-139 in MG-63 and HOS cells, and the microRNA-4325p inhibitor reversed the effect of resveratrol on MG-63 and HOS cells, indicating that resveratrol has a function in the induction of apoptosis in HOS and MG-63 cells, which may be related to miR-139.
It has been noted that OS has a high metastatic potential that has been associated with a poor survival rate for patients. Despite this, the intricate link between OS and the microenvironment in which it thrives has not yet been fully understood. For example, autocrine and paracrine secretions have a significant effect in developing cell-to-cell contact within the microenvironment of a tumor. 28 In addition to this, these substances promote angiogenesis, which promotes tumor development and allows metastatic spread.29,30
A variety of growth factors have been implicated in the process of angiogenesis. These molecules—VEGF (vascular endothelial growth factor), fibroblast growth factor-2, and PDGF (platelet-derived growth factor)—are regarded as particularly important. A significant amount of PDGF is secreted by tumor cells and stromal cells, and accumulating evidence implies that PDGF signaling plays a noticeable contribution under angiogenesis both under normal conditions and OS tumor conditions of OS and fibrosarcoma; tumor cells produce the growth factor and its receptor, ultimately resulting in an accelerated rate of tumor progression through autocrine regulation. 31 There is evidence that sequestration of PDGFB by melanoma improves the overall number of pericytes present within the tumor's microenvironment, which displays a higher tumor progression rate. 32 There is evidence that resveratrol prevents tumor growth by phosphorylation of the PDGFR gene and suppresses the angiogenesis triggered by PDGFB, thereby inhibiting the growth of tumors. Most of these expressions are cytoplasmic and, in addition to, tumors. These expressions may also be identified in minor blood vessels and dispersed cells within the tumor and its surroundings.
A significant amount of PDGFB was expressed in tumor cells in our study. Several investigations have revealed that PDGFB affects angiogenesis initiation, although the mechanism by which it does so is not well known. The fact that actions could be performed effectively by triggering the PDGF signaling pathway supports this hypothesis. Furthermore, they might be triggered indirectly by dramatically increasing the synthesis of stromal VEGF. It is crucial to suggest alternate prognostic and treatment options for OS patients. MicroRNAs are essential in cancer progression and may be used as potential biomarkers in the future. There is substantial evidence that microRNAs govern various cancers, including OS.8,19,32
There is a possibility that microRNA-4325p can control the production of other angiogenesis factors through mechanisms that are independent of PDGFB. Angiogenesis is the process of endothelial cells proliferating, migrating, and forming lumens to generate new blood vessels. 33 The angiogenesis pathway is critical for hyperproliferative disorders such as malignancies, hypoxic mice retinas, liver cirrhosis, inflammation, and nephritis.34,35 In our work, the conditioned medium of microRNA-4325p mimic-transfected OS cells inhibited endothelial cell movement, endothelial cells, and sprouting relative to control miRNA or microRNA-4325p inhibitor transfection. This study indicates that microRNA-4325p suppresses angiogenesis in human OS cells through its PDGFB-dependent expression. This investigation further demonstrated that miR-432-5p/PDGFB signaling could restrict endothelial cell growth and sprouting to affect tumor angiogenesis in a model derived from a chick embryo.
The size of the tumor requires the presence of 2 distinct processes for a functional vasculature. Angiogenesis occurs in 2 phases: the emergence of new vessels from either modifying or adapting existing ones and the protrusion of the inner vessel wall into the lumens to generate 2 vessels through one. 34 Therefore, our findings infer that microRNA-4325p signaling regulates angiogenesis during tumor growth, which are supported by an increase in angiogenesis-related expressions of genes both in vitro and in vivo.
Conclusion
In conclusion, we observed that pre-microRNA-432 expression in OS was reduced, and functional studies confirmed this result of microRNA-4325p, suggesting that these 2 microRNAs target PDGFB directly. Furthermore, there is evidence that microRNA-4325p regulates PDGFB signaling in OS, which suppresses tumor angiogenesis, as shown by reduced proliferation of endothelial cells, movement of endothelial cells, and aorta ring development assays. Moreover, miR-432-5p/PDGFB signaling is important to regulate tumor angiogenesis in OS, as shown in chick embryos. In conclusion, we hypothesize that targeting the miR-432-5p/PDGFB signaling pathways in conjunction with other molecules may be a more efficient therapeutic option for controlling the angiogenesis contributed by the OS microenvironment in tumors. This would be accomplished by specifically targeting the mechanisms related to angiogenesis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
Not applicable.
Statement of Informed Consent
Not applicable.
