Abstract
Objectives
This study investigated the Catharanthus roseus phytochemicals by chromatography-mass spectrometry (GC-MS), and in vitro effects on diabetic wound healing based bioassays, molecular docking, and dynamics.
Methods
Fresh C. roseus leaves were extracted using methanol and subjected to preliminary phytochemical screening and GC-MS analysis. The extract was evaluated for antioxidant, anti-inflammatory, antidiabetic, and antimicrobial activities using in vitro assays. Molecular docking and dynamics simulations were performed to analyse interactions of key bioactive compounds with microbial virulence proteins and wound-healing targets.
Results
Phytochemical screening revealed the presence of flavonoids, alkaloids, tannins, saponins, phenols, steroids, carbohydrates, and glycosides. GC-MS identified 63 bioactive compounds, including D-sorbitol, myo-inositol, and palmitic acid etc The extract showed potent antioxidant activity comparable to Vitamin C and significant anti-inflammatory effects via protein denaturation inhibition (89 ± 5.4%, similar to Diclofenac at 91 ± 6.1%). It also inhibited α-amylase and α-glucosidase (69 ± 3.9% and 75 ± 5.4%) respectively, confirming antidiabetic potential. Antimicrobial assays revealed strong inhibition of Streptococcus mutans (MIC: 0.312 mg/mL) and Enterococcus faecalis (0.62 mg/mL). Molecular docking and dynamics revealed that vincristine and vinblastine bind strongly to both microbial virulence proteins and wound-healing targets (VEGF, NF-κB, IL-10), suggesting dual antimicrobial and pro-healing potential. Vincristine showed the highest affinity (–8.9 kcal/mol), indicating stronger therapeutic relevance in diabetic wound repair.
Conclusion
These findings highlight C. roseus bioactive compounds as a promising therapeutic candidate for diabetic wound healing via antioxidant, anti-inflammatory, antidiabetic and antimicrobial activities.
This is a visual representation of the abstract.
Introduction
Permanent hyperglycaemia is a hallmark of diabetes mellitus, a chronic metabolic condition that can cause serious side effects, such as poor wound healing, neuropathy, nephropathy, and cardiovascular disorders, if untreateted. 1 The incidence of diabetes is on the rise worldwide, both developing and developed countries, where a growing number of cases are linked to high mortality rates and vascular complications.2,3 About 10% of people with diabetes develop diabetic foot ulcers (DFUs) at some point during their illness, making them a serious concern among these complications.4,5 People with diabetes type 1 and type 2 are more likely to sustain chronic wounds because of immunological dysregulation, prolonged inflammation, and poor angiogenesis, which all contribute to delayed tissue repair.6,7
Homeostasis, inflammation, proliferation, and remodelling are all components of the intricately coordinated process of wound healing. 8 A successful wound healing response requires these stages to occur in the correct sequence and within an appropriate timeframe. 9 In patients with diabetes there is severe disruption of this process, resulting in prolonged inflammation, oxidative stress, and inadequate collagen deposition, ultimately leading to chronic wounds. 10 Vascular endothelial growth factor (VEGF) plays a crucial role in angiogenesis, promoting new blood vessel formation and tissue repair. 11 However, diabetes is associated with reduced VEGF expression, impairing neovascularization and contributing to chronic wound pathology.12,13 Recent studies highlight that modulating the HIF-1α/VEGF pathway, by inhibiting miR-217 can enhance angiogenesis and accelerate wound healing in diabetic ulcers. 14
Bioactive phytochemicals with antibacterial, anti-inflammatory, wound healing, and antioxidant qualities are abundant in medicinal plants. Plant based sources are account for more than 25% of contemporary medications, demonstrating their potential to provide accessible and affordable treatments. 15 Catharanthus roseus L. (Apocynaceae), also referred to as Vinca rosea or Madagascar periwinkle, is a medicinal subshrub that is well-known for its pharmacological qualities, especially in the treatment of wounds and diabetes.16,17 There are two flower-coloured variants of this plant, C. roseus (pink) and C. alba (white), identified in Mesopotamian records. C. roseus, which is well-known for their high alkaloid content, especially staphyloxanthin, vinblastine and vincristine, and essential to worldwide medical care. 18 The glossy leaves, pink-to-white flowers, and paired follicle fruits of C. roseus, an evergreen subshrub, grow up to 1 meter tall. 19 The plant's leaf extracts have shown high antibacterial action, especially against Salmonella typhimurium, Pseudomonas aeruginosa, and Staphylococcus aureus, indicating that they may help prevent several infections. 20 Compared to C. alba (white flower), an ethanolic root extract of C. roseus showed a higher antioxidant capacity, with benefits growing with concentration. 21 It is used to treat cancer, particularly childhood leukaemia, because of alkaloids like vincristine and vinblastine. 22 This plant also helps heal wounds and treats skin disorders,23,24 insulin resistance, and high cholesterol levels, tissue regeneration and tissue repair. 10 Also, lowers blood glucose, improves wound healing, and increases tensile strength and hydroxyproline. 25 By lowering triglycerides, total cholesterol, VLDL-c, and LDL-c and enhancing the structural characteristics of the heart, liver, and kidney, C. roseus extract from leaves exhibited strong anti-atherosclerotic properties. 26 Flower extract greatly increased tissue weight, may be used topically to treat wounds. 27 Our study aims to identify the key phytochemicals present in C. roseus extract, and to evaluate its antioxidant, anti-inflammatory, and antimicrobial activities, and to analyse molecular interactions with target proteins involved in antimicrobial and wound healing. The findings may contribute to the development of optimized, plant-based wound care treatments, bridging the gap between traditional medicine and modern therapeutic applications.
Materials and Methods
Chemicals and Reagents
All chemicals and reagents were of analytical grade. Methanol (HPLC grade) was obtained from Merck (USA), and standard phytochemical reagents, including Mayer's, Dragendorff's, Wagner's reagents, ferric chloride, lead acetate, sodium hydroxide, and concentrated sulfuric acid, and Mueller–Hinton agar and broth were from Hi-Media Laboratories (Mumbai, India). DPPH was purchased from Nanjing Duly Biotech Co., Ltd (Nanjing, China). BSA, α-amylase, α-glucosidase, p-nitrophenyl-α-D-glucopyranoside, diclofenac sodium, and ascorbic acid were purchased from Sigma-Aldrich (USA). Streptococcus mutans ATCC-25175, Enterococcus faecalis ATCC-14506, Pseudomonas aeruginosa ATCC- 15442, and Staphylococcus aureus ATCC – 25923, were used as bacterial strains. Protein structures for docking were retrieved from the Protein Data Bank, ligands from PubChem, and analyses were performed using AutoDock Vina, GROMACS, and related bioinformatics tools.
Extract Preparation
Fresh leaves of C. roseus were collected, washed, and shade-dried. The dried material was powdered and subjected to extraction using methanol by maceration extraction. The extract was concentrated under reduced pressure using a rotary evaporator and stored at −20 °C for further analysis (Figure 1).

Sequential process of Catharanthus roseus plant extraction.
Phytochemical Screening
Phytochemical investigations were performed to verify the existence of different chemicals in plant extracts. Whereas flavonoids created a green precipitation with 10% ferric chloride, alkaloids produced a reddish-brown precipitate with Wagner's reagent. 5% ferric chloride caused phenols to become blue-black or dark green. 28 A brown ring at the contact was used to identify glycosides using glacial acetic acid, FeCl₃, and H₂SO₄.29,30 With chloroform and H₂SO₄, terpenoids and steroids produced a reddish-brown hue. 31 When combined with ferric chloride, tannins were seen as either blue (gallic) or greenish-black. 32 After shaking with water, saponins created a 1 cm layer of foam.29,33 Proteins were coloured purple with ninhydrin, whereas carbohydrates created a violet ring with sulphuric acid and α-naphthol.34–36
GC-MS Analysis
GC/MS analysis was performed using an Agilent 5977B system equipped with a DB5-MS column (29 m × 0.25 mm × 0.25 µm) and a 10 m DuraGard column. The oven temperature was ramped from 60 °C to 325 °Cover 37.5 min, with helium as the carrier gas at a constant flow rate of 1 mL/min. Sample injection was performed at 250 °C under both split less conditions (helium purge 10.5 mL/min for 1 min) and split mode (1:10, split flow 10.3 mL/min). The MSD was set to a source temperature of 230 °C and a quadrupole temperature of 150 °C, scanning a mass range of 50-600 u after a 5.90 min solvent delay. Retention times were standardized using d27-myristic acid with Agilent's RTL system for consistency. Metabolite identification was carried out using the NIST metabolomics library. 37
Antioxidant Activity-DPPH Radical Scavenging Assay
The scavenging activity of the DPPH radical was evaluated according to the method described by Rajesh and Natvar (2011).
38
Briefly, DPPH solution was prepared in 3.3 mL of methanol and protected from light using aluminium foil. Different concentration of the extracts were individually mixed with 150 µL of the DPPH solution, and the final volume was adjusted to 3 mL with methanol. After incubation for 15 min at room temperature in the dark, the absorbance was measured at 517 nm using a Shimadzu UV-1601 UV–visible spectrophotometer, and methanol was used as the blank. The following formula was used to calculated inhibition percentage:
Anti-inflammatory Activity by Protein Denaturation Assay
A volume of 1 ml of extracts or of diclofenac sodium at different concentrations 0.025, 0.05, 0.1, 0.15 and 0.2 mg/mL was homogenised with 1 ml of aqueous solution of bovine serum albumin (5%) and incubated at 27 °C for 15 min. The mixture of distilled water and BSA constituted the control tube. Denaturation of the proteins was caused by placing the mixture in a water bath for 10 min at 70 °C. The mixture was cooled at room temprature, and the activity each mixture was measured at 660 nm.
39
Each test was done in three times. The following formula was used to calculated inhibition percentage:
Anti-diabetic Activity: α-Amylase & α-Glucosidase Inhibitory Activity
The α-Amylase inhibitory activity was determined using the 3,5-dinitrosalicylic acid (DNS) method. Briefly, a reaction mixture containing phosphate buffer, α-amylase enzyme, and varying concentrations of the plant extract were pre-incubated under controlled conditions. The reaction was initiated by the addition of starch substrate and incubated to allow enzymatic hydrolysis. The enzymatic reaction was terminated by the addition of DNS reagent, followed by heating to develop colour. After cooling, the optical density was measured spectrophotometrically, and extent of α-amylase inhibition was calculated. The α-Glucosidase inhibitory activity was evaluated by pre-incubating a mixture of phosphate buffer, α-glucosidase enzyme, and plant extract. The reaction was initiated by adding p-nitrophenyl-α-D-glucopyranoside (p-NPG) as the substrate and incubated for a defined period. The reaction was terminated by the addition of sodium bicarbonate, and the absorbance of the released p-nitrophenol was measured spectrophotometrically. Acarbose was used as the standard reference inhibitor in both assays, and percentage inhibition was calculated in comparison with the control. 40
Antimicrobial Activity: Determination of Minimum Inhibitory Concentration (MIC)
The minimum inhibitory concentration (MIC) of C. roseus was determined using the micro dilution technique at concentrations ranging from 10 mg/mL to 0.019 mg/mL and 23,5-triphenyltetrazolium chloride (TTC) was used as the viability indicator. Viable bacterial cells reduce the colourless TTC to a red-coloured insoluble formazan. After incubation, wells showing no colour development indicated complete inhibition of bacterial growth and were considered as MIC.
Molecular Docking
Protein–ligand interactions were investigated using molecular docking analysis. Initially, the three-dimensional structure of the target protein was retrieved from the Protein Data Bank (PDB) and prepared for docking analysis. Ligand preparation (staphyloxanthin (STX), vinblastine (VBL), and vincristine (VCR)) involved obtaining the three-dimensional structures either from the PubChem database or generating them using appropriate computational tools. During protein preparation, only polar hydrogen atoms were added to facilitate accurate intermolecular interactions. For ligand preparation, Gasteiger charges were assigned and non-polar hydrogen atoms were added. Rotatable bonds within the ligand molecules were identified and optimized, after which each prepared ligand was individually uploaded into the docking software. Molecular docking simulations were performed using AutoDock version 1.5.6, and the resulting protein–ligand interactions were analyzed and visualized in both two- and three-dimensional formats using BIOVIA Discovery Studio. 42
Molecular Dynamics
Molecular dynamics (MD) simulations were performed using the GROMACS software package, with additional computational support provided by the WebGRO server (https://simlab.uams.edu/). The docked protein–ligand complex was separated into receptor (protein) and ligand components to facilitate system preparation. Protein topology files were generated in GROMACS using the AMBER99SB-ILDN force field, while ligand topology parameters were obtained using the PRODRG server. Subsequently, the individual topologies were combined to reconstruct the protein–ligand complex for simulation. The complex was placed in a dodecahedral simulation box with a minimum distance of 1.0 nm between the complex and the box boundaries. The system was solvated using the TIP3P water model, and electrostatic neutrality was achieved by the addition of 0.15 M NaCl. MD simulations were carried out for 10 ns under standard conditions using the GROMACS command-line interface on an Ubuntu operating system. Post-simulation analyses were performed using the generated trajectory (XTC) files to evaluate the root mean square deviation (RMSD) and root mean square fluctuation (RMSF). These analyses were used to assess the conformational stability and dynamic behaviour of the protein–ligand complex throughout the simulation period, thereby confirming the structural integrity of the complex under dynamic conditions.
Statistical Analysis
The data were statistically assessed using one-way analysis of variance (ANOVA) and Duncan's multiple comparison test using computerized software (SPSS 7.5 using Windows student version) to determine the significance of difference between different groups. The experiment was conducted with a significance criterion of p < 0.05.
Results
Phytochemical Analysis
The phytochemical analysis of C. roseus shows the presence of several bioactive compounds that aid diabetic wound healing (Figure 2 and Table 1). Flavonoids, saponins, tannins, phenols, and carbohydrates are abundant. Alkaloids, steroids, and glycosides are moderately present. Terpenoids and proteins are either absent or present in negligible amounts. These compounds are known to work together to enhance healing by reducing oxidative stress, fighting infections, and supporting tissue repair.

Various types of phytochemicals found in the methanol extract of C. roseus. 1. Proteins, 2. Carbohydrates, 3. Flavonoids, 4. Phenols, 5. Terpenoids, 6. Glycosides, 7. Alkaloids, 8. Saponins, 9. Tannins and 10. Steroids.
Preliminary Phytochemical Study of C. roseus Methanol Extracts.

Phytochemicals were represented by the symbols “+, ++, and +++” to indicate their presence low, moderate, and high, whereas “-“denoted their absence.
GC-MS Analysis of a Methanolic Extract of Catharanthus Roseus
GC–MS analysis of the methanolic extract of Catharanthus roseus revealed the presence of multiple bioactive constituents. The identification of compounds was performed by comparing the obtained mass spectra with those available in the NIST mass spectral library. Several major constituents were detected, including D-fructose, 1,3,4,5,6-pentakis-O-(trimethylsilyl)-, O-methyloxime, D-galactose, D-(+)-talose, D-sorbitol, myo-inositol, and palmitic acid. The reported match or similarity indices for these compounds were high hit score. Our preliminary phytochemical test showed the presence of alkaloids and many literature explored that staphyloxanthin, vinblastine and vincristine were abundant in C. roseus, however our GC–MS conditions not amenable to these phytochemicals. The identified compounds are known to exhibit diverse biological activities relevant to diabetic wound healing. Sugars and sugar alcohols, such as fructose, galactose, sorbitol, talose, and myo-inositol, play important roles in cellular energy metabolism, osmotic balance, and tissue hydration, thereby supporting wound repair processes. Palmitic acid contributes to membrane integrity and may influence inflammatory signaling. Collectively, these bioactive constituents suggest that the methanolic extract of C. roseus possesses significant antioxidant and anti-inflammatory potential. The extract may promote diabetic wound healing by reducing NF-κB-mediated inflammatory responses, enhancing angiogenesis through VEGF regulation, and facilitating tissue regeneration. The GC-MS chromatogram and bioactive compounds present in the extracts were summarized in Figure 3 and Table 2.
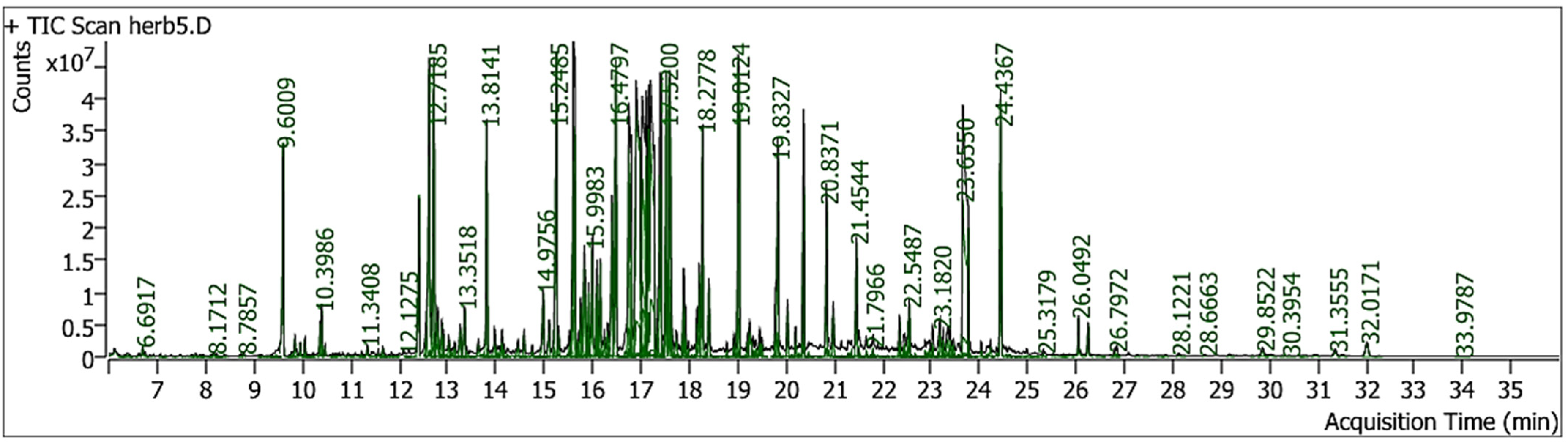
GC-MS chromatogram of methanolic extract of the C. roseus.
Bioactive Compounds Identified from GC-MS Analysis of Methanolic Extract of C. roseus.

Effects of Antioxidant DPPH Activity of Extract
The DPPH radical scavenging assay demonstrated a marked, concentration-dependent antioxidant activity of the C. roseus extract (Figure 4). The percentage of inhibition increased from approximately 16% at the lowest tested concentration to about 79% at 0.2 mg/ml, indicating a strong free radical–quenching ability. This activity was comparable to the standard antioxidant Vitamin C, which showed 86% inhibition at the same concentration. The close similarity in scavenging efficiency at higher doses highlights the potent antioxidant potential of the extract, suggesting its effectiveness in reducing oxidative stress, a key factor impairing tissue repair and diabetic wound healing (Table S.T1).

Antioxidant activity of the extract assessed by the DPPH radical scavenging assay. The extract exhibited strong free-radical scavenging activity comparable to the standard, as determined by percentage inhibition of DPPH. Data are expressed as mean ± SD of triplicates, and statistical significance was considered at p < 0.05, and a, b, c & d represents significance difference among different concentrations.
Effects of Anti-Inflammatory Activity of Extract
The anti-inflammatory activity assessed by the protein denaturation assay showed a strong concentration-dependent inhibitory effect of the extract (Figure 5). Inhibition increased steadily from approximately 22% at 0.025 mg/ml to about 89% at 0.2 mg/ml, demonstrating effective prevention of protein denaturation. This response was comparable to the standard diclofenac, which exhibited inhibition ranging from ∼28% at the lowest concentration to 91% at 0.2 mg/ml. At higher concentrations (0.1-0.2 mg/ml), the extract showed no significant difference from the standard drug, indicating potent anti-inflammatory efficacy. These findings suggest that the extract possesses strong anti-inflammatory potential, supporting its therapeutic relevance in managing inflammation during diabetic wound healing (Table S.T2).

Protein denaturation assay showing the anti-inflammatory activity of C. roseus extract at various concentrations. Values are expressed as mean ± SD of triplicates. Statistical significance was determined at p < 0.05, and a, b, c & d represents significance difference among different concentrations.
Effects of α-Amylase Inhibition Activity of Extract
The α-amylase inhibitory assay revealed that the extract exhibited a clear concentration-dependent antidiabetic effect (Figure 6). The percentage inhibition increased from approximately 19% at 0.1 mg/ml to about 69% at 0.5 mg/ml, indicating its ability to suppress carbohydrate-digesting enzyme activity. Although the inhibitory effect was slightly lower than that of the standard drug acarbose, which showed inhibition ranging from ∼20% to 80% over the same concentration range, the extract demonstrated statistically significant activity at higher doses. These findings suggest that the extract can effectively slow carbohydrate digestion and postprandial glucose release, supporting its potential role in glycemic control and the management of diabetes-associated wound healing (Table S.T3).
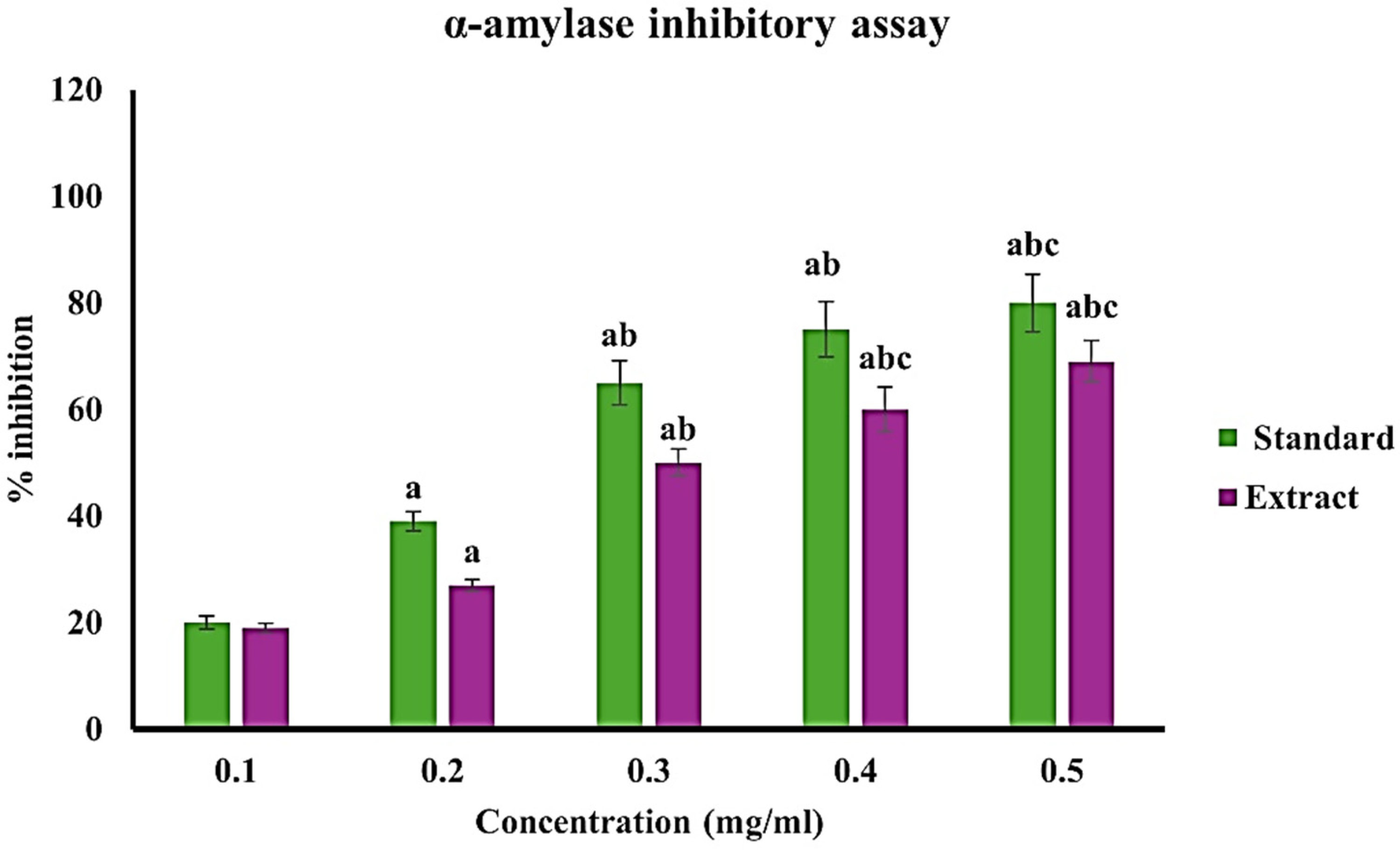
α-amylase inhibitory activity of C. roseus extract at various concentrations. Values are expressed as mean ± SD of triplicates. Statistical significance was determined at p < 0.05, and a, b, & c represents significance difference among different concentrations.
Effects of α-Glucosidase Inhibition Activity of Extract
The α-glucosidase inhibition assay demonstrated a significant, dose-dependent antidiabetic activity of the extract (Figure 7). The extract inhibited α-glucosidase from approximately 16% at 0.1 mg/ml to about 75% at 0.5 mg/ml, indicating effective suppression of intestinal carbohydrate-digesting enzymes. Although its activity was marginally lower than the standard drug acarbose, which showed inhibition ranging from ∼18% to 79% over the same concentration range, the extract exhibited comparable efficacy at higher doses. These findings suggest that the extract can effectively delay carbohydrate breakdown and glucose absorption, contributing to improved glycemic control and supporting its potential role in the management of diabetes-associated wound healing (Table S.T4).
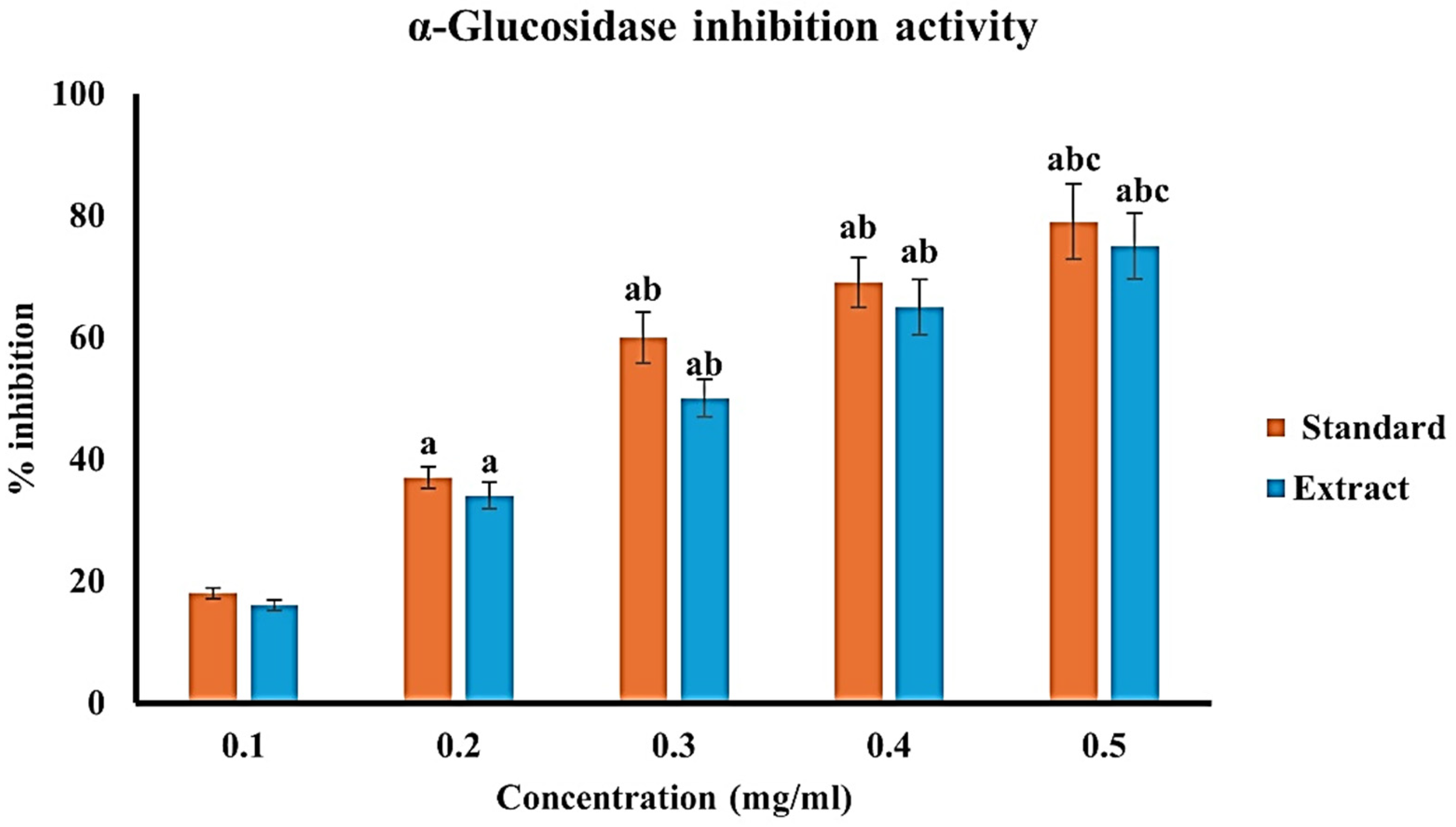
α-glucosidase inhibitory activity of C. roseus extract at various concentrations. Values are expressed as mean ± SD of triplicates. Statistical significance was determined at p < 0.05, and a, b, & c represents significance difference among different concentrations.
Minimum Inhibitory Concentration
The antimicrobial effects of C. roseus extract demonstrated in table 3. Strong antibacterial activity was observed against S. mutans and E. faecalis at higher concentrations (10 to 0.62 mg/mL). At 0.312 mg/mL, S. mutans exhibit no growth, while E. faecalis growth was observed in this concentration. At lower concentrations (0.156 mg/mL and below), both bacterial strains exhibit growth, showing the extract's loss of antimicrobial activity due to less concentration of bioactive compounds. The extract had no effect on P. aeruginosa and S. aureus. The minimum inhibitory concentrations were 0.312 mg/mL for S. mutans and 0.62 mg/mL for E. faecalis.
Anti-bacterial activity of methanol extracts of C. roseus. Concentrations (mg/mL), (-) no Bacterial Growth and (+) Bacterial Growth.

In-silico Analysis - Docking Studies with Microbial Proteins
According to docking studies, the microbial enzymes glycosyltransferase, glucosidase, mannosidase, elastase, and serine protease are firmly bounded by vincristine and vinblastine with binding energies ranging from −7.4 to −9.7 kcal/mol. These interactions imply that protease-mediated tissue injury and bacterial glucose metabolism are inhibited. The chemicals improve VEGF-mediated angiogenesis and indirectly modify TNF-α by decreasing microbial pathogenicity and excessive protease activity. Therefore, under diabetic circumstances, C. roseus has both antibacterial and wound-healing properties (Table 4).
Vinblastine and Vincristine, Bioactive Compounds of C. roseus, Molecularly Target with Bacterial Proteins.

Docking Studies with Wound Healing Proteins
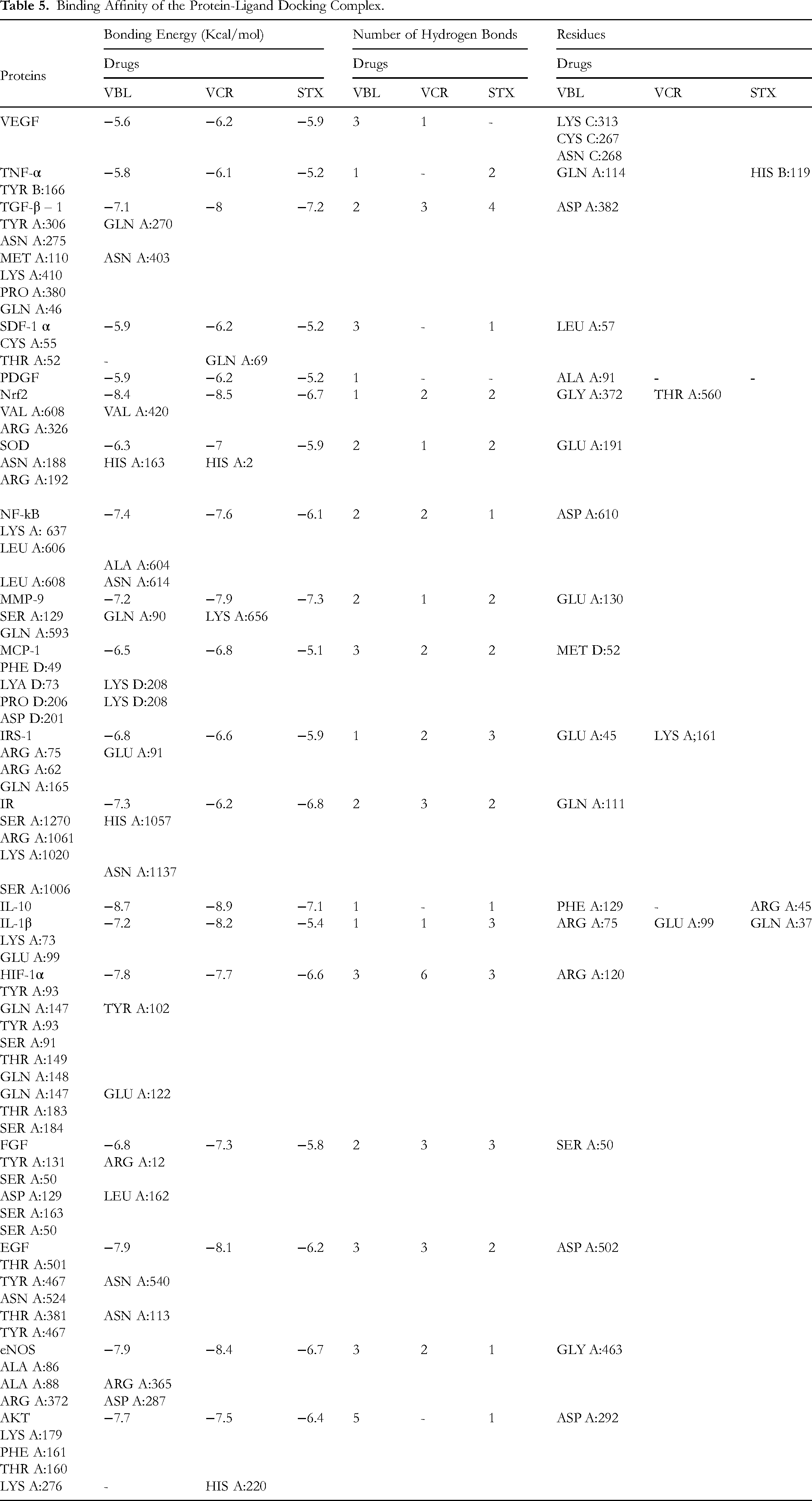
STX, VBL, and VCR exhibited notable binding affinities toward key proteins involved in angiogenesis, inflammation, insulin signalling, and wound repair, including AKT, VEGF, EGF, eNOS, FGF, HIF-1α, IL-1β, IL-10, IR, IRS-1, MCP-1, MMP-9, NF-κB, PDGF, SDF-1α, SOD, TGF-β1, and TNF-α. STX showed moderate binding energies ranging from −5.1 to −7.3 kcal/mol and formed one to three hydrogen bonds per protein, primarily involving ARG, LYS, and GLN residues, contributing to stable protein–ligand interactions. In comparison, VBL demonstrated stronger binding affinities (−5.6 to −8.7 kcal/mol) and formed conventional hydrogen bonds with proteins such as IL-10, AKT, and HIF-1α, involving key residues including LYS, ASP, GLU, and TYR. VCR exhibited the highest interaction stability, forming up to six hydrogen bonds with targets such as TGF-β1, EGF, and Nrf2, mediated by residues including ASN, SER, GLN, and ARG. Collectively, these results indicates that bioactive compounds from C. roseus, particularly vinblastine and vincristine, possess strong multi-target binding potential, supporting their therapeutic relevance in modulating inflammatory, angiogenic, and insulin-related pathways associated with diabetic wound healing (Table 5 and Figures S.5–7).
Binding Affinity of the Protein-Ligand Docking Complex.
Molecular Dynamics
Molecular dynamics (MD) simulations conducted for 10 ns further confirmed the stability of the identified bioactive compounds against key diabetic wound-related targets. The MMP-9–STX complex exhibited a stable RMSD trajectory (∼0.14-0.17 nm) throughout the simulation, with minimal deviations, suggesting strong conformational stability. Similarly, the Serine Protease–VBL complex displayed a consistently low RMSD (∼0.11-0.13 nm), further validating the structural integrity of the binding interaction. RMSF analysis supported these observations, revealing only minor fluctuations in most residues (<0.2 nm), with higher flexibility limited to loop and terminal regions. In MMP-9, local fluctuations reached up to 2.5 nm in selective segments, yet the binding site residues remained stably anchored, while the Serine Protease complex showed negligible flexibility with fluctuations remaining below 0.3 nm. These findings collectively indicate that C. roseus phytoconstituents form dynamically stable interactions with diabetic wound-associated targets, modulating TNF-α/VEGF signalling to enhance wound repair mechanisms and suppress pathogen-associated proteolytic activity (Figure 8).
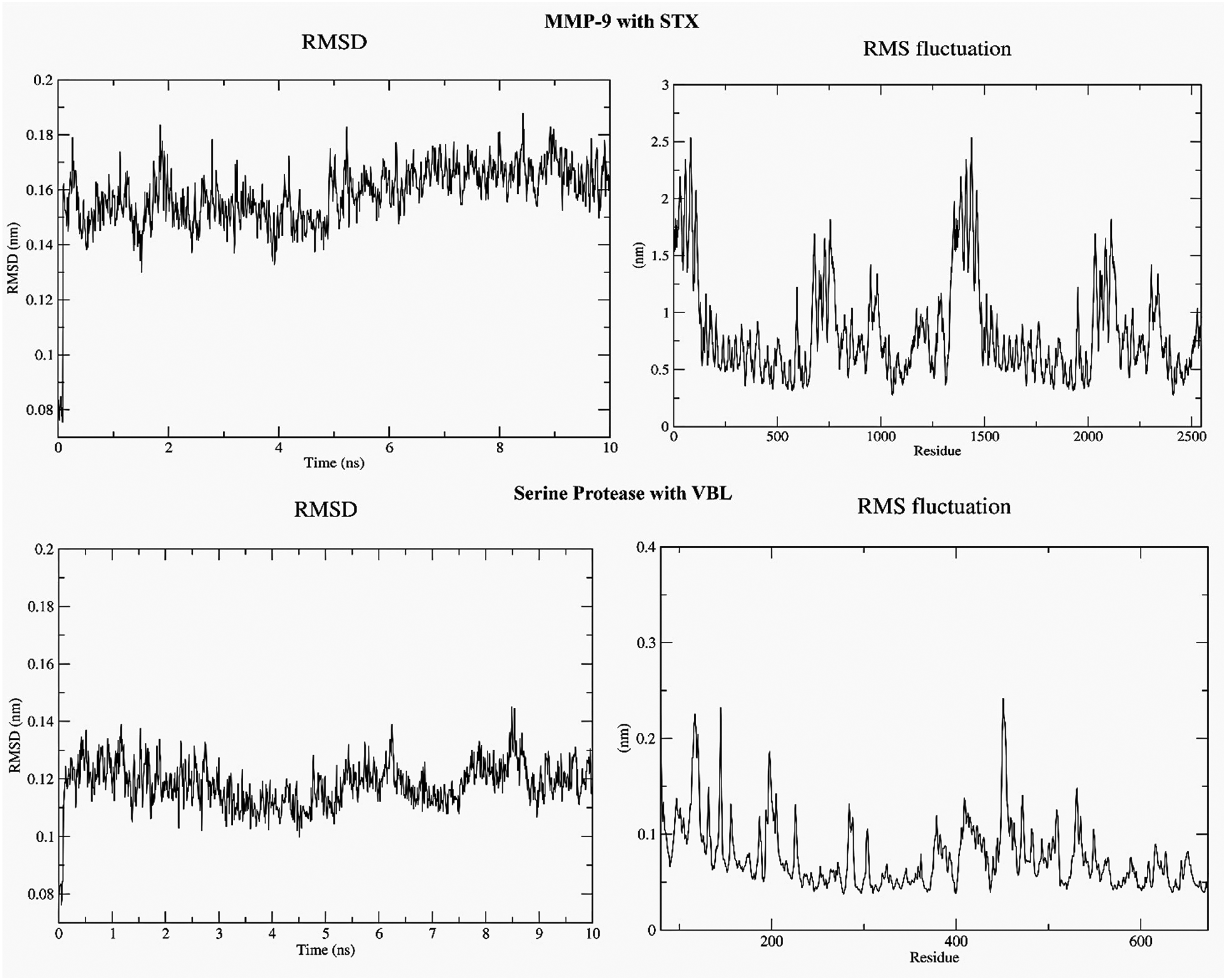
RMSD and RMSF plots of MMP-9 with STX and serine protease with VBL over 10 ns md simulations, showing stable protein–ligand interactions. Minor residue fluctuations were confined to loop regions, confirming complex stability in regulating TNF-α/VEGF-mediated diabetic wound healing.
Discussion
Wound healing is a complex and dynamic process that involves the interplay of various cell types, signalling molecules, and extracellular matrix components. In diabetic conditions, this repair mechanism is markedly impaired due to chronic inflammation, oxidative stress, and poor angiogenesis, often resulting in non-healing ulcers and amputations. 41 Although herbal ointments have shown promise in reducing amputation rates in diabetic wounds, 42 systematic research is needed to validate their efficacy and establish molecular mechanisms. In this context, the present study investigated the therapeutic potential of C. roseus in diabetic wound healing, comparing its bioactive properties with previous reports. Phytochemical profiling of C. roseus extracts revealed the presence of alkaloids, flavonoids, tannins, saponins, carbohydrates, phenols, steroids, and glycosides, compounds well-known for their pharmacological relevance (Figure 2 and Table 1). These findings align with earlier studies,43–46 which documented more than 400 alkaloids in this plant, including vinblastine and vincristine, with established pharmacological and anticancer effects. Previous studies have demonstrated that C. roseus exhibits part-specific phytochemical enrichment with relevance to metabolic and angiogenic regulation. Leaf extracts predominantly contain terpenoids such as phytol, while flower extracts are enriched with fatty acid esters, including D-sorbitol, myo-inositol, and palmitic acid. 47 These metabolites have been associated with antioxidant, anti-inflammatory, and metabolic regulatory effects, which are critical in diabetes-associated vascular impairment. Furthermore, Amna Parveen et al (2019) 48 reported that natural products can interact with VEGF-promoting factors through multiple signalling pathways, supporting the role of plant-derived compounds in modulating angiogenesis-linked metabolic dysfunction. Phenolic and alkaloid constituents such as vincristine and vinblastine have also been reported to influence angiogenesis and inflammatory signaling, 49 processes closely linked to VEGF-mediated vascular repair in diabetic conditions. Previous literature reports part-specific phytochemical enrichment in C. roseus, with leaf extracts rich in terpenoids such as phytol (57.47%) and flower extracts enriched in fatty acid esters, including palmitic acid (34.65%) and methyl 9-octadecanoate (29.88%).50,51 Methanolic leaf extract profiling further identified D-sorbitol, myo-inositol, and palmitic acid, along with phenolics and alkaloids such as staphyloxanthin, vincristine and vinblastine. The presence of these VEGF-associated and metabolically relevant compounds supports the conclusion that C. roseus may exert antidiabetic effects through regulation of VEGF-linked signalling pathways.
Oxidative stress is one of the major contributors to delayed wound healing in diabetes. C. roseus extracts exhibited strong antioxidant capacity in a dose-dependent manner. Previous research reported 55% DPPH inhibition for leaf extract in ethanolic solvent.50,52,53 Unlike prior studies, our research provides a direct comparison with vitamin C, underscoring the plant's potential for diabetic wound care. Extracts of C. roseus leaves and flowers have demonstrated dose-dependent hypoglycaemic effects, enhancing liver glucose utilization.54–56 These extracts showed significant hypoglycaemic, hypolipidemic, and anti-hyperglycaemic properties by improving glucose metabolism, enzymatic activity, insulin levels, and sensitivity in diabetic rats.53,55 The DPPH radical scavenging activity increased from 16 ± 0.9% to 79 ± 4.6%, values comparable to Vitamin C (86 ± 6.2%) (Figure 4). These results are consistent with earlier reports, 57 which demonstrated similar free-radical scavenging activity. In addition, C. roseus extracts improved antioxidant enzyme activity (CAT, SOD, GPx, GST) and enhanced glycogen storage, highlighting its role in mitigating oxidative stress and supporting metabolic balance.
The C. roseus extracts exhibited concentration-dependent anti-inflammatory and protein denaturation inhibition activities, with the composite showing slightly lower effectiveness than diclofenac.58,59 In the present study, the anti-inflammatory potential of C. roseus was further confirmed through protein denaturation inhibition assays. The extract demonstrated a concentration-dependent effect, with inhibition values ranging from 22 ± 1.7% to 89 ± 5.4%. At higher concentrations (0.15-0.2 mg/ml), activity was comparable to diclofenac, a standard anti-inflammatory drug (Figure 5). These findings suggest that C. roseus effectively reduces inflammation, a key pathological barrier in diabetic wound repair. In addition, moderate inhibitory activity exhibited against both α-glucosidase and α-amylase enzymes. At a concentration of 0.5 mg/ml, α-amylase inhibition reached 69 ± 3.9%, suggesting their potential in controlling postprandial glucose spikes, while α-glucosidase inhibition increased to 75 ± 5.4%, indicating a role in reducing glucose absorption in the small intestine (Figure 6 & 7). These findings highlight the plant's potential in managing blood sugar levels. The antimicrobial activity of C. roseus was also evident, with methanolic extracts showing strong inhibitory activity against E. faecalis (MIC: 0.62 mg/mL) and S. mutans (MIC: 0.312 mg/mL), however no significant effects was observed against P. aeruginosa and S. aureus (Table 3). These results are consistent with earlier research57,60 that showed C. roseus had antibacterial activity against B. subtilis, E. coli, and S. typhi. However, at 20%–40% concentrations, moderate inhibition against S. aureus and E. coli, as reported by Kabesh et al (2015). 61 Importantly, our study demonstrated that the antibacterial effects of C. roseus are concentration-dependent, adding clarity to earlier findings.
Many studies reported that staphyloxanthin, vincristine and vinblastine compounds abundant in C. roseus, thus we performed docking analysis with these compounds. The results demonstrated strong hydrogen bond interactions with active site residues and high binding affinity to the target proteins. Compounds from C. roseus, such as vinblastine, ajmalicine, vindolinine, and reserpine, have the potential to control hypertension and diabetes by modifying HPA activity.62,63 C. roseus compounds were found to directly bind to several proteins, including AKT, VEGF, EGF, eNOS, FGF, HIF-1α, IL-1β, IL-10, IR, IRS-1, MCP-1, MMP-9, NF-κB, PDGF, SDF-α, SOD, TGF-β1, and TF-α. IL-10, TGF-β1, EGF, eNOS, MMP-9, and Nrf2 with strong hydrogen bonds. Effective interactions were shown by compounds with binding energies, including staphyloxanthin, vinblastine, and vincristine. Compared to other compounds, vincristine showed the strongest binding, particularly with IL-10, and formed stable interactions through multiple hydrogen bonds, with key residues such as GLU, ARG, ASN, and TYR playing a crucial role. These interactions highlighted the compounds’ potential to modulate growth factors and inflammatory pathways, which makes them potential therapies for diabetic wound healing (Tables 4 & 5). In vivo studies further supported these findings, showing that the ethanol extract of C. roseus accelerated wound healing in diabetic rats by enhancing angiogenesis, epithelization, antimicrobial activity, and tensile strength. It also improved biochemical markers such as hexuronic acid, hydroxyproline, hexosamines, lysyl oxidase, and tissue protein levels, indicating its potential as a topical therapeutic.10,27 MD simulations confirmed the stability of these interactions, with the MMP-9–STX complex maintaining RMSD values around ∼0.14–0.17 nm, while the Serine Protease–VBL complex displayed even greater stability (∼0.11-0.13 nm). RMSF analysis revealed that fluctuations were limited mainly to loop and terminal regions, while residues at the binding sites remained stable. These results suggest that C. roseus phytochemicals can effectively suppress proteolytic activity associated with diabetic wound pathogens. Collectively, these findings highlight their role in regulating the TNF-α/VEGF signalling axis to promote angiogenesis and wound repair. Overall, C. roseus exhibits significant therapeutic potential in diabetic wound healing via antioxidant, anti-inflammatory, and antidiabetic properties. Its effects are reinforced by enzyme inhibition, antimicrobial activity, and molecular-level interactions with wound-healing targets. Nevertheless, further studies needed to confirm the safety and efficacy in humans. Future research should prioritize well-designed trials to validate these findings and fully establish their therapeutic applications.
Limitation of the Study
Despite promising in vitro and in silico results, this study has certain limitations. Docking and short-term molecular dynamics simulations provide predictive insights but may not fully represent the complex diabetic wound microenvironment. The findings are limited to in vitro assays, with no in vivo wound-healing validation. Although vincristine and vinblastine showed strong target affinity, cytotoxicity and dermal safety evaluations need to be evaluate. Further in vivo, safety, and mechanistic studies are required to establish clinical relevance.
Conclusion
C. roseus shows remarkable potential in diabetic wound healing due to diverse phytochemical profile, including alkaloids, flavonoids, and tannins. The bioactive compounds, implicated in modulating the NF-κB and VEGF pathways. These metabolites exhibited strong antioxidant, anti-inflammatory, and antimicrobial properties, which collectively promote angiogenesis, reduce inflammation, and accelerate tissue regeneration. Molecular docking studies further supported its therapeutic role by demonstrating interactions with key wound-healing proteins, indicating its capacity to regulate oxidative stress, improve glucose metabolism, and enhance wound closure. Specifically, vincristine and vinblastine showed strong binding with microbial enzymes, suggesting inhibition of pathogen virulence factors and modulation of TNF-α and VEGF signalling. Together, these findings highlight C. roseus as a cost-effective natural candidate for diabetic wound management, though further investigations essential to validate its safety and therapeutic efficacy in humans.64,65
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261433097 - Supplemental material for Phytochemical and Biological Investigation of Catharanthus roseus: Diabetic Wound-Healing Approach
Supplemental material, sj-docx-1-npx-10.1177_1934578X261433097 for Phytochemical and Biological Investigation of Catharanthus roseus: Diabetic Wound-Healing Approach by Mohammad A. Alshuniaber, Magdi A. Osman and Rajapandiyan Krishnamoorthy in Natural Product Communications
Footnotes
Acknowledgments
The authors thank the Ongoing Research Funding Program, (ORF-2026-1226), King Saud University, Riyadh, Saudi Arabia, for funding this research.
Ethics Approval and Informed Consent
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for Publication
Not applicable
Authors’ Contribution
MA; Project concept developed, Funding and supervising, MO; data validations, Original Manuscript writing and review, RK: Experimental, data validations, Original Manuscript writing and review,
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ongoing Research Funding Program, (ORF-2026-1226), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used during the current study available from the corresponding author on reasonable request
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


