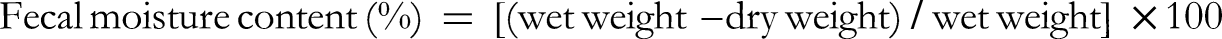Abstract
Constipation is a multifactorial health problem resulting from systemic or neurologic disorders including medication. Peach blossom, an edible flower, could promote defecation, urination, and improve abdominal pain. However, the active components and mechanisms of action remain unclear. In the present study, we established a loperamide-induced mice constipation model to validate the anti-constipation activity of peach blossom. Some classical biomarkers for constipation, including fecal water content, gastrointestinal transit ratio, and fecal pellet weight were used to evaluate the efficacy of peach blossom. Then, the chemical compounds present in peach blossom were screened out by Ultra-performance liquid chromatography (UPLC)-Orbitrap fusion mass spectrometer combined with diagnostic ions and database searching. Finally, the potential mechanism of peach blossom for the treatment of constipation was explored by network pharmacology on a system level. Peach blossom significantly reduced the fecal water content, gastrointestinal transit ratio, and fecal pellet weight, which were increased in the model group. Histological analysis showed that peach blossom could alleviate histological changes of the colon in constipated mice. In UPLC-MS analysis, 40 compounds, mainly flavonoids and phenolic acids, were identified. Network pharmacology analysis results indicate that 27 compounds exhibited anti-constipation activity by neurotransmitter receptor activity, carboxylic acid binding, flavin adenine dinucleotide binding, adrenergic receptor, and nuclear receptor activity. In this study, we found that peach blossom might alleviate the pathological process of constipation by regulating multi-targets. Our study serves as a theoretical basis for the utilization and further development of peach blossom as a promising health food for ameliorating constipation.
Introduction
Constipation is an extremely prevalent functional gastrointestinal disorder with a wide spectrum of symptoms such as abdominal discomfort, bloating sensation, difficult defecation, pelvic pain, gut obstruction, and nausea. 1 However, the symptoms linked with constipation are frequently intermittent and mild, but they may be prolonged, hard to treat, and debilitating. 2 Overall, these symptoms have a severe impact on the patients’ quality of life. Though this disease is commonly caused by inadequate dietary fiber intake, insufficient fluid intake, reduced physical activity, and so on, increases in fluid intake and physical activity do not relieve chronic constipation, except in dehydrated patients.2,4 Nowadays, anthraquinones, diphenols, and osmotic laxatives are widely used for the treatment of constipation. These drugs are suggested to facilitate gastrointestinal motility, but their extended use results in adverse effects. Therefore, it is essential to identify effective and easy-to-mplement strategies to counter constipation.
Recently, dietary intervention is regarded as an important strategy to relieve constipation symptoms. Many foods have been reported to possess laxative properties, such as banana-resistant starch, 5 mulberry, 6 and Foeniculum vulgare M. 7 Peach blossom, originated from the flower of Prunus persica (L.) Batsch, has been cultivated on a large scale in China. Because of its high nutritional and therapeutic effect, peach blossoms are generally consumed as tea or wine in China and Korea and are believed to be beneficial for human well-being. 8 Previous literature 9 also reported that peach blossom possessed prokinetic activity on smooth muscle isolated from colonic tissue of rats. Besides, some researchers also showed that peach blossom extracts have antioxidant, anti-inflammatory, 8 and tyrosinase inhibitory activity.10,11 Peach blossoms also possess protective effects against DNA and protein damage. 8 All these studies led us to believe that peach blossom extracts might alleviate constipation.
Due to having multiple components, the natural product could control body homeostasis using multiple targets. 12 However, clarification of the active ingredients of a natural product and their targets has been the bottleneck for the application of natural products in drug development. As a promising method to reveal the active ingredients of natural products, an “effect-components-target” strategy based on HPLC-MS and network pharmacology has become prevalent.13,14 Ultra-performance liquid chromatography along with high-resolution mass spectrometry (UPLC-HR MS) has been extensively used in many fields, such as foodstuffs and medicine.15,17 Based on the components identified by UPLC-HR MS, network pharmacological analysis is used to identify the essential targets related to these components. Moreover, the network pharmacology method could simplify the action mechanism of drugs completely and be applied to the research of the material basis of natural products.
Here, we first evaluated the effects of peach blossom extract on loperamide-induced constipation of mice. Then, a UPLC-Orbitrap fusion MS method combined with a HR-MS/MS database was used for the identification of the components in the extract of peach blossom. Finally, the potential anti-constipation mechanism of peach blossom was further studied by network pharmacology. To our knowledge, this is the first report of an investigation of the comprehensive chemical profile of peach blossom with anti-constipation effects.
Materials and Methods
Chemicals, Reagents, and Materials
Dried peach blossoms were purchased from markets in Shandong, China. The voucher number TH09 was authenticated as the dry flowers of Prunus persica (L.) Batsch by Assistant professor Yu Fu from Henan University of Chinese Medicine, Zhengzhou, China. A picture of the peach blossoms is shown in Figure S1. The voucher specimens were deposited in the Department of Pharmacognosy. Acetonitrile was procured from Merck (Darmstadt, Germany). Purified water obtained using a Milli-Q system from Millipore (Billerica, USA) was used for UPLC analysis. Chlorogenic acid, ferulic acid, rutin, isoquercitrin, and hyperoside were obtained from Chengdu Must Biotechnology Co. Ltd (Chengdu, Sichuan, China).
Preparation of Peach Blossom Extract
Dry peach blossoms were pulverized into powder form, and then 20 g of powder was accurately weighed and reflux extracted twice with 200 mL distilled water at 100 °C for 1 h. After filtration, the two filtrates were combined, evaporated and freeze-dried at −20 °C for 72 h to dryness; 3.4 g freeze-dried powder was collected. The freeze-dried powder was then stored at 4 °C for further studies.
Animal Experiments
Sixty SPF male Kunming mice (16-18 g) were purchased from Beijing Sipeifu Biotechnology Co. Ltd. The mice were housed at 25 ± 5 °C, with a 50%-60% relative humidity, in a 12-h light/12-h dark cycle. Before starting the experiments, mice were kept for 7 days to adjust to their environment with a standard diet and water. This study was carried out following EU Directive 2010/63/EU for animal experiments. The present study protocol was approved by the Animal Care and Ethics Committee of the Henan University of Chinese Medicine (No. DWLL202103141).
Mice were randomly placed into six experimental groups: (1) The normal group (Nor.) was given saline solution by gavage (n = 10), (2) The model group (Mod.) was fed with 8 mg/kg loperamide by gavage (n = 10), (3) The positive (Pos.) group was fed with 8 mg/kg loperamide with phenolphthalein tablets (50 mg/kg) (n = 10), (4) The peach blossom-low dose (PB.-L) group was given 8 mg/kg loperamide with peach blossom extract (200 mg/kg) (n = 10), (5) The peach blossom-middle dose (PB.-M) group was given 8 mg/kg loperamide with peach blossom extract (300 mg/kg) (n = 10), and (6) The peach blossom-high dose (PB.-H) group was given 8 mg/kg loperamide with peach blossom extract (400 mg/kg) (n = 10). All samples were intragastrically administered for 7 days (once daily). All the mice were then sacrificed on the eighth day through CO2 aspiration. The transverse colons were then acquired and stored in Eppendorf tubes at −70°C until further assay.
Analysis of Food Intake, Water Consumption, and Body Weight
The food weight, water volume, and body weight of mice were measured daily at 08:00 am during the experimental period, with a measuring cylinder (for water volume) and electrical balance (for food and body weight). The average food intake and water consumption were calculated for the group and body weight was calculated for an individual. All measurements were performed thrice to ensure accuracy.
Measurement of Stool Parameters and Gastrointestinal Transit Ratio
After oral administration on the seventh day, all mice were kept in metabolic cages for stool collection. Fresh stool pellets from each mouse were collected into round-bottomed stoppered tubes every 20 min from 08:00 am to 20:00 pm, and the total weights from each mouse were logged. Fecal samples were collected and freeze-dried to determine the fecal moisture content. The moisture content was calculated by the following equation:
Histopathological Analysis
Transverse colons collected from all mice were fixed with 10% formalin for 48 h, embedded in paraffin wax, and then sectioned into 4 μm thick slices. These slices were then stained with hematoxylin and eosin (H&E, Sigma-Aldrich Co., St. Louis, MO). Light microscopy was used to observe the morphological features of these sections.
UPLC-MS Conditions for Qualitative Analysis
Chromatographic and MS analyses were conducted as previously described. 19 Briefly, chromatographic analysis was performed on a Dionex Ulti-Mate 3000 UPLC system. Chromatographic separation was carried out at 25 °C on an Agilent InfinityLab Poroshell SB-C18 column (2.1 mm × 15 cm, 2.7 μm). The mobile phase consisted of 0.1% formic acid water (A) and ACN (B) using a gradient elution of 5% B from 0 to 3 min; 5-10% B from 3 to 8 min; 10-16% B from 8 to 17 min; 16% B from 17 to 18 min; 16-20% B from 18 to 19 min; 20% B from 19 to 25 min; 20-30% B from 25 to 35 min; 30-40% B from 35 to 40 min; 40-100% B from 40 to 50 min; and 100% B from 50 to 55 min. The flow rate was maintained at 0.5 mL/min. The injected sample volume was set at 2 μL.
The identification of constituents in peach blossoms was carried out using a Thermo Fisher Orbitrap fusion mass spectrometer (Thermo Fisher Scientific, Bremen, Germany) equipped with an H-ESI source. The spray voltage was set at 3500 and 2500 V for positive and negative ion modes. Sheath gas was set at 50 Arb, Aux gas at 10 Arb, and Sweep gas at 1 Arb. The ion transfer tube temperature was set at 325°C, and the vaporizer temperature at 350°C. Full MS scans from 100 to 1500 m/z were performed in the Orbitrap at a resolution of 70 K. The most intense ions detected in the full-scan spectrum were selected for the data-dependent scan, and a stepped NCE mode (20, 40, 60 V) was used. The data were recorded and then processed using Xcalibur 3.0 software (Thermo Fisher Scientific) and Compound Discovery 3.0 software (Thermo Fisher Scientific).
Network Pharmacology Analysis
The components of peach blossom were acquired from the UPLC-MS analysis, and targets in constipation treatment were searched in different databases.
Screening the Targets of the Identified Compounds of Peach Blossom
The biological targets for the identified constituents were screened using Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, http://tcmspw.com/) and Swiss Target Prediction database (http://www.swisstargetprediction.ch/). The components-targets network was then established using Cytoscape 3.6.1 software (Bethesda, MD, USA).
Screening the Targets Related to Constipation
Information on constipation-associated targets was collected from three databases, Therapeutic Target Database (TTD, http://bidd.nus.edu.sg/group/cjttd/), Cortellis Drug Discovery Intelligence Database (https://www.cortellis.com/drugdiscovery), and Comparative Toxicogenomics Database (CTD, http://ctdbase.org/). “Constipation” was selected as the keyword, and the species was limited to “Homo sapiens”. The targets that were repeated at least twice were collected.
PPIs Network Construction and Topological Analysis
STRING database (https://string-db.org/) was applied to discover the interactions among the shared proteins from compound-related and disease-related proteins. To filter the possible protein–protein interactions (PPIs), the attained PPIs with high confidence (combine score > 0.98) were retained for network construction and analysis. Then, the screened proteins were imported to Cytoscape 3.6.1 to construct a PPIs network and screen key proteins. The degree value was used to appraise the importance of the protein. Finally, the proteins with a degree > 7 (average degree = 6.7) were selected as key proteins and a network was constructed.
Statistical Analysis
GraphPad Prism version 7.0 was used for descriptive statistical analyses. Data were expressed as mean ± SD and analyzed by the t-test to test for variability between each trial, with P ≤ 0.05 being taken as significant.
Results
Body Weight and Water and Food Intake of the Mice
During the experiment, no mouse died, indicating better security and operability of the animal model. As shown in Figure 1, body weight (A), food intake (B), and water intake (C) of the model group significantly decreased in comparison to those of the normal group, which suggests that the administration of loperamide could affect the behavior of the mice. In terms of food intake, oral administration of loperamide significantly reduced the food intake of the mice, but the amount of food consumed was improved in the peach blossom group. The water consumption of both the positive and peach blossom groups also increased compared to the model group. These results suggest that peach blossom treatment could improve the feeding behavior of constipated mice.

Body weight, feeding behavior, and fecal parameters following intragastric administration either with or without peach blossom extract in mice with loperamide-induced constipation. (A) Body weight, (B) water intake, (C) food intake, (D) fecal weight, (E) fecal water loss, and (F) intestinal transit ratio; all were measured at the same time during the experiment. Six mice per group were assayed in triplicate for analysis. Data represent the means ± SD from three replicates. **P < 0.01 compared to the Nor. group; #P < 0.05 and ##P < 0.01 compared to Mod. group.
Fecal Parameters and Intestinal Charcoal Transit
To elucidate the laxative effects of peach blossom on constipated mice, the present study examined the fecal weights, fecal water contents, and intestinal charcoal transit of the mice. As shown in Figure 1, the fecal weight (D) and fecal water content (E) were significantly reduced by 70% and 34%, respectively, after administration of loperamide compared with the normal group. After administration of phenolphthalein tablets and different concentrations of peach blossom extract, the fecal weight and fecal water content of the animals were significantly increased (P < .05).
The charcoal meal transit test was performed to examine whether the increase in the stool excretion was accompanied by changes in gastrointestinal motility. A significant decrease in the intestinal charcoal transit ratio was noticed in the model group as compared to the normal group (Figure 1F). After treatment with peach blossom extract, there was no significant difference in the peach blossoms-low and -middle groups compared with the model group, but the propulsion was completely recovered in the peach blossom-high treated groups. These results indicate that the increased excretion induced by peach blossom treatment is linked with increased gastrointestinal motility in the loperamide-induced constipation model.
Histological Alteration of Transverse Colon
The histological alterations after treatment with peach blossom were examined in the mice colons with loperamide-induced constipation through H&E staining. As shown in Figure 2, in the normal group (A), the tissue structure of the mucous layer, submucosal layer, muscular layer, and epithelium mucosae were intact, and the intestinal mucosa contained a large number of intestinal glands. In addition, the muscular layer consisted of smooth muscle cells and was arranged closely, whereas the tissue in the model group (B) showed a marked loss of colon epithelium, goblet cell depletion, and intestinal gland damage. Moreover, there was plenty of proliferation of connective tissues (black arrows) with infiltration of inflammatory cells (red arrows). Capillarectasia and congestion were also common (yellow arrows). Following treatment with peach blossom in low dose (D), moderately damaged epithelial cells were found in comparison to the model group. The colons of mice treated with high-dose peach blossom (F) showed intact epithelial and goblet cells, which were comparable to those of the normal group. These results indicate that peach blossom could induce recovery in histological changes of the colon in constipated mice.
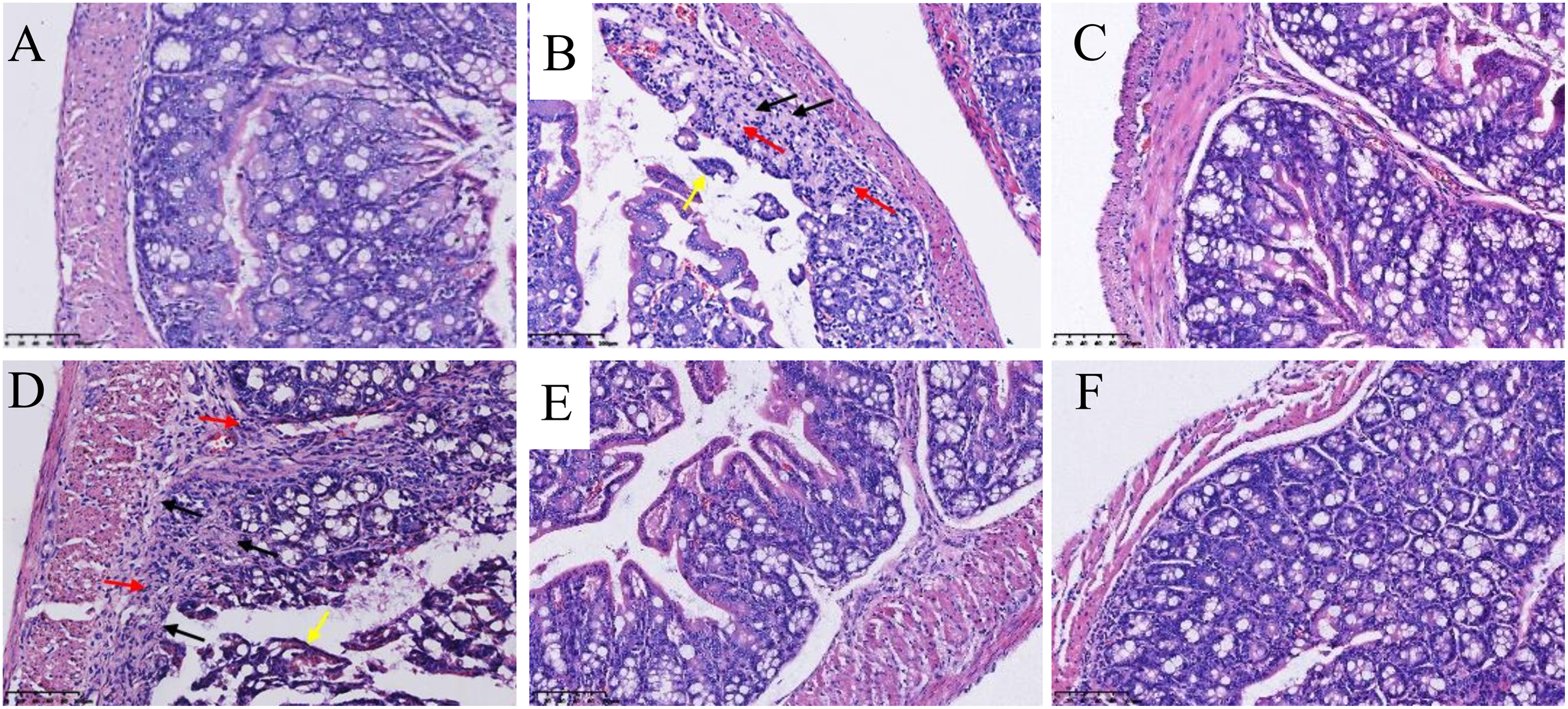
H&E staining for assessment of histological changes in the mice colon. (A) Nor. group, (B) Mod. group, (C) Pos. group, (D) PB.-L group, (E) PB.-M group, and (F) PB.-H group. Black arrows point to proliferation of connective tissues, red arrows point to infiltration of inflammatory cells, and yellow arrows point to capillarectasia and congestion.
Constituent Analysis by UPLC-MS
Optimized UPLC-Orbitrap fusion MS both in negative and positive ionization modes were used to explore the chemical constituents of peach blossom. Because of more abundance of precursor ions and fragment ions, negative ion mode was mainly used for structure identification. The total ion chromatogram (TIC) in negative ion mode of peach blossom is shown in Figure 3. MS/MS fragments, OTCML database, mzCloud database, and reference standards were applied to ascertain the chemical constituents from the chemical profile of peach blossom. We explored the MS/MS diagnostic ions information of the constituents in the 70% methanol extract of peach blossom in our previous paper, 19 and this information helped us elucidate the constituents of the water extract. A total of 40 compounds were identified, mainly phenolic acids and flavonoids (Figure 3, Table 1). Among these compounds, chlorogenic acid (9), ferulic acid (13), rutin (14), isoquercitrin (18), and hyperoside (19) were confirmed by comparison with reference standards. Others were identified from diagnostic ions, OTCML database and mzCloud databases. The similarity scores of the compounds identified by OTCML and mzCloud database were more than 80, which shows higher reliability. Therefore, these compounds could be used for network pharmacology analysis.

The total ion chromatogram (TIC) of peach blossom extract.
The MS Information of Compounds in Peach Blossom.

Standard reference.
Compound-Target Network Construction
By searching the TCMSP and Swiss Target Prediction databases, 27 compounds and 153 corresponding targets were acquired (Tables S1 and S2). The compound–target interaction network was constructed by Cytoscape software (Figure 4). The visualized network consisted of 180 nodes and 463 edges. The nodes represent components and target proteins, which are depicted as a green circle and a yellow rhombus, respectively. The degree of the nodes was defined by the magnitude. As a result, 13 compounds from peach blossom linked with more than 10 targets. Among them, quercetin could act on 87 targeted proteins. Besides, among the 153 compound-related targets, 7 targets (ACHE, ADORA1, ADORA2A, CHRM1, CYP3A4, IL 6, PTPN1) were the target for constipation.

The compound–target interaction network of peach blossom. The compounds of peach blossom were placed in green circles, and the targets in a yellow rhombus.
PPIs Network Construction and Molecular Function Analysis
Next, we constructed a relationship between the compound-related targets and disease-related targets. Fifty-one disease-related proteins and 153 compound-related proteins were uploaded to the STRING database, and the protein–protein interactions (PPIs) were constructed with Cytoscape software. By comparing the combined scores of the PPIs, 501 interactions (combined scores >0.9) were selected for PPIs network construction via Cytoscape software. Then, we identified the key proteins according to the criteria as degree >7 (average degree = 6.7). The key protein PPIs network is shown in Figure 5, and consists of 56 nodes and 295 edges. The nodes placed in a rectangle with a yellow rhombus represent disease-related proteins, while the nodes placed in green circles represent constituent-related proteins. Among these key proteins, CXCL8, TACR1, OPRM1, OPRD1, JUN, HSP90AA1, and CHRM2 possessed a greater degree of connectivity in the PPI analysis, and they could be considered as the potential key proteins in treating constipation.
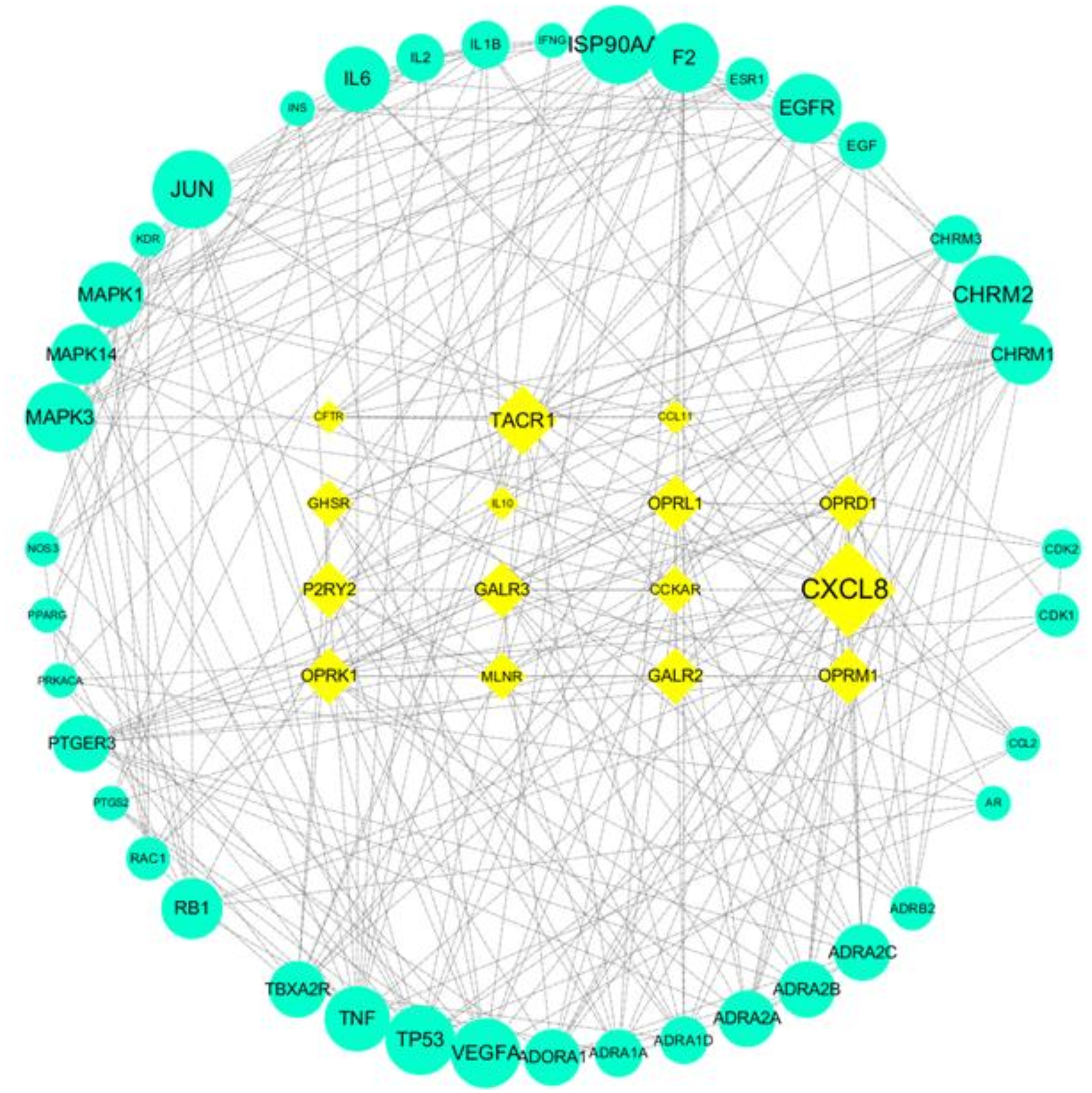
The PPIs network of key proteins. The yellow rhombus represent disease-related proteins, green circles represent compound-related proteins.
To decipher the molecular functions of 56 key proteins, ClueGO, an extensively used Cytoscape plugin, was used to identify biological interpretation. As shown in Figure 6, the molecular functions were mostly comprised of five groups (exceeding 50%): neurotransmitter receptor activity, flavin adenine dinucleotide binding, carboxylic acid binding, adrenergic receptor, and nuclear receptor activity. It is suggested that the above molecular functions play important role in the anti-constipation of peach blossom.

Cluego analysis of the predicted targets.
Discussion
Constipation is often multifactorial, resulting from either systemic or neurological disorders, as well as medication. 2 To date, a series of animal models have been used to examine the therapeutic effects of medicine on constipation, including drug and diet induction models. 20 Among drug-induced models, loperamide administration has been shown to cause constipation successfully in many previous studies.21,22 Loperamide, a μ-opioid receptor agonist causes compulsive contractions in the gastrointestinal tract. In the present study, after the administration of loperamide, body weight, food intake, and water intake of the model group were significantly decreased in comparison with the normal group. As classical biomarkers, fecal water content, fecal pellet weight, and gastrointestinal transit ratio were significantly decreased in the model group in comparison with the normal group. After treating with peach blossom, gastrointestinal transit ratio and fecal indexes were significantly improved. This result suggested that peach blossom improved defecation and intestinal motility. Histological studies also confirmed that loperamide administration caused significant alterations in the transverse colon, for example, decreases in epithelium and damage of intestinal glands. Epithelial cells constitute colonic mucosa, which primarily promote colonic motility by regulating electrolytes and luminal water absorption. 22 Intestinal glands can produce sulfomucin-rich acid mucin which is a typical secretion for protecting the intestinal mucosa. Meanwhile, accumulation of feces in the colon constricts the intestine, and leads to capillarectasia and infiltration of inflammatory cells. Following treatment with peach blossom in high-dose, the colons of mice displayed intact goblet cells and epithelial cells, which were comparable to those of the normal group. Therefore, the protective effects against these pathological states of the colon were a direct indication of the laxative effects of peach blossom.
Investigation of the mechanism of action of a natural product is a daunting task because of its complicated constituents and multi-targets. In our research, because the peach blossom was extracted with water, numerous polar components were present in the extract, such as carbohydrates, glycosides, and phenolic acids. Literature 23 reports that these components all possess anti-constipation activity. Carbohydrates, 24 such as fructan and galacto-oligosaccharides, have been proved to have a positive effect on the composition and function of gut microbiota. Glycosides and phenolic acids 25 also could effectively decrease the symptoms of constipation in the loperamide-induced mice model. In the present study, we focused on glycosides and phenolic acids in peach blossom. UPLC-MS combined with network pharmacology was employed to elucidate the possible mechanisms of peach blossom against constipation. Public databases and literatures have been used for searching herbal compounds in many studies.26,27 However, the compounds acquired from the databases may not exist in the herbal extracts that are administrated to animals and patients. For example, some nonpolar or thermally unstable constituents may not exist in the water extracts of herbal medicine. Here, UPLC-MS was used to analyze the extract of peach blossom, which provided “real herbal compounds” for subsequent analysis. A total of 40 compounds were identified, mainly phenolic acids and flavonoids. In our previous study, 19 MSn fragmentation behaviors and diagnostic ions of phenolic acids and flavonoids were investigated. According to these diagnostic ions, flavonoids and phenolic acids were identified. Besides, combined with database searching, the accuracy of the identified compounds was promoted.
Network pharmacology, considered a favorable method to study the complicated mechanism of a TCM, offers a different method from the current “one target, one drug” mode to a novel “network targets, multi-components” mode for target identification assisting research on the underlying mechanisms of TCM. In our study, 28 compounds were recorded in the TCMSP and Swiss Target Prediction databases, and these compounds could intensively act on 153 targets. Among these constituents, quercetin could act on 87 proteins. Literature 18 also reports that quercetin could promote gastrointestinal motility and mucin secretion in loperamide-induced constipation of SD rats by regulating the mAChRs downstream signal. Among constituent-related targets, ACHE, ADORA1, ADORA2A, CHRM1, CYP3A4, IL 6, and PTPN1 were reported to be directly correlated with constipation. PTPN1, which directly relates to 11 constituents of peach blossom, could dephosphorylate the insulin receptor and leptin receptor-linked enzyme Janus kinase 2, and it is also related to constipation. 28 Though most compound-related proteins are not directly correlated with constipation, the PPI analysis predicted that these proteins could interact with disease-related proteins for the treatment of constipation. The results of GO enrichment analysis showed neurotransmitter receptor activity plays an important role in the anti-constipation activity of peach blossom. Literature 29 reports that some neurotransmitters, such as serotonin, could regulate the central nervous system, enteric nervous system development, and long-term functions, as well as gastrointestinal motility. Besides, activation of the adrenergic receptor sites could also relieve constipation.
Conclusion
In conclusion, the present study shows that the extract of peach blossom induces gastrointestinal motility, recovery of stool excretion, and maintains histopathology in the loperamide-induced constipation model. Moreover, UPLC-Orbitrap fusion MS in negative ionization modes was employed to explore the chemical constituents of peach blossom. In total, 40 constituents were identified by UPLC-MS. Finally, network pharmacology analysis results indicate that 27 compounds exhibited anti-constipation activity by neurotransmitter receptor activity, carboxylic acid binding, flavin adenine dinucleotide binding, adrenergic receptor, and nuclear receptor activity.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231161184 - Supplemental material for Exploring the Active Constitutions of Peach Blossom for Amelioration of Loperamide-Induced Mice Constipation by Ultra-Performance Liquid Chromatography-Mass Spectrometry Combined with Network Pharmacology
Supplemental material, sj-docx-1-npx-10.1177_1934578X231161184 for Exploring the Active Constitutions of Peach Blossom for Amelioration of Loperamide-Induced Mice Constipation by Ultra-Performance Liquid Chromatography-Mass Spectrometry Combined with Network Pharmacology by Yu Fu, Xinya Zhang, Zhenguang Song, Lili Wang, Peng Zhao and Suiqing Chen in Natural Product Communications
Footnotes
Authors Contributions
Yu Fu took on conceptualization, data curation, methodology, and manuscript writing—review and editing. Xinya Zhang took on project administration and writing—the original draft. Zhenguang Song took on data curation and investigation. Lili Wang took on methodology and validation. Peng Zhao took on formal analysis, methodology, and project administration. Suiqing Chen took on conceptualization and funding acquisition.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The present study protocol was approved by the Animal Care and Ethics Committee of the Henan University of Chinese Medicine (No. DWLL202103141).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2021 Henan Science and Technology key projects (grant number 212102311103).
Statement of Animal Rights
This study was carried out following EU Directive 2010/63/EU for animal experiments. The present study protocol was approved by the Animal Care and Ethics Committee of the Henan University of Chinese Medicine (No. DWLL202103141).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.