Abstract
Background
Qinteng Tongbi decoction (QTTBD) was an empirical prescription that could effectively prevent and treat rheumatoid arthritis (RA), but there was no report of pharmacological studies on this prescription. The purpose of this paper was to report the effects of QTTBD on the proliferation, migration, and apoptosis of synovial fibroblasts in adjuvant arthritis model rats, and to reveal its anti-RA regulatory mechanism.
Methods
To divide the fibroblast-like synovial (FLS) cells of experimental rats into 6 groups (blank control group, model control group, positive drug group, QTTBD high, medium, and low dose groups) and cultured with serum-containing drugs. And using Cell Counting Kit-8 to detect the proliferation rate of FLS cells, flow cytometry to detect the apoptosis of FLS cells, the enzyme-linked immunosorbent assay method to detect the levels of interleukin (IL)-6, IL-1β, and tumor necrosis factor-α (TNF-α), Western Blot to detect phosphatidylinositol 3-kinase (PI3K), AKT1, p-AKT1, Bax and Bcl-2 gene and protein expression.
Results
Experimental results showed that QTTBD-containing serum could effectively inhibit the proliferation of FLS cells (p <0 .05), induce the apoptosis of FLS cells, reduce the expression levels of inflammatory factors such as IL-6, IL-1β, and TNF-α (p <0 .05), reduce expression of PI3K, AKT1, p-AKT1, and Bax (p <0 .05), while the Bcl-2 expression increased (p <0 .05).
Conclusion
QTTBD could effectively regulate the proliferation, migration, and apoptosis of FLS cells in adjuvant-induced arthritis (AIA) rats, and its mechanism might be related to regulating the level of inflammatory factors and intervening in the PI3K/protein kinase B (AKT) signaling pathway.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic progressive autoimmune disease characterized by erosive arthritis. It mainly erodes small joints such as hands and wrists and surrounding tissues to cause chronic inflammation. It is often manifested as joint swelling and pain in joints, accompanied by synovial hyperplasia and inflammatory cell infiltration.1‐3 At present, the global prevalence rate of RA is about 0.5% to 1.0%, involving about 1% to 2% of the population and their families. RA is prone to disease due to its recurrence and high disability rate, coupled with its poor prognosis leading to a long treatment cycle, which seriously affects people's quality of life. 4 The pathogenesis of RA has not yet been completely clear, and there are no specific drugs and treatments that can be completely cured. The mainstream western medicine treatment is mainly nonsteroidal antiinflammatory drugs, antirheumatic drugs (disease-modifying antirheumatic drugs), glucocorticoids, and biological agents. 5 The short-term treatment of western medicine is effective, but long-term medication is prone to many adverse reactions and a high recurrence rate after stopping the drug, and it is difficult for most ordinary families to adhere to long-term medication due to the increased financial burden caused by the high price of such drugs. 6 Traditional Chinese Medicine's (TCM’s) understanding and records of RA have a long history. The earliest description of the word “bi” appeared in the “Yellow Emperors Internal Classic (Huang Di Nei Jing).” Later, there were relevant discussions in medical books such as “Treatise on Febrile and Miscellaneous Diseases” and “Synopsis of the Golden Chamber.”7,8 With the continuous deepening of TCM research in recent years, TCM's understanding of the etiology and its development mechanism, as well as dialectical treatment, is also deepening. The advantages of TCM in multilink, multitarget, and multiway treatment of RA have gradually become prominent, and because of its low price and small toxicity and side effects, the vast majority of families can bear and are willing to accept it. Therefore, under the background of people advocating science and approaching nature, TCM has shown great potential and prospects in the prevention and treatment of RA.
Qinteng Tongbi decoction (QTTBD) is obtained by the increase or decrease of famous old TCM experience prescriptions in the Department of Rheumatology and Immunology of Ruikang Hospital Affiliated with Guangxi University of Traditional Chinese Medicine, Guangxi Zhuang Autonomous Region, China, based on the basic theory of TCM combined with their own decades of clinical experience. It has been widely used in clinical practice. The prescription of QTTBD was composed of 11 TCMs and Zhuang and Yao medicine: Gentiana macrophylla Pall. (Qinjiao, QJ), Clematis chinensis Osbeck (Weilingxian, WLX), Cissus hastata Miq. (Sifangteng, SFT), Sinomenium acutum (Thunb.) Rehder & E.H. Wilson (Qingfengteng, QFT), Stephania tetrandra S. Moore (Fangji, FJ), Astragalus membranaceus (Fisch.) Bunge (Huangqi, HQ), Epimedium brevicornu Maxim. (Yinyanghuo, YYH), Aconitum carmichaelii Debeaux (Fuzi, FZ), Ephedra sinica Stapf (Mahuang, MH), Poria cocos (Schw.) Wolf. (Fuling, FL), and Glycyrrhiza uralensis Fisch. (Gancao, GC). The plants studied were recorded in the Chinese Pharmacopoeia (Part 1 of the 2020 edition) except for the SFT recorded in the Quality Standards for Zhuang Medicines in Guangxi Zhuang Autonomous Region of China (2008 edition). After preliminary basic research, it was found that QTTBD had the effect of dispelling wind and dehumidification, dispelling cold and removing bi, and dredge meridians and relieving pain, and could effectively treat RA. Its mechanism might be related to the intervening Oxytocin signaling pathway, cGMP-protein kinase G signaling pathway, Rap1 signaling pathway, PI3K–Akt signaling pathway, mitogen-activated protein kinase (MAPK) signaling pathway, Wnt signaling pathway, and regulating the expression levels of AKT1, interleukin (IL)-1β, IL-2, phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit gamma (PIK3CG), tumor necrosis factor (TNF), IL-10, IL-4, IL-6, and other genes. 9 However, after searching the literature, it was found that the research on QTTBD's anti-RA was still in the initial stage, and there has been no report on the relevant pharmacology and the mechanism of its action. Therefore, this recent study mainly aims to report the regulatory effect of QTTBD on the proliferation, migration, and apoptosis of fibroblast-like synovial (FLS) cells in the adjuvant-induced arthritis (AIA) rat model and tries to clarify its anti-RA mechanism for academic exchanges.
Materials and Methods
Plant Material and Extraction
QJ (No. 20190601), WLX (No. 20190802), SFT (No. 20190102), QFT (No. 20190201), FJ (No. 20190101), HQ (No. 20190803), YYH (No. 20190802), FZ (No. 20190702), MH (No. 20190201), FL (No. 20190801), and GC (No. 2019001) were purchased from Guangxi Xianzhu Traditional Chinese Medicine Technology Co. Ltd (Nanning, China) and the specimen of the samples were kept in the Drug Research and Development Center of Ruikang Hospital Affiliated to Guangxi University of Traditional Chinese Medicine (Nanning, China). All TCM decoction pieces were extracted 3 times according to the prescribed amount. For the first time, add 10 times the amount of water to soak for 30 min and then decoct for 1.5 h. For the second and third times, add 8 times the amount of water to decoct for 1.0 h. The 3 times of decoction were filtered and vacuum-condensed into a dry extract, then crushed into 80 mesh fine powder for later use.
Animals
Male Sprague-Dawley rats (160–200 g) were purchased from the China Three Gorges University with the laboratory animal license number: SCXK (E) 2018-0104 and animal certificate number: No. 42010200005421. All animal experiments received ethical approval from Hubei Provincial Animal Management Committee according to the relevant regulations of its “Experimental Animal Ethics Certificate.” The animals were maintained in a specific pathogen-free environment with a temperature of 22 °C (±2 °C) and relative humidity of 50% to 60% and were subjected to a 12 h light/dark cycle. Provide adequate food and water during the rearing period, and change bedding and the cage positions at a fixed time every week. Model construction was performed after 7 days of adaptive feeding.
Fingerprint of QTTBD extraction 10
Ten batches of QTTBD prescription medicinal herbs were weighed in parallel and extracted, concentrated, dried, and crushed into 80 mesh powder for later use according to the above extraction method. Precisely weighing QTTBD powder 5.0417 g in a conical bottle, adding 50 mL methanol (Chengdu Cologne Chemical Co., Ltd, batch number 2021091602) ultrasound for 30 min, taking it out and cooling to room temperature, replenishing the weight with methanol, filtering, taking 5 mL of continuous filtrate and steam dry, dissolved with 10 mL chromatographic methanol (TEDIA, batch No. 14060185) and set the capacity in a 10 mL bottle, then filtered with 0.22 μm microporous filter membrane (Tianjin Jinteng Experimental Equipment Co., Ltd) to obtain the sample solution for later use.
The analysis was performed with a Waters Symmetry Shield RP18 (250 mm × 4.6 mm, 5 μm) chromatographic column in the high-performance liquid chromatography (HPLC; Shimazu, Japan, LC-20A). The flow phase consists of chromatographic acetonitrile (A) (TEDIA, batch number 14060412) and 0.01% aqueous phosphate solution (B) (National Pharmaceutical Group Chemical Reagents Co., Ltd, batch number T20160324), with an injection volume of 10 μL, a flow rate of 1.0 mL/min, a column temperature of 30 °C, an ultraviolet detector detection wavelength of 365 nm, and the gradient elution conditions are shown in Table 1.
Gradient Elution Conditions.

Model Establishment and Preparation of Drug-Containing Sera 11
Sixty male rats were randomly divided into 6 groups, namely, blank control group, model group, methotrexate group, QTTBD high-dose group, medium-dose group, and low-dose group, each group had 10 animals. Except for the blank control group, other groups were subcutaneously injected with 0.1 mL of Complete Freund`s Adjuvant (F5881, Sigma, USA) on the plantar part of the right foot, which was boosted once on day 14. The blank control group was injected with the corresponding volume of normal saline, and the AIA model was formed after 4 weeks. After successful modeling, the high-dose, medium-dose, and low-dose groups were given QTTBD with 17.833, 8.917, and 4.458 mg/g by gavage every day, respectively, methotrexate group was given methotrexate (036200601, Shanghai Shangyao Pharmaceutical Xinyi Pharmaceutical Co., Ltd, Shanghai, China) 0.07 mg/g, blank control group and model group were given the corresponding volume of normal saline with dosing twice a day for 21 consecutive days. One hour after the last administration, the animals were anesthetized by intraperitoneal injection of 1% sodium pentobarbital, and blood was collected from the abdominal aorta under sterile conditions. If the animal did not die, then use 1% pentobarbital sodium to overdose to death (100 mg/kg). After anesthesia, the animal was judged to be dead if there was no breathing or heartbeat. After standing for 2 h at room temperature, rat blood was centrifuged at 4 °C and 2000 r/min for 15 min, and the supernatant was taken. After inactivation in a water bath at 56 °C, it was stored in a −80 °C refrigerator for later use.
Synovial Fibroblasts Isolation and Culture 12
The synovial layer tissue taken out under sterile conditions after the experimental rats were sacrificed was placed in precooled sterile phosphate-buffered saline (PBS) containing 5% double antibody, and the culture dish was washed on ice to remove blood stains, adventitia, and adipose tissue. Place the synovial tissue in a sterile cell culture dish, and rinse the tissue 3 times with 5% double antibody-containing PBS (P1010, Solarbio, Beijing, China) solution, transfer the washed tissue to a new sterile culture dish, add a small amount of Dulbecco's Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F12; SH30023.01, Hyclone, USA), and chop into small pieces with a volume of approximately 1 mm × 1 mm × 1 mm, and transfer the chopped tissue pieces to a Petri dish, add 5 times the tissue volume of 0.2% collage II (prepared with DMEM/F12 medium), and incubate at 37 °C for 120 min (shake and observe every 15 min), the first tube of cells was collected after 30 min: the cell suspension was filtered through a 200-mesh cell strainer, centrifuged at 400 g for 6 min, the supernatant was collected, and the unfiltered tissue was added to continue to be used for digestion, and observed every 15 min. After 60 min, collect the second tube of cells, filter the cell suspension through a 200-mesh cell strainer, centrifuge at 400 g for 6 min, wash twice with DMEM/F12, resuspend with DMEM/F12 each time, and centrifuge at 400 g for 6 min. The cells were observed and counted with a counting plate for the last time, and the cell seeding density was adjusted to 2 × 105 cells/mL, inoculated into culture flasks for culture passage, and after passage to the third passage, trypsinized for use.
The cells were divided into a blank control group, model group, methotrexate group, QTTBD high-dose, middle-dose, and low-dose groups. The blank control group was normal rat synovial fibroblasts, and the other groups were model group rat synovial fibroblasts. The blank control group and model group were cultured with 10% normal rat serum, the methotrexate group was cultured with the drug-containing serum of the 10% methotrexate group, and the QTTBD high, medium, and low dose groups were cultured with high, medium, and low drug-containing serum with 10% QTTBD, respectively.
Detection of Proliferation of FLS Cells13‐15
We use the Cell Counting Kit-8 (CCK8) detection kit and operate according to the instructions to detect the cell proliferation rate. Collecting the cells and adjusting the concentration of cell suspension to divide into 96-well plates for 3 × 103 cells/well and 100 µL per well, and culture overnight in a 37 °C, 5% CO2 incubator to make cells adherent. Then treating the cells according to the pregrouped groups and continue to culture for 24, 48, and 72 h, taking out the cell culture plate and adding 10 μL of CCK8 solution to each well to continue culture for 4 h, and measuring the absorbance of each well at 450 nm in an enzyme-linked immunosorbent assay (ELISA) (A). The calculation formula was: proliferation inhibition rate = [(Ablank group − Aadministration group)/Ablank group] × 100%.
Detection of Apoptosis of FLS Cells 13
We followed the steps of annexin V conjugated with fluorescein isothiocyanate/propidium iodide (Annexin V-FITC)/propodium iodide (PI) apoptosis detection kit (556547, BD Biosciences, USA) instructions. First, collect cells from each group, take 1 × 106 cells resuspended in medium and centrifuge at 400 g for 5 min at 4 °C, discard the supernatant, add 1 mL of precooled PBS, and mix the cells gently by pipetting, and then take 400 g of cells at 4 °C centrifuge for 5 min, discard the supernatant, resuspend the cells in 200 μL of PBS, add 10 μL of Annexin V-FITC and 10 μL of PI, mix gently, and incubate at 4 °C for 30 min in the dark before adding 300 μL of PBS, followed by flow detection. Analysis was performed using NovoExpress analysis software, and the calculation formula was: apoptosis rate (%) = early apoptosis rate + late apoptosis rate.
Detection of Expression Levels of IL-6, IL-1β, and TNF-α 16
Prepared all reagents according to the instructions in advance and mix thoroughly to avoid foam (IL-6 [RA20607], IL-1β [RA20020], TNF-α [RA20035], Bioswamp, Wuhan, China). The FLS cells culture supernatant of each group was collected after drug intervention at 72 h and centrifuged at 1000 r/min for 10 min at 4 °C to collect the supernatant which was added to the ELISA plate (100 μL per well) and incubated at 37 °C for 30 min, and the ELISA method was used to detect the expression levels of IL-6, IL-1β, and TNF-α in strict accordance with the instructions of each kit.
Detection of Expression of PI3K, AKT1, p-AKT1, Bax, and Bcl-217,18
The tissues of each group were separated and cut into small pieces, and 200 μl of radioimmunoprecipitation assay (RIPA) lysis buffer (R0010, Solarbio, Beijing, China) was added to each 20 mg of tissue to extract the total tissue protein, and the protein was quantified by the bicinchoninic acid assay (BCA) method. The amount of protein loaded in each well was 12 μg protein, and the protein was separated by 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel (SDS-PAGE; S8010, Solarbio, Beijing, China), transferred to polyvinylidene fluoride (PVDF) membrane (IPVH0010, Millipore, USA), and placed in 5% nonfat milk powder at room temperature to block and then left at 4 °C overnight. Primary antibody: dilute the antibody according to the instructions: PI3K (1:1000, PAB30084, Bioswamp), AKT1 (1:1000, PAB30596, Bioswamp), p-AKT1 (1:1000, 4060T, CST), Bax (1:1000, PAB30727, Bioswamp), Bcl-2 (1:1000, PAB30041, Bioswamp), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; 1:1000, PAB36269, Bioswamp). Adding the antibody to the blocking solution to dilute to the desired concentration, and incubate with the membrane for 1 h at room temperature; Secondary antibody: the membrane incubated with the primary antibody was washed 3 times with PBS plus 20% Tween-20 (PBST; T8220-100, Solarbio, Beijing, China) for 5 min each time, and then the horseradish peroxidase-labeled secondary antibody was diluted 1:10,000 according to the dosage, incubated with the membrane for 1 h at room temperature, and then washed with PBST 3 times for 5 min each time. Enhanced chemiluminescence solution (WBKLS0500, Millipore, USA) was added dropwise, developed in the dark, and the film was placed in an automatic chemiluminescence analyzer for detection. With GAPDH as the internal reference, the gray value of the relevant bands was read by TANON GIS software.
Statistical Analysis
SPSS 23.0 software was used for statistical processing, measurement data were expressed as
Results
HPLC Fingerprint Establishment
The prefabricated sample solution was injected into the HPLC and analyzed according to the above-mentioned chromatographic conditions. Ten batches of QTTBD HPLC fingerprints were obtained and introduced into the Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine (Version 2012A). S5 was selected as the reference fingerprint, using the median method, and the width of the time window was 0.5. The chromatographic peak was selected through multipoint calibration and automatically matched to generate the overlay fingerprint of QTTBD. QTTBD fingerprint map (Figure 1) and reference fingerprint map “R” (Figure 2) were obtained through chromatographic analysis. Chromatographic Peak 6 was selected as the reference peak (S) because it was common in all test samples and was well separated. Feature peaks were marked with consecutive numbers (1, 2, 3, …, n), marking a total of 28 feature peaks, which account for more than 90% of the total area of the total peak area. Therefore, these 28 feature peaks were identified as common feature peaks in QTTBD's HPLC fingerprint.

HPLC fingerprint chromatogram of 10 batches of QTTBD.

HPLC control fingerprint diagram of QTTBD.Abbreviations: HPLC, high-performance liquid chromatography; QTTBD, Qinteng Tongbi decoction.
The similarity of 10 batches of QTTBD HPLC fingerprint chromatography was analyzed, and the data was imported into the Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine (Version 2012A). After the peak matching of chromatography, the standard fingerprint chromatography “R” was generated, and the similarity between fingerprints and standard fingerprints of different batches of samples was calculated. The similarity and standard fingerprints of 10 batches of QTTBD samples were 0.912, 0.868, 0.925, 0.755, 0.923, 0.919, 0.926, 0.917, 0.836, 0.801, and 0.959, respectively, indicating that the similarity of the fingerprints of 10 batches of QTTBD samples was similar.
Identification of FLS Cells
Some typical morphological patterns of rats after successful molding are shown in Figure 3. Through microscopic observation, it was found that most of the FLS cells observed in this study were quasar-shaped and spindle-shaped, with clear cell borders, many secretions were seen around them, and the expression of vimentin was positive. According to the cell source, morphological observation, and immunocytochemical detection results, it could be confirmed that the cells obtained by tissue isolation and culture were RA-FLS cells. The results are shown in Figure 4.

Experimental comparison of the degree of foot swelling in each group.
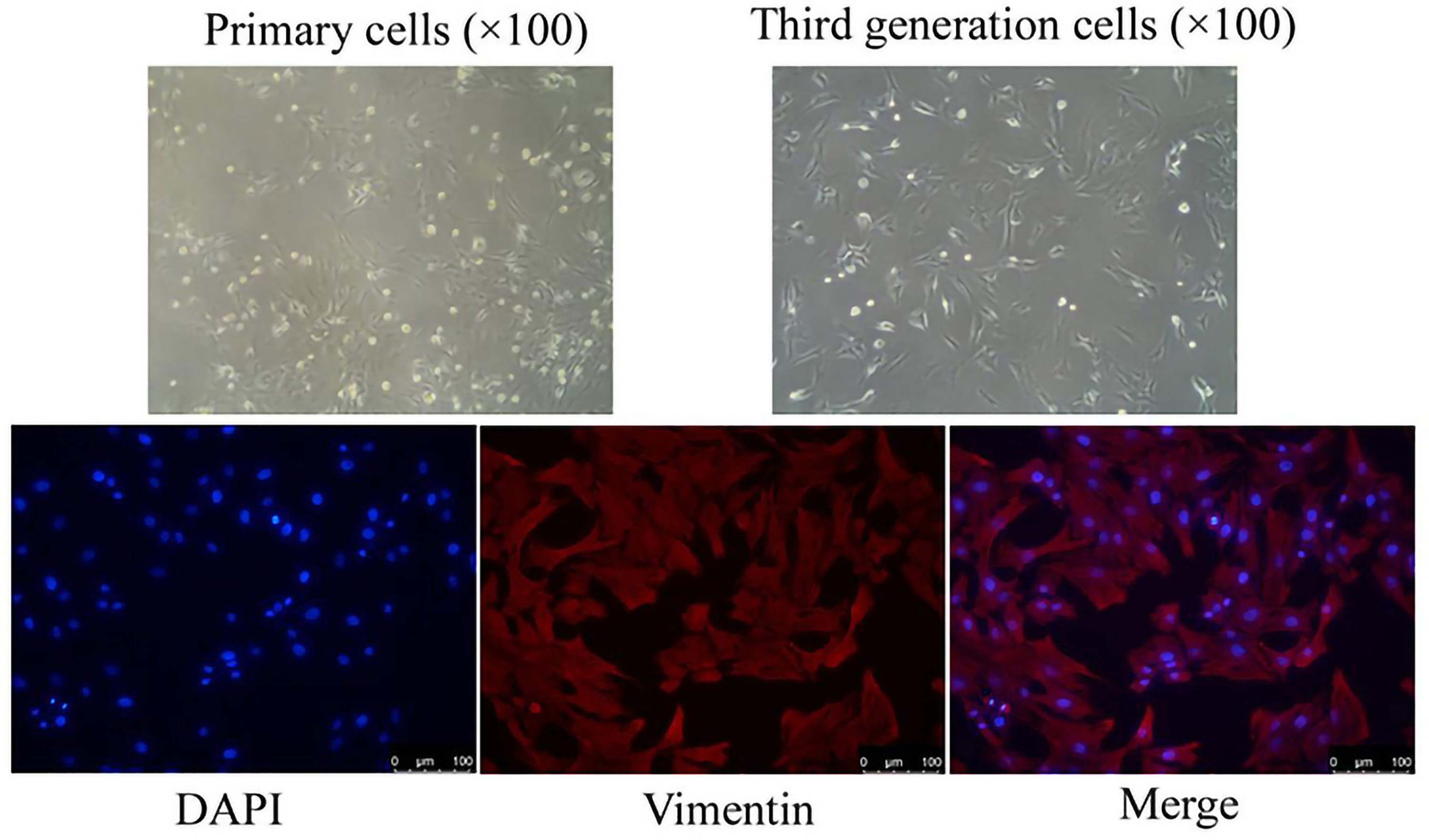
Schematic diagram of passaged cells and their immunofluorescence single-label detection.
Effects of QTTBD-Containing Serum on the Proliferation of FLS Cells
Compared with the model group, the drug-containing serum of each dose group of QTTBD had an inhibitory effect on the proliferation of FLS cells, and there was a statistically significant difference in the apoptosis rate between the groups (p <0 .05). The inhibition rate of cell proliferation increased after 24 h of treatment with drug-containing serum (p <0 .01), among which, the inhibition rate of cell proliferation in the low-dose QTTBD group was the lowest at 12.24%, and the cell proliferation inhibition rate in the high-dose QTTBD group was the highest at 17.03%. After 48 h of treatment, the inhibition rate of cells in each dose group increased (p <0 .01), among which the inhibition rate of cell proliferation in the low dose group was 14.97%, and the inhibition rate of cell proliferation in the high dose group was 21.05%. After 72 h of treatment, the inhibition rate of cells in each dose group was higher than that of the previous 2 time points (p <0 .01). The results showed that with the increase of the dose of QTTBD, the inhibition rate of cell proliferation also increased correspondingly, and the inhibition rate of cell proliferation showed an increasing trend with the increase of intervention time. The results are shown in Figure 5.

The effect of QTTBD-containing serum on the proliferation of FLS cells (x ± s, n = 10). **p <0 .01.
Effects of QTTBD-Containing Serum on Apoptosis of FLS Cells
Compared with the model group, the drug-containing serum of each dose group of QTTBD could induce the apoptosis of FLS cells (p < .01), and the apoptosis rate increased with the increase of dose, showing a dose-dependent manner. The difference between the rates was statistically significant (p < .05). The results showed that QTTBD could induce the apoptosis of FLS cells, and the inhibition rate increased with the increase of dose, and the difference was statistically significant (p < .01). The results are shown in Figure 6.

Effect of QTTBD-containing serum on apoptosis of FLS cells (
Effects of QTTBD-Containing Serum on the Expressions of IL-6, IL-1β, and TNF-α
Compared with the blank control group, the expressions of IL-6, IL-1β, and TNF-α in the model group were significantly increased (p <0 .01). And compared with the model group, the expression of IL-6, IL-1β, and TNF-α in FLS cells in the drug-containing serum of the QTTBD high, medium, and low dose groups were decreased (p <0 .05); The results implied that QTTBD could significantly reduce the expressions of IL-6, IL-1β, and TNF-α in FLS cells of AIA rats, and the difference was statistically significant (p <0 .05). The results were shown in Figure 7.

The effect of QTTBD-containing serum on the expressions of IL-6, IL-1β, and TNF-α (
Effects of QTTBD-Containing Serum on the Expressions of PI3K, AKT1, p-AKT1, Bax, and Bcl-2
Compared with the blank control group, the expressions of PI3K, AKT1, p-AKT1, and Bax in the model group were significantly increased (p <0 .01), and the expression of Bcl-2 was decreased (p <0 .01). And compared with the model group, the expressions of PI3K, AKT1, p-AKT1, and Bax in the drug-containing serum FLS cells of the QTTBD high, medium, and low dose groups were decreased (p <0 .05), and the expression of Bcl-2 was increased (p <0 .05). There was statistical significance (p <0 .05). The results are shown in Figure 8.

Effects of QTTBD-containing serum on the expressions of PI3K, AKT1, p-AKT1, Bax and Bcl-2 (
Discussion
RA is a systemic chronic systemic immune disease. It is common that the body's FLS cells develop abnormally, resulting in progressive inflammation and destruction of tissues around the joints, resulting in disability. RA has the characteristics of high morbidity and disability rate, low cure rate, and high recurrence rate. The treatment of RA has become an urgent problem to be solved in rheumatology.19,20
The pathogenesis of RA is not completely clear, but one of the important factors that usually lead to diseases such as joint deformity and dysfunction in RA patients is the excessive proliferation and apoptosis of FLS cells, as well as the large infiltration of inflammatory cells, and the inflammatory response plays an important role in its pathological process. 21 During the pathology of RA, activated synovial cells recruit lymphocytes such as T cells, B cells, and natural killer (NK) cells in the joint cavity, as well as macrophages and neutrophils, to respond to each other. The immune response is to take inflammatory factors as the key link, and then the inflammatory factors continue to stimulate the target cells to form a continuous inflammatory cycle, and then continue to erode the bone joint to aggravate the disease. 22 IL-6 is a 22–29ku glycoprotein produced by various cells such as T cells, B cells, mononuclear or macrophages. It is mainly regulated by the upstream MAPK signaling pathway and nuclear factor-κB signaling pathway and participates in the pathological process of RA by regulating the expression of various cytokines, immune regulatory molecules, and inflammatory mediators. In addition, IL-6 and its soluble receptors can induce bone resorption by increasing receptor activator of NF-κB ligand or directly inducing osteoclast formation and act as one of the effective promoters of osteoclast differentiation and bone resorption. 23 IL-1β is one of the subtypes of IL-1 in the IL family, which is produced by IL-1β precursors under the cutting of aspartic acid protein hydrolase cysteine-containing. In arthritis, IL-1β is secreted by a variety of cells dominated by macrophages, stimulating T cells, B cells, etc, to release cytokines and antibodies, regulating a variety of cytokines and other targets to act on joint cells to produce collagenase and protease, which induces the proliferation of synovial cells and activate osteoclasts to cause erosion and cartilage destruction.22,24 TNF-α is a member of the TNF family. It is an inflammatory cytokine produced by monocytes and macrophages. It can also be produced by T cells and NK cells under certain stimuli and participate in the whole process of immune and inflammatory responses. The main functions of TNF-α are to kill and inhibit tumor cells, promote neutrophil phagocyte phagocytosis, promote the differentiation of myeloid leukemia cells into macrophages, promote cell proliferation and differentiation, and participate in the occurrence and development of certain autoimmune diseases. 25 In the process of RA pathological development, TNF-α can improve the expression of adhesion molecules of endothelial cells, activated monocytes, and adjust the ability to release small molecular inflammatory media such as cytokines and prostaglandins, which will eventually lead to the continuation of inflammatory reactions. Studies have shown that TNF-α has synergistic effects with IL-6 and IL-1β, both of which play a key role in RA autoimmune pathways.26‐28 In addition, based on the fact that synovial hyperplasia and synovial inflammation are the main pathological characteristics of RA, the PI3K/Akt signal transduction pathway is considered to be a bridge between FLS cells proliferation and apoptosis imbalance. 29 Some scholars have experimentally confirmed the high expression of PI3K, Akt, and other signals in RA synovial tissue and have a certain impact on the excessive migration of FLS cells. 30 So far, more and more scholars believe that the PI3K/Akt pathway is involved in the inflammatory response process of RA, so by intervening in the treatment of the PI3K/Akt signaling pathway, new methods and ideas can be provided for the treatment of RA.
RA belongs to the category of “bi” disease in TCM. According to the basic theory of TCM, the etiology and pathogenesis of paralysis generally have 2 aspects: insufficient righteousness and foreign evil invasion. Inadequate righteousness and qi are the internal factors of paralysis, and the invasion of external evils is one of the important pathogenic factors. The evils of wind, cold, dampness, and heat often abuse each other before they can become sick. In the pathogenesis of RA, positive deficiency is an intrinsic factor, and foreign evil qi, which plays an important role, repeatedly invades and suppresses the body's righteous qi and invades limb joint muscles, making meridians blocked, and blood and fluid do not operate smoothly, causing “bi” to become a recurrent chronic disease. Therefore, in the treatment of “bi,” the treatment methods of TCM can be summarized as dispelling evil spirits and supporting righteousness, including clearing away heat, dispelling cold and dehumidification, detoxification and collaterals, etc. Studies have confirmed that TCM can play a key role in preventing and treating RA through multiple components and channels. 31 The alkaloids, organic acids, glycosides, volatile oils, and other immunoreactive substances contained in TCM also can enhance or inhibit the immune function of the body. 32 Its pharmacological effects are related to pain relief, improvement of inflammation, regulation of immune function, protection of cartilage, reducing vascular formation, inhibition of synovial hyperplasia, etc. 33 In addition, monomers and compounds included in TCM could exert therapeutic effects by acting on immune cells and inflammatory factors. 34 QTTBD was created based on the basic theory of TCM above and contained flavonoids, alkaloids, lignans, saponins, terpenes, coumarins, anthraquinones, steroids, organic acids, volatile oils, polysaccharides, trace elements, and other active ingredients, which all had antiinflammatory, analgesic, immune regulation, and body protection effects. And the whole formula of QTTBD has the functions of dispelling wind and dampness, dispelling cold and eliminating paralysis, dredging collaterals and relieving pain, and can effectively prevent and treat RA. 9
The results of this recent study showed that compared with the model group, the proliferation of FLS cells in the QTTBD drug-containing serum treatment group was effectively inhibited (p <0 .01), and its apoptosis rate increased significantly (p <0 .05), which was positively correlated with the drug dosage and intervention time; the levels of IL-6, IL-1β, and TNF-α after QTTBD drug-containing serum intervention decreased significantly (p <0 .05), the expression of PI3K, AKT1, p-AKT1, and Bax protein was also lower than that in the model group (p <0 .05), while the expression of Bcl-2 increased (p < 0.05)
To sum up, we could conclude that QTTBD could effectively inhibit the level of inflammatory factors of RA, effectively regulate the expression of related proteins, and relieve RA symptoms. However, there are still some problems that require researchers to make breakthroughs in future studies, including the small sample size of clinical case studies of drug treatment RA, role targets related to drug treatment, in-depth research on the mechanism of drug action, etc. Therefore, expanding the sample data of clinical research, and finding specific targets for drug action and its mechanism will be the focus of future research, which will certainly provide a more complete theoretical basis for the research of QTTBD's prevention and treatment of RA.
Conclusions
QTTBD could effectively regulate the proliferation, migration, and apoptosis of FLS cells in AIA rats. Its mechanism might be related to the regulation of inflammatory factors levels and intervention in PI3K/AKT signaling pathways.
Footnotes
Acknowledgements
The pharmacological experiments in this study were greatly supported and helped by Wuhan Myhalic Biotechnology Co., Ltd, Wuhan, China.
Author Contributions
GCL and XFP contributed to the conception and design of the work. GCL and SYC contributed to data collection, analysis, and interpretation. GCL, XFP, and JKF contributed to pharmacological experiment operation. GCL and XFP contributed to statistical analysis. GCL and SYC contributed to drafting the manuscript. XFP contributed to the critical revision of the manuscript. All authors have read the manuscript and agreed to the conclusion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this study was obtained from the “Experimental Animal Ethics Certificate” of the Hubei Provincial Animal Management Committee, Hubei Province, China. Approval number: HLK-20210228-01.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic Ability Improvement Project of Young and Middle-Aged College Teachers, Department of Education, Guangxi Zhuang Autonomous Region, China (Grant Nos. 2017KY0290, 2021KY0322, and 2021KY0286).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Ethical Review System for Laboratory Animal Welfare of the Wuhan Myhalic Biotechnology Co., Ltd, China, and approved by the Administration Committee of Experimental Animals, Hubei Province, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
